Abstract
Background:
Madariaga virus (MADV), a member of the eastern equine encephalitis virus (EEEV) complex, circulates in Latin America and exhibits distinct evolutionary and ecological features compared to the North American EEEV. While published data have shed light on MADV ecology, several key aspects remain unknown.
Methods:
In this study, we compiled data on virus isolation, vector competence, and animal serology collected over six decades in Latin America to identify critical knowledge gaps on MADV transmission and ecology.
Results:
Specific vertebrate animals serving as amplifying hosts and the mosquito species acting as enzootic and epizootic vectors have not yet been identified. Other aspects that remain unclear are the virus current geographic distribution, the role of equines as hosts in epizootic cycles, and the full impact of MADV on human health in endemic regions.
Conclusions:
The numerous knowledge gaps surrounding MADV, its widespread distribution in Latin America, and its potential to cause severe disease in animals and humans emphasize the urgent need for increased research efforts, heightened awareness, and intensified surveillance towards this potential emerging threat.
Madariaga Virus, a Member of the Eastern Equine Encephalitis Virus Complex
In 1963, a meticulous serological experiment led to the identification of two antigenic varieties (or subtypes) of the eastern equine encephalitis virus (EEEV) (Casals, 1964). These varieties were classified as North American (NA) and South American (SA) based on the geographic origins of the viruses used in the experiments. The author noted that the SA EEEV isolates exhibited greater antigenic heterogeneity than the NA group, a finding that was subsequently confirmed in other studies.
Further serological and phylogenetic analyses of 18 NA EEEV and 29 SA EEEV isolates led to the identification of four distinct EEEV subtypes, classified into Lineages I–IV (Brault et al., 1999). Lineage I comprises isolates from the United States and Mexico (North America), whereas Lineages II and III include isolates from various countries in Central and South America and one isolate from Trinidad and Tobago in the southern Caribbean. Lineage IV consists of a single isolate from Brazil (South America).
Expanding on the findings of these earlier studies, a more thorough genetic analysis of EEEV strains revealed distinct genetic and evolutionary features of Lineage I EEEV compared with Lineages II–IV EEEV. While Lineage I EEEV isolates are highly conserved among each other and exhibit a monophyletic, temporal relationship, viruses within Lineages II–IV are highly divergent, display a polyphyletic pattern, and cluster geographically. The evolutionary pattern of Lineage I EEEV is consistent with the ecology of a virus spread by birds, which allows efficient dispersion across geographic barriers. In contrast, Lineages II–IV EEEV show an evolutionary pattern indicative of a virus with ground-dwelling hosts, resulting in limited dispersion and independent evolution. Interestingly, the evolutionary pattern of Lineages II–IV strains closely resembles that of the Venezuelan equine encephalitis virus (VEEV), a virus that has rodents as the main enzootic hosts. The higher diversity of vectors in tropical regions may also have contributed to the diversity observed among Lineages II–IV strains (Arrigo et al., 2010). According to analysis of the structural polyprotein open reading frame, viruses clustering into Lineage I differ from those clustering into Lineages II–IV by 23–24% in the nucleotide sequence and by 9–11% in the amino acid sequence. Based on the genetic and ecological distinctions among EEEV strains, the authors proposed that EEEV isolates belonging to Lineages II–IV should be classified as a separate species within the EEEV complex, named Madariaga virus (MADV) (Arrigo et al., 2010). Potential differences in the pathogenicity for humans between Lineage I and Lineages II–IV EEEV were also considered in the proposition of the latter as a new species (Arrigo et al., 2010).
Phylogenetic trees showing the divergence patterns of EEEV strains belonging to Lineages I–IV, including some that have incorporated recently isolated viruses, are available elsewhere (Arrigo et al., 2010; Brault et al., 1999; Carrera et al., 2013; Gil et al., 2021).
Because NA EEEV has never been confirmed south of Mexico and all the EEEV detected in Central and South America that have been tested have shown to be SA strains of EEEV, now known as MADV, we will refer to all the detections of EEEV in Central/South America as MADV.
Virus Isolation from Mosquitoes
The vectors responsible for transmitting MADV in Latin American countries remain unknown, but a few mosquitoes have been identified as potential candidates.
During an equine epizootic in North Brazil in 1960, MADV was isolated from three out of 27 pools of Aedes taeniorhynchus (Causey et al., 1962). The authors noted that this species was highly abundant in the farms affected by the outbreaks and suggested its potential role as the epizootic vector. Another study conducted in North Brazil between 1955 and 1989 reported the isolation of MADV from several mosquito species, including Ae. taeniorhynchus, but most isolations were obtained from Culex (Melanoconium) pedroi (Vasconcelos et al., 1991). Two other works report MADV isolation from Cx. (Mel.) spp. in Brazil (Shope et al., 1966; Sirivanakarn and Jakob, 1981) (Table 1).
Detection of Madariaga Virus in Mosquitoes in Latin American Countries
Virus isolation was performed through cell culture assays or inoculation in naïve animals, whereas virus RNA was detected through molecular assays.
Confirmed by virus isolation.
The most extensive work on MADV isolation in mosquitoes was performed in the Amazon basin of Peru, when 539,694 mosquitoes captured in Loreto Department from 1996 to 2001 were screened for various arboviruses (Turell et al., 2005). MADV isolation was obtained from numerous mosquito species; however, nearly all the isolations (95.0%) were from Cx. (Mel.) spp., with nearly all of those (92.0%) from Cx. (Mel.) pedroi. In another study in Peru, specifically targeting Cx. (Mel.) pedroi, MADV RNA was detected by RT-PCR in five out of 117 mosquito pools (O’Guinn et al., 2004) (Table 1).
MADV has been isolated from Cx. spp. in Trinidad and Tobago (Spence et al., 1961) and from Cx. (Mel.) spp. in Panama (Dietz et al., 1980; Srihongse and Galindo, 1967) and Venezuela (Walder et al., 1984). In Argentina, MADV RNA was detected in Cx. spp. through molecular assays, but virus isolation was not pursued (Stechina et al., 2019) (Table 1).
However, the mere isolation of a virus from a mosquito species does not mean that it is a competent vector. Laboratory experiments on vector competence serve as a complement to field observations. Following the previous research in Peru that isolated MADV in multiple mosquito species (Turell et al., 2005), the vector competence of nine Peruvian mosquito species for MADV was further investigated (Turell et al., 2008). In this work, live chickens and hamsters were used to expose the mosquitoes to MADV and access the transmission potential from mosquitoes back to susceptible hosts. The results showed that Cx. (Mel.) pedroi, Psorophora albigenu, Ae. serratus, and Ps. ferox were able to transmit the virus, with Cx. (Mel.) pedroi being the most efficient (Turell et al., 2008).
Two observations regarding Cx. (Mel.) spp. are noteworthy. First, Cx. (Mel.) spp. such as Cx. (Mel.) pedroi are important vectors of enzootic strains of VEEV, which geographically overlaps with MADV in multiple regions. This overlapping distribution suggests the potential involvement of Cx. (Mel.) pedroi in the transmission of both viruses. Second, studies conducted in Brazil have shown that Cx. (Mel.) spp. have a broad host preference range and may display a certain level of adaptation to human environments (Forattini et al., 1991; Forattini et al., 1987), both of which can be indicative of bridge vectors involved in spillover events.
Nevertheless, although Cx. (Mel.) spp. have been identified as potential vectors for MADV in Latin American countries, other species, such as Ae. taeniorhynchus, may also play a role as enzootic and epizootic vectors for the virus (Fig. 1). Further research is needed to investigate the vector competence and involvement of different mosquito species in the transmission dynamics of MADV.
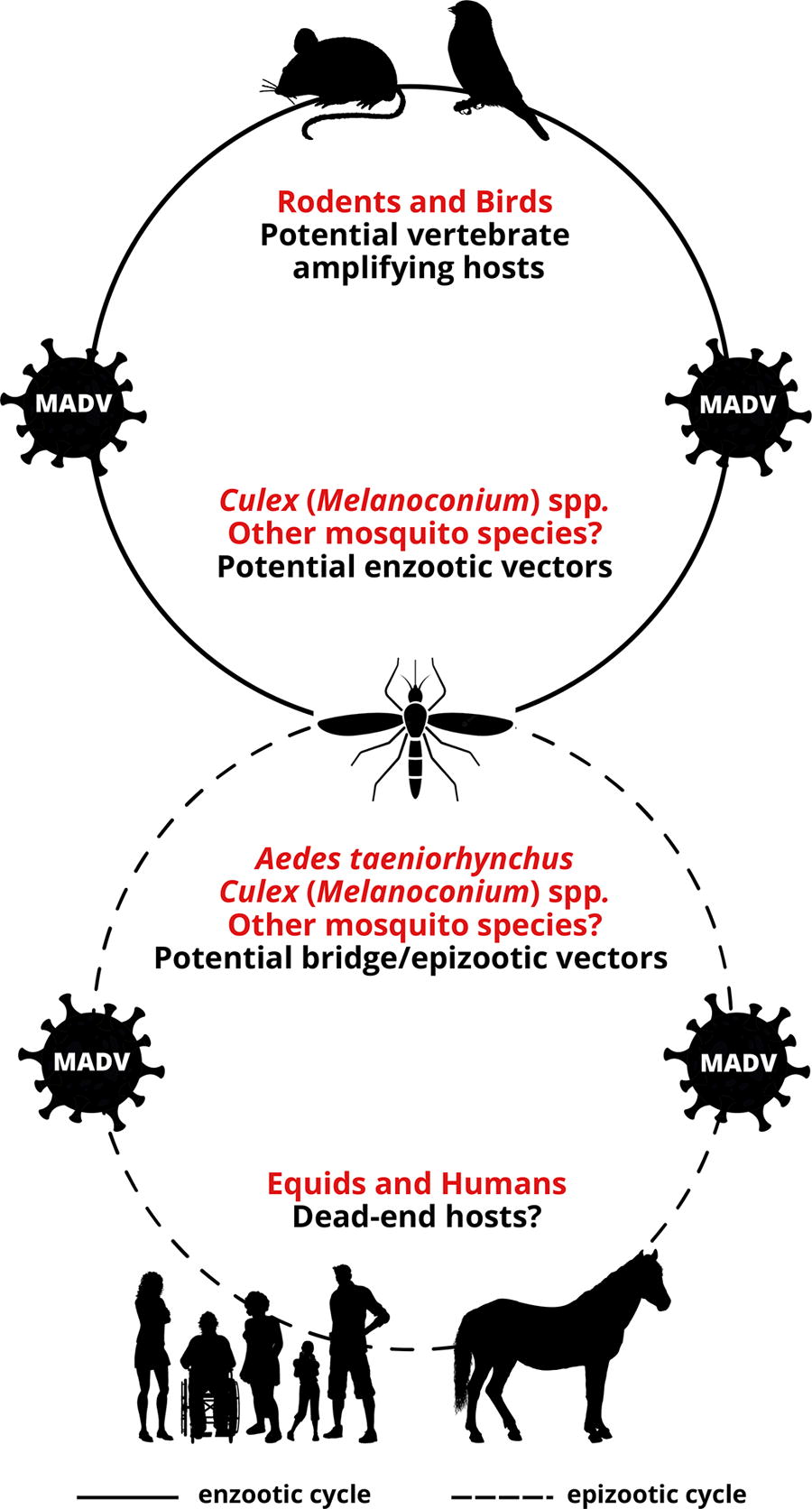
Uncertainties in the transmission cycle of Madariaga virus (MADV). Although birds are the main vertebrate hosts of the North American eastern equine encephalitis virus (EEEV), it has been suggested that ground-dwelling mammals, such as rodents, may serve as vertebrate hosts for Madariaga virus (MADV). However, birds and other vertebrates should also be considered possible hosts. Regarding the vectors, the isolation of MADV from Culex (Melanoconium) spp. in various Latin American countries implies the potential involvement of this mosquito group in MADV transmission cycles, but other mosquito species, such as Aedes taeniorhynchus, could also play a role as vectors. Equids and humans are susceptible to MADV infection and disease, but whether they are dead-end hosts, similar to the EEEV transmission cycle, is unknown.
Virus Isolation and Antibodies in Vertebrate Animals
The vertebrate hosts for MADV also remain unknown; however, genetic and ecological comparisons among NA EEEV, MADV, and VEEV, as well as serological and virus isolation studies in animals, suggest that rodents and birds could potentially serve as hosts for MADV (Fig. 1). Nonetheless, field studies aimed at identifying animal hosts for MADV are limited and scarce. The following provides a brief overview of the available studies on vertebrate animals associated with MADV (equine data presented in a separate section).
MADV has been isolated from wild birds, rodents, marsupials, and reptiles (lizard), as well as from and sentinel animals (chicks/chicken, mice, and non-human primates) in Brazil (Causey et al., 1961; Shope et al., 1966; Vasconcelos et al., 1991). In Colombia, MADV was isolated in 1969 from an unspecified animal source and in 1976 from a sentinel hamster (Groot et al., 1996). In Ecuador, MADV was isolated during field surveys during 1974–1978, but the animal source was not mentioned in the study (Calisher et al., 1983). In Guatemala (Ordonez et al., 1971), Peru (Kondig et al., 2007), and Venezuela (Walder et al., 1984), MADV was isolated from sentinel hamsters in 1968, 1998, and 1980–1981, respectively.
In North Brazil, during an equine epizootic in 1960, anti-MADV antibodies were detected in three wattled jacanas (Jacana jacana) and one opossum (species not identified) (Causey et al., 1962). In a study conducted between 1955 and 1989 in the same region, several bird species were seropositive for MADV and the overall seroprevalence rate among birds was 1.3%, with the white-shouldered antshrike (Thamnophilus aethiops) having the highest (3.5%) seroprevalence rate (Vasconcelos et al., 1991). Low seropositivity (1.4–2.9%) in birds was also reported in North Brazil in another work conducted in the 1960s (Shope et al., 1966). Anti-MADV antibodies have also been found in wild boars in Brazil (Kmetiuk et al., 2020).
In Panama, a serosurvey was conducted 2 years after a 2010 MADV outbreak to identify potential vertebrate hosts. In addition, sera collected from birds 3 years before the outbreak were also assayed for antibodies against the virus. Although no bird serum (collected before the outbreak) tested positive for MADV-reactive antibodies, four rodent and two bat species collected after the outbreak tested positive by plaque-reduction neutralization test, with the short-tailed cane mouse (Zygodontomys brevicauda) yielding the highest (8.3%) seropositivity rate (Vittor et al., 2016). This study stands out as one of the few serosurveys conducted on a significant number of relevant vertebrate animals, providing valuable insights into potential hosts of MADV in Panama.
A publication involving various countries of Central America reported seropositivity in domesticated pigs in Belize, Honduras, and Costa Rica and in wild birds in Guatemala during field studies conducted during 1961–1971—the authors mention that these areas were near the Guatemalan region where MADV was isolated in 1968 (Scherer et al., 1977). Antibodies against MADV were detected in opossums, lizards, and turtles in the field studies conducted in the 1980s in Venezuela (Walder et al., 1984).
Overall, there is a need for additional investigations in endemic regions to identify the vertebrate amplifying hosts for MADV. Also, given the significant variation in biomes among endemic regions, and the independent evolutionary patterns observed for MADV (Arrigo et al., 2010), it is likely that different species serve as hosts in the distinct geographic areas where MADV circulates.
Equine Cases and Epizootics in Latin American Countries
Once MADV enzootic transmission amplifies in natural sylvatic cycles or following anthropogenic disturbances, spillover epizootics may occur in equids (horses, donkeys, and mules). Several Latin American countries have reported equine MADV epizootics and/or virus isolation from horses, some of them dating back to the early 20th century.
In Brazil, numerous studies conducted between 1943 and 2015 have reported seropositivity rates for MADV in equines. Rates during inter-epizootic periods range from 6.7% to 56.6%, while those during or immediately following epizootic periods range from 39.5% to 69.0%. It is important to note that during epizootics, many animals may have died before being tested, which reduces the seroprevalence rate. Epizootics have been observed in states across all five regions of Brazil (North, Northeast, Central-West, Southeast, and South), indicating that MADV is widespread in that country and has adapted to diverse Brazilian biomes (Aguiar et al., 2008; Campos et al., 2013; Causey et al., 1962; Cunha et al., 2009; Fernandez et al., 2000; Iversson et al., 1993; Lennette and Fox, 1943; Melo et al., 2012; Nunes et al., 2021; Oliveira et al., 2014; Pauvolid-Correa et al., 2015; Pauvolid-Correa et al., 2010; Silva et al., 2011; Vasconcelos et al., 1991). Reported case fatality rates in equines range from 55.5% to 98.8% (Campos et al., 2013; Lennette and Fox, 1943; Silva et al., 2011). In addition, the virus has been isolated from or detected through molecular assays in samples from the central nervous system of horses on multiple occasions (Campos et al., 2013; Causey et al., 1962; Oliveira et al., 2014, Silva et al., 2011), including a recent isolation in 2019 in the northeast region (Gil et al., 2021).
In Panama, equine epizootics were documented in five provinces in 1962 (Medina et al., 1965). Although the exact case fatality rate was not indicated, the authors reported approximately 200 equine deaths and very few recoveries from disease. Another equine epizootic caused by MADV occurred in 1973, with a case fatality rate of 40.0% (Dietz et al., 1980). In 2010, an epizootic was reported in the Darién Province, coinciding with an outbreak caused by VEEV (Carrera et al., 2013). The case fatality rates in this study were reported as 76.0% and 83.0% in two different regions, although it was not explicitly specified how many deaths were attributed to MADV or VEEV. A separate study analyzing serum samples from horses collected during the 2010 MADV epizootic reported a seroprevalence rate of 26.3% for MADV among animals exhibiting neurological symptoms (Carrera et al., 2018).
In the studies conducted by Brault et al. (Brault et al., 1999) and Arrigo et al. (Arrigo et al., 2010), MADV strains isolated from horses in Argentina and Venezuela, obtained from 1936 to 1996, were included in the analysis. Although it is likely that these viruses were isolated from the animals during epizootics, there is no available report in the literature detailing the outbreaks. The first documented equine epizootic in Argentina caused by MADV occurred in 1981, with an estimated case fatality rate of 61.0% (Sabattini et al., 1991). Still, in Argentina, a serosurvey conducted from 1977 to 1980, during inter-epizootic periods, showed that several horses were seropositive for MADV (Monath et al., 1985).
In Peru, while MADV has been isolated from sentinel animals during a VEEV surveillance program conducted in 1970-1971 (Scherer et al., 1975), and from mosquitoes in subsequent studies, there is no documented case in the scientific literature of equine epizootics caused by the virus. However, a serosurvey conducted in an indigenous community area during 2007–2008 showed a seropositivity rate of 1.5% in horses (Perez et al., 2019), indicating that animals in this region have been exposed to MADV.
In Colombia, there are references to serological evidence suggesting the circulation of MADV in 1957 within a region where equine encephalitis outbreaks were reported (Groot et al., 1996). However, it is unclear whether the samples tested were obtained from horses. The only documented case of MADV equine infection in Colombia occurred between 1992 and 2002, with a case fatality rate of 66.6% in a 1992 epizootic (Mesa et al., 2005).
In Guiana, MADV was isolated from a horse in 1959 during an epizootic (Spence et al., 1961). In Ecuador, 1.3% of surveyed equines had antibodies against MADV in a seroprevalence study conducted in 2009–2011, and the authors refer to previous equine encephalitis cases caused by MADV in the region (Gutiérrez-Vera et al., 2021).
In Trinidad, a seroprevalence study conducted between 2006 and 2009 in domesticated animals reported that 4.3% of horses were seropositive for MADV (Thompson et al., 2012). In Costa Rica, 3.0% of horses were seropositive for MADV in 2013 (Leon et al., 2020).
Overall, these studies underscore the high pathogenicity of MADV to equids.
Human Cases
Until 2010, there were only three reported cases of encephalitis caused by MADV in humans: two cases in Trinidad and Tobago (Corniou et al., 1972) and one case in Northeast Brazil (Alice, 1956). Two of these cases, one in each country, were fatal. Owing to the low number of reported human neurological cases associated with MADV, it has been generally believed that the virus is less pathogenic to humans than NA EEEV.
An outbreak in Panama in 2010 that led to the detection of several cases of human encephalitis, particularly in children, challenged this paradigm (Carrera et al., 2013). Clinical symptoms of MADV infection in people included fever, headache, vomiting, status epilepticus, somnolence, and altered mental status. In two confirmed MADV infections, the clinical symptoms and brain images closely resembled those of NA EEEV cases, but brain images of other patients differed from NA EEEV infection. In addition, the authors highlighted the high frequency of seizures leading to status epilepticus. One fatal case was considered a “suspected” MADV infection and three of the hospitalized MADV cases were discharged with moderate to severe neurological sequelae (Carrera et al., 2013). In 2017, another fatal case caused by MADV infection was identified by the Ministry of Health of Panama (Carrera et al., 2020).
Seroprevalence studies conducted in humans residing in or near endemic regions of Latin American countries have reported varying seropositivity rates, ranging from 2.2% to 25.0% in Brazil (Causey et al., 1962; Causey and Theiler, 1958; Romano-Lieber and Iversson, 2000; Vasconcelos et al., 1991), from 4.8% to 19.4% in Panama (Carrera et al., 2018; Carrera et al., 2020; Vittor et al., 2016), and from 1.5% to 3.0% in Peru (Aguilar et al., 2007; Perez et al., 2019). In Ecuador, 8.6% of humans tested positive for MADV in a serosurvey (Gutiérrez-Vera et al., 2021). A study conducted in the 1970s in Panama following an equine epizootic did not find any seropositivity for MADV among the 1,700 human sera screened (Dietz et al., 1980). These seroprevalence findings highlight the varying levels of risk for MADV infection in human populations and emphasize the importance of further research to identify risk factors associated with infection.
Importantly, MADV has been recently detected in human febrile cases in Brazil (Costa et al., 2019), Venezuela (Blohm et al., 2018), and Haiti (Lednicky et al., 2019) during surveillance efforts aimed at other arboviruses.
The historical human fatal cases in Brazil (Alice, 1956) and Trinidad and Tobago (Corniou et al., 1972), the 2010 human encephalitis outbreak in Panama (Carrera et al., 2013), the 2017 fatal case in Panama (Carrera et al., 2018), and the unexpected detection of MADV in individuals with acute febrile illnesses in Brazil (Costa et al., 2019), Haiti (Lednicky et al., 2019), and Venezuela (Blohm et al., 2018) provide evidence of the virus’ pathogenicity to humans.
Potential Threat
MADV represents a significant threat to both animal and human health, as supported by the aforementioned data. There are several additional considerations that further emphasize its potential risks.
First, MADV may pose a higher threat in endemic countries than currently recognized, as infections and associated diseases in animals and humans may be misdiagnosed or go unnoticed. The co-circulation of several arboviruses among human populations in MADV-endemic regions, including some that may lead to neurological symptoms, could contribute to misdiagnosis. Thus, an important question to ask is whether diseases caused by MADV (acute febrile or neurological illnesses) are going unnoticed or being misdiagnosed and thus being underreported.
Second, as pathogens increasingly cross geographic barriers, MADV also presents a risk to regions where it is not currently known to be circulating, including the United States. Once introduced to new areas or countries, MADV may cause sporadic epizootics and human outbreaks or quickly adapt to local vectors, hosts, and environments, establishing sustained transmission. These risks are also associated with the fact that alphaviruses possess a remarkable ability to rapidly adapt to new ecological niches, vectors, and hosts, sometimes through a single genetic mutation (Weaver and Reisen, 2010). These mutations can also lead to changes in virulence or pathogenicity. It would be important to evaluate if different MADV strains are more virulent than others because of vector or host adaptations.
Also important is the fact that equine encephalitis viruses are considered biothreat agents because of their potential to cause severe disease and death, high infectivity, ease of production, stability, and potential for aerosol transmission. Experimental studies in animals have demonstrated that aerosolized MADV leads to severe disease and mortality rates similar to the outcomes observed with NA EEEV (Roy et al., 2009).
Finally, there is no vaccine available for humans or animals specifically targeting MADV. Although the commercially available trivalent equine encephalitis vaccines (against NA EEEV, WEEV, and VEEV) (Stromberg et al., 2020) may potentially offer cross-protection against MADV in animals, the efficacy of these vaccines in protecting animals against disease caused by MADV to control or prevent an outbreak is unknown. Similarly, the efficacy of the inactivated NA EEEV human vaccine (Pierson et al., 2021) in protecting people against MADV infection and disease is unknown. A study conducted in the 1990s with three volunteers who received the inactivated NA EEV showed that vaccinees developed extremely low levels of neutralizing antibodies against MADV envelope 2 protein (Strizki and Repik, 1995), raising questions about the cross-protective effect of the elicited immune response.
Conclusions
Ecological and serological data point to ground-dwelling mammals, in particular rodents, as potential amplifying hosts for MADV in enzootic cycles, and seropositivity data indicate various types of animals are susceptible to infection and able to mount an immune response against the virus. However, specific vertebrate animals capable of sustaining MADV viremia sufficient to infect local vectors and serving as amplifying hosts have not been identified. Because birds are the main amplifying vertebrate hosts for NA EEEV and have been found to be seropositive for MADV in Central American and SA countries, their role as MADV hosts remains to be determined.
Numerous studies highlight Cx. (Mel.) spp. as likely enzootic vectors of MADV in certain regions, and these species could also act as bridge or epizootic vectors. Nevertheless, other species, such as Ae. taeniorhynchus, might also serve as vectors in enzootic or epizootic cycles. A concerning issue is the lack of information regarding the physiological status of mosquitoes in various studies reporting MADV isolation from the insects. This is a crucial information as virus isolation from engorged female mosquitoes within tested mosquito pools may have derived from infected host blood rather than infected mosquito tissues. Likewise, more studies on the host-feeding patterns of putative vectors would help narrow down candidate vertebrate amplification hosts. Nevertheless, as MADV seems to have efficiently adapted to various biomes across Central and South America (Fig. 2), the transmission cycles may differ across the different regions.
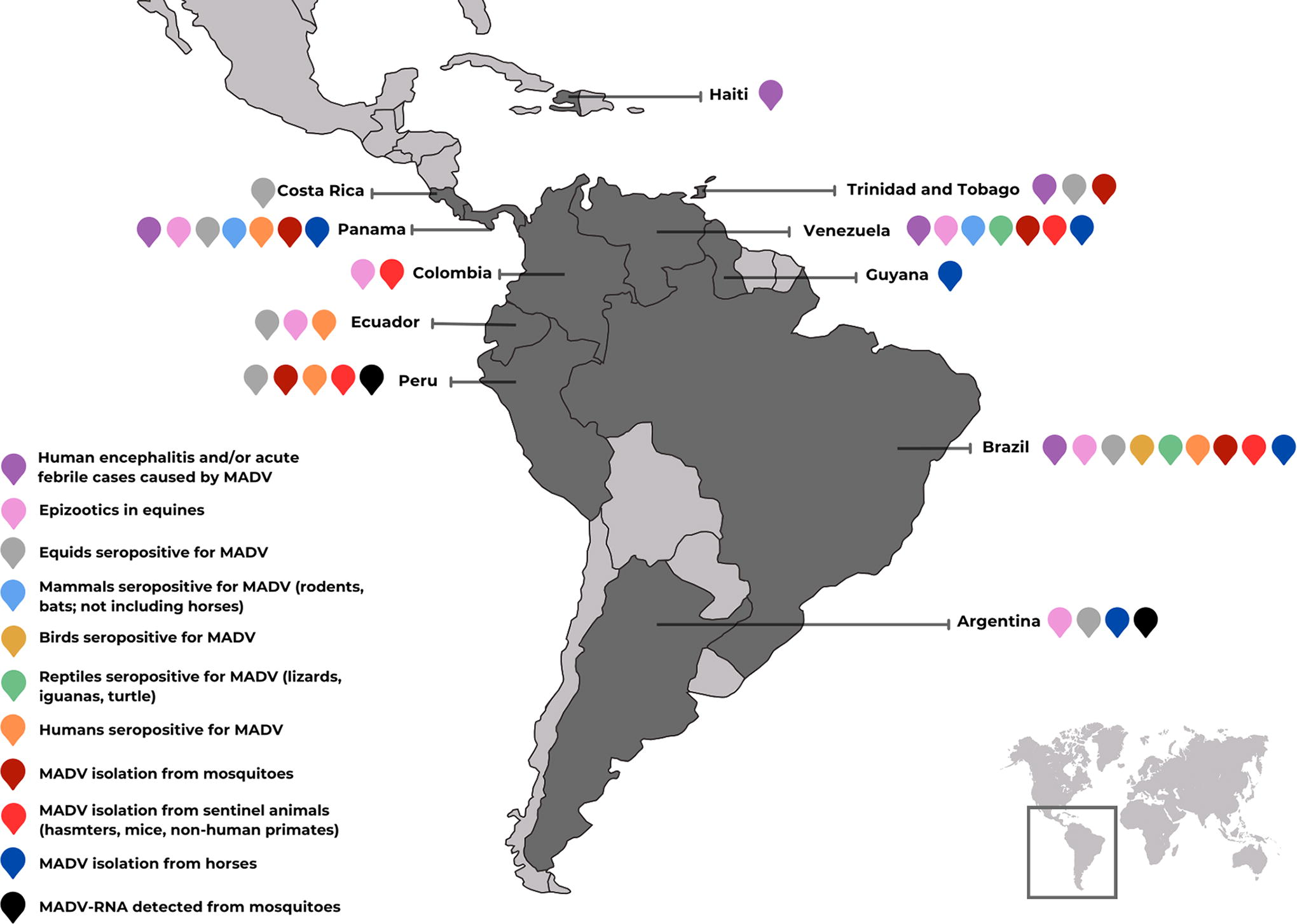
Compilation of Madariaga virus (MADV) data from published literature. The map illustrates the countries in Latin America where equine epizootics and human cases have been reported, as well as countries where antibodies against MADV have been detected in vertebrates and virus isolation has been obtained from mosquitoes and vertebrates. Sequencing data show that MADV Lineages II and III predominate in Latin America, and only one isolate of Lineage IV has been found in Brazil in 1985. More detailed information is available in the main text.
Finally, even though equids and humans can seroconvert to MADV and become sick after infection, whether they are dead-end hosts, like in the NA EEEV transmission cycle, or amplifying hosts, as in the VEEV transmission cycle, also represents an important knowledge gap.
The numerous knowledge gaps regarding MADV, its widespread presence in Latin America and its potential to cause severe disease in both animals and humans, highlight the urgency for more research efforts, heightened awareness, and intensified surveillance toward this potential emerging threat.
Footnotes
Authors' contribution
Study conception: T.M., M.J.T. Data collection and analysis: T.M., G.L.H., M.J.T. Draft manuscript preparation: T.M., G.L.H., D.C.L., V.M.L.R., M.J.T. All authors reviewed the results and approved the final version of the manuscript.
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
This work received support from Texas A&M University, Texas A&M AgriLife Research, and the United States Department of Agriculture National Institute of Food and Agriculture (Hatch proposal 6070). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the article.
