Abstract
Background:
Tick-borne encephalitis (TBE) is caused by the tick-borne encephalitis virus (TBEV). TBEV infection can cause symptoms of central nervous system (CNS) inflammation and result in severe consequences including death. TBE is an increasing health threat in the Czech Republic and elsewhere in Europe. In 2020, 23% of 3734 TBE cases reported to the European Centre for Disease Prevention and Control were from the Czech Republic. TBE vaccination is universally recommended in the Czech Republic, but a full analysis of TBE vaccine effectiveness (VE) in the Czech Republic has not been published.
Methods:
TBE is a notifiable disease in the Czech Republic with mandatory reporting of cases (i.e., laboratory-confirmed TBEV infected patient with symptoms of CNS inflammation) and vaccination history to public health authorities. TBE VE was estimated using the screening method utilizing public health surveillance data from 2018 to 2022 and online household surveys of the general population on TBE vaccine uptake conducted in 2019−2022.
Results:
In 2018−2022, 3648 TBE cases were reported in the Czech Republic; 98.1% (3105/3166) of TBE cases with known vaccination history were unvaccinated. Among 42,671 persons surveyed from the general population who had known TBE vaccination history, 66.5% were unvaccinated. VE against TBE was 97.6% (95% confidence interval 95.7−98.7). When stratified by age group, VE was 97.1% (88.4−99.3) in 1−15 years of age, 97.9% (95.3−99.0) in 16−59 years of age, and 96.9% (90.5−99.0) in ≥60 years of age. TBE vaccination averted an estimated 1020 TBE cases in the Czech Republic from 2018 to 2022.
Conclusions:
This first published study with a full analysis of TBE VE in the Czech Republic showed that vaccination was highly effective for the prevention of TBE including in children, an age group with increasing TBE disease burden. Vaccination averted hundreds of TBE cases and hospitalizations despite the relatively low compliance with TBE vaccine recommendations. To prevent additional TBE cases in the Czech Republic, enhanced efforts to increase TBE vaccine uptake are needed.
Introduction
Tick-borne encephalitis (TBE) is caused by the tick-borne encephalitis virus (TBEV). TBEV is a flavivirus that can cause symptoms of central nervous system (CNS) inflammation (Lindquist and Vapalahti, 2008). TBE is endemic in northern, eastern, and central Europe, including the Czech Republic, and parts of Asia (Lindquist and Vapalahti, 2008; Süss, 2011). Although TBEV infections may be asymptomatic, infections can cause a febrile illness with fatigue, malaise, headache, and body pains (Lindquist and Vapalahti, 2008). Some infections have a biphasic course with mild to severe neurological symptoms (i.e., meningitis, encephalitis, and encephalomyelitis) and can result in long-term neurological sequelae and death. TBEV is transmitted to humans predominately by the bite of an infected Ixodes tick and, less commonly, by consumption of unpasteurized dairy products (Lindquist and Vapalahti, 2008; Süss, 2011).
TBE is an increasing health threat in the Czech Republic and in Europe (Kriz et al., 2012; Riccardi et al., 2019). There were 3734 TBE cases reported to the European Centre for Disease Prevention and Control (ECDC) by 24 European Union/European Economic Area countries in 2020, an incidence (0.9/100,000 population) 125% higher than that reported in 2015 (0.4/100,000); similarly, the incidence in the Czech Republic was 139% higher in 2020 (7.9/100,000) than in 2015 (3.3/100,000) (ECDC, 2015; 2020). In 2020, 23% of the ECDC-reported TBE cases were from the Czech Republic (ECDC, 2020). Reasons for the increased incidence of TBE in the Czech Republic and Europe are not fully understood but may include climate and environmental changes, geographic expansion of the tick vector and of TBEV, and changes in human behaviors leading to increased exposure to ticks (Kriz et al., 2012; Riccardi et al., 2019).
Vaccination offers the most effective protection against TBE (WHO, 2011). Two TBE vaccines, FSME-IMMUN® (Pfizer Europe, Brussels, Belgium) and Encepur® (Bavarian Nordic, Hellerup, Denmark), are used in the Czech Republic. FSME-IMMUN ® has been used in the Czech Republic since 1991, and Encepur ® has been used in the Czech Republic since 1993. The executive board of the Czech Vaccination Society of the Czech Medical Association of J. E. Purkyně recommends TBE vaccination for all persons residing in or visiting areas with high TBE incidence (Czech Vaccination Society of the Czech Medical Association of J. E. Purkyně, 2016). Despite this recommendation, there is low uptake of TBE vaccine in the Czech Republic. In a 2020 survey, only 33% of the general population in the Czech Republic reported receiving at least one dose of a TBE vaccine (Pilz et al., 2023). For comparison, similar surveys in southern Germany and Austria, which have a comparable endemicity of TBEV as the Czech Republic, reported TBE vaccine uptakes of 60% and 81%, respectively (Pilz et al., 2023). It appears likely, therefore, that the low TBE vaccine uptake in the Czech Republic contributes to the high TBE incidence in the country.
Several factors contribute to the low TBE vaccine uptake in the Czech Republic. Although there is public awareness of TBE and the vaccine, acceptance of TBE vaccination is low despite the severity of the disease and its frequent long-term consequences. Promotion or provision of TBE vaccination by some health care providers is limited. Out-of-pocket costs are an additional barrier to vaccination for many consumers. Although persons in occupations with a high risk of exposure to ticks (foresters, etc.) are offered free TBE vaccination by their employer, for other persons living in areas with high TBE incidence there is no governmental funding for TBE vaccination and only partial reimbursement of vaccine costs by some health insurance agencies. In 2022, to increase TBE vaccine uptake health insurance programs began providing TBE vaccine free-of-charge for persons ≥50 years of age. An estimated 96% of the almost 1.2 million doses of TBE vaccines administered in the Czech Republic from 2018 to 2022 were FSME-IMMUN ® (Sprinx Pharma Portal at https://pharma.sprinx.com/).
When TBE vaccination was introduced in Austria in 1979, TBE cases rapidly declined (Kunz, 2003). Studies have reported estimates of TBE vaccine effectiveness (VE) of ≥95% against TBE in multiple countries in Europe, but a full analysis of TBE VE data in the Czech Republic has not been published (Erber et al., 2022; Nygren et al., 2022; Santonja et al., 2023; Zens et al., 2022). In addition, VE data are needed to evaluate whether the interval between TBE vaccine boosters can be modified: currently, most countries including the Czech Republic follow the product label and recommend an interval of ≤5 years between boosters for persons <60 years of age and an interval of ≤3 years between boosters for persons ≥60 years of age, but some other countries have recently extended the recommended interval to ≤10 years between boosters for all ages (Steffen et al., 2021). Finally, a full analysis of the public health impact of TBE vaccination (i.e., the number of TBE cases averted due to TBE vaccination) has not been published for the Czech Republic.
The objective of this study was to estimate TBE VE and impact in the Czech Republic, a country with a high TBE incidence.
Methods
Study population
In this observational study in the Czech Republic, vaccine histories of TBE cases reported to the public health surveillance system were compared with the vaccine histories of survey respondents from the general population to estimate VE using the screening method. The study population included all persons in the Czech Republic ≥1 year of age (population 10,405,048 in 2022).
Procedures
As part of routine clinical practice, blood and/or cerebrospinal fluid (CSF) specimens were collected from predominately hospitalized patients presenting with symptoms of CNS inflammation or, less commonly, fever and unspecified influenza-like symptoms with a history indicating potential TBEV infection (i.e., tick exposure or raw milk consumption in the 2 weeks before symptom onset). Specimens were submitted to one of approximately 100 clinical diagnostic laboratories or the National Reference Laboratory for Arboviruses (NRL). At the laboratories, specimens were tested by enzyme-linked immunoassay or indirect immunofluorescence for anti-TBEV IgG and IgM antibodies. In accordance with ECDC case definitions, a laboratory-confirmed TBEV infection is a patient with anti-TBEV IgG and IgM antibodies in blood, seroconversion or a four-fold increase in anti-TBEV antibodies in paired serum samples, or anti-TBEV IgM antibodies in CSF (ECDC, 2018). At the NRL, virus neutralization tests were used to confirm the serological tests in patients recently vaccinated against TBE or other flaviviruses or recently returned from endemic regions of the other flaviviruses.
In accordance with ECDC case definitions, a TBE case was a laboratory-confirmed TBEV-infected patient with symptoms of CNS inflammation (ECDC, 2018). TBE is a notifiable disease in the Czech Republic with mandatory reporting of TBE cases by clinicians to the Regional Public Health Authorities (Czech Republic Parliament, 2000; Kriz et al., 2012). Information collected about a reported TBE case includes age, gender, region of residence, place of probable exposure, clinical course, vaccination history, tick bite history, and history of consumption of unpasteurized dairy products. Vaccination history was obtained directly from the patient, the patient’s general practitioner, or specialized vaccination centers. Epidemiologists at each of the 14 Regional Public Health Authorities compiled the surveillance data and reported it via a nationwide electronic reporting system. Maintenance and analysis of the public health surveillance data at the national level are conducted by the National Institute of Public Health in Prague.
Online household surveys on TBE and TBE vaccine knowledge, attitudes, and practices were conducted in 2019, 2020, 2021, and 2022 by Ipsos GmbH (Hamburg, Germany) (Erber and Schmitt, 2018; Pilz et al., 2023). Participants were recruited to participate in the online survey using sampling proportions for gender, region, occupational status, and household size, based on Eurostat data (https://ec.europa.eu/eurostat). Initial participants were 18–65 years of age and provided information for all household members ≥1 year of age. Participants in previous surveys were excluded from participating in later surveys. Surveys took 5–15 minutes and gathered information on TBE awareness, TBE vaccination knowledge, and TBE vaccine history. Respondents consulted vaccination cards for TBE vaccine history. Survey results were weighted, using national census data, to derive national prevalence estimates. Estimates for the population unvaccinated in 2018 were calculated as a projection of the 2019 survey data utilizing the number of participants that reported receiving a first TBE vaccine dose in 2019.
This study analyzed nationwide surveillance data on TBE cases that are routinely collected by the National Institute of Public Health under authority of a Czechoslovakian ministerial decree of 1970 that was reiterated in Act 258 of the Czech Republic, which was passed in 2000 (Czech Republic Parliament, 2000). Only aggregate data, with no personal identifiers, were utilized in this analysis. The analysis plan for this study was internally reviewed by the National Institute of Public Health and found to be compliant with all regulatory requirements including guidelines for use of public health surveillance data and human subject research.
Statistical analysis
To estimate VE, a person was considered vaccinated in accordance with the vaccine schedule if he/she received the three-dose primary series and was not late for the first booster (i.e., did not receive the booster >3 years after last dose) or if he/she received ≥1 boosters and was not late for the subsequent booster (i.e., did not receive the subsequent booster >3 years after last dose for persons ≥60 years of age or did not receive the subsequent booster >5 years after last dose for persons <60 years). A person receiving a partial primary series or late in receiving the first or subsequent boosters was considered partially vaccinated. Estimates of VE against TBE (with 95% confidence interval [CI]) for persons who were vaccinated were determined by comparing the proportion of TBE cases who were vaccinated in accordance with the vaccine schedule (PCV) and the proportion of persons in the general population surveys who were vaccinated in accordance with the vaccine schedule (PPV), excluding persons who were partially vaccinated, using the screening method formula VE = 1-[PCV/(1-PCV)]/[PPV/(1-PPV)] (Farrington, 1993). Estimates of VE against TBE for persons who received ≥4 vaccine doses (i.e., 3-dose primary series and at least the first booster) at ≤10 years and >10 years after receipt of the last dose were similarly determined.
The number of TBE cases averted by vaccination were estimated by calculating the incidence in the unvaccinated population, derived from surveillance and the population survey, under a counterfactual scenario whereby persons who were vaccinated did not receive vaccination; cases averted is the difference in the number of TBE cases in the counterfactual scenario and the actual observed number of TBE cases. The number of deaths averted was estimated by multiplying estimated number of averted TBE cases by the observed case fatality rate. TBE cases that would have been prevented if the population was vaccinated were estimated as the product of the number of TBE cases in the counterfactual scenario and VE against TBE for persons who were vaccinated.
Results
Description of TBE cases from surveillance
There were 3648 reported TBE cases in 2018–2022 (an average of 730 cases per year) (Table 1). Information on whether a patient was hospitalized was available for 3517 (96.4%) of the TBE cases, of which 3324 (94.5%) were hospitalized. Of the 3648 TBE cases, 20 were reported as deceased for a case fatality rate of 0.55%. Of the 3648 TBE cases, the TBE vaccine history could be determined for 3166 (86.8%). The mean age of the 3166 cases with and 482 cases without a known vaccine history was 43.7 and 43.9 years, respectively. A higher proportion of the cases with a known vaccine history, compared to cases without a known vaccine history, were hospitalized (93.9% and 89.6%, respectively), but cases with and without a known vaccine history did not differ in the proportion with meningoencephalitis, or in the proportion admitted to the intensive care unit (data not shown). Of the TBE cases with a known TBE vaccine history, 3105 (98.1%) were unvaccinated and 61 (1.9%) received ≥1 doses of a TBE vaccine, of which 11 (18.0%) were vaccinated in accordance with the vaccine schedule and 50 (82.0%) were partially vaccinated.
Information on Tick-Borne Encephalitis (TBE) Cases, Including Number and Incidence by Year and Age Group, Patient Outcomes of TBE Cases, and TBE Vaccination History, Czech Republic, 2018–2022
Population ≥1 year of age.
NA, not applicable; TBE, tick-borne encephalitis.
Vaccine history from general population survey
For the population survey, 46,855 people (11,517 in 2019, 11,878 in 2020, 11,335 in 2021, and 12,125 in 2022) were selected from the general population to participate, of whom 42,669 (91.1%) provided information on TBE vaccine history (Table 2). Among persons reporting vaccine history, 28,398 (66.5%) were unvaccinated and 14,271 (33.5%) received ≥1 doses of a TBE vaccine: Of those receiving ≥1 dose, 6305 (44.2%) had a known date of vaccination, of whom 1873 (29.7%) were vaccinated in accordance with the vaccine schedule.
TBE Vaccine History from the Ipsos General Population Survey, Czech Republic, 2019–2022
TBE, tick-borne encephalitis.
TBE incidence by age group and region
From 2018 to 2022, the incidence of TBE across all age groups, including children <1 year of age, was 6.9/100,000 population per year. The incidence of TBE in the population ≥1 year of age (where the vaccine can be used) was 7.0/100,000 (Table 1). By year, the highest incidence was in persons ≥1 year of age in 2020 (8.1/100,000 population), and by age group, the highest incidence was in persons ≥60 years of age (7.3/100,000 population per year). The highest incidence across all age groups was in Jihočeský region (17.8/100,000 population per year) located in the southwestern part of the Czech Republic (Fig. 1). Applying the general population survey results to the census to estimate the size of the unvaccinated population, the incidence of TBE in the population ≥1 year of age was 8.9/100,000 population per year in the unvaccinated population from 2018 to 2022.
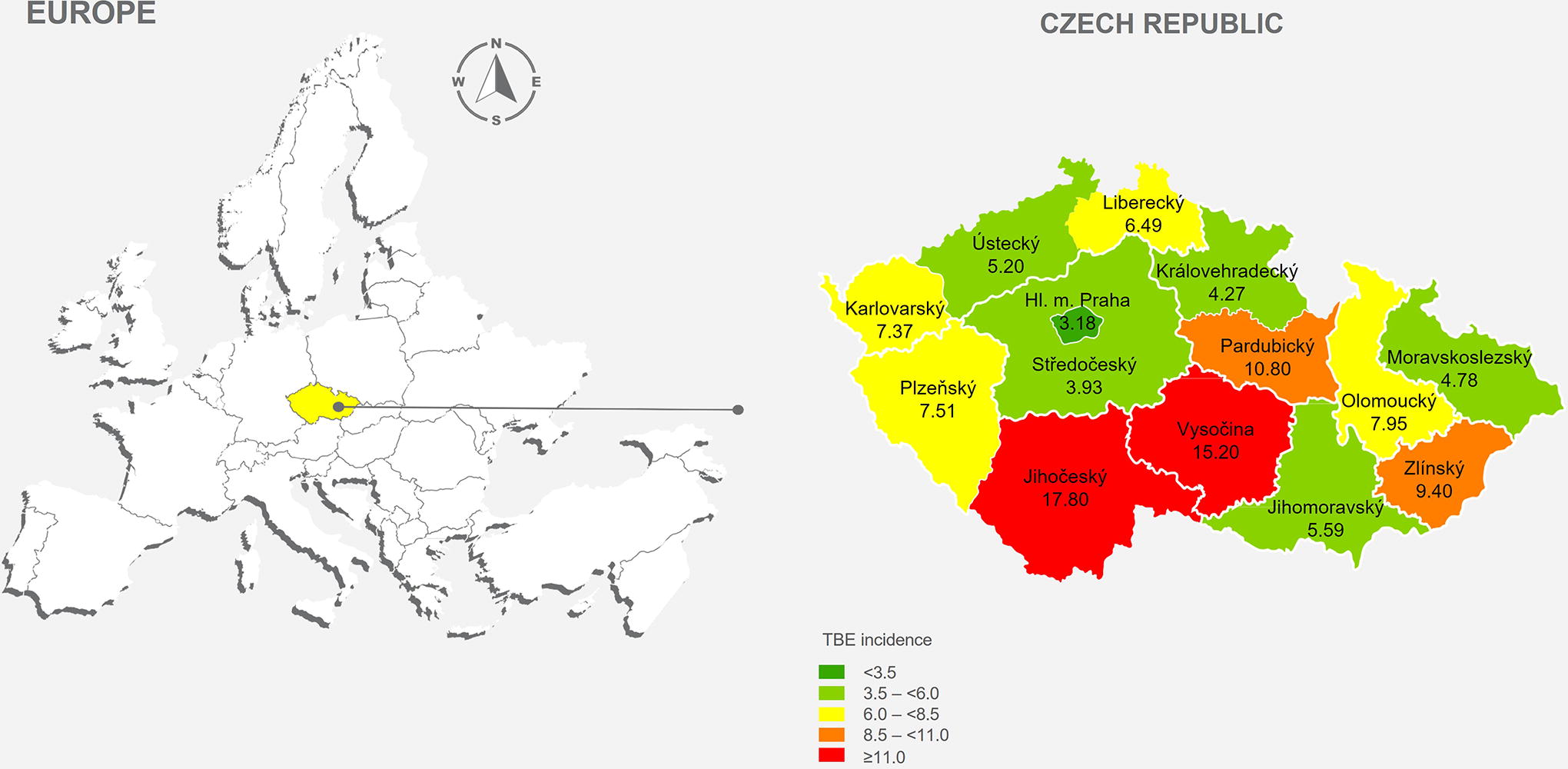
Incidence per 100,000 population per year of tick-borne encephalitis cases by region, Czech Republic, 2018–2022. Tick-borne encephalitis incidence is reported across all age groups, including children <1 year of age.
TBE vaccine effectiveness
VE against TBE of being vaccinated with ≥3 doses of a TBE vaccine in 2018−2022 was 97.6% (95% CI: 95.7–98.7) (Table 3). Stratified by the age group, VE against TBE for ≥3 doses of a TBE vaccine was 97.1% (95% CI 88.4–99.3) in persons 1–15 years of age, 97.9% (95% CI: 95.3–99.0) in persons 16–59 years of age, and 96.9% (95% CI: 90.5–99.0) in persons ≥60 years of age. VE against TBE for ≥4 doses of a TBE vaccine in 2018−2022 was 97.9% (95% CI: 95.0–99.1). Stratified by time after receipt of last dose, VE against TBE of being vaccinated by ≥4 doses of a TBE vaccine was 98.1% (95% CI: 95.5–99.2) at ≤10 years and 95.7% (95% CI: 69.2–99.4) at >10 years after receipt of the last dose (Table 4).
Vaccine Effectiveness of ≥3 Doses of a TBE Vaccine by Age Group, Czech Republic, 2018−2022
CI, confidence interval; TBE, tick-borne encephalitis; VE, vaccine effectiveness.
Vaccine Effectiveness of ≥4 Doses of a TBE Vaccine, Czech Republic, 2018–2022
CI, confidence interval; TBE, tick-borne encephalitis; VE, vaccine effectiveness.
Estimation of TBE cases averted by vaccination
In a scenario with no TBE vaccination in the Czech Republic, it is estimated that there would have been 4668 TBE cases from 2018 to 2022 (Table 5). Since 3648 TBE cases were observed during this period, it is estimated that TBE vaccination averted 1020 TBE cases in the Czech Republic from 2018 to 2022, which would have resulted in 964 hospitalizations and 6 (1020 × 0.5%) deaths. With an observed VE of 97.6% for the prevention of TBE cases, if the entire population in the Czech Republic was vaccinated, TBE vaccination would have prevented 4556 TBE cases and, with an observed case fatality rate of 0.5%, 25 deaths.
Estimated TBE Cases Averted Due to Vaccination, Czech Republic, 2018–2022
TBE incidence in unvaccinated (8.9/100,000 population per year) × population.
VE, vaccine effectiveness; TBE, tick-borne encephalitis.
Discussion
TBE vaccination was highly effective for the prevention of TBE in the Czech Republic. The effectiveness of TBE vaccines against TBE has been previously demonstrated in observational studies in Austria, Germany, Latvia, and Switzerland (Erber et al., 2022; Nygren et al., 2022; Santonja et al., 2023; Zens et al., 2022), but this is the first published report with a full analysis of the effectiveness of TBE vaccination in the Czech Republic, a country with increasingly high TBE incidence and one of the highest TBE incidence in Europe (Kriz et al., 2012). Furthermore, the protective effect of TBE vaccines in the Czech Republic was evident in all age groups. The need to increase TBE prevention in children has been recently highlighted as the proportion of reported TBE cases in the Czech Republic that occurred in children and teenagers increased from 11.3% in 2015 to 17.7% in 2021 (Orlikova and Kyncl, 2022). The effectiveness of TBE vaccination for the prevention of TBE in children 1–15 years of age in this study (97.1% [95% CI: 88.4–99.3]) emphasizes the protective effect of TBE vaccination in children and highlights the need for further availability of TBE vaccines for children in the Czech Republic.
Despite the effectiveness of TBE vaccines, during the 5-year period of this study, there were 3648 reported TBE cases (7.0/100,000 population ≥1 year of age per year) in the Czech Republic. The TBE disease burden predominantly affected the unvaccinated. In this study, of the 3166 surveillance-reported TBE cases from 2018 to 2022 for whom a TBE vaccine history could be determined, only 1.9% received ≥1 doses of a TBE vaccine, and only 0.3% were vaccinated in accordance with the vaccine schedule. The increasingly high TBE incidence in the Czech Republic has been attributed to a geographic expansion of the tick vector and changes in human behaviors leading to increased exposure to ticks in the Czech Republic (Kriz et al., 2012). The results from this study, with the high VE of TBE vaccines and with almost all TBE cases occurring among persons who were unvaccinated, demonstrated that the low TBE vaccine uptake in the Czech Republic is an important contributing factor to the high incidence of TBE in the Czech Republic. Furthermore, the low TBE vaccine uptake in the Czech Republic is also likely contributing to the greater increase in TBE incidence in the Czech Republic compared to that in neighboring southern Germany or Austria, areas where TBE is also endemic but where TBE vaccine uptake is notably higher.
Nevertheless, without TBE vaccination, the TBE disease burden in the Czech Republic would have been even higher. We estimate that during the study period, TBE vaccination averted 1020 TBE cases, including 964 hospitalizations, in a population of 10.4 million persons. In the Czech Republic, vaccination against TBE is recommended for all persons at risk. However, during the study period, only 10% of the population was vaccinated with a TBE vaccine in accordance with the vaccine schedule and two-thirds of the population was unvaccinated. If the entire population had been vaccinated with a TBE vaccine in accordance with the vaccine schedule, TBE vaccination would have prevented 4556 TBE cases and 25 deaths during the 5-year study period. To prevent life-threatening TBE, further efforts are needed to increase TBE vaccine uptake in the Czech Republic (Kunze et al., 2022).
In the Czech Republic, the recommended interval between TBE boosters is ≤5 years for persons <60 years of age and ≤3 years for persons ≥60 years of age. In contrast, the recommended interval between boosters in Switzerland and Finland is ≤10 years for all age groups (Steffen et al., 2021). An evaluation in Switzerland concluded that there was an overall public health benefit of prolonging the booster interval from 5 years to 10 years due to the rarity of TBE cases in persons who received ≥3 vaccine doses and the increased TBE vaccine uptake associated with schedule simplification (Steffen et al., 2021). In the Czech Republic, we found a relatively small difference (98.1% vs. 95.7%) in VE between those who had received their latest booster ≤10 or >10 years previously, with VE >95% for both time intervals. These data support the recommendation to extend booster intervals to ≤10 years for all boosters after the first booster in all age groups. This recommendation would simplify the TBE vaccine schedule, potentially helping to increase TBE vaccine uptake and improve overall population protection against TBE in the Czech Republic and other endemic areas in Europe.
This study has some limitations. The VE estimates were derived via the screening method which compared the vaccine history of patients with TBE with that of survey responders selected from the general population. The potential for selection bias of the survey responders was minimized by using survey methods conducted by an experienced market surveying company. It should be noted, however, that the TBE vaccine uptake reported by general population survey respondents in 2019−2022 in our study (33%) is higher than the TBE vaccine uptake reported from a similarly performed but smaller survey conducted in the Czech Republic in 2015 (24%) (Erber and Schmitt, 2018). A potential consequence of an overestimated population TBE vaccine uptake could be an overestimated VE. However, given the rigorous survey methodology and large sample size of our population survey (46,855 respondents), it appears unlikely that our survey overestimated TBE vaccine uptake. The demographics of the survey respondents matched the general population in the Czech Republic, but the potential for selection bias remains.
Differential misclassification bias may also be present in our analysis since different approaches were used to ascertain the vaccine history of TBE cases and of the surveyed general population. Analytically, the screening method does not enable the evaluation of potential confounding variables. We were able, however, to stratify the VE estimates by age groups and estimate similar VE in all groups, indicating that age does not appear to be an important confounder of this analysis. Another limitation is that TBE vaccination history could not be determined for 13.2% of the TBE cases and 8.9% of the survey participants. Although reported demographic characteristics of the participants with and without vaccine history are similar (data not shown), we do not know if individuals with missing vaccine histories were more or less likely to have received a TBE vaccine. A final limitation of this study is that we were unable to estimate product-specific VE for FSME-IMMUN ® vs. Encepur ® . However, during the study period, an estimated 96% of TBE vaccines sold in the Czech Republic was FSME-IMMUN ® .
Conclusions
This first published study with a full analysis of TBE VE in the Czech Republic showed that TBE vaccination was highly effective for the prevention of TBE. Furthermore, the effectiveness of TBE vaccination in children, an age group with increasing TBE disease burden, was demonstrated. Finally, high TBE VE was observed both ≤10 years and >10 years after the last booster in individuals who had received four or more doses of vaccine, suggesting an extension of the booster interval may be considered for all age groups. Vaccination averted hundreds of TBE cases and hospitalizations, and several deaths despite the relatively low TBE vaccine uptake and compliance with TBE vaccine recommendations. To prevent additional TBE cases in the Czech Republic, enhanced efforts to increase TBE vaccine uptake are needed.
Footnotes
Acknowledgments
We acknowledge Melissa Furtado, MPH, ISMPP CMPPTM, of Pfizer, for providing medical writing support and editorial assistance funded by Pfizer Inc. for this article. We acknowledge Anja Gilvad of Ipsos for her leadership and assistance with the population surveys.
Authors’ Contributions
J.K.: conceptualization; methodology; data curation; formal analysis; writing—original draft; writing—review & editing. F.J.A.: conceptualization; methodology; project administration; formal analysis; writing—original draft; writing—review & editing. H.O.: writing—original draft; writing—review & editing. P.Z.: methodology; formal analysis; writing—review & editing. I.V.: data curation; writing—review & editing. M.M.: data curation; writing—review & editing. D.K.: writing—review & editing. L.R.H.: project administration; writing—review & editing. J.E.: project administration; writing—review & editing. C.B.: formal analysis; writing—review & editing. A.P.: methodology; writing—review & editing. W.E.: writing—review & editing. H.M.: supervision; writing—review & editing. J.C.M.: supervision; writing—review & editing. All authors read and approved the final version of the article for submission.
Authors Disclosure Statement
F.J.A., P.Z., D.K., L.R.H., J.E., C.B., A.P., W.E., H.M., and J.C.M. are employees of Pfizer Inc. and hold stock and stock options in Pfizer Inc. J.K., H.O., I.V., and M.M. declare that they have no competing interests.
Funding Information
This study was conducted as a collaboration between The National Institute of Public Health in Prague, Czech Republic, and Pfizer.
