Abstract
Abstract
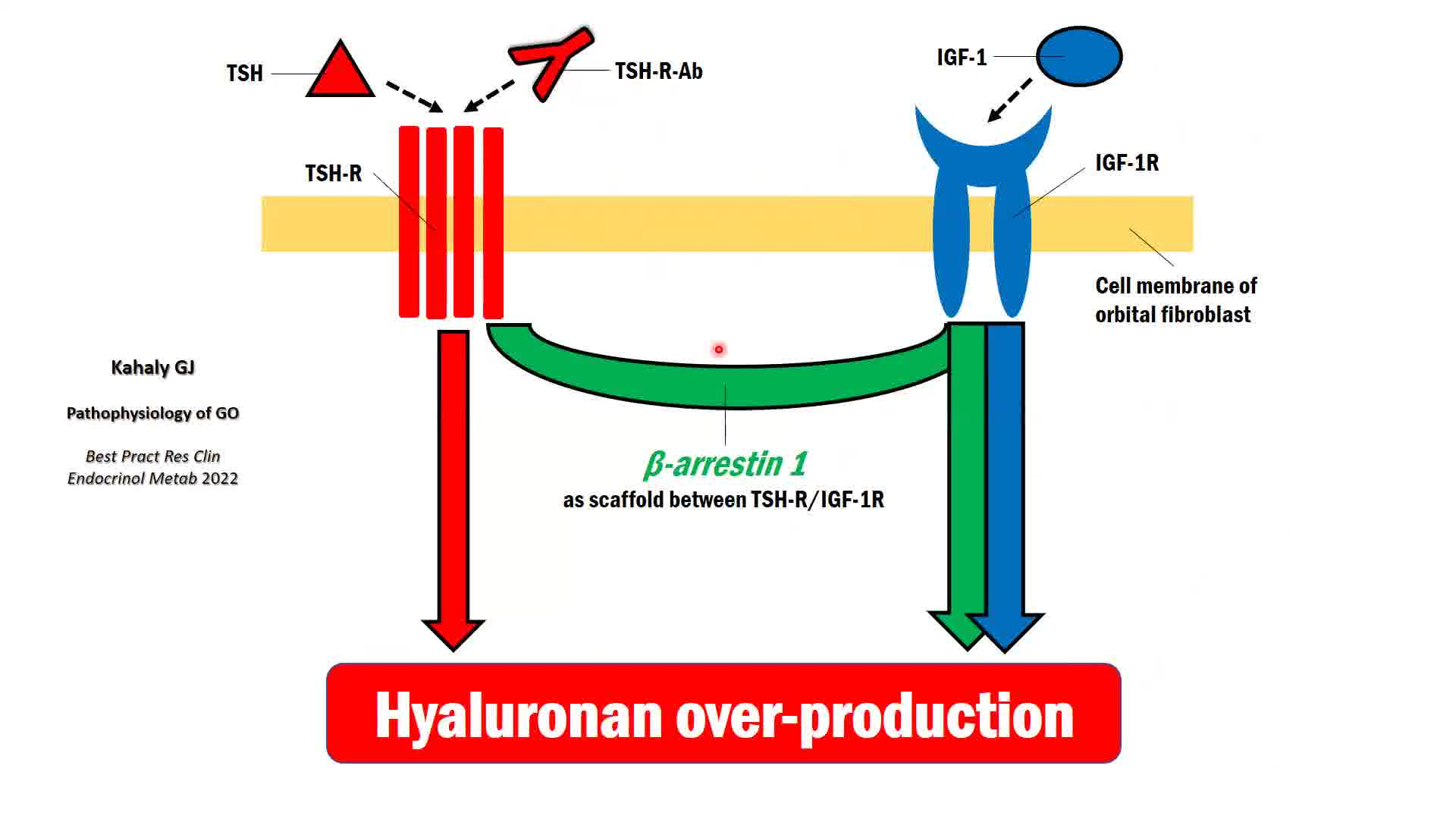
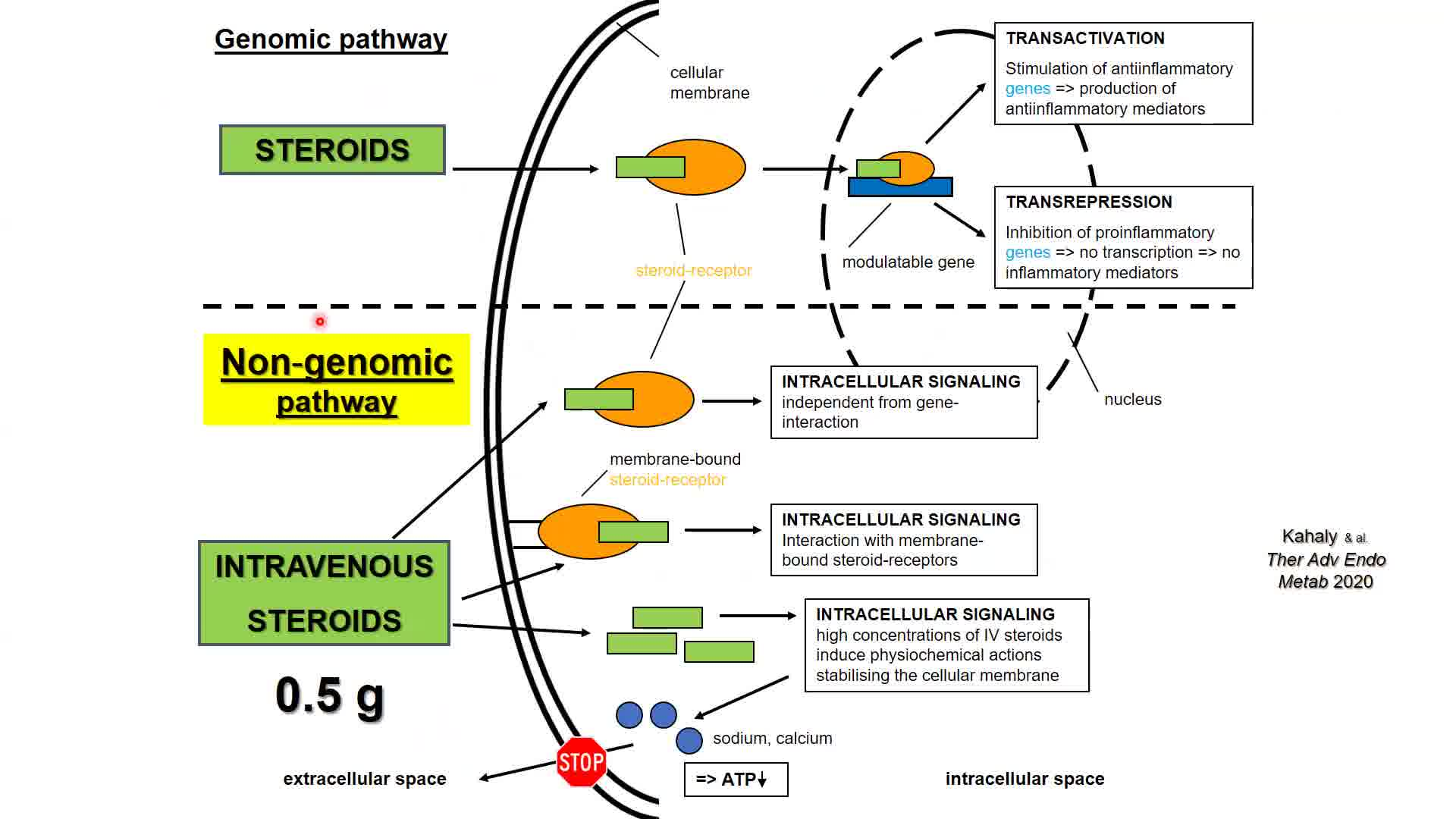
Graves' orbitopathy (GO), the most common extrathyroidal manifestation of autoimmune-induced Graves' disease, is characterized by orbital inflammatory infiltration and activation of orbital fibroblasts, which mediates de novo adipogenesis, excessive production of hyaluronic acid, myofibroblast differentiation, and ultimately tissue fibrosis. Interactions among T cells, B cells, and orbital fibroblasts result in their activation and perpetuation of orbital inflammation as well as tissue remodeling. T helper 17 cells belong to a newly identified pathogenic CD4+ T cell subset, which possesses prominent pro-inflammatory and profibrotic capabilities. Thyrotropin receptor (TSH-R)/insulin-like growth factor-1 receptor (IGF-1R) crosstalk and the downstream signaling pathways of both receptors represent the major mechanisms leading to activation of orbital fibroblasts. TSH-R autoantibody is the disease-specific biomarker of great clinical relevance and utility. There is growing evidence that oxidative stress, gut microbiome, and epigenetics also play a role in the pathogenesis and their manipulation may represent novel therapeutic strategies. For the past few decades, the scientific community has witnessed major advances in its understanding of the pathogenesis of GO. The identification of new potential therapeutic targets drives the development of more effective treatment of GO. The characterization of TSH-R/IGF-1R crosstalk signaling pathways and the clinical efficacy of the IGF-1R inhibiting monoclonal antibody teprotumumab are excellent examples on how translational research optimizes patient care. The international academic multidisciplinary consortium European Group on GO (EUGOGO) has recently published the European Society of Endocrinology endorsed guidelines for the medical management of GO (Bartalena, Kahaly, et al. Eur J Endocrinol 2021). 1 Hence, the thyroid community is optimistic that effective armamentaria in fight against GO, which target the disease-specific mechanisms, will continue to expand.
The Johannes Gutenberg-University (JGU) Medical Center, Mainz, Germany has received research-associated funding from the JGU Medical Faculty, Byondis, Glycoera, Horizon Therapeutics, Immunovant, ISAR, Mediomics, Merck, Novartis, Quidel and Roche. G.J.K. consults for Glycoera, Immunovant, ISAR, Merck, Novartis, Quidel and VasaraGen.
The author has received and archived patient consent for video recording/publication in advance of video recording of procedure. The author has secured permission to reuse the images included in the video from the original copyright holder.
Runtime of video: 13 mins 35 secs

Get full access to this article
View all access options for this article.
