Abstract
People living with HIV (PLWH) beginning antiretroviral therapy (ART) retain a high burden of cytomegalovirus (CMV). CMV has been implicated in atherosclerosis in healthy adults, and a role in PLWH is plausible. Atherosclerosis has also been linked with γδ T cells and CMV seropositivity with altered γδ T cell profiles in other populations. In our cohort of PLWH starting ART in Jakarta (Indonesia), metrics of the CMV burden correlated with altered profiles of Vδ2− γδ T cells. Here CMV DNA was sought by RT-PCR as PLWH began ART. γδ T cell subsets were immunophenotyped using flow cytometry, and CMV-reactive antibodies were quantitated by ELISA after fixed intervals on ART. Carotid intima-media thickness (cIMT) was used to assess atherosclerosis. PLWH retained higher levels of CMV-reactive antibody than healthy controls (p = 0.001–0.04), and 50% began ART with detectable CMV DNA. cIMT values rose between 6 and 12 months on ART. At 6 months, cIMT correlated with CMV-reactive antibodies and proportions of activated Vδ2− γδ T cells (r = 0.56–0.57; p = 0.035–0.042) in PLWH who began ART with detectable CMV DNA. Hence, a high burden of replicating CMV may promote atherosclerosis in PLWH after a period on ART, and the role of activated Vδ2− γδ T cells warrants further study.
Introduction
Associations between cytomegalovirus (CMV) and markers of cardiovascular disease (CVD) in the general population have been confirmed by meta-analyses (Wang et al., 2017). Atherosclerosis is a common pathology underlying CVD, and plaque rupture can lead to chronic inflammation and stroke (Cristescu et al., 2022). T cells attract an inflammatory response against pathogens that initiate atherosclerotic plaque and progression (Galkina and Ley, 2009). This can include CMV, as CMV DNA was found in smooth muscle cells from patients with aortic aneurysms and atherosclerotic plaques from patients with carotid artery stenosis (Beyaz et al., 2019). In Indonesian people living with HIV (PLWH) beginning antiretroviral therapy (ART), we identified variable associations between levels of CMV antibody and carotid artery intimal thickness (cIMT) (Karim et al., 2017). cIMT is a noninvasive measure of endothelial dysfunction and an early indicator of atherosclerosis.
CMV reactivations drive the clonal expansion of terminally differentiated αβ and γδ T cells in healthy adults and transplant recipients (Halary et al., 2005; Lee et al., 2017). Indeed, there is evidence that populations of Vδ2− γδ T cells expanded by CMV may control CMV replication in transplant recipients (Halary et al, 2005). However, identification of associations between CMV, CVD, and γδ T cells in PLWH may require different metrics of CMV burden because all individuals are seropositive, and CMV DNA positivity is common. Proportions of Vδ2− γδ T cells were high in Indonesian PLWH and declined on ART, while proportions of Vδ2+ γδ T cells were low and correlated inversely with levels of CMV DNA and CMV-reactive antibody (Ariyanto et al., 2021).
There is growing evidence to support a role for γδ T cells in the early stages of atherosclerosis (Xu et al., 2024). Accordingly, studies in animal models showed γδ T cells can promote vascular injury (Caillon et al., 2017). γδ T cells are present in atherosclerotic plaques (Kleindienst et al., 1993), and numbers of circulating γδ T cells are reduced in individuals with a history of coronary artery disease (Adamski et al., 2014), suggesting recruitment to the vascular endothelium. Here, we examine associations between γδ T cells, cIMT, and metrics of the high burden of CMV found in Indonesian PLWH beginning ART.
Materials and Methods
Clinical assessments
The JakCCANDO study (Jakarta CMV Cardiovascular ART Neurology Dentistry Ophthalmology) at the HIV/AIDS clinic of Cipto Mangunkusumo Hospital (Jakarta, Indonesia) recruited 81 ART-naive PLWH of age 18–40 years, with <200 CD4+ T cells/μL, between 2013 and 2015. Blood samples were collected before ART (V0) and after 6 and 12 months on ART (V6, V12). Healthy adult controls (n = 20) were recruited from hospital staff and colleagues. The study was approved by the ethics committees of the Faculty of Medicine Universitas and Cipto Mangunkusumo Hospital, Indonesia, and informed consent was obtained from all participants. Stratification was based on the presence of CMV DNA at V0 in buffy coat samples, and 20 CMV DNA+ and 20 CMV DNA– participants were selected randomly from the cohort. Cardiovascular examinations included Doppler sonography to evaluate cIMT in the left and right carotid arteries (Karim et al., 2017).
CMV-reactive antibody and CMV DNA
CMV-reactive IgG was quantitated in 96-well plates coated with a lysate of human foreskin fibroblasts infected with the CMV strain AD169. Optical densities were compared with a standard plasma assigned a value of 1,000 arbitrary units and diluted over seven 3-fold dilutions. Patient samples were tested over four dilutions, starting at 1:10,000. Briefly, plates were blocked, plasma samples were added, and binding was detected using goat anti-human IgG—horseradish peroxidase (Sigma–Aldrich, St Louis, MI). Results were computed relative to the standard and are presented as Log10 values (Ariyanto et al., 2018). The in-house quantitative polymerase chain reaction assay targeting the UL54 gene has been described (Karim et al., 2017).
γδ T cell immunophenotyping
γδ T cell subsets were determined using peripheral blood mononuclear cells cryopreserved at V0 and V6. PBMC were stained with Fixable Viability Stain 620 and established as Tube 1: CD3 BV510 (clone UCHT1), CD27 PE (clone L128), CD8 PE-Cy7 (clone SK1), CD45RA APC-H7 (clone HI100), CD27 PE-Cy7 (clone L128), HLA-DR BV421 (clone G46-6), TCRγ/δ−1 PECy7 (clone 11F2), and Vδ2 FITC (clone B6) and Tube 2: TCRγ/δ−1 PECy7 (clone 11F2), Vδ2 FITC (clone B6), CD16 APC-H7 (clone 3G8) (Becton Dickinson Biosciences, San Diego, CA). Cells were then fixed with BD Cytofix/cytoperm, and eight-color data were acquired on a FACS Canto II (Becton Dickinson) and analyzed using FlowJo software (TreeStar, Ashland, OR) (Ariyanto et al., 2021).
Statistical analyses
Nonparametric statistical analyses were performed using Prism 8 (GraphPad, San Diego, CA). Wilcoxon paired tests were used to evaluate changes over time; Mann–Whitney unpaired tests were used to compare groups, and results are reported as a median (range). Correlation analyses were conducted using Spearman’s rank tests. A significance level of p < 0.05 marks statistically significant differences, but values of 0.05 < p < 0.10 are noted when they conform to a trend evident from other data.
Results and Discussion
Levels of CMV-reactive antibody and proportions of activated Vδ2− γδ T cells were high in PLWH
As described previously, most JakCCANDO patients (n = 81) cleared HIV RNA and displayed improved CD4+ T cell counts on ART. Here, 40 PLWH were selected on the basis of detectable CMV DNA at V0 (i.e., 20 CMV DNA+ and 20 CMV DNA–). This mirrors the frequency of CMV DNA positivity in the JakCCANDO cohort and marks a high and moderate burden of CMV, respectively, as all individuals were CMV-seropositive (Ariyanto et al., 2018).
The PLWH had higher levels of CMV-reactive antibody than healthy adults. They displayed increased proportions of Vδ2− γδ T cells at V0, with some resolution by V6. Activation of γδ T cells was assessed via the HLA-DR mean fluorescence intensity, and proportions of Vδ2− γδ T cells expressing CD8 or CD16 were also elevated at V0 and declined by V6 (Table 1). No significant correlations were evident between levels of CMV-reactive antibodies or CMV DNA and proportions of Vδ2− γδ T cells (Ariyanto et al., 2021).
Levels of CMV Reactive Antibody and Proportions of Activated Vδ2− γδ T Cells Were High in PLWH
Data are presented as median (range). Bold: p-value ≤ 0.05.
p-value: Mann–Whitney test.
p-value: Wilcoxon test.
MFI, median fluorescence intensity; cIMT, carotid intima-media thickness.
In the entire JakCCANDO cohort, PLWH had higher cIMT values than healthy adults (Ariyanto et al., 2018; Karim et al., 2017), suggesting inferior vascular health. In the subset studied here, high values were evident after 12 months on ART (Table 1).
Levels of CMV-reactive antibodies and proportions of activated Vδ2− γδ T cells correlated with elevated cIMT in HIV patients with high CMV burden
Following stratification based on the presence of CMV DNA at V0, the 20 CMV DNA+ and 20 CMV DNA− PLWH had similar baseline CD4+ T cell counts (p = 0.16), levels of CMV-reactive antibody (p = 0.24), and cIMT values (left p = 0.88, right p = 0.44) (data not shown). Six months later, the CMV DNA+ PLWH had marginally higher levels of CMV-reactive antibody (Log10: 3.01 [1.59–3.47] vs. 2.70 [1.19–3.47]; p = 0.09) and left cIMT (0.57 [0.39–0.70] vs. 0.51 [0.32–0.64]; p = 0.09). Although this suggested a link between CMV and cIMT, no differences achieved statistical significance. We therefore examined the relationship after stratification by the presence of CMV DNA at V0. At V6, we found positive correlations between cIMT and levels of CMV antibody (Figs. 1A and B) and expression of HLA-DR (Figs. 1C and 1D), CD8 (Fig. 1E), and CD16 (Fig. 1F) on Vδ2− γδ T cells in individuals who started ART with detectable CMV DNA. These data link the activation of Vδ2− γδ T cells at V6 with CVD in PLWH with a high CMV burden.
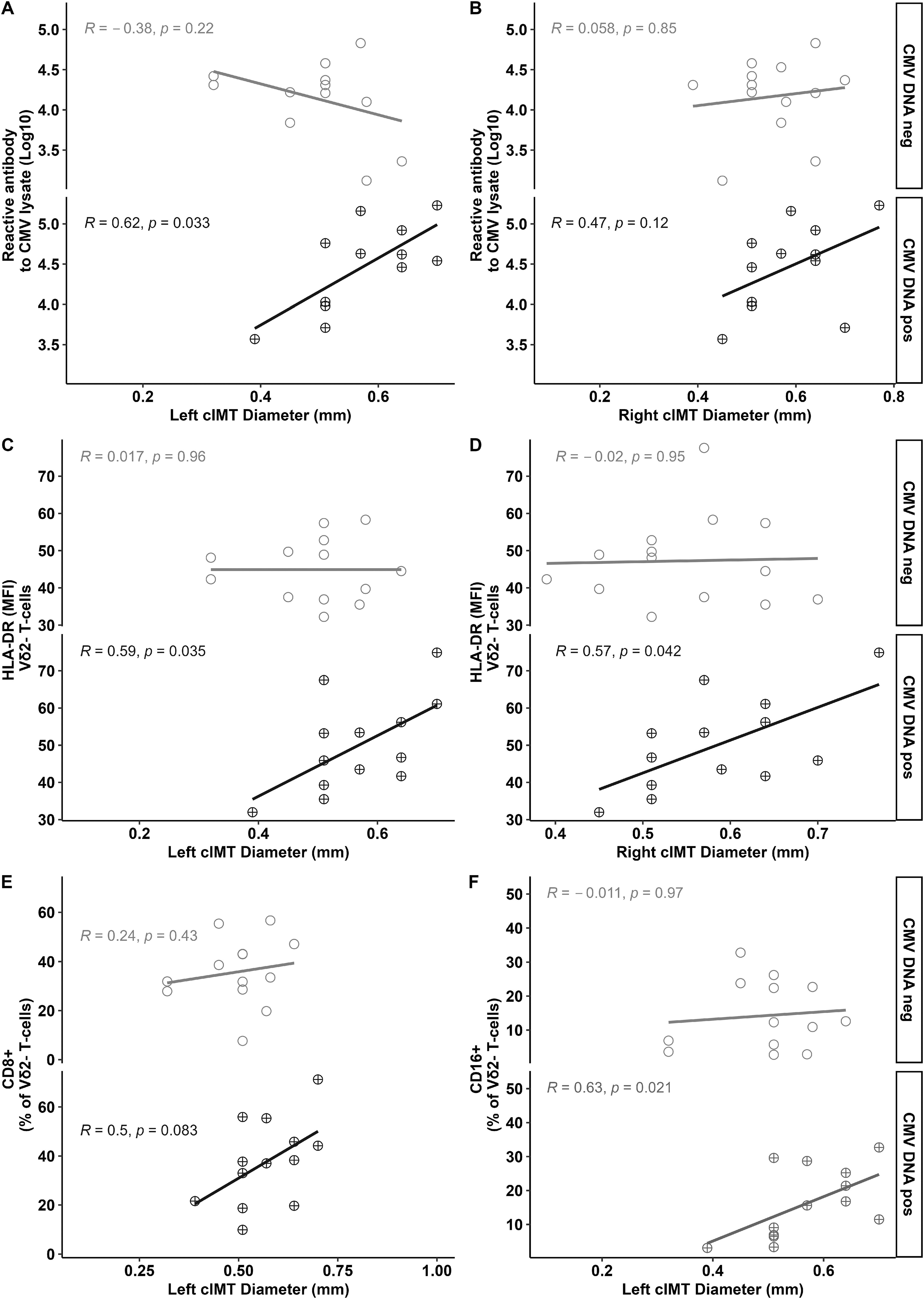
cIMT associates directly with levels of CMV-reactive antibody and activated Vδ2− γδ T cells in PLWH at V6 who began ART with detectable CMV DNA. Levels of CMV antibody at V6 correlated with left
We then examined the elevated cIMT values seen at V12 in more detail. At V12, cIMT values were no longer correlated with CMV antibody levels in CMV DNA+ PLWH (right cIMT, r = 0.21, p = 0.40; left cIMT, r = −0.11, p = 0.66) and were similar in PLWH who were CMV DNA+ and CMV DNA− respectively (right cIMT: 0.70 [0.45–0.8] vs. 0.67 [0.45–1.00] p = 0.35; left cIMT: 0.65 [0.45–0.80] vs. 0.70 [0.45–0.80] p = 0.14).
Conclusions
We describe a transient association between CMV and cIMT, preceding a rise in cIMT values after 12 months on ART. At V6, the association of cIMT with CMV antibodies was mirrored by an association with markers of the activation of Vδ2− γδ T cells. This finding supports further studies of the role of γδ T cells in CVD in individuals with very high burdens of CMV. As high CMV burdens are probably common in PLWH throughout the developing world, the metrics used to assess CMV must encompass high values. High levels of replicating virus may invoke pathogenic pathways not seen during healthy ageing in Australia and Europe.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The research was funded by a Universitas Indonesia PUTI Grant (NKB-634/UN2.RST/HKP.05.00/2024) and Curtin University. The authors express our gratitude to all PLWH and healthy controls who took part in JakCCANDO.
