Abstract
Retrospective serological and case diagnostic data of endemic bluetongue virus (BTV) and epizootic hemorrhagic disease virus (EHDV) provide evidence of viral transmission among livestock and wildlife from 2016 in Kansas and Nebraska. Serological testing of mature cattle in nine distinct regional zones of Kansas revealed 76% to 100% had detectable antibodies to BTV and/or EHDV. Specimens tested in the Kansas Veterinary Diagnostic Laboratory (55 submissions) were 51% test positive for antibodies to BTV and/or EHDV. Specimens tested in the Nebraska Veterinary Diagnostic Center (283 submissions) were 25% test positive for antibodies to BTV and/or EHDV. Low disease incidence in white-tailed deer and other susceptible wild ungulates was observed during 2016. However, there were no confirmed reports of disease in livestock in either state. The reasons for emergence of significant clinical disease in livestock and wildlife populations remain undefined.
Bluetongue virus (BTV) and epizootic hemorrhagic disease virus (EHDV), family Reoviridae, genus Orbivirus, infect both domestic and wild ruminants and are transmitted by Culicoides (Diptera: Ceratopogonidae) biting midges (Bonneau et al., 2002; MacLachlan, 1994; Mecham, 1997). The primary vector species for much of the United States is Culicoides sonorensis (Purse et al., 2005). BTV and EHDV are genetically distinct viruses, but have overlapping reservoir hosts, vectors, and exhibit similar clinical signs of disease. BTV has been responsible for numerous disease outbreaks in North America, particularly in sheep flocks of the western United States (Parsonson et al., 1985). In addition to livestock, BTV also infects wildlife (Drolet et al., 2013; Nettles and Stallknecht, 1992). Some North American wild cervid species (e.g., white-tailed deer, mule deer) appear to be more susceptible to BTV than Eurasian deer species (Drolet et al., 2013).
EHDV is the causative agent of EHD, an acute, infectious, and often fatal disease of wild ruminants. In North America, the most severely affected ruminant is the white-tailed deer (Odocoileus virginianus), although EHDV may also infect mule deer, black-tailed deer, elk, bighorn sheep, and pronghorn antelope, as well as some nonnative ruminant species (Mayo et al., 2020). A northward expansion of orbiviral hemorrhagic disease in white-tailed deer (O. virginianus) has been documented in the upper midwestern and northeastern United States (Orange et al., 2021; Stallknecht et al., 2015). Widespread EHD has been observed in cattle herds in both Kansas and Nebraska (Kvasnicka, 1985; Stevens et al., 2015).
We completed retrospective studies of serological data and some wildlife disease data to estimate serological prevalence and disease presence in 2016 in Kansas and Nebraska. Initially, a set of serum specimens was collected from mature cows from ranches selected in the zones as indicated in the Kansas map during the fall of 2016 (Fig. 1 and Table 1). The sera (50 samples per zone) were separated and tested for antibody activity to BTV and EHDV using serogroup-specific enzyme-linked immunosorbent assay (ELISA) according to the manufacturer's instructions * . This first study utilized sera from a study completed in 2016 by the Kansas Veterinary Diagnostic Laboratory. Most of the animals were antibody test positive (by ELISA). There was a consistently high positive test rate across the state in this study, even in western counties where disease in cattle was rarely reported (Table 1). It is not clear from these data when exposure and transmission may have occurred.
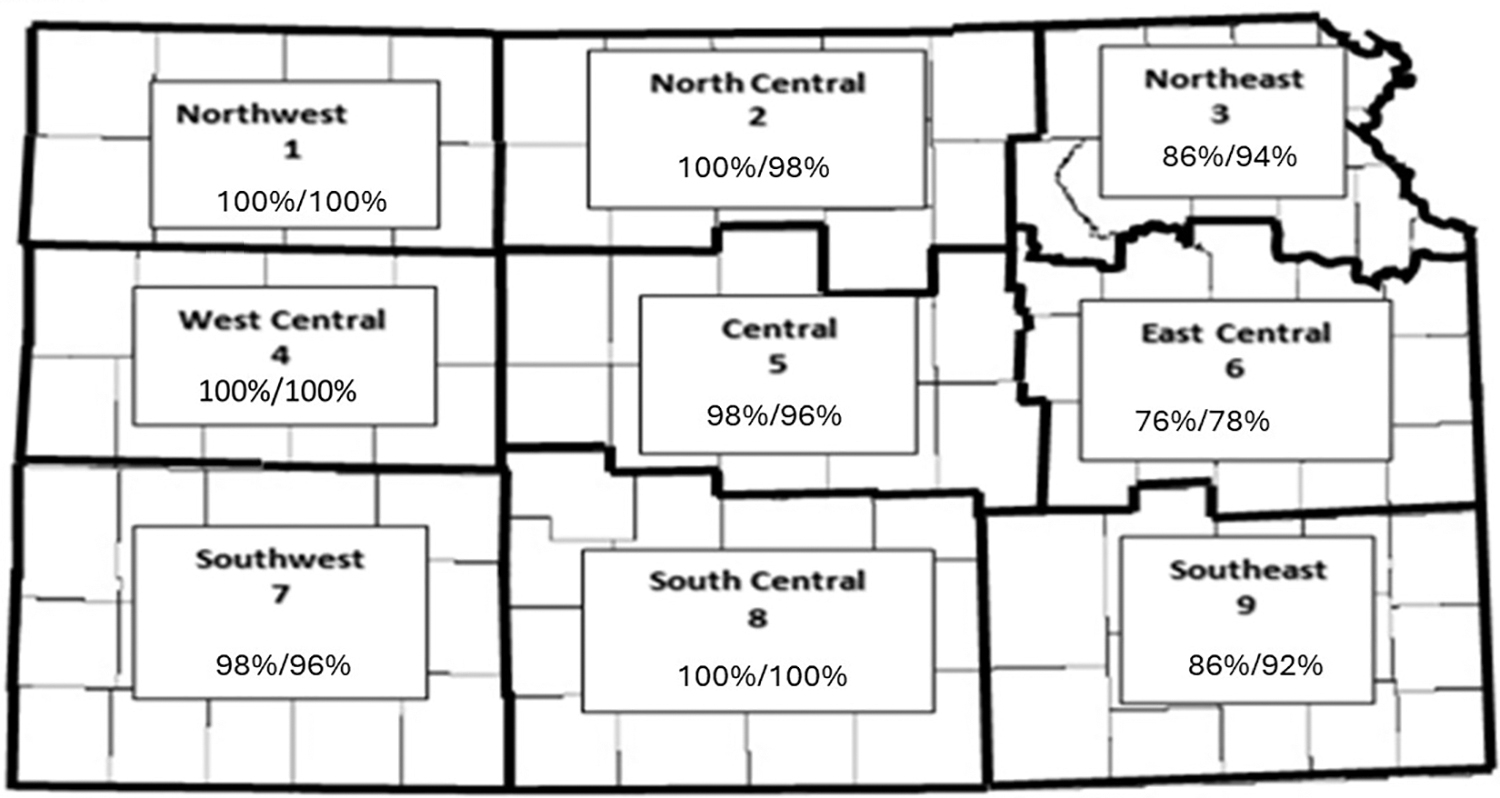
Map of Kansas showing nine distinct regions of surveillance for antibodies to epizootic hemorrhagic disease virus (EHDV) and bluetongue virus (BTV). Sera obtained from cattle (adult cows, n = 50) within each region on approximately 10 operations per region. Sera were originally obtained as part of an anaplasmosis study. Antibody prevalence for BTV and EHDV in cattle serum samples collected from nine zones in Kansas are indicated within each zone on the Figure (BTV seropositive%/EHDV seropositive%). Sera were tested using serogroup-specific enzyme linked immunosorbent assays (ELISA) Bluetongue Virus Antibody Test Kit, cELISA v2, VMRD, Inc., Pullman, WA; EHDV Screen EHDV Competition, IDVet, Grabel, France.
*
Antibody Prevalence for Bluetongue and Epizootic Hemorrhagic Disease Virus in Cattle Samples Collected from Nine Regions in Kansas (Fig. 1.)
BTV, bluetongue virus; EHDV, epizootic hemorrhagic disease virus;
Additional case and data sets from both the Kansas and Nebraska veterinary diagnostic laboratories were reviewed for the years 2016. The number and distribution of positive tests in both states indicate transmission of both BTV and EHDV (Fig. 2A, B, Tables 2 and 3). Most of these specimens were submitted in support of commercial livestock sales and interstate movement, not for disease diagnostics. Specimens tested in the Nebraska Veterinary Diagnostic Center (283 submissions) were 25% test positive for antibodies to BTV and/or EHDV. We also reviewed descriptive information regarding disease and death in wild ungulates in both Kansas and Nebraska, as follows:
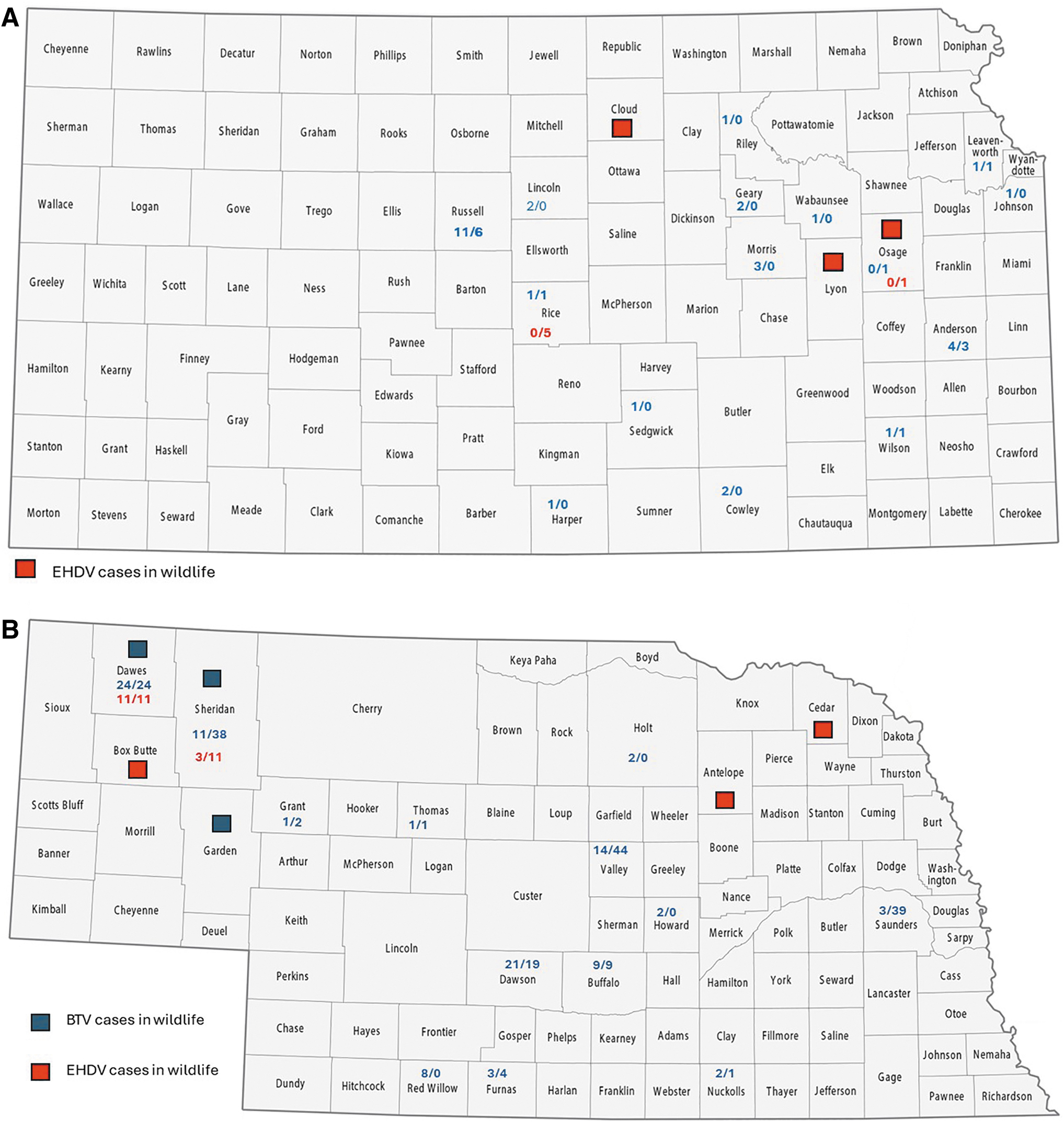
Maps of Kansas
*
Serological Results for Kansas Cattle (KVDL) and Wildlife Cases (SCWDS) by Counties from 2016 and 2017.
The %positive data were only listed if more than two specimens were tested.
NS = no submission; KVDL, Kansas Veterinary Diagnostic Laboratory; SCWDS, Southeastern Cooperative Wildlife Disease Study.
*
Serological Results for Nebraska Cattle (NVDC) and Wildlife Cases (SCWDS) by Counties from 2016 to 2017
The %positive data were only listed if more than two specimens were tested.
NS = no submission; NVDC, Nebraska Veterinary Diagnostic Center; SCWDS, Southeastern Cooperative Wildlife Disease Study.
*
From Kansas during 2016, 10 samples were received at the Southeastern Cooperative Wildlife Disease Study (University of Georgia), all from white-tailed deer. EHDV-2 was isolated from 3 counties: Osage, Lyon, Cloud (Fig. 2A).
From Nebraska during 2016, 26 samples were received at SCWDS representing a variety of species (white-tailed deer, mule deer, pronghorn, elk, bighorn sheep). Viruses were isolated from seven animals (four EHDV-2 and three BTV-17) in the following counties: EHDV-2: Cedar, Antelope, Box Butte; BTV-17: Garden, Sheridan, Dawes (Fig. 2B).
For passive, retrospective serological surveillance, the ELISA is a useful tool, but there are some unfortunate restrictions interpreting results as detection is virus serogroup specific but not serotype specific. Antibody usually appears within 7 to 10 days after infection of the orbiviruses (Afshar et al., 1987a; Afshar et al., 1987b; Stott and Osburn, 1990). Antibody activity, particularly neutralizing antibody activity, decreases after viral clearance (Koumbati et al., 1999), but the absolute duration of titers is not known. Serotype-specific protective immunity may last for years, but the actual duration of measurable titers and the impact of potential re-exposure is not definitively known. Given the high numbers of animals with detectable antibodies to the orbiviruses, serological surveillance of yearling animals would likely provide the best evidence of more recent transmission.
A duplex microsphere assay to differentially detect-specific antibody to BTV or EHDV in a single serum sample has recently been developed (Drolet and Reister-Hendricks, 2021) although it is not yet available in diagnostic laboratories. This platform offers the potential advantages of enhanced laboratory efficiency and the ability to add multiple serotype-specific antigens. Serum neutralization testing is generally not feasible for surveillance studies and routine diagnostics due to technical requirements for virus propagation. Epitope and serotype specificity of antibody responses and disease protection correlations to specific infecting viruses would be very useful.
Transmission of BTV and EHDV typically increases through the warm weather months as midge populations (and the proportion of infected midges) increase. In the presence of infected midges, wildlife and livestock are continually at risk for BTV or EHDV infection (Möhlmann et al., 2021) and within-farm transmission rates can be high (Hagenaars et al., 2021). Endemic boosting from infected midges may contribute to immunity and low incidences of observed disease in some years (Flacke et al., 2004; Ribeiro et al., 2020). It is not clear what factors contribute to emergence of more significant clinical disease in livestock and wildlife populations.
Surveillance of white-tailed deer as sensitive sentinels for orbivirus transmission and disease would be effective, as reviewed by Clarke et al. (2022). Integrating entomological surveillance (Dinh et al., 2021; McDermott and Lysyk, 2020; McGregor et al., 2021), molecular taxonomic tools (Shults et al., 2022), and habitat characterization (Erram et al., 2019; Martin et al., 2019; Mayo et al., 2014) would also be useful to describe the ecology of the Culicoides vectors relative to competent vector density in the United States. These questions warrant further investigation and the enhancement or initiation of active livestock and wildlife serosurveillance programs.
Footnotes
Disclaimer
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Author Disclosure Statement
The investigators declare no conflicting interests. Raw data will be available to the public through Ag Data Commons within 30 months of publication.
Funding Information
These studies were supported by the USDA, ARS, Animal Health National Program NP-103, #3020-32000-020-00D. The Kansas Veterinary Diagnostic Laboratory (Kansas State University, Manhattan, KS), the Southeastern Cooperative Wildlife Disease Study (University of Georgia, Athens, GA), and the Nebraska Veterinary Diagnostic Center (University of Nebraska Lincoln, Lincoln, NE) provided collaborative support and data.
