Abstract
Crimean Congo Hemorrhagic Fever (CCHF) is an emerging tick-borne zoonotic viral disease with the potential of causing public health emergencies. However, less is known about the role of wildlife and livestock in spreading the virus. Therefore, we aimed to assess how the interactions between African buffalo (Syncerus caffer) and cattle may influence the seroprevalence of CCHF across livestock-wildlife management systems in Kenya. The study included archived sera samples from buffalo and cattle from wildlife only habitats (Lake Nakuru National Park and Solio conservancy), open wildlife-livestock integrated habitats (Maasai Mara ecosystem and Meru National Park), and closed wildlife-livestock habitats (Ol Pejeta Conservancy) in Kenya. We analyzed 191 buffalo and 139 cattle sera using IDvet multispecies, double-antigen IgG enzyme-linked immunosorbent assay (ELISA). The seroprevalence toward Crimean Congo hemorrhagic fever virus (CCHFV) was significantly higher for buffalo compared to cattle (75.3% and 28.1%, respectively, p < 0.001). We obtained the highest seroprevalence among buffalo of 92.1% in closed wildlife only systems compared to 28.8% and 46.1% prevalence in closed-integrated and open-integrated systems, respectively. The regression coefficients were all negative for cattle compared to buffalo in both closed-integrated and open-integrated compared to wildlife only system. Our results show that CCHFV circulates among the diverse animal community in Kenya in spatially disconnected foci. The habitat overlap between cattle and buffalo makes cattle a “bridge species” or superspreader host for CCHFV and increases transmission risks to humans. The effect of animal management system on prevalence is depended on tick control on the cattle and not the animal per se. We conclude that buffalo, a host with a longer life span than livestock, is a reservoir and may serve as a sentinel population for longitudinal surveillance of CCHFV.
Introduction
Crimean-Congo Hemorrhagic Fever (CCHF), which first emerged as a human disease in the Crimea in 1944 and later in Africa in 1956, is caused by a virus in the family Nairoviridae, genus Orthonairovirus family (Simpson et al. 1967, Hoogstraal 1979). Since then, sporadic cases occur in Africa and Eurasia without attracting International response or attention (Fisher-Hoch 2005, Bente et al. 2013). Considering that the virus is highly contagious with case-fatality rates of 5–30% (Bente et al. 2013) and its geographical expansion coupled with lack of a widely approved antiviral drugs or effective vaccines, CCHFV is potentially a public health risk of international concern.
Recently, the World Health Organization (WHO) listed CCHFV among diseases of public health priority for research and development (Mehand et al. 2018). Although Bulgaria has domesticated use of CCHFV vaccine within the country, the vaccine yields a low amount of neutralizing antibodies against CCHFV in immunized healthy people (Papa et al. 2011, Mousavi-Jazi et al. 2012). To advance research in the development of a vaccine that may offer a wider coverage, a global representative of CCHFV genotypes must be studied and virus evolution and phylogeography understood.
Currently, genetic data of CCHFV from Africa are too few in the global databases to be informative (Lukasheve et al. 2016). Interestingly, most of the European isolates, which belong to genotype V, are highly conserved than CCHFV circulating elsewhere (Deyde et al. 2006, Zehender et al. 2013, Sherifi et al. 2014). Besides, except for Seroprevalence studies, little information is known about the CCHFV host–pathogen interaction, a component that is pertinent when developing prevention and control strategies.
The information available about the ecology and epidemiology of the CCHFV is that the virus is maintained and transmitted by hard ticks (family Ixodidae) mainly by the genus Hyalomma (Logan et al. 1989, Gonzalez et al. 1992, Spengler and Estrada-Peña 2018). Despite the virus's detection in other tick genera, their vectorial capacity and reservoir role have not been confirmed (Gargili et al. 2017). The spread and emergence of CCHFV in new regions such as India, Spain, Greece, and Morocco (Papa et al. 2010, Messina et al. 2015, Al-Abri et al. 2017) are of public health interest, given the low awareness of the disease and lack of strategic preparedness in case of an outbreak. The annual intercontinental migration of birds harboring virus-infected ticks is likely mediating the spread to new locations (Chumakov et al. 1974, Hoogstraal 1979, Estrada-Peña et al. 2012, Spengler et al. 2016b).
CCHFV may infect most terrestrial vertebrates without clinical symptoms; hence, some species serve as virus reservoirs. Thus, the enzootic tick-vertebrate-tick cycle is critical in the maintenance of the virus. Human transmission is mainly by infected tick bites, and other transmission routes include direct contact with the blood of an infected animal (Nabeth et al. 2004b), sexual human-to-human transmission, and nosocomial spread (Ergonul and Battal 2014, Pshenichnaya and Nenadskaya 2015, Pshenichnaya et al. 2016, Papa et al. 2017).
The high diversity and demography of free-ranging wild and domestic animals in sub-Sahara Africa coupled with lack of tick control make Africa a critical location to evaluate the epidemiology and ecology of CCHFV. Most of the earliest studies on the epidemiology and clinical profiles of CCHFV were from Africa (Gear et al. 1982, Joubert et al. 1985, Swanepoel et al. 1987, 1989, Burt et al. 1997), although recent case reports from Africa are becoming fewer compared to Eurasia (Goldfarb et al. 1980, Baskerville et al. 1981, Ergonul et al. 2006, Ergonul 2007, Bente et al. 2010, Pshenichnaya et al. 2017). The disease is barely known across the animal and human health sectors in Africa; thus, no diagnosis or surveillance is targeted (Burt 2011, Mazzola and Kelly-Cirino 2019, Papa 2019). Moreover, given that CCHF is a febrile illness, it is likely masked by other well-known febrile diseases, such as malaria.
Kenya is one of the African countries where CCHFV circulates silently among humans, animals, and ticks. For instance, evidence of human infection (Dunster et al. 2002) and the detection of the virus in Hyalomma and Rhipicephalus ticks are documented (Sang et al. 2011, Chiuya et al. 2020). Human seroprevalence studies (Lwande et al. 2012, Tigoi et al. 2015) provide evidence that the circulation of CCHFV occurs in a wider geographical area in Kenya (Nyataya et al. 2020).
Human exposure and risk for infection by CCHFV in Kenya are predictably linked to the increasing contact with the tick-animal reservoirs. However, apart from a study that reported a lack of antibodies to CCHFV in the wild baboon population (Kirya et al. 1972), there is no other seroprevalence study in Kenya's wildlife. Similarly, studies on prevalence of CCHFV in livestock in Kenya are scarce, with the surveillance in camels conducted three decades ago being the only apparent reference (Morrill et al. 1990).
This study aims to unravel the epidemiology of CCHFV in the wildlife-livestock systems in the Kenyan rangelands. Specifically, the study sought to determine seroprevalence among African buffalo's (Syncerus caffer) different subpopulations across protected areas in Kenya. In addition, since CCHFV is a tick-borne pathogen, and previous studies have shown that the management systems of wildlife and livestock influence tick abundance in the environment (Keesing et al. 2013, Allan et al. 2017), we further tested the effect of buffalo-cattle management systems on the seroprevalence of CCHFV. Surveillance data from animals are highly recommended by the WHO (Mehand et al. 2018), and findings from this study are expected to reveal virus circulation, distribution, and epidemiology of CCHFV in animals in Kenya.
Materials and Methods
Study areas and animal populations
The animal population studied are cattle and buffalo populations in rangelands across multiple Counties in Kenya (Fig. 1). The five locations: Maasai Mara ecosystem (MME), Lake Nakuru National Park (LNNP), Meru National Park (MNP), Solio Conservancy (SC), and Ol Pejeta Conservancy (OPC) (Fig. 1) occur in the agropastoralist zones of Kenya. The region is rich with megaherbivores, including African elephants, zebra, African buffalo, and highly diverse antelope species. The area has a high population of livestock that includes cattle, goats, sheep, and camels, which are reared under intensive commercial system or transhumance.
Map of Kenya (inset), with the study area (highlighted section) showing locations of populations of cattle and African buffalo (Syncerus caffer). Color images are available online.
Overall, wildlife numbers have remarkably declined on average by 68%, while livestock have significantly increased across Kenya's rangelands between 1977 and 2016 (Ogutu et al. 2016). For instance, the biomass for livestock was 8.1 times greater than wildlife in 2011–2013 compared to 3.5 times in 1977–1980 (Ogutu et al. 2016). The interaction between livestock and wildlife at the interface is variable depending on the management systems.
OPC is a fenced ranch that integrates free interaction between wildlife and cattle. Cattle is mainly raised for commercial beef production, and the main breed is improved boran. The close interaction between wildlife and livestock necessitates intensive disease prevention and control on cattle, which includes vaccinations, deworming, tick-control, culling, and treatment of the sick. Tick control is done by mechanized spray race, which ensures that every individual animal is thoroughly sprayed by acaricide. The spraying is carried out twice weekly in all the herds.
The MME is a wildlife protected area that comprises the Maasai Mara National Reserve (MMNR) and dispersal areas contiguous to the pastoral community land. The ecosystem, which is on the south-western border between Kenya and Tanzania, is in Narok County, which holds about 30% of all Kenya's wildlife (Ogutu et al. 2016). The County is dominated by the Maasai pastoralist community whose livelihood is livestock depended. They keep large herds of cattle, which are communally grazed, across landscapes in search of water and pasture (Omondi et al. 2021). Considering that the MMNR is not fenced, livestock–wildlife interaction is common, which results in frequent conflicts and also increases risk of vector and disease transmission.
The MNP is a wildlife-protected area in the North-Eastern part of Kenya. The agropastoralists, mainly the Borana, Somali, and Meru communities, have settled around the park, and they periodically drive animals into the park to graze. A lack of perimeter fence coupled with the seasonal demand for pasture leads to an incursion of livestock into the protected area and allows for interaction between livestock and wildlife.
According to Mugambi et al. (2012), the tick-borne diseases are a huge production burden among the pastoralists in Kenya, including those in MME and MNP. The pastoralists apply acaricides using hand sprays, once a week on animals seen with ticks, rather than on the whole herd because of cost constraints (Mugambi et al. 2012). It is interesting that due to cost constraints and large herds, over 90% of farmers incorrectly dilute the acaricides (Mugambi et al. 2012), which may lead to resistance or low effectiveness.
The LNNP, located on the central Rift Valley of Kenya, is completely ring-fenced and exclusive for wildlife. SC and OPC are situated in the Laikipia-Samburu ecosystem; however, SC has a wildlife only ring-fenced system, while OPC has a mixed wildlife-livestock population ring-fenced system.
Blood sampling
This study is based on archived sera collected between 2008 and 2018 in cattle and African buffalo populations in the five locations (Fig. 1) during surveillance studies of other diseases and routine wildlife management interventions.
Cattle and buffalo sera were collected concurrently in 2015 in OPC and also in 2018 in MME. Other buffalo samples were collected from populations in SC in 2008, LNNP in 2010, and MNP in 2012. We retrieved a total of 191 buffalo sera for analysis: SC (n = 79), LNNP (n = 35), MME (n = 24), OPC (n = 47), MNP (n = 6). For cattle, we retrieved a total of 139 sera for analysis: OPC (n = 78) and MME (n = 61). The serological tests were carried out at Kenya AIDs Vaccine Initiative Institute of Clinical Research (KAVI-ICR), University of Nairobi.
Briefly, during sampling, about 15 mL of blood was drawn from sedated buffalo (using a mixture of Etorphine and Azaperone; Wildlife Pharmaceuticals) and physically restrained cattle. Blood was drawn in plain tubes (coated with coagulant activator), centrifuged at 1500 g for 15 min to harvest sera. The sera were labeled and preserved in liquid nitrogen during transportation and in −80°C freezers for long-term storage until processing.
Serology—CCHF enzyme-linked immunosorbent assay
Previously, different studies have used various assays for serological detection of antibodies against CCHFV in cattle and buffalo. In this study, we used a CCHF double-antigen, multispecies Enzyme-Linked Immunosorbent Assay (ELISA) Kit (IDScreen®; IDvet, Grabels, France), which according to the manufacturer has a sensitivity of 99.8% and specificity at 100%, both measures under 95% confidence interval (CI). We tested IgG antibodies in 191 buffalo and 139 cattle sera according to the manufacturer's instructions. Briefly, 50 μL of the kit dilution buffer was added onto each well of the 96-well microplates coated with CCHFV recombinant nucleoprotein followed by 30 μL of each test sample or control on designated test and control wells. The plate was covered and incubated for 45 min at 25°C.
After a washing procedure, 50 μL of the reconstituted nucleoprotein-Horseradish Peroxidase conjugate was added to each test well, followed by incubation for 30 min at 25°C. A second washing procedure was then performed, and 100 μL of substrate added, followed by incubation for 15 min at 25°C. To stop the reaction, 100 μL of stop solution was added to each well. We determined optical density value for each test sample or the control using a spectrophotometer at 450 nm. Sample to positive control ratio percent (S/P %) was then calculated by dividing the optical density obtained for the tested sample by the test's positive control, multiplied by 100. The S/P % was used for result interpretation; serum presenting an S/P % less than or equal to 30% was negative and those greater than 30% were positive (Grech-Angelini et al. 2020).
Data analysis of risk factors
To test whether there is an association between buffalo and cattle interaction, we categorized the locations (study areas) where sera samples of buffalo and cattle were sourced. We used presence/absence of interaction between livestock and wildlife and whether the area is fenced off or lacks movement barriers to categorize the ecosystems. LNNP and SC are closed wildlife systems because they host wildlife with no livestock interaction in a ring-fenced ecosystem. OPC is a closed integrated system as wildlife and livestock continually interact within a fenced ecosystem. MME and MNP are open integrated systems as livestock periodically interact with wildlife due to open borders or lack of ring-fence.
We used a General Linear Model (GLM) with a binomial link to assess the association between the different risk factors (species—buffalo/cattle) and area of sampling classification (closed wildlife, closed integrated, and open integrated) and prevalence of CCHFV, with a p value of <0.05 considered statistically significant.
Ethical approvals
The archived samples of cattle and buffalo were collected during a study permitted by the Kenya Wildlife Service (KWS/BRM/5001) and the Narok County government (NCG/HEALTH/GEN/VOL.1/2). The ethical approval for the study protocols was obtained from both KWS and the University of Minnesota IRB ID (STUDY00000837).
Results
Out of the 191 buffalo and 139 cattle samples, we obtained an estimated prevalence of CCHFV at 75.3% (95% CI: 68.65–81.32) and 28.1% (95% CI: 20.77–36.30) for buffalo and cattle, respectively. Prevalence in cattle herds in MME was 31.1%, while herds in OPC was 25.6%. Seroprevalence of CCHFV in buffalo populations across locations was spatially variable (Fig. 2). Compared to buffalo, cattle were less likely to be positive for CCHFV (p < 0.001). Analysis of sampled areas revealed a prevalence of 92.1% in closed wildlife ecosystems (LNNP and SC) compared to 28.8% and 46.1% prevalence in closed integrated (OPC) and open integrated systems (MME and MNP), respectively (Table 1). Prevalence in buffalo was highest in habitats with no cattle interaction (LNNP and SC) (Table 2). The regression coefficients were all negative for cattle compared to buffalo (Tables 3 and 4) while closed integrated and open integrated compared to wildlife only, indicating that cattle presence is protective although the effect is much stronger in a closed integrated system (Table 4).
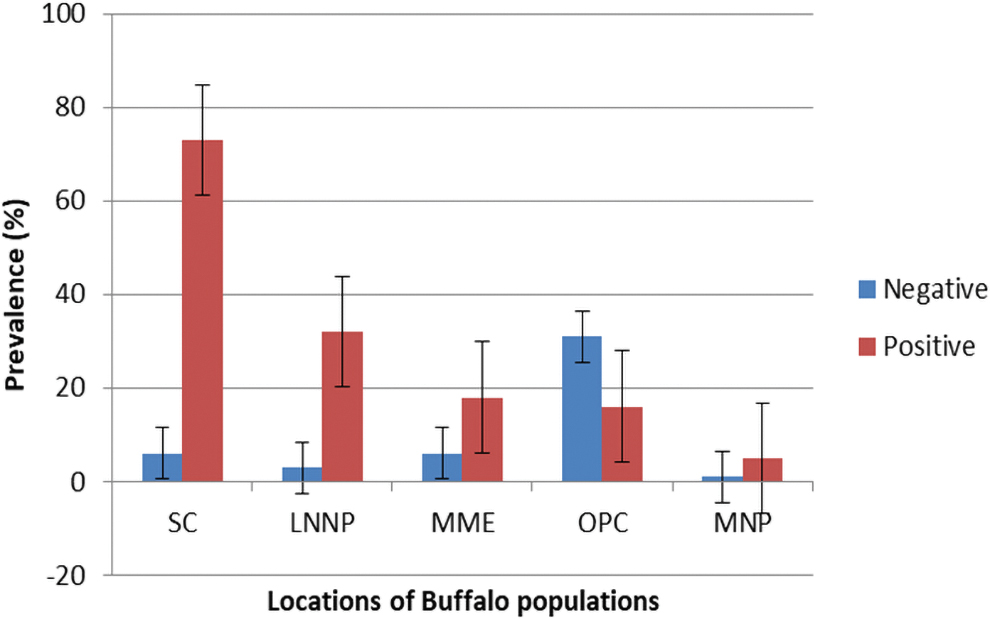
Seroprevalence of CCHFV in Buffalo populations in five different locations in Kenya (error bars). Key: LNNP, Lake Nakuru National Park; MME, Maasai Mara Ecosystem; MNP, Meru National Park; OPC, Ol Pejeta Conservancy; SC, Solio Conservancy. Color images are available online.
Seroprevalence of CCHFV in African Buffalo and Cattle in Relation to Livestock-Wildlife Management Systems in Kenya
Key: LNNP, Lake Nakuru National Park; MME, Maasai Mara ecosystem; MNP, Meru National Park; OPC, Ol Pejeta Conservancy; SC, Solio Conservancy.
Prevalence of CCHFV in African Buffalo (Syncerus caffer) Populations in Relation to Livestock-Wildlife Management Systems in Kenya
Results of the General Linear Model on the Effects of Animal Species and the Livestock-Wildlife Management Systems on the Prevalence of CCHFV in Kenya (n = 330 Observations)
SE, standard error.
Results of the Final General Linear Model on the Effects of Management Systems on the Prevalence of CCHFV in African Buffalos (n = 191 Observations)
Discussion
The study's overall objective was to determine the prevalence of CCHFV in both domestic and wild animal communities in Kenya. Thus, our results confirm that both wild and domestic animals are exposed to CCHFV, with an overall seroprevalence in cattle at 28.1% and buffalo at 75.3%. Cattle is one of the mammals highly investigated for CCHFV, with over 75 studies worldwide with overall seroprevalence at 19% (Spengler et al. 2016a). In contrast, there are fewer studies on CCHFV prevalence among livestock in Africa (Spengler et al. 2016a). For instance, the only significant study on CCHFV prevalence in Kenya among livestock is on camels (n = 499), which showed 26% seropositivity (Morrill et al. 1990). Therefore, our results report the first study in Kenya on CCHFV seroprevalence in cattle.
It appears that seroprevalence of CCHFV in cattle is relatively high across Africa, especially when we compare our results to those in Uganda (75%) and Sudan (57%), which were also based on IDvet ELISA (Mangombi et al. 2020, Balinandi et al. 2021). Overall, the exposure to CCHFV in cattle is heterogeneous across herds in different African regions, and factors driving such variations are not well clarified.
Our study also provides the first record of CCHFV seroprevalence in the African buffalo in Kenya, and the third survey across Africa, following the only two previous studies that were conducted in South African populations (Shepherd et al. 1987, Burt et al. 1993). The seroprevalence of CCHFV in South African buffalo populations was 10% and 20% based on reverse passive hemagglutination inhibition (RPHI) assays and competitive ELISA (Shepherd et al. 1987, Burt et al. 1993).
The difference in test assays between our study and the two previous ones makes it difficult to compare prevalence in buffalo populations, but the seropositivity suggests that the African buffalo are highly susceptible to CCHFV infection. African buffalo is one of the widespread bovids across sub-Sahara Africa, and their grazing requirements tend to overlap with cattle, which facilitates cross-transmission of several infectious diseases and ectoparasites (Miguel et al. 2013, Kock et al. 2014). Moreover, the two bovid species are common in integrated wildlife-livestock ranching systems in Africa. In this study, the prevalence of CCHFV in buffalo was higher than cattle, and because of the range overlap, buffalo likely serves as a source of infection to cattle.
The role of wildlife, especially African buffalo, on the epidemiology of CCHFV goes beyond its reservoir role and includes a potential source of direct human transmission. The illegal hunting, slaughter, and consumption of buffalo in Kenya (Kimwele et al. 2012) may cause CCHFV transmission to the hunters as they handle infected blood (Nabeth et al. 2004a).
Our results also showed spatial heterogeneity of CCHFV prevalence (Fig. 2) in discrete buffalo populations across Kenya (Fig. 1). Analysis of sampled areas revealed a positivity rate of 92% in closed wildlife ecosystems compared to 29% and 46% positivity rate in closed integrated and open integrated systems, respectively. The buffalo populations in closed wildlife systems, SC and LNNP, have no contact with cattle, while populations in MNP and MME (open integrated) and OPC (closed integrated) interact with cattle. Thus, our results suggest that cattle presence is protective, especially in closed integrated systems, which explains the very low (29%) seroprevalence in OPC buffalo.
The variation in CCHFV prevalence between open and closed integrated systems in Kenya is likely influenced by the tick-vertebrate-tick cycle of CCHFV and the relative tick abundance coupled with associated animal seroprevalence. According to Keesing et al. (2018), wildlife-livestock integration has ecological benefits in reducing tick abundance and transmission of tick-borne pathogens (Keesing et al. 2018). A study carried out in the Laikipia-Samburu ecosystem, which includes our study sites of OPC and SC, showed that tick abundance was higher in wildlife properties and lower in cattle properties (Keesing et al. 2013). However, in wildlife-livestock integrated properties, tick abundance was 75% lower than in wildlife-alone properties (Keesing et al. 2018). The reduced tick abundance on these habitats is likely associated with pesticides used to control ticks and mites (acaricides) on livestock.
Acaricide-treated livestock serves as biological sprinklers, releasing droplets as they disperse in grazing field and consequently killing larval tick stages, and also as traps that kill ticks attempting to feed (Keesing et al. 2013, 2018, Allan et al. 2017). Therefore, the relatively lower prevalence of CCHFV in OPC buffalo (closed integrated system) compared to MME (Open integrated system) may have been influenced by the varied frequency and application methods of the acaricide treatment on livestock.
According to Amulyoto (2020), OPC applies acaricides on all the cattle through the automatic cattle spray races, which is more efficient than hand spraying of randomly selected animals, commonly used by pastoralist communities that occupy areas adjacent to MME and MNP. In addition, OPC applies the acaricides twice a week, while application by pastoralists is nonscheduled and acaricides not correctly diluted (Mugambi et al. 2012). We did not test for cross-reactivity within Nairoviruses, especially Hazara virus, which is in the same serogroup as CCHFV. However, based on previous records of CCHFV in Kenya and this Double-Antigen ELISA, which has been validated for multiple species across different mammalian ancestries (Sas et al. 2018), it provides preliminary data on animal CCHFV prevalence in Kenya.
Our results thus suggest that in environments without tick control, such as wildlife-alone spaces, exposure to CCHFV is high, and movement of livestock to and from those spaces disseminates (through infected ticks) the virus to human spaces.
Conclusion
Our results indicate that CCHFV is prevalent in Kenya's animal community, with wildlife maintaining a permanent source of the virus.
Footnotes
Authors' Contributions
V.O., B.A., I.B.-P., C.A., M.E., O.W.L., and V.O. conceived the study and wrote the draft of the article. V.O., B.A., I.B.-P., C.A., M.E., and V.O. participated in discussions affecting the contents and further improvements of the article. V.O., I.A.M., E.K., and G.P.O. performed laboratory analysis. All authors have read and approved the final article.
Acknowledgments
The authors appreciate the researchers, veterinarians, and technicians who assisted in fieldwork, laboratory analysis, and sample management in Kenya. Specifically, the authors are grateful to Elsie Wambui Maina for her technical assistance during laboratory work.
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
The project was funded by the Swedish Research Council (2019-04366). The funding agency had no role in any part of the study or in the decision to publish the results.
