Abstract
Introduction:
Lyme disease (LD) surveillance yields useful information to monitor the disease trends and spatial distribution. However, due to several factors, the Manitoba Health surveillance system, as with other systems, could be subject to underreporting.
Objectives:
To estimate the number and incidence of clinician-diagnosed LD over the study period in Manitoba, describe the epidemiology of clinician-diagnosed LD, and compare the findings with Manitoba Health LD surveillance data during the same period to estimate the extent of underreporting.
Methods:
A retrospective analysis of administrative health data was performed to calculate the number and incidence of clinician-diagnosed LD from 2009 to 2018 in Manitoba and describe the epidemiological characteristics using the International Classification of Diseases, Tenth and Ninth Revision (ICD-10-CA and ICD-9-CM) codes, and antimicrobial drug prescriptions.
Conclusion:
Of the 1,629,698 registrants within the Manitoba Health Insurance Registry followed over 10 years, 1658 LD events were identified. Most of the cases occurred from May to July and corresponded to the peak activity of the nymphal stage of the blacklegged tick in the province. LD events presented a bimodal distribution with a peak in children between 5 and 9 years of age for both sexes, and a peak in adults from 65 to 84 and from 50 to 74, respectively, for males and females. We estimated that an average of 165 LD events occurred annually in Manitoba (mean annual incidence of 10.17 per 100,000 population), compared with 30 cases per year reported in the surveillance system; therefore, the LD surveillance yields an important underreporting.
Introduction
Lyme disease (LD), the most reported vector-borne disease in the Northern Hemisphere, is caused by the spirochete Borrelia burgdorferi sensu stricto, which is transmitted by Ixodes scapularis in eastern and central Canada and Ixodes pacificus in British Columbia. The geographic range of ticks I. scapularis has expanded northward in the central and eastern provinces, probably due to climate change and ecological factors (Leighton et al., 2012; Tardy et al., 2021), resulting in a significant increase in the incidence of human LD. The number of LD cases reported by the provinces to the
LD is a multisystemic disease that evolves in progressive stages (Hatchette et al., 2014; Steere and Sikand, 2003). Early clinical manifestations may present with an erythema migrans (EM) rash and can accompany flu-like symptoms such as fever, fatigue, headache, myalgia, or arthralgia. At the early localized stage, the disease can generally be treated successfully with antimicrobial treatment (Steere and Sikand, 2003). If left untreated, stage 2, or early-disseminated infection, can develop, and more serious LD complications can occur (Hatchette et al., 2014). Manifestations of this phase may include multiple secondary EM lesions, neurological manifestations (e.g., facial palsy and meningitis), and cardiac symptoms (e.g., heart block), which may, on rare occasions, be fatal (Centers for Disease Control and Prevention, 2013). Untreated early-disseminated infection can progress to the late-disseminated stage, 6 months to several years later, affecting several organs, with many cases developing articular manifestations (Bacon et al., 2008; Steere et al., 1977).
LD became nationally notifiable in 2009 and is reported through a passive surveillance system in Canada. Provincial and territorial public health authorities report confirmed and probable LD case counts by age and sex to the PHAC through the Canadian Notifiable Diseases Surveillance System. In addition, the Lyme Disease Enhanced Surveillance system captures clinical symptoms, exposure history, and laboratory data collected by some provinces and shared with PHAC (Public Health Agency of Canada, 2023b). A revised national case definition was implemented in 2016 that defined several methods to identify LD risk areas according to the context of the emergence of the disease in each region of the country; however, case classifications (e.g., probable, confirmed) remain similar to the previous case definition (Public Health Agency of Canada, 2017). In Manitoba, since 2009, LD cases that meet the confirmed and probable PHAC surveillance case definition were reported by the laboratories and/or the health professionals to regional health departments (Manitoba Health, Seniors and Active Living, 2016) under the province’s Public Health Act (Government of Manitoba, 2009). Manitoba Health uses these data, in combination with vector surveillance data, to assess and communicate to the population LD transmission risk through annual reports (Manitoba Health, Seniors and Active Living, 2020; Manitoba Health, Seniors and Active Living, 2017; Manitoba Health, Seniors and Active Living, 2016). From 2009 to 2018, the incidence of LD cases reported by Manitoba Health increased from 0.40 per 100,000 population in 2009 to 3.97 per 100,000 population in 2018, representing a ninefold increase (Manitoba Health, Seniors and Active Living, 2020; Manitoba Health, Seniors and Active Living, 2017). A study from the United States using administrative health data suggests that up to a 10-fold underreporting of LD may be present (Nelson et al., 2015). No comparable studies have yet been carried out to investigate to what extent underreporting of LD is occurring in Canada. Ogden et al. suggested that approximately one-third of LD cases are reported (Ogden et al., 2019). Unlike the studies in the United States, the study by Ogden et al. was mainly based on the extrapolation of human LD surveillance data as well as tick (vector) and the LD pathogen surveillance data, and not on administrative health data. LD reporting is challenging for numerous reasons (Tseng et al., 2015). During the early phase of the disease, the recommended two-tier testing has poor sensitivity (Theel, 2016). Many clinical manifestations may be nonspecific, and laboratory tests of patients who live in areas where the disease is rare have a low positive predictive value (Lantos et al., 2015; Tugwell et al., 1997). Misclassification of cases reported by clinicians may also occur. Reported LD diagnoses may be classified as noncases, and confirmed cases may be classified as probable cases and vice versa if the case definition criteria are not specific enough or local health units do not have the resources to investigate the cases and collect the information needed.
The overall objective of this study is to assess the utility of applying LD diagnosis codes and prescribed antimicrobial algorithms to administrative health data (AHD) to estimate clinician-diagnosed LD in Manitoba. These estimates will help assess the level of underreporting of LD surveillance in Manitoba. More specifically, this descriptive analysis aims to use the Manitoba Centre for Health Policy (MCHP) database to (1) estimate the number and incidence of clinician-diagnosed LD over the study period in Manitoba, (2) describe the epidemiology of clinician-diagnosed LD, and (3) to compare the findings with Manitoba Health LD surveillance data during the same period to estimate the extent of underreporting.
Methods
Data sources
To estimate the incidence of clinician-diagnosed LD, we conducted a population-based retrospective cohort study using a linked population-level administrative health database housed at the MCHP in Manitoba, Canada (Smith et al., 2015). This database allows data pertaining to unique individuals to be linked through a deidentified (scrambled) personal health identification number.
Counts of the Manitoba population by year were obtained using the Manitoba Health Population File, which provides a count of the Manitoba population by age and sex yearly (Government of Manitoba, 2023). It represents a snapshot of Manitoba residents, usually on December 31 of each year. This dataset is built from the Manitoba Health Insurance Registry, which is a longitudinal population-based registry of all individuals registered to receive health services in the province.
The study included registrants in the Manitoba Health Insurance Registry from January 1, 2009, to December 31, 2018. Patient demographics (e.g., age, sex) from the Manitoba Health Insurance Registry were linked with administrative health datasets at the individual level as described elsewhere (Smith et al., 2015).
The discharge diagnoses coded using the International Classification of Diseases, tenth edition, Canadian modification (ICD-10-CA) system, captured in the Discharge Abstract Database (DAD) and claims including the codes for a physician-assigned diagnosis captured in the Physician Medical Claims database, were searched for registrants. In Manitoba, the 5-digit ICD, ninth revision, clinical modification (ICD-9-CM) codes in medical claims were phased in from 2015 to 2018. For those years, we searched for the appropriate code based on the precision available in the data. Discharge diagnoses and physician claims for LD were identified using the ICD-9-CM and ICD-10-CA codes 088 or 088.81 and A69.2, respectively. This method has been used in the United States to estimate the number of LD cases per year using information from an extensive nationwide medical claims database (Nelson et al., 2015). Each instance of these codes was defined as a potential LD event. For each event, the date of admission or first visit to the medical clinic that met the study inclusion criteria was considered the date of the event.
LD diagnosis and associated treatment were then pooled at an aggregate level. Diagnoses occurring in the same year were aggregated into potential LD events for each patient; therefore, the first diagnosis in each year that met the study definition was considered the incident potential LD event. Antimicrobial drugs recommended for LD treatment, prescribed with a minimum of 7-day supply, and dispensed within a 30-day window of the onset of each potential LD event, were searched in the Drug Program Information Network database. This approach has been used in several studies to estimate the prevalence of clinician-diagnosed LD in the United States (Kugeler et al., 2021; Nelson et al., 2015; Schwartz et al., 2021). If one or more such dispensations were found in the prescription drug database, the potential LD events were upgraded to LD events.
LD-confirmed and probable cases from Manitoba Health’s surveillance system for the 2009–2018 period published in different annual reports (Manitoba Health, Seniors and Active Living, 2020; Manitoba Health, Seniors and Active Living, 2017; Manitoba Health, Seniors and Active Living, 2016) were compared with LD events defined above. Demographics and seasonality information were not available for Manitoba Health’s surveillance data.
Data analysis
Descriptive analyses were performed for demographic characteristics (age and sex), the year and month of admission or first visit to the medical clinic. Incidence rates per 100,000 population for LD events defined above and LD cases recorded in Manitoba Health’s surveillance system were calculated by year, age group, and sex using the province’s annual population obtained using the Manitoba Health Population File (Government of Manitoba, 2023). As the number of registrants in the Manitoba Health Population File was similar to the population estimate for Manitoba published by Statistics Canada on July 1 of each year (Statistics Canada, 2024), no further analysis was required to extrapolate the findings in this article to the Manitoba population.
Data analyses were performed using SAS Enterprise Guide (EG) version 7.15 (SAS Institute Inc., USA).
Ethics review
This study has been approved by Health Canada and
Results
Trends over the years
During the study period, there were 1,629,698 registrants within the Manitoba Health Insurance Registry. Half of the registrants were female (50.4%) and the mean age was 34.80 years.
Over the 10-year study period, 5160 potential LD events were identified; among these, 53 were DAD records with ICD-10-CA code A69.2 and 5107 were physician claims with ICD-9-CM code 088 (previous LD diagnostic code) or 088.81 (current LD diagnostic code). Using the first diagnostic occurrence per patient in each year, 3317 incident potential LD events were identified; the remaining 1843 potential LD events were excluded to avoid duplication. Among those 3317 incident potential LD events, 1658 did receive one or more prescriptions of antimicrobial drugs recommended for LD treatment and were upgraded to LD events. Among the registrants with LD events, 51% (n = 845) were male and 49% (n = 813) were female (Figs. 1, 2) (Table 1).
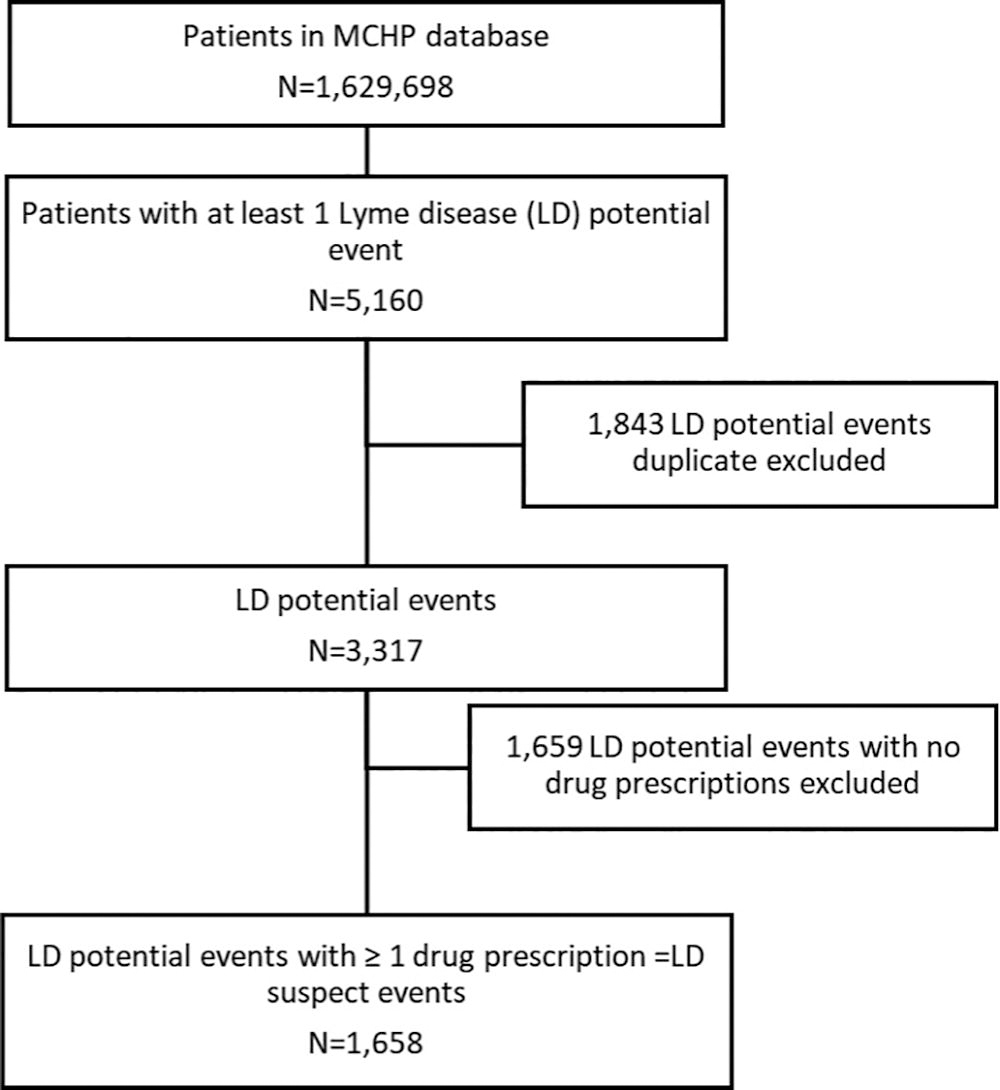
Study population and number of clinician-diagnosed Lyme disease events identified using the Manitoba Population Research Data Repository, Manitoba, 2009–2018.
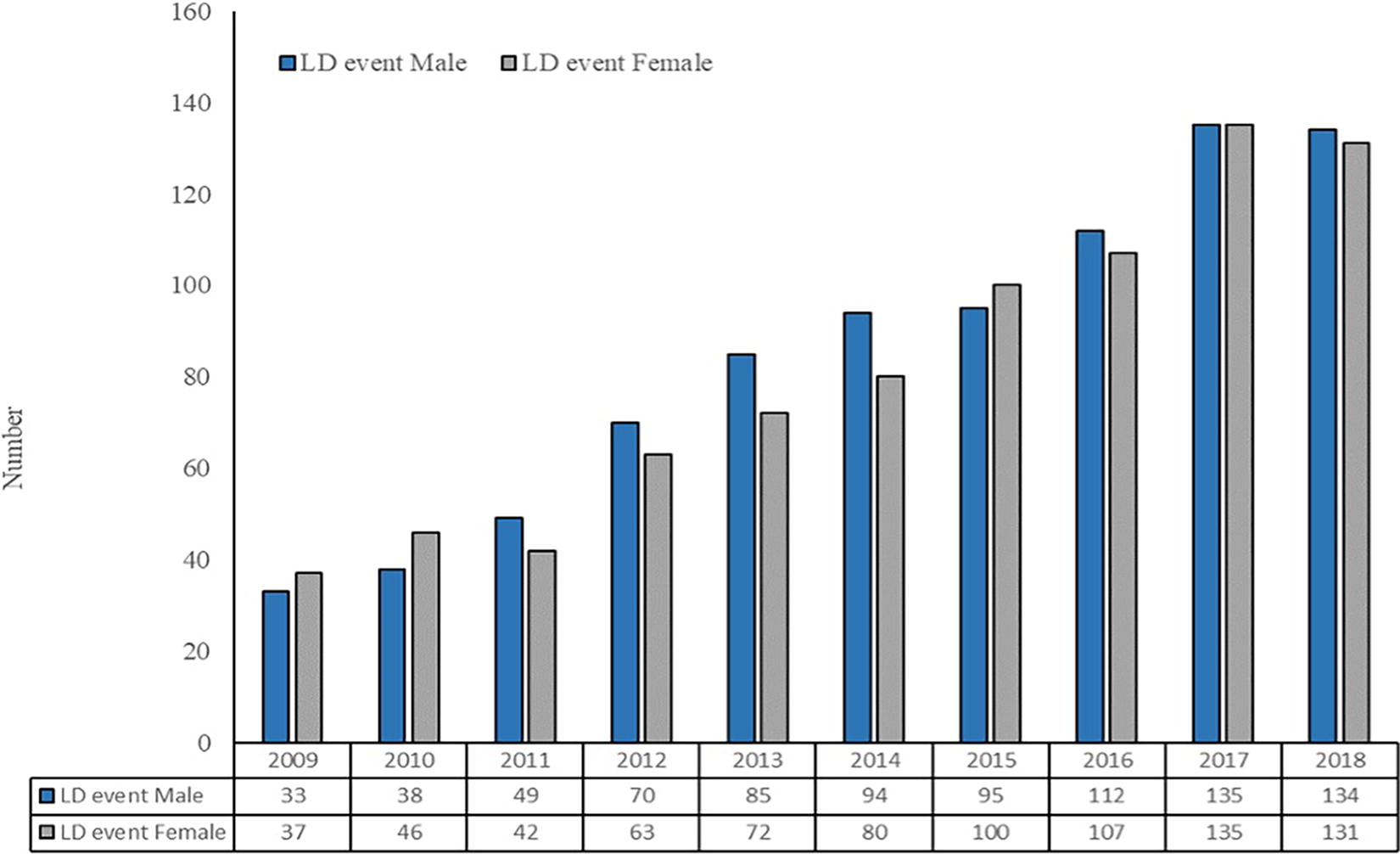
Number of clinician-diagnosed Lyme disease events identified using the Manitoba Population Research Data Repository, by year and sex, Manitoba, 2009–2018.
Number and Incidence per 100,000 Population of Clinician-Diagnosed Lyme Disease Events Identified Using the Manitoba Center for Health Policy Database, by Age Group and Sex, Manitoba, 2009–2018
LD, Lyme disease.
Over the same period, there were 302 reported probable and confirmed cases in Manitoba Health surveillance system. Therefore, the number of LD events (n = 1658) identified using the MCHP database was higher than the total number of probable and confirmed cases (n = 302) reported to the Manitoba Health surveillance system over the study period. On average there were 165 LD events identified annually in the MCHP database compared with 30 LD cases per year reported in the Manitoba Health surveillance system.
The number of LD events increased progressively over the study period, trending up from 70 events in 2009 to 265 events in 2018 (Fig. 2). LD cases reported by the Manitoba Health surveillance system increased from 5 in 2009 to 54 cases in 2018 (Manitoba Health, Seniors and Active Living, 2017, 2020).
The incidence of clinician-diagnosed LD events increased more than threefold over the study period, from 5.72 per 100,000 population in 2009 to 19.35 per 100,000 population in 2018 (Fig. 3). During the same period, the incidence of LD cases reported by Manitoba Health increased from 0.40 per 100,000 population in 2009 to 3.97 per 100,000 population in 2018, representing a ninefold increase (Manitoba Health, Seniors and Active Living, 2020; Manitoba Health, Seniors and Active Living, 2017).
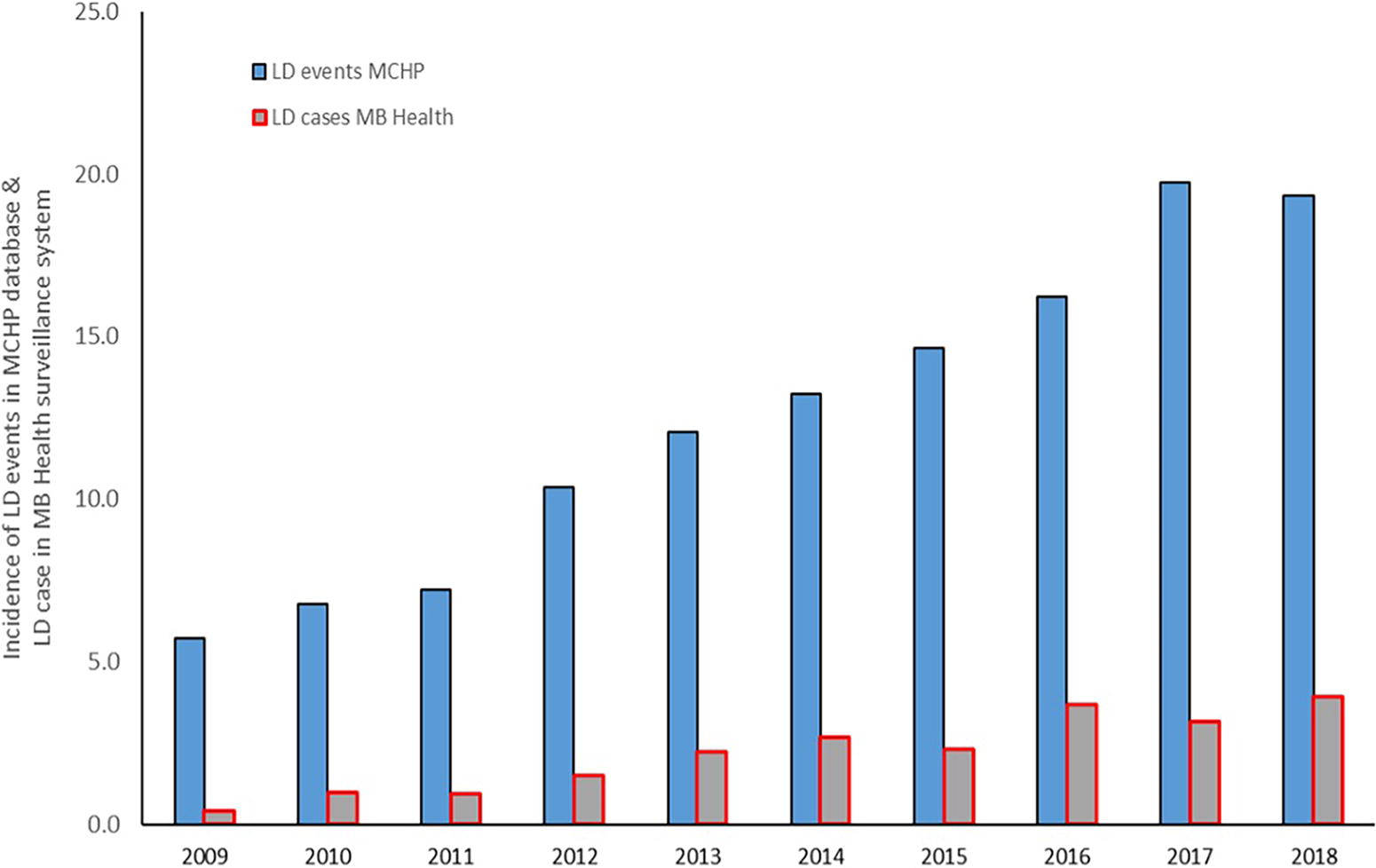
Annual incidence per 100,000 population of clinician-diagnosed Lyme disease (LD) events identified using the Manitoba Population Research Data Repository and LD cases reported to Manitoba Health’s surveillance system, Manitoba, 2009–2018.
Seasonality
Clinician-diagnosed LD events identified using the MCHP database, based on the likely date of admission or first outpatient visit, occurred mainly between May and July. The number of LD events reported during these 3 months represents 68.6% of all the LD events identified during the study period (Fig. 4).
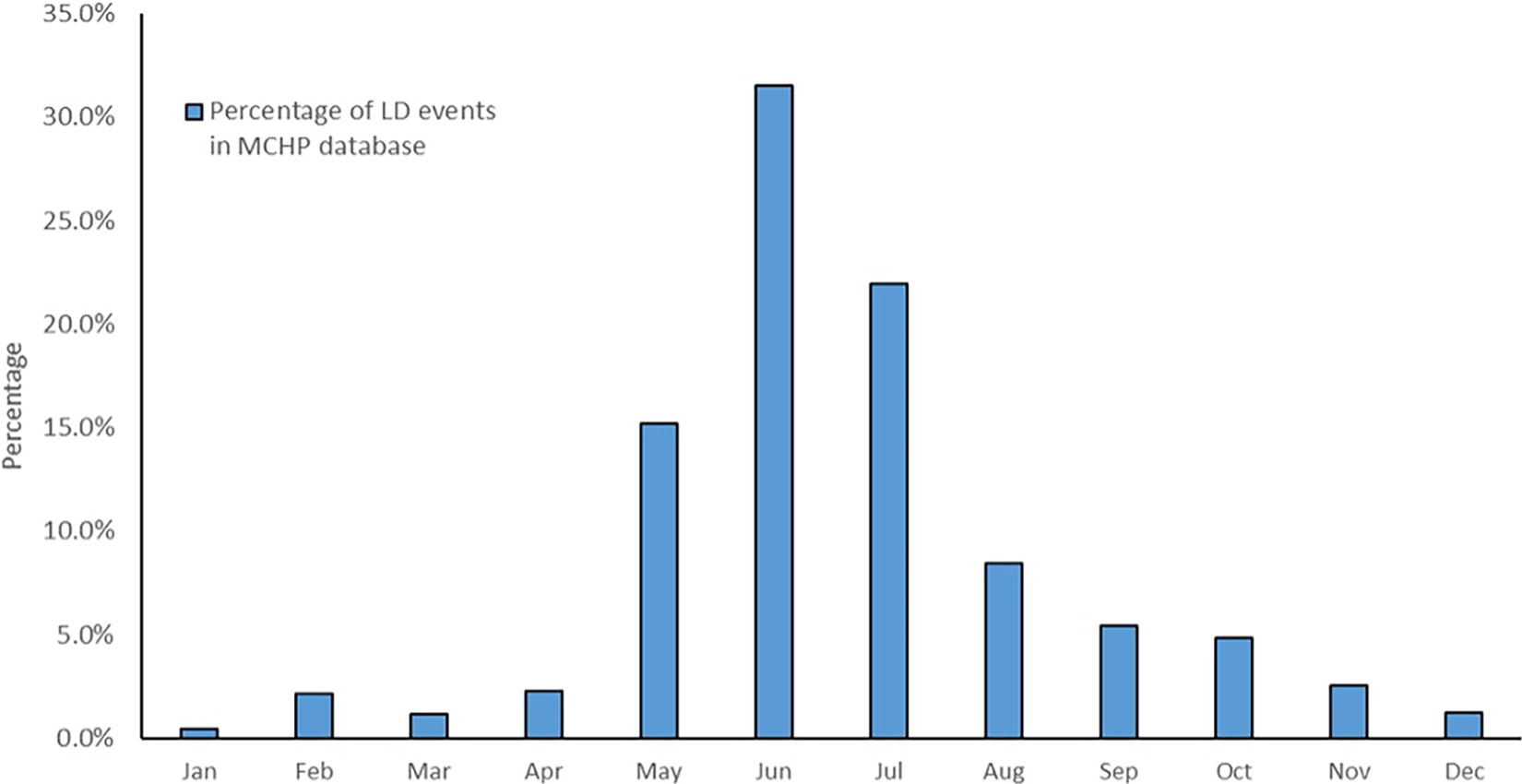
Seasonal distribution of clinician-diagnosed LD events identified using the Manitoba Population Research Data Repository by month, Manitoba (2009–2018).
Demographic characteristics
For both males and females, the incidence of LD events suggested a bimodal distribution, with a peak in childhood followed by a decline in early adulthood before the largest peak in later adulthood. The peak in childhood was seen between ages 5 and 9 and was similar for males and females. Whereas for adults, the peak was between ages 65 and 84 for males and between ages 50 and 74 for females (Fig. 5). In most age groups younger than 65 years, the incidence in females is slightly higher than in males. However, in the elderly, the incidence in men is higher than in women.
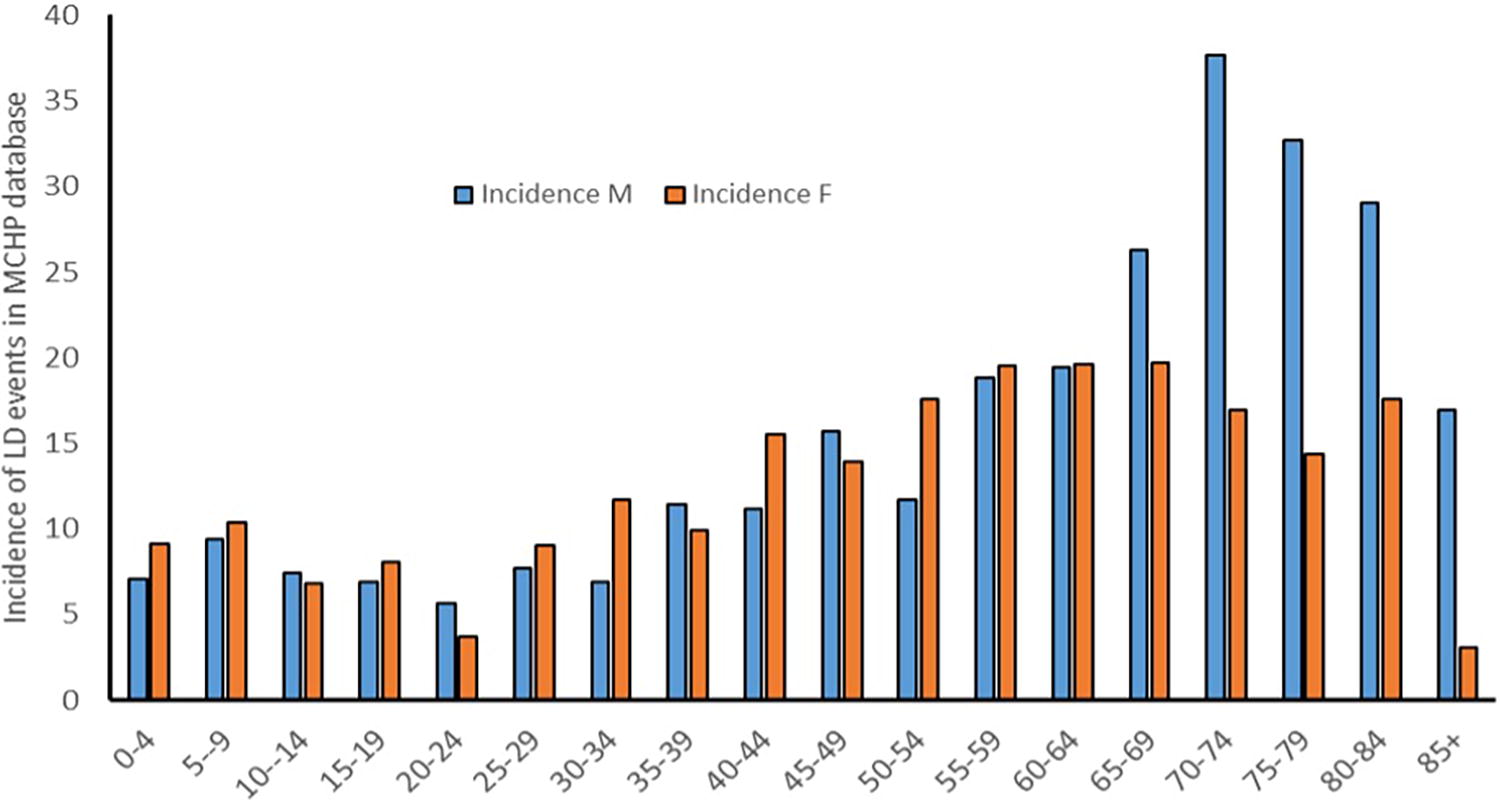
Incidence per 100,000 population of clinician-diagnosed Lyme disease events identified using the Manitoba Population Research Data Repository, by age and sex, Manitoba, 2009–2018.
Discussion
This study estimated that there were 1658 clinician-diagnosed LD events over the study period in the province of Manitoba. Comparatively, the number of cases reported to Manitoba Health was 302 over the study period (Manitoba Health, Seniors and Active Living, 2020). This suggests that the true burden of LD in Manitoba is likely higher than what is captured by the public health surveillance system. Our results indicate that LD cases could be underreported, although some important considerations should be highlighted. This finding is consistent with the results of the U.S. study that found a 10-fold underreporting of surveillance data (Nelson et al., 2015). However, our estimate for the province of Manitoba is lower.
Likely, some of the LD events identified in this study would not meet the national LD case definition. Overdiagnosis of LD is common in Lyme-endemic areas (Rose et al., 1994) because the diagnosis is often clinical (Lantos et al., 2021) with a credible history of exposure to infected blacklegged ticks (Wormser et al., 2006). Individuals in Lyme-endemic areas who receive tick bites are at greatest risk, but not all tick bites transmit LD. The risk of getting an LD infection even after a tick bite is small (ranging from less than 1% to 6%) (Huegli et al., 2011). Early localized LD also does not require diagnostic testing before antibiotic therapy is started. Therefore, some of the 1658 events identified with ICD-9-CM and ICD-10-CA codes that were prescribed LD-recommended antimicrobial drugs may not have been confirmed using diagnostic testing. Due to the nature of LD presentation, unless there is a clear history of tick exposure, along with the most common symptoms and signs, establishing a diagnosis of LD at the first patient visit is not always assured. In many cases, empirically administered antibiotics would resolve symptoms without a clear diagnosis (Joseph et al., 2010).
In Canada, the use of hospitalization data recorded in the DAD and the ICD-10-CA codes for carrying out population studies has been validated (Joseph et al., 2010; Kramer et al., 2006; Satia et al., 2020). However, a study from Wisconsin in the United States that reviewed LD patient charts with the ICD-9-CM code 088.81 found that 55.2% of those patients were classified as noncases (Naleway et al., 2002). This finding underscores the importance of validating clinician-diagnosed LD algorithms applied to administrative data by adding additional criteria. In our study, in addition to the presence of ICD-9-CM and ICD-10-CA codes for LD recorded in the DAD or in the physician billing system, a prescription of antimicrobial drugs recommended in the Infectious Diseases Society of America guidelines was required (Lantos et al., 2021; Wormser et al., 2006). The addition of antimicrobial drug prescription criterion increases the probability of identifying actual infections in our dataset, compared with the study in Wisconsin in which the ICD code was the only criterion (Naleway et al., 2002). The claim-based algorithm, combining diagnosis code and prescription of antimicrobials recommended by clinical guidelines (Lantos et al., 2021; Wormser et al., 2006) for estimating LD cases and incidence, has been utilized in several studies in the United States (Cocoros et al., 2023; Kugeler et al., 2021; Nagavedu et al., 2024; Nelson et al., 2015; Schwartz et al., 2021). These studies demonstrated the effectiveness of this methodology in accurately identifying LD cases. Furthermore, the performance of this algorithm has been rigorously assessed by Cocoros et al. (2023) and Nagavedu et al. (2024), both of whom concluded that the method has a high sensitivity and predictive positive value and, therefore, reliable for epidemiological research. This body of evidence supports the validity of the claim-based approach in accurately estimating LD diagnoses.
The seasonality of LD events identified using the MCHP database was similar to what was observed in the Manitoba Health surveillance system. Over two-thirds of the LD events occurred in late spring/early summer (i.e., May, June, and July). This time frame corresponds to the peak activity period for the nymphal stage of the blacklegged tick in the province (Manitoba Health, Seniors and Active Living, 2020). Nymphs are smaller and, therefore, harder to see and remove, compared with adults.
Similarities exist between demographic characteristics of registrants with LD events identified using the MCHP database and LD cases reported to the Manitoba Health surveillance system, as well as the national LD surveillance data. The findings of this study indicate that the incidence of LD in females is slightly higher than the incidence in males in most groups younger than 65 years. A significant disparity is observed in the elderly population, from the age groups of 65–69 to 85 years and older, where the incidence among males is considerably higher than that among females.
The study showed a bimodal distribution, with two peaks, in childhood and in later adulthood. However, the incidence within the adult cohort aged 30 to 50 years is lower than that documented in Canadian and U.S. national data (Bacon et al., 2008; Gasmi et al., 2022). Manitoba surveillance data from 2009 to 2016 (Manitoba Health, Seniors and Active Living, 2017) showed a bimodal distribution of LD cases, with one peak in patients younger than 20 years and a second peak in adults 40–59 years old.
The incidence of LD events increased moderately over the study period. However, this increase is less than that observed in the Manitoba Health routine surveillance system (Manitoba Health, Seniors and Active Living, 2017).
Limitations
Given the nature of data collected for administrative purposes, records may be incomplete, datasets may vary in data quality, and data are subject to differences in clinical coding. Therefore, several misclassifications can occur in this study. First, LD events identified in this study are based on codes and it is possible that these events are not true infections. Steps were taken to address this limitation by adding the inclusion criteria of a prescribed recommended antibiotic in an attempt to reduce overestimation of potential occurrences as much as possible. Second, diagnostic codes may also under detect cases reported by clinicians. Third, reporting may be subject to the severity of the disease or stage at which people seek treatment, as well as the population eligible for service. Finally, misdiagnosis is also possible as the earlier stage of the disease can express as flu-like symptoms and other symptoms that are not specific to LD.
Conclusions
The true burden of LD is unknown; however, combining clinical information, laboratory test results, and drug prescription administrative datasets may enhance LD surveillance system data to inform public health policy in specific regions. Similar to the U.S. study and using a comparative methodology, this study has found a possible underreporting of LD cases reported through routine surveillance processes. Higher estimated rates of LD using administrative data are expected compared with reported rates through traditional surveillance due to the nature of the disease, testing limitations, and differential overdiagnosis. Moreover, case reporting by clinicians is traditionally low unless the disease is perceived to be of increased public health risk (Gibbons et al., 2014). As LD is an important vector-borne disease in Canada, further research would be beneficial to explore the level of underreporting in other provinces to enhance our understanding of the true burden of this infection across Canada.
Footnotes
Acknowledgments
The authors thank all those involved with data collection at the regional and provincial levels and those who contributed to the Manitoba Health surveillance data. They also thank Heather P. from MCHP for data extraction and analysis.
Authors’ Contributions
R.R., J.K.K., S.G., and G.R.D. — Formal analysis, visualization, writing–original draft, writing–review and editing, A.C.B., M.W., K.S., and D.B. — Writing–review and editing.
Disclaimer
The opinions expressed by the authors contributing to this journal do not necessarily reflect the opinions of the institutions with which the authors are affiliated.
Biographical Sketch
Dr Richard Rusk is a public health physician and clinician working in Winnipeg, Canada. Aside from his work as a clinician, his main interests focus on the epidemiology of infectious diseases and the prevention and control of vector-borne diseases.
Author Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Information
This research was generously supported by the Public Health Agency of Canada.
