Abstract
Introduction:
Approximately 20% of thyroid nodules that undergo fine needle aspiration have an indeterminate cytology result, with a 10%–30% risk of malignancy.1-3 Molecular testing has been considered to better define the risk of malignancy in indeterminate thyroid nodules, with the goal of avoiding unnecessary diagnostic surgery for benign nodules.4-6
Materials and Methods:
This is a counterpoint debate providing evidence that molecular testing may not be necessary for indeterminate thyroid nodules. Data from the Mayo Clinic are reviewed.
Results:
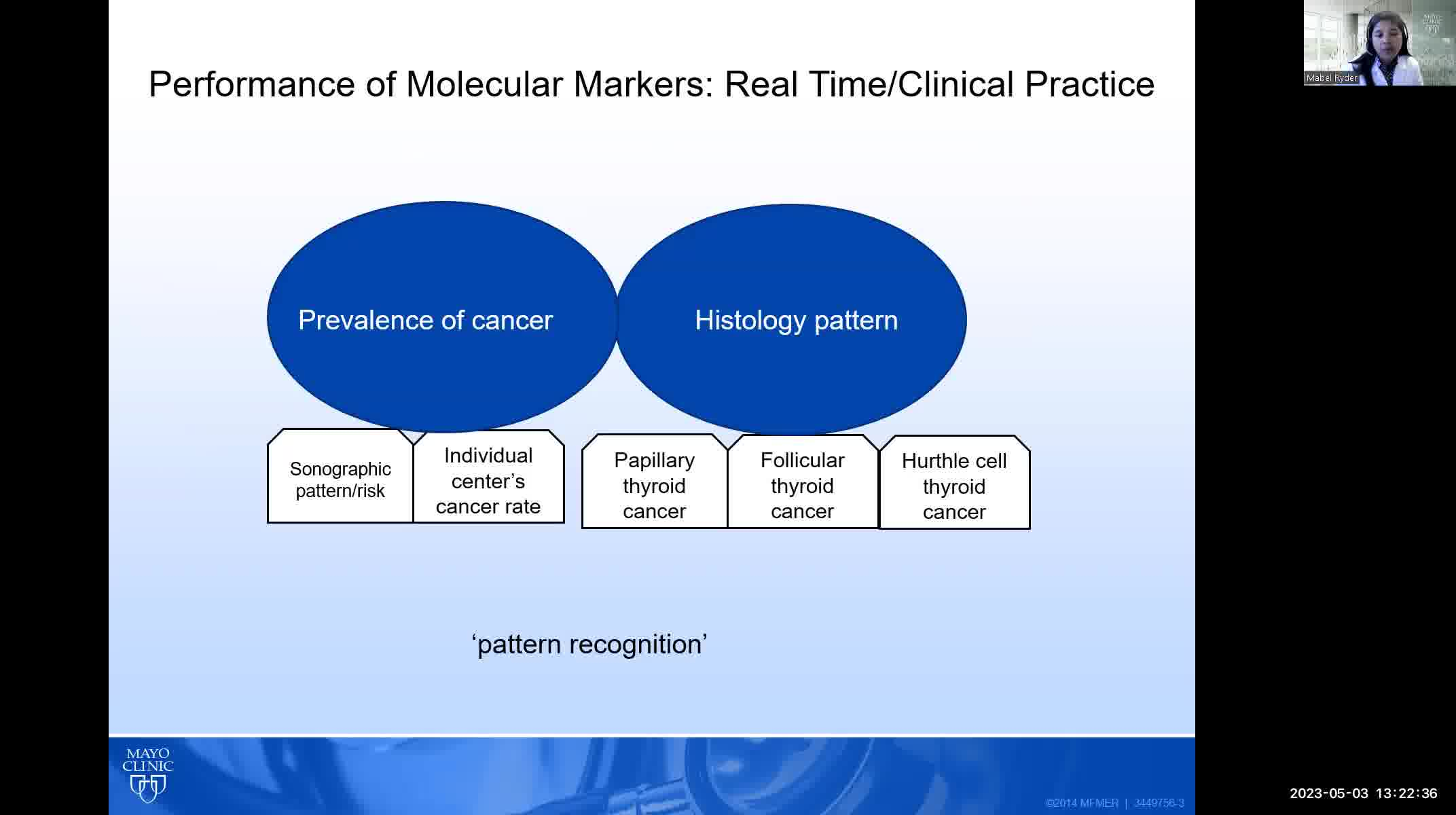
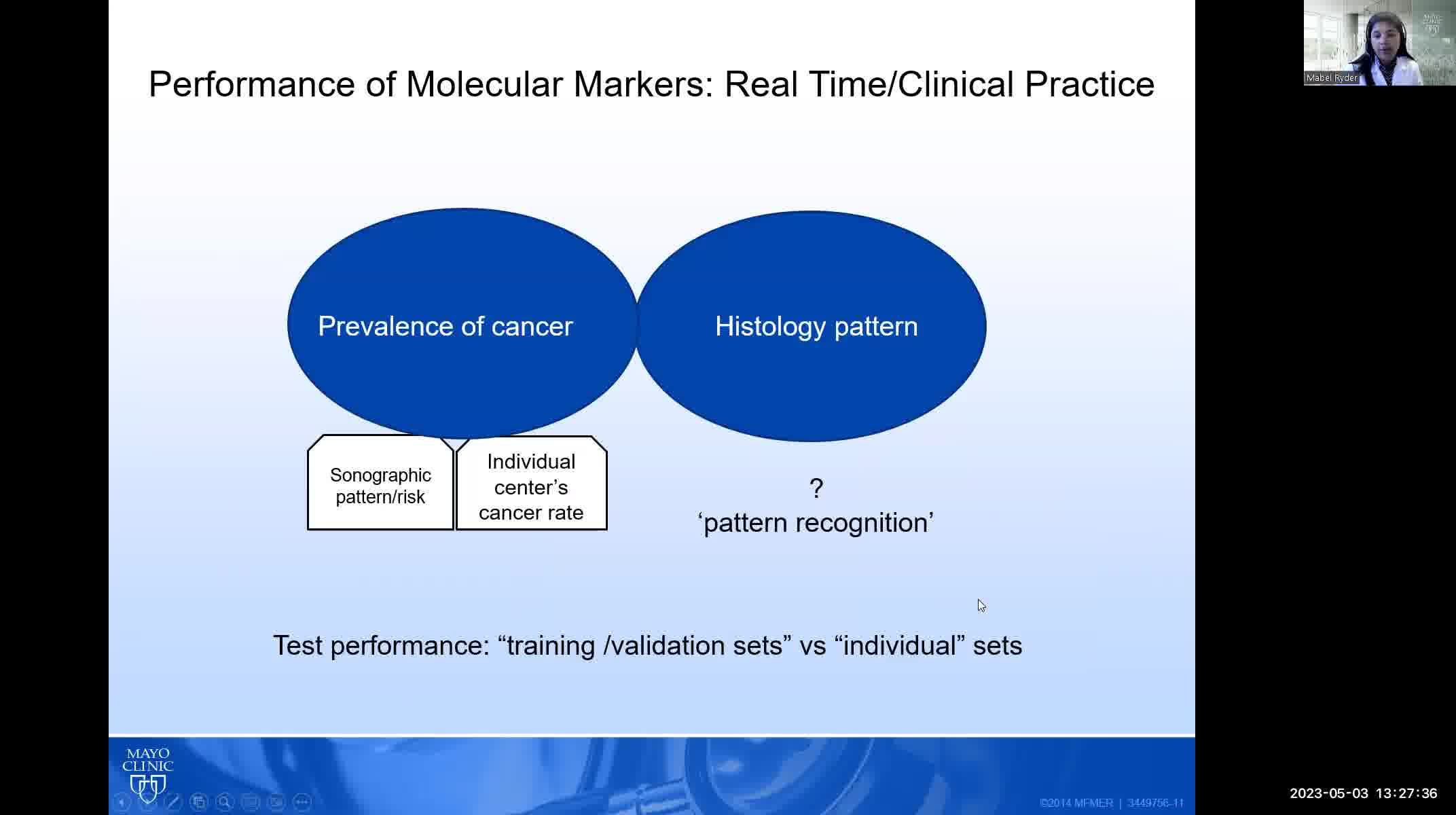
The three molecular tests that are commercially available in the United States (Afirma, Thyroseq, and ThyGeNEXT/ThyraMIR) were all trained primarily on indeterminate thyroid nodules that, when malignant, consisted mostly of papillary thyroid cancers. The performance of these tests in follicular and Hurthle cell cancers may be significantly worse than reported. Diagnostic performance of molecular testing is also dependent on the prevalence of malignancy in a given population. Most validation studies were performed at a prevalence of malignancy of ~20%; if the malignancy rate is significantly higher in a given institution, the negative predictive value will be lower resulting in more false negative cases.
Conclusions:
The prevalence of malignancy and patterns of both cytology and histopathology must be evaluated in each practice pattern to determine whether molecular testing will be accurate and useful.
No competing financial interests exist.
Runtime of video: 13 mins 52 secs

Keywords
Get full access to this article
View all access options for this article.
