Abstract
We call into question the established dogma that viruses with envelopes and RNA genomes have limited stability by demonstrating the staggering long-term viability, ∼2 years, of chikungunya virus when stored in liquid environments at +4°C in the dark. We contend that our understanding of the infectivity of a variety of enveloped viruses requires a new approach to identify under standardized conditions the primary determinants of their viability.
Introduction
Viruses are broadly categorized by the presence or absence of a lipid-bilayer envelope and the DNA/RNA nature of their genome. This has for decades structured virologists' thinking regarding virion viability, with envelope and RNA genomes being commonly associated with virus lability (Feng et al. 2013, McKnight and Lemon 2017). In this study, we call this established dogma into question by demonstrating the staggering long-term stability of chikungunya virus (CHIKV), when stored in liquid environments at +4°C in the absence of light.
CHIKV (genus Alphavirus) has a protein-encapsidated positive-sense RNA genome, enclosed by a lipid-bilayered envelope protein. Epidemic CHIKV is primarily transmitted by domesticated Aedes aegypti mosquitoes and is responsible for iterative explosive epidemics worldwide (Geographic Distribution | Chikungunya virus | CDC 2019). CHIKV viremia can occur in presymptomatic and asymptomatic individuals, such as blood donors (Brouard et al. 2008, Appassakij et al. 2013, 2014, Gallian et al. 2014, Simmons et al. 2016).
This situation creates a risk of transfusion-related transmission of blood product recipients—although such incidents have never been formally reported (Petersen and Epstein 2014). During the 2014 CHIKV outbreak in the French West Indies, locally collected packed red blood cells (PRBCs) were subjected to a 3-day quarantine to enable donors to report symptoms after donation and to facilitate RT-qPCR screening (Gallian et al. 2014).
Moreover, PRBCs maintained at +4°C may be stored for up to 42 days postdonation, during which period they undergo progressive deterioration representing the so-called storage lesion (Koch et al. 2013). To minimize the risk of blood-borne transmission of CHIKV, the issue of decreasing this “fragile” virus infectivity has been raised (Petersen et al. 2010, Petersen and Epstein 2014). Here we investigate the hypothesis of a limited stability of CHIKV in blood products and provide an answer to the relevance of implementing a quarantine period for blood products to limit CHIKV infectivity.
Methods
Preparation and storage of PRBCs, plasma, and CHIKV-spiked water aliquots
In February 2014, an asymptomatic viremic donor was identified in France, French Departmental Area of Martinique, by RT-qPCR screening of blood products as previously described (Gallian et al. 2014). The blood sample was collected by personnel at the French Blood Bank with agreement from the donor for nontherapeutic use of blood donation, including medical research. A plasma sample and a PRBC bag from the donor were obtained and used for this study. From the PRBC bag, stored at +4°C for 52 days postsampling, we prepared twenty 2-mL aliquots; the plasma sample stored at −20°C was divided into twenty-four 2-mL aliquots.
The infectious virus strain from the plasma sample was isolated, sequenced, and deposited in the European Virus Archive collection (“UVE/CHIKV/2014/MQ/EFS_1_plasma,” Ref-SKU: 001v-EVA1556). The freeze-dried strain (titer: 10E6.82 TCID50/mL) was diluted in pH = 7 sterile distilled water (final experimentally determined titer: 10E5.92 TCID50/mL) and twenty-four 1-mL aliquots were prepared.
All aliquots (PRBCs, plasma, and CHIKV-spiked water) were subsequently stored at +4°C in the dark, mimicking the PRBC storage conditions before transfusion.
Molecular testing
Viral loads of these aliquots of PRBCs/plasma/CHIKV-spiked water were monitored over time by RT-qPCR. Regularly, a new aliquot underwent nucleic acid extraction using the automated assay EZ1 Virus Mini Kit (QIAGEN). RT-qPCR amplification was performed with GoTaq RT-qPCR kit (PROMEGA), a QuantStudio 12K Flex thermocycler (ThermoFisher) and protocol according to Pastorino et al. (2005). Negative and positive controls were included in each RT-qPCR run; samples with a Ct value <40 were considered positive.
Infectivity assay
In parallel with molecular testing, residual infectivity assays were performed using aliquots as inoculum. For virus isolation experiments, 200 μL of a new aliquot of stored PRBCs/plasma/CHIKV-spiked water diluted 1:2 in heat-inactivated fetal bovine serum (FBS) was inoculated onto a 12 · 5 cm2 monolayer of Vero cells. After 90 min, cells were washed and incubated under standard culture conditions (10% FBS-supplemented Minimum Essential Medium, 5% CO2) at 37°C. Infectious virus was detected based on observation of cytopathic effect (CPE) on cell monolayers and the results of RT-qPCR were performed on cell culture supernatant fluids.
Criteria to identify infectious aliquots were (1) a distinct CPE at, or before, day 7 postinfection (pi) and (2) at day 7 pi, lower RT-qPCR Ct values in culture supernatant fluid than in the inoculum, indicating replication of viral RNA in the Vero cells.
The protocol for preparation of PRBCs, plasma, and CHIKV-spiked water aliquots, and monitoring of viral load and residual infectivity is summarized in Fig. 1.

Flowchart of tests on PRBCs, plasma, and CHIKV-spiked water samples. CHIKV, chikungunya virus; PRBCs, packed red blood cells. Color images are available online.
Results
Residual infectivity of PRBCs, plasma, and CHIKV-spiked water
At the time of reception of the PRBC bag, 52 days postsampling, it still contained infectious virus that could be isolated and amplified to high titers in Vero cells. Thus, delaying the quarantine period by weeks before transfusion would not have resolved transfusion safety issues. We, therefore, investigated the longevity of infectivity over time and surprisingly, infectious CHIKV was isolated from a PRBC aliquot stored at +4°C in the dark, 47 weeks (329 days) after blood collection. Although lysis of erythrocytes developed progressively in PRBC aliquots, we assumed that the persistent infectivity of CHIKV was due to the specific biological environment in PRBCs, including the presence of red blood cells. Accordingly, we tested the infectivity in plasma aliquots, which represent a protein-rich acellular medium.
Remarkably, infectious CHIKV was isolated from a plasma aliquot stored at +4°C 71 weeks (497 days) after defrosting the original plasma unit. Moreover, the viral load in the plasma was higher than that in the PRBC bag (Fig. 2), which may have contributed to its longer period of detectable infectivity. We speculated that plasma proteins might preserve CHIKV infectivity. Hence, we tested the longevity of CHIKV infectivity stored in water. Surprisingly, CHIKV was isolated from a spiked-water aliquot stored for 77 weeks (539 days) at +4°C in the dark (RT-qPCR Ct values for CHIKV-spiked water aliquots were similar to those of plasma aliquots). Results of residual CHIKV infectivity in PRBCs, plasma, and CHIKV-spiked water samples are summarized in Fig. 3.
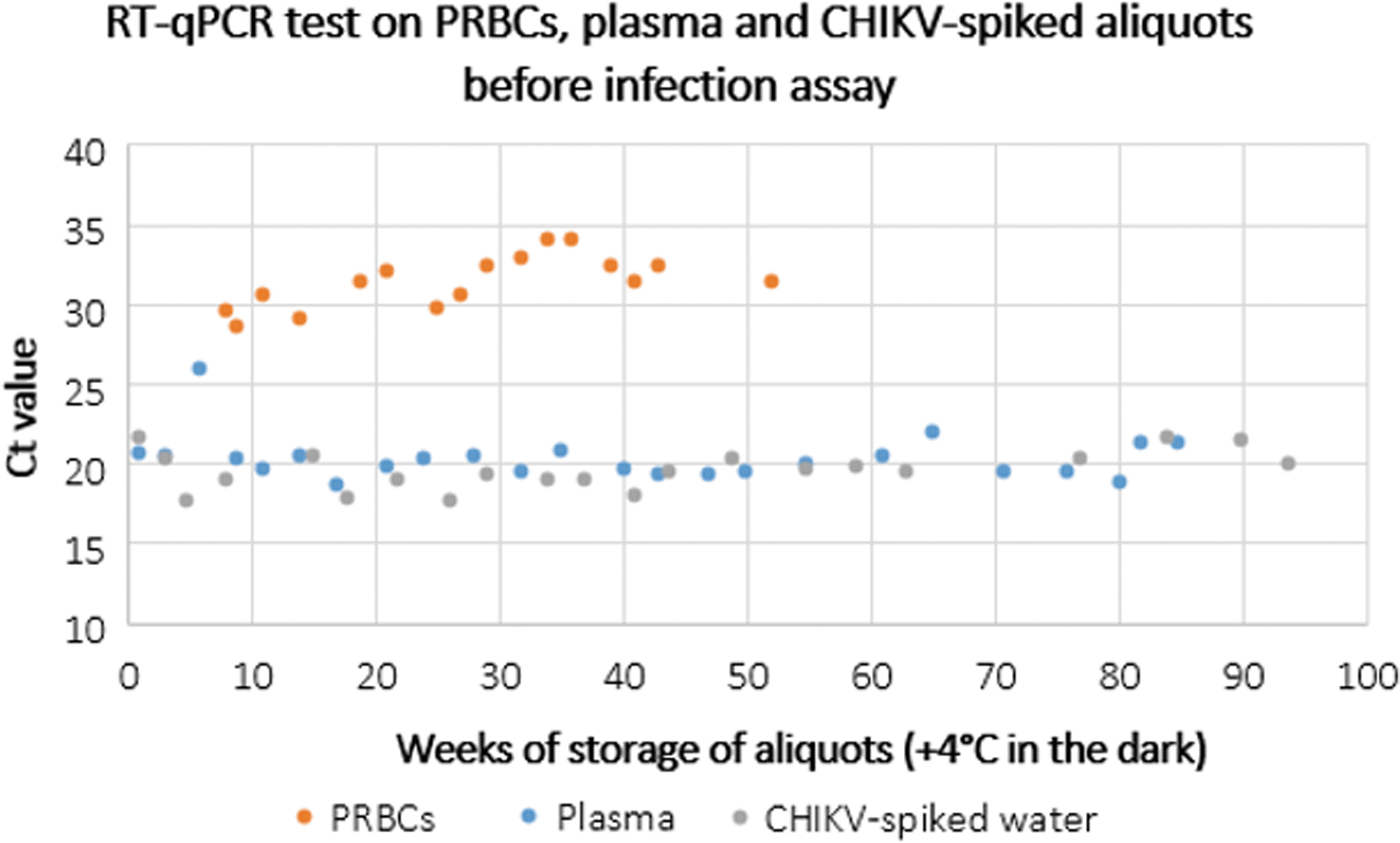
RT-qPCR Ct values, represented by dots, for aliquots of PRBCs, plasma, and CHIKV-spiked water over time. Color images are available online.
CHIKV infectivity versus storage time in aliquots of PRBCs, plasma, and CHIKV-spiked water. Dots (•) represent aliquots with confirmed infectivity;
Virus infectivity was confirmed by the progressive appearance of CPE when aliquots were inoculated onto Vero cells (Fig. 3) and also by the detection of CHIKV-specific RT-qPCR product in the corresponding Vero cell supernatant fluids (Fig. 2).
Molecular testing of PRBCs, plasma, and CHIKV-spiked water
RT-qPCR tests on aliquots, performed at the time of infectivity assay, showed that Ct estimates remained constant over time in all media tested (Fig. 3, Supplementary Figs. 1–3), with mean Ct values of 31.3, 20.1, and 19.4 for PRBCs, plasma, and CHIKV-spiked water, respectively.
Discussion
This study suggests that CHIKV molecular load and infectivity (which reflect the integrity of the nucleocapsid complex and of the envelope, respectively) can be maintained for months at 4°C in the dark in various liquid media such as PRBCs, plasma, and water. This appears to contradict conventional thinking and our intuitive understanding of the (in)stability of enveloped RNA viruses. These results indicate the need to identify the primary determinants of CHIKV stability and by extension, also of other enveloped viruses. Our observations are not without precedent.
Recently, another Alphavirus (Tonate virus [TONV]) has been isolated from a bat serum collected 10 years before despite low viral load in the original sample, suggesting that the infectious potential was preserved over time (Fischer et al. 2021). The enveloped DNA hepatitis B virus (Shikata et al. 1978) and enveloped RNA viruses including influenza, Ebola, and Marburg viruses and human immunodeficiency virus (Stallknecht et al. 1990, Dublineau et al. 2011, Shigematsu et al. 2014) are surprisingly stable under similar conditions to those described herein for CHIKV, notably in a liquid environment at +4°C and even in some cases at 60°C (Huang et al. 2015). The importance of the absence of light in preservation of viral survival has also been demonstrated (Sagripanti et al. 2010).
These preliminary findings indicate that quarantining blood products at +4°C in the dark may not be an effective strategy with which to mitigate the risk of transfusion-related CHIKV infection. Paradoxically, the preservation of infectivity under such basic conditions suggests that we should reconsider the primary determinants of conservation of infectious viral material. Although freezing at −80°C or freeze drying will remain the most convenient solutions for long-term preservation of viruses, it remains to be investigated whether conservation at 4°C could offer an effective alternative for shorter periods of time. This would generate several technical and economic benefits (e.g., absence of freeze–thaw cycles, simplification of safety procedures, and shipment of infectious material without the need for expensive and heavy dry ice).
Conclusions
In light of these findings, we call for more systematic investigations of the stability of enveloped viruses under standardized conservation conditions. This should provide new clues for understanding the key biochemical and structural determinants of the stability of enveloped virions and, in turn, open the way to optimized protocols for the conservation and/or inactivation of enveloped virus strains in infectious samples or culture media.
Footnotes
Authors' Contributions
Conceptualization of the study was done by X.d.L. and P.G.; methodology was taken care of X.d.L. and O.P.; validation was done by O.P. and M.G.; data analysis was carried out by L.P., O.P., I.D., P.M.S.V., and T.F.; writing, original draft preparation, was done by L.P. and E.A.G.; writing, review and editing, was done by X.d.L., O.P., M.G., P.M.S.V., I.D., T.F., and P.G.; supervision was taken care of X.d.L. and P.G.; project administration was done by X.d.L.; funding acquisition was by X.d.L. and P.G. All authors have read and agreed to the published version of the article.
Author Disclosure Statement
No conflicting financial interests exist.
Funding Information
No funding was received for this article.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
