Abstract
Background:
In the United States, West Nile virus (WNV) was first identified in 1999 in New York City and in Kansas in 2002. How the virus was introduced remains uncertain, although several potential methods have been hypothesized. This review presents an overview of what has occurred with WNV since 1999 and the potential for establishment of related viruses, such as Japanese encephalitis virus, if they were to be introduced in the United States.
Materials and Methods:
We have compiled human West Nile case data from Kansas over time with the purpose of illustrating how this virus has become endemic in the center of North America in a representative Midwestern state that is seasonal and does not have year-round mosquito vector activity. Case data were compiled from multiple sources including the U.S. Center for Disease Control and Prevention, the U.S. Department of Agriculture, and the Kansas Department of Health and Environment.
Results:
The annual reported numbers of human WNV infections have fluctuated widely in Kansas since the introduction in 2002. Between 2002 and 2022, there were a total of 715 reported cases in Kansas, with an average of 34 cases per year. Among the 715 cases, 73% were from 23 counties, but 4 counties accounted for 32% of this total with clusters of cases. Sedgwick County, which is the second most populous county in Kansas, represented 15% of the 715 total cases.
Conclusion:
The reasons for fluctuations in the number of cases reported each year and the distribution of these cases is uncertain, but they are most likely due to changes in the mosquito vector populations, which are influenced by environmental factors, such as seasonal rainfall, temperature, and humidity. With regards to the future, WNV is here to stay with annual fluctuations that are difficult to predict. The establishment of WNV in the U.S. and Kansas should serve as a warning for the possibility of establishment of other mosquito-vectored diseases, including Japanese Encephalitis virus.
Introduction: Mosquito-Borne Viruses in Kansas
In the publication “Arboviral infections in the United States” (Tsai, 1991), it was stated that of the 66 arboviruses that had been isolated in the United States and Canada, 16 caused human illness. These viruses were: California encephalitis (CEV), Colorado tick fever (CTFV), Eastern equine encephalitis (EEEV), Jamestown Canyon (JCV), LaCrosse (LACV), Lymphocytic choriomeningitis (LCMV), Oklahoma tick fever, Powassan (POWV), Rio Bravo (RBV), Seoul (SEOV), snowshoe hare (SSHV), St. Louis encephalitis (SLEV), trivitattus (TVTV), Venezuelan equine encephalitis (VEEV), Vesicular stomatitis (VSV), and Western equine encephalitis (WEEV). In terms of importance based on the case fatality rate, they were ranked as EEEV (>30%), SLEV (7%), SSHV (5%), VSV (1–5%), and WEEV (3%), with the fatality rate for others being 1% or less. The distribution of the viruses was not discussed in detail, although maps in the publication showed that WEEV and SLEV occurred in Kansas. Other viruses mentioned included two hantaviruses (Prospect Hill, Leakey virus), the arenavirus Tamiami, and two flaviviruses (Rio Bravo, dengue).
Calisher’s publication “Medically important arboviruses of the United States and Canada” (Calisher, 1994) provides the geographic distribution for CTFC, EEEV, LACV, POWV, SLEV, VEEV, and WEEV, and of these, cases of WEEV and SLEV had been reported in Kansas. The incidence of human WEEV cases in Kansas between 1955 and 1984 was 68 per 100,000 (Reisen and Monath, 1988). SLEV was first isolated from the large epidemics in St. Louis and Kansas City in 1933 (Webster and Fite, 1933). The incidence of human SLEV cases in Kansas between 1964 and 1984 was reported as 123 per 100,000 (Tsai and Mitchell, 1988). Between 2014 and 2018, no neuroinvasive cases of JCV, EEEV, or SLEV were reported in Kansas (Piantadosi and Kanjilal, 2020). Data for SLEV collected by the Centers for Disease Control and Prevention showed two human cases of SLE in Kansas in the period between 2002 and 2022, including one human case in Ellis County and another in Sedgwick County in 2004. In addition, West Nile virus (WNV) activity that was not associated with human cases was reported in Sedgwick County in 2019 and Reno County in 2018 (CDC, 2024b). A map published by Piantadosi and Kanjilal (Piantadosi and Kanjilal, 2020) suggested that LACV occurred in Kansas, albeit fewer than 10 neuroinvasive cases were reported in the period between 2014 and 2018.
WNV in the United States—a case of mistaken identity
Following the occurrence of eight human cases of encephalitis in New York City in August of 1999, it was concluded that the causative agent was SLEV based on serum analysis by enzyme-linked immunosorbent assay (CDC, 1999; Nash et al., 2001). However, the etiological agent was subsequently identified as WNV (Roehrig, 2013). How the virus was introduced remains uncertain, although several potential methods have been hypothesized (Rappole et al., 2000; Roehrig, 2013). WNV is a flavivirus that causes a zoonotic disease of significant public health concern in the United States and Kansas. WNV was confined historically to Africa and Europe, primarily transmitted by Culex sp. mosquitoes with birds being the major amplifying host. The introduction of WNV into New York in 1999 was the first time the virus was identified in the Western hemisphere (Kramer et al., 2019).
There was uncertainty about the virus’ ability to overwinter (Ronca et al., 2021), and although Lanciotti et al. (1999) stated that “it cannot be predicted whether the WN virus will reappear in the year 2000 …,” it was subsequently detected in a bird (Garmendia et al., 2000) and thereafter spread to New Jersey and Connecticut. The virus survived the winter months, perhaps in hibernating vector mosquitoes (Nasci et al., 2001), and then was subsequently disseminated, likely by avian hosts (Rappole et al., 2000). Despite control efforts, the virus spread throughout the continental U.S. and by 2012 had been detected in all 48 continental states and the District of Colombia (Garcia et al., 2015; Ronca et al., 2021).
Following the introduction of WNV into the United States, the virus displayed a remarkable capacity to infect species to which it had previously been unexposed. The virus has been isolated from over 60 species of mosquitoes in many genera and over 300 species of birds (Petersen et al., 2013). To date, millions of birds have likely been infected and killed by the virus. Horses are a dead-end host that do not contribute to the transmission cycle; however, almost 30,000 equine cases, many of them fatal, have been reported. Additionally, almost 3000 human fatalities have been attributed to WNV in the United States, with over 30,000 neurological cases (CDC, 2024e) and over 5 million potential human infections. Since most human cases result from exposure to infected mosquitoes, WN is a seasonal disease, with cases and incidence being correlated with the presence and abundance of competent mosquito species.
Human WNV infections in Kansas
Cases of human WNV have occurred in Kansas every year since 2002, with 779 cases causing 411 cases of neuroinvasive disease, with 317 positive blood donors identified from 2002 to 2023 (CDC, 2024d) (Table 1). However, between 70% and 80% of infections are asymptomatic, with 20–30% resulting in West Nile fever, and less than 1% are neuroinvasive (CDC, 2024c). These percentages, combined with no active surveillance for WNV in Kansas, suggest that the actual number of WNV infections was at least 10-fold higher than the number reported. Although there are four WNV vaccines approved for horses (AAEP, 2024; Saiz, 2020), despite many vaccine trials (Ronca et al., 2021), there is no approved vaccine to protect humans from infection (Gould et al., 2023).
Human Cases of West Nile Virus Reported in Kansas (Kansas Department of Health and Environment and U.S. Center for Disease Control and Prevention Data) Since It Was Introduced into the United States in 1999
Distribution of human WNV cases in Kansas
WNV was first reported in Kansas during 2002 that included 22 cases distributed across 13 of Kansas’ 105 counties (Clay, Douglas, Ellis, Franklin, Harper, Johnson, Pratt, Reno, Rice, Sedgwick, Shawnee, Smith, and Wyandotte). The annual reported numbers of human infections with WNV based on reports have fluctuated widely since the introduction in 2002 (Table 2).
Human cases of West Nile virus reported in Kansas by county (Kansas Department of Health and Environment and U.S. Center for Disease Control and Prevention data) since it was introduced into the United States in 1999
Human cases of WNV have been reported every year since 2002, although the numbers of cases reported annually have varied considerably between just 4 in 2011 and the maximum of 91 in 2003 and 2013. Of the 105 counties in Kansas, 14 have had no cases (Allen, Anderson, Bourbon, Brown, Cherokee, Coffey, Elk, Jefferson, Labette, Miami, Morton, Sheridan, Wilson, and Woodson). In the 21-year period between 2002 and 2022, there have been a total of 715 reported cases, with an average per year of 34 cases.
Of the 91 counties that have reported human cases, 15 have only had cases during a single year. However, there does not seem to be any obvious pattern or trend with these single events occurring in 12 of the 21 years: Lyon (2003), Geary (2004), Cheyenne and Neosho (2006), Greenwood and Montgomery (2007), Washington and Wichita (2008), Doniphan and Grant (2011), Edwards (2012), Chautauqua and Ottawa (2013), Comanche (2014), Nemaha and Osage (2015), Wabaunsee (2017), and Rawlins (2018).
Among the 715 cases, 73% were from 23 counties. However, 4 counties accounted for 32% of this total with clusters of cases (Barton with 33 total cases in 10 of the 21 years, Johnson with 59 cases in 19 of the years, Reno with 35 cases in 13 of the years, Sedgwick with 107 cases in 18 of the years). The 107 cases in Sedgwick County represented 15% of the 715 total cases. With regards to resident populations (census data for 2020), Johnson and Sedgwick are the two most populous Kansas counties (>600,000 and >500,000 people, respectively). Reno has over 60,000 residents while Barton has only 25,000. Of the 105 counties, 23 have populations over 25,000, but only 5 have more than 100,000. The Kansas county map in Fig. 1 was generated to represent relative risk of infection in each county, based on the average number of human cases in each county calculated as the total number reported since 2002 divided by the number of years since WNV was first reported in the county.
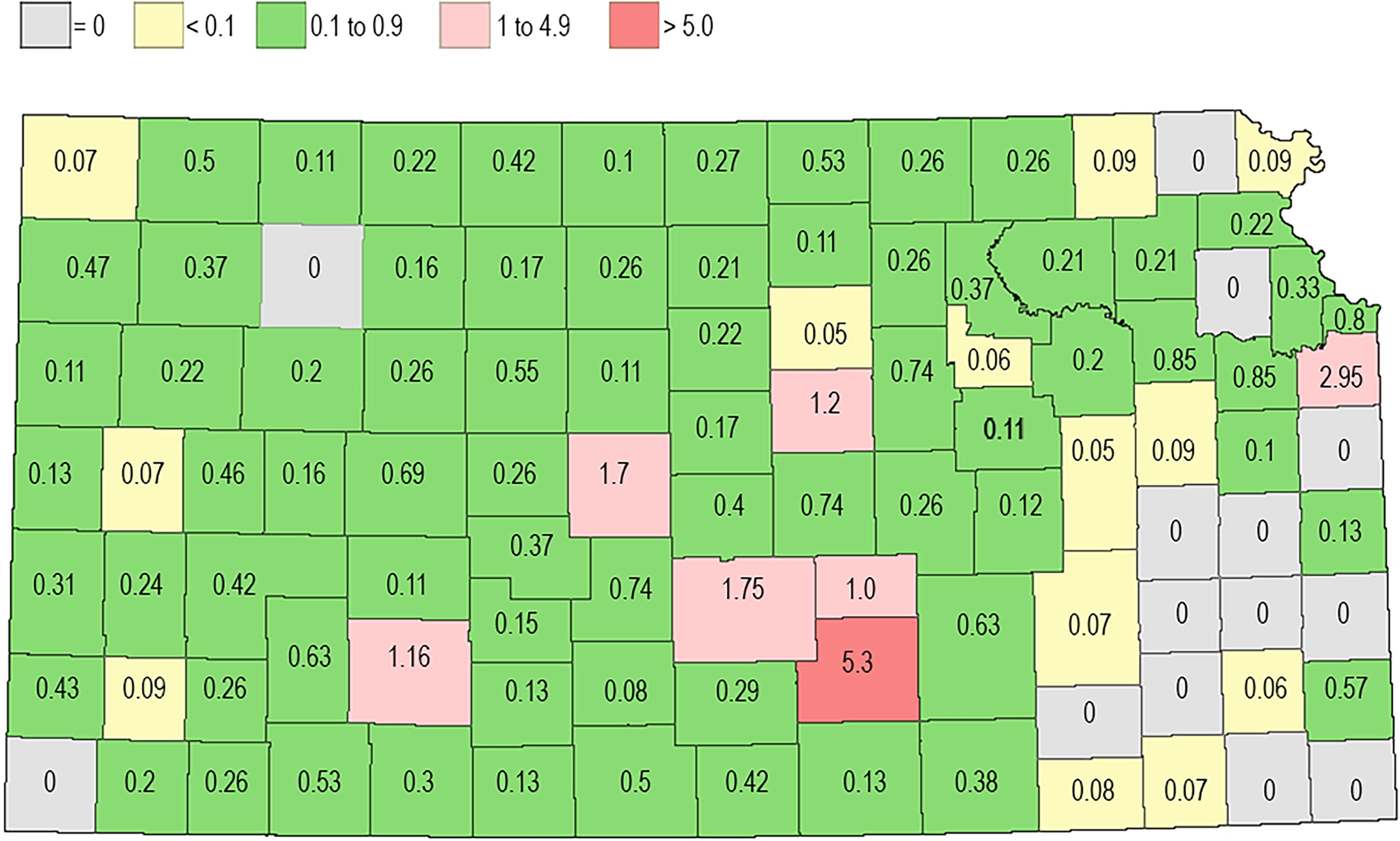
The map shows the average number of human cases in each county calculated as the total number reported since 2002 divided by the number of years since WNV was first reported in the county. WNV, West Nile virus.
Equine cases of WNV in Kansas
The number of WNV cases among equine in the United States have ranged from 25 in 1999 to a peak of 15,257 in 2002, with a total of 29,364 cases from 1999 to 2023. In Kansas, 793 cases were reported in 2002, followed by an additional 152 cases through 2021, ranging from no cases during 9 years and from 1 to 81 cases during 12 years. After the peak number of cases during 2002 in the United States, there were 5181 in 2003, 1406 in 2004, 1088 in 2005, and 2086 in 2006. After 2006, annual case numbers have always been below 700. This dramatic decline is likely attributed to the availability and widespread use of approved vaccines to protect horses from WNV infection. According to United States Deparment of Agriculuture (USDA) Animal and Plant Health Inspection Service data, equine cases in Kansas are as follows: 2003 (81 equines), 2004 (9), 2005 (14), 2006 (4), 2007 (5), 2008 (0), 2009 (0), 2010 (0), 2011 (0), 2013 (9), 2014 (0), 2015 (0), 2016 (2), 2017 (1), 2018 (7), 2019 (0), 2020 (3), 2021 (3), 2022 (2), and 2023 (13) (Statista, 2024; USDA, 2024).
Limitations of the Study
The data used to produce this report were obtained from various sources, primarily websites of the U.S. Center for Disease Control and Prevention (CDC), the USDA, and the Kansas Department of Health and Environment (KDHE). During analysis, some discrepancies were observed between numbers published by the CDC and those published by the KDHE. Some of these differences may be due to changes in how case definitions were defined over the years or due to retrospective updates of data as tests were verified. The CDC website, from which some data were obtained, actually states “surveillance data have several limitations that should be considered when using and interpreting the data,” with five specific issues that may result in limitations on the ArboNET data (CDC, 2024e). There is also some ambiguity with regards to whether or not numbers of, for example, neuroinvasive cases exclude fatalities that are reported as deaths, or whether data for seropositive blood donors were included in total positive case numbers. In comparing totals from the CDC and numbers from the KDHE, it certainly seems that fatal cases were included in the totals for some years. Data from the KDHE were only available back to 2012. Where data were not available from one source, we extrapolated to calculate the numbers consistently. Because of these inconsistencies and ambiguities, some of the numbers in Table 1 do not add up correctly. However, they are, we believe, as close to accurate as possible and do not impact our interpretations and conclusion. During the preparation of the article, the availability of some data available on websites changed. For example, a critical link for the CDC website from which data were obtained was updated and some of the data are no longer available. Since the acceptance of this publication, the availability of some federal data on websites changed and some links are currently disabled. These include https://wwwn.cdc.gov/arbonet/maps/ADB_Diseases_Map/index.html, http://www.aphis.usda.gov/vs/nahss/equine/wnv/wnv_distribution_maps.htm, and https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/sa_animal_disease_information/sa_equine_health/sa_west_nile_virus/ct_wnv_index/. The data spreadsheets and maps were, however, downloaded and are available from the authors on request.
Discussion
As reported in this review, since the introduction of WNV to the United States in 1999, there has been widespread occurrence of variation in annual epidemics and in the number of cases in Kansas as well as other states where WNV occurs (Petersen et al., 2013). Although there is strong evidence for the role of temperature as a driver of such variation, a modeling study revealed that climatic droughts rather than independent seasonal or winter temperatures, or precipitation were the main driver of increased epidemics of WNV (Paull et al., 2017). According to the findings, drought increased epidemics because of changes in prevalence rate of mosquito infection rather than mosquito abundance, and that acquired human immunity resulted in limited transmission. These observations were consistent with the results of a study in Suffolk County, New York, that showed WNV activity based on the yearly percentage of WNV-positive Culex pools to be greater when summer temperatures were hotter and during the drier years (Shaman et al., 2011). Although there is a wealth of literature implying that climate change associated with increasing temperature and precipitation are key determinants of the expansion of the geographic range and abundance of arthropod vectors that leads to an increase in the incidence of vector-borne diseases, further studies are warranted to understand more specifically the mechanism leading to any climate-induced changes in the incidence of vector-borne diseases.
With respect to WNV in Kansas, the reasons for the observed fluctuations in the number of cases reported each year and the distribution of these cases remain uncertain, but they are most likely due to changes in the mosquito vector populations. These population changes are influenced primarily by environmental factors, specifically seasonal rainfall, temperature, and humidity. Ecologically, there are inevitably some differences between counties, for example, Barton County is home to wetland habitats, at least in rainy years. The Playa Lake system that stretches from Nebraska to Texas occurs in counties in the West half of Kansas (https://kaws.org/projects/playa-lakes/). Although some Kansas rainfall data are available (https://mesonet.k-state.edu/climate/precip/county/), given the period covered in this review, we were unable to analyze data for each of the 105 counties. There are no data to link the annual fluctuations with climate change per se, although there are many speculative publications that suggest that vector-borne diseases may change with changing climate trends.
WNV is undoubtedly the best example of how a foreign zoonotic, mosquito-borne disease could be introduced into the United States and become established here. Nobody anticipated it, initial investigations misidentified it (Roehrig, 2013), and predictions that it would not survive the winter were wrong (Reisen and Wheeler, 2019). Its introduction revealed a lack of preparedness on a grand scale. Despite efforts to expand capabilities, and despite clear indications of the potential for widespread dispersal of chikungunya and Zika viruses, both viruses were introduced into the United States during 2016. Initially transmission occurred locally, but local transmission has not occurred since 2017 (Castro et al., 2017). Fortunately, intense mosquito control efforts have prevented these viruses from becoming established in North America, although this is not the case in some countries, for example, Brazil, in which these viruses have become endemic. In the United States, Zika and chikungunya have predominately been traveler-associated, with the exception of local transmission of Zika in Florida and Texas during 2016 and 2017 (Castro et al., 2017). Since its introduction into the United States in 1999, WNV has become the most common mosquito-borne virus in the United States and Kansas (Roehrig, 2013), with a total of 59,141 human cases reported in the United States and 2958 deaths (CDC, 2024a). The recent hospitalization of Dr. Anthony Fauci because of his infection with WNV has certainly put a spotlight on the disease, but before this, the Kansas Department of Health was issuing public warnings about risks associated with WNV and by August 23, 2024, had placed every county on the high-risk level even though several counties have never reported cases of WNV.
With regards to the future, clearly WNV is here to stay, and without an approved vaccine for humans (Gould et al., 2023), cases will continue to occur with annual fluctuations that seem difficult to predict (Paull et al., 2017). Now the remaining questions are, what other viruses may be introduced into the United States, and will we be adequately prepared to rapidly identify a new virus and respond more effectively so that it can be controlled and eliminated before it spreads (Gould et al., 2023)? Japanese encephalitis is certainly a virus of concern as shown by its introduction and perhaps permanent establishment in Australia (Williams et al., 2022). Based on experimental data, we know that some North American species of mosquitoes are susceptible to infection and can transmit the virus (Huang et al., 2016a; Huang et al., 2016b). Furthermore, U.S. breeds of domestic swine can be infected with Japanese encephalitis virus (JEV) and could therefore be amplifying hosts (Lyons et al., 2018). Feral phenotype swine, which are continuing to spread and increase in abundance in the United States, have also been shown to be susceptible to JEV infection and might act as an important wildlife component of the JEV transmission cycle (Park et al., 2023).
Footnotes
Acknowledgment
The authors would like to thank Kerri Cole for assistance in collating and tabulating data.
Authors’ Contributions
S.H.: Writing—original draft, conceptualization, investigation, and writing—review and editing. S.M.H.: Resources and writing—review and editing. A.M.A.: Review and editing. N.M.I.: Review and editing. D.L.V.: Conceptualization, writing—review and editing, and funding acquisition.
Author Disclosure Statement
S.H. is editor of Vector-Borne and Zoonotic Diseases. All other authors have no competing financial interests.
Funding Information
