Abstract
Introduction:
Congenital diaphragmatic hernia (CDH) is a disease with varying degrees of severity, from severe cases found prenatally who need fetal surgery to mild cases found after birth who undergo elective treatment. Regarding the repair of the diaphragm, thoracic and abdominal techniques through both open and minimally invasive approaches are available. In neonate cases, thoracoscopic repair is recommended because of the ease and safety for moving the organs from the thoracic cavity to the abdominal cavity. 1 However, applicable cases of endoscopic surgery is limited because of the varying degrees of severity. Pediatric surgeons, therefore, do not have the opportunity to perform many cases of thoracoscopic repair of CDH, which can reduce their readiness and familiarity with the procedure. We developed a training simulator of neonatal CDH that can reproduce patient-specific anatomical conditions. 2 Such simulator training for neonatal CDH was found to be effective in training inexperienced pediatric surgeons, 3,4 as shown in this video.
Methods:
A zero-day-old girl was transferred to our hospital suspected of having CDH. She was born at full term through vaginal delivery and was not found to have CDH prenatally. She showed cyanosis because of respiratory failure after birth. Chest X-ray showed intestinal herniation into the thoracic cavity. After intubation and ventilation, we planned to perform elective minimally invasive surgery by an inexperienced pediatric surgeon under the instruction of an expert surgeon.
Operative Findings and Procedure:
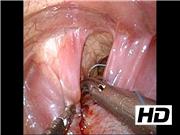
Before the operation, the surgeon underwent simulator training. We created a physical three-dimensional model of the thoracic cavity of the patient covered with rubber skin using her computed tomography data. Ultrasonography showed that the herniated organs were only the intestine, not the liver or spleen. We then reproduced this situation in the simulator model. The model was placed in a right lateral position, and three trocars were inserted (5 mm, middle axillary fourth intercostal space (ICS) for the endoscope; 5 mm, anterior axillary fifth ICS for the operator's right hand; 3 mm, posterior axillary fifth ICS for the operator's left hand). The training menu comprised the reduction of the intestine and repair of the diaphragm. After gentle reduction of the intestine, the defective diaphragm (3 × 1 cm in size) was closed using 3-0 BB Ethibond sutures (Ethicon, Cincinnati, OH) with Roeder's knot technique. After the operator had acquired sufficient skills, he performed the clinical operation. The patient's position and trocar placement were the same as in the simulator training. Artificial pneumothorax by CO2 inflation (5 mm Hg, 1 L/min) was established. Under inspection using a 5-mm 30° endoscope, the herniated organs were confirmed to be the small and large intestines that were then reducted. The defect of the diaphragm was 3.5 × 1.5 cm and was closed using BB Ethibond sutures in the same manner as in the simulator training.
Conclusion:
There were no intra- or postoperative complications. The patient was extubated 2 days after the operation, and no recurrence occurred. The postoperative course was uneventful. Simulator training depicting patient-specific anatomical conditions is effective, even for inexperienced surgeons and even when performed only shortly before the actual surgery.
Acknowledgment:
We thank Brian Quinn for his comments and help with the article.
No competing financial interests exist.
Runtime of video: 5 mins 3 secs


Get full access to this article
View all access options for this article.
