Abstract
Introduction:
Standards for intraoperative nerve monitoring (IONM) during thyroid and parathyroid surgery are well established.1,2 Most monitoring systems use endotracheal tube-based electrodes. Applications of nerve monitoring include intraoperative neural identification and mapping, neural dissection, identification of impending neurologic injury, and neural prognostication at the conclusion of surgery, among others. Intermittent IONM (I-IONM) assesses the nerve at surgeon-directed specific timepoints using a hand-held probe. Continuous IONM (C-IONM) assesses the functional status of the nerve every few seconds without surgeon input required. C-IONM may be associated with lower early and permanent postoperative vocal fold paralysis compared with I-IONM. 3
Materials and Methods:
This 10-minute 49-second video summarizes the role of IONM for thyroid and parathyroid surgeries. Particular attention is paid to the differences between C-IONM and I-IONM. An instructional case demonstrating the use of C-IONM is included.
Results:
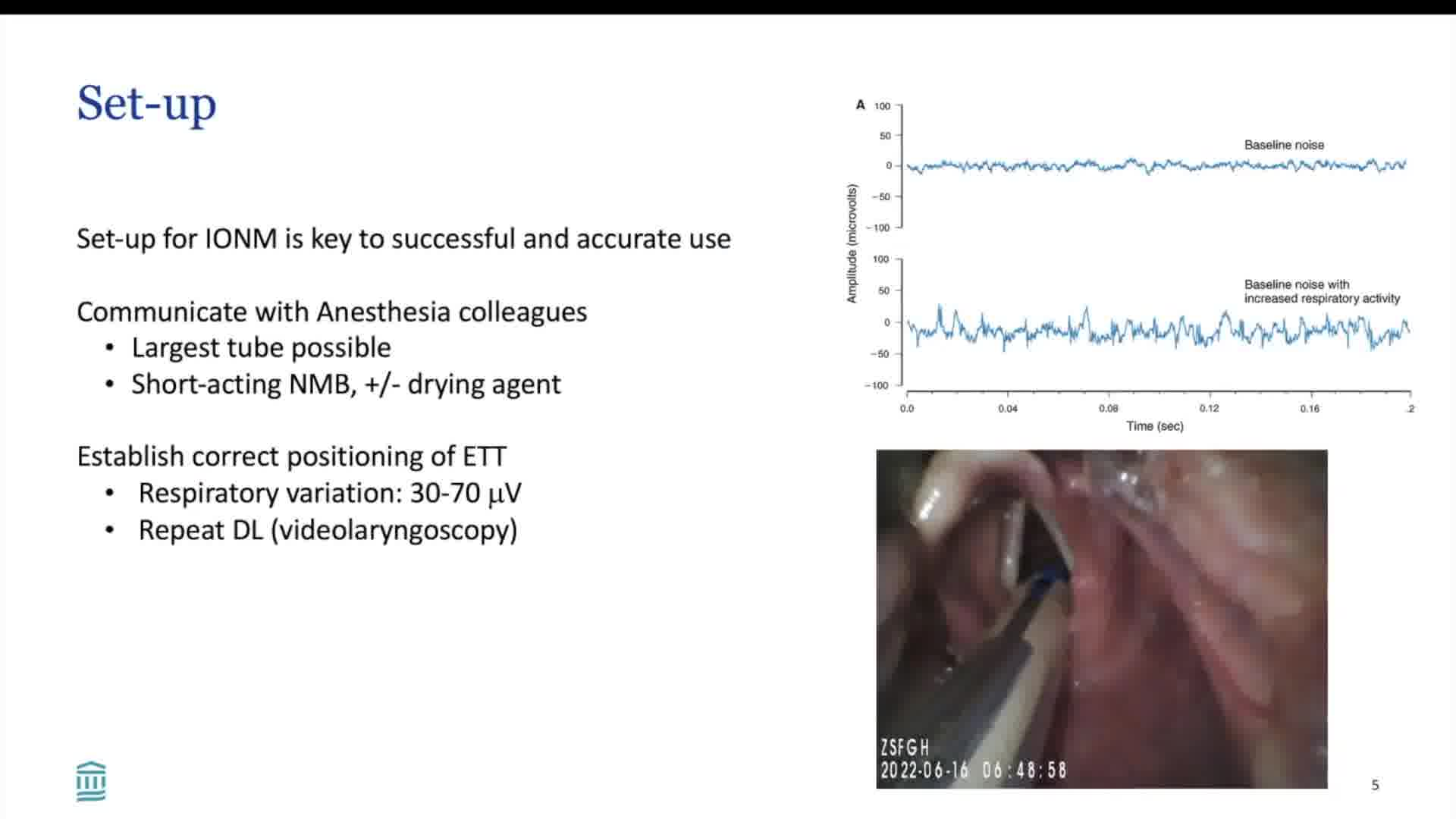
A baseline electromyographic (EMG) signal should be obtained to confirm a working monitoring system and an intact nerve. An adequate baseline includes an amplitude of at least 500 μV at a 1 to 2 mA stimulus, as well as a detectable laryngeal twitch. A severe combined event, marking impending neuropraxia, is defined as a concurrent decrease in EMG amplitude by at least 50% from baseline, and an increase in latency of at least 10%. C-IONM allows for real-time tracking of amplitude and latency changes, which can help direct the surgeon to avoid or cease maneuvers that precipitate adverse EMG changes. C-IONM does not lend itself to nerve mapping and identification, which are features of I-IONM. I-IONM and C-IONM are complementary in their functions.
Conclusions:
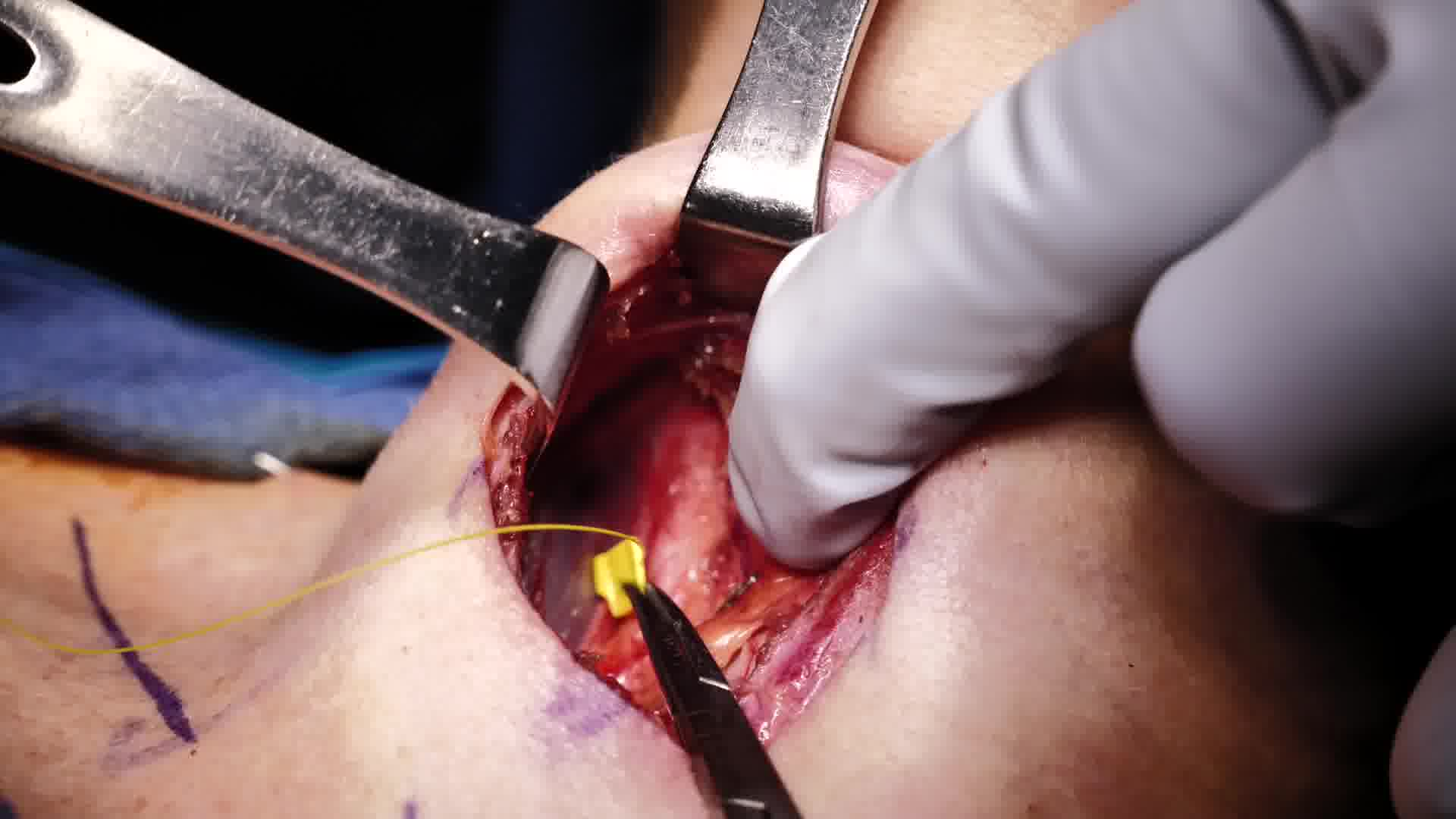
C-IONM has the ability to provide real-time feedback, potentially allowing avoidance of a slowly evolving nerve injury. The use of C-IONM with a vagal electrode is demonstrated in this video.
G.W.R. receives research institutional grants from Eisai, Fluoptics, and Medtronic. He is also a Medtronic consultant. All other authors have no relevant disclosures.
Runtime of video: 10 mins 49 secs

Get full access to this article
View all access options for this article.
