Abstract
Trachoma is the leading cause of infectious blindness worldwide. Ocular infection by the obligate intracellular pathogen, Chlamydia trachomatis, causes the eyelashes to turn in and scratch the cornea, leading to blindness if left untreated. The disease is most prevalent in poor, rural communities that lack the infrastructure for basic hygiene, clean water, and proper sanitation. Infection is often spread through infected clothes, contaminated hands, and face seeking flies. The goal of this research was to understand the biological role of Musca domestica flies in the transmission of C. trachomatis. PCR, tissue culture, and immunofluorescence microscopy were used to determine the presence, viability, and the anatomical location of C. trachomatis within the digestive tract of M. domestica. Flies were fed with C. trachomatis and then harvested at various time intervals after feeding. The data confirmed the presence of C. trachomatis DNA and viable elementary bodies (EBs) in fly crops, up to 24 h postfeeding. C. trachomatis DNA was also isolated from the upper portions of the alimentary tract of flies up to 48 h postfeeding. In addition, DNA was isolated from the regurgitation material from fly crops up to 12 h postfeeding. The viability of isolated C. trachomatis EBs was repeatedly confirmed between 12 and 48 h and up to 7 days in ex vivo crops stored at room temperature. Our data suggest that eye-seeking flies such as M. domestica can ingest C. trachomatis during regular feeding. Because M. sorbens does not occur in continental United States, we did not use it in any of our studies. These data also confirm, for the first time, that ingested chlamydia remains viable inside the flies for 24–48 h postfeeding. We further show that these flies can regurgitate and transmit the trachoma agent at their next feeding. We believe that these findings reveal an opportunity for efficient intervention strategies through fly vector control, especially as we near new target date for global elimination of trachoma.
Introduction
C
According to the WHO report (WHO 2020), trachoma is a public health problem in 44 countries and accounts for 1.9 million people having blindness or visual impairment. Untreated, trachoma infections can cause severe scarring of the inside of the eyelid, resulting in the eyelashes scratching the cornea (trichiasis). In addition to being painful, trichiasis permanently damages the cornea and can lead to irreversible blindness (Wright et al. 2007). Infections often spread through contact with dirty clothes, contaminated hands, and eye-seeking flies. It was reported that by decreasing the number of flies, the number of cases of trachoma also decreased (Emerson and Bailey 1999, Emerson et al. 2000), thus flies somehow must be critical in trachoma cases.
Houseflies are often abundant, especially in tropical and temperate climates, where human activities, such as food markets, restaurants, slaughter houses, poultry, livestock farms, and even hospitals are high. They constitute not just a nuisance to humans, but also act as potential vectors of diseases. Musca domestica is currently widely known as a mechanical vector for an array of pathogens, including viruses, bacteria, fungi, and parasites, some of which cause serious diseases in humans and domestic animals. The type of pathogen carried by houseflies depends on the location where the flies exist (Khamesipour et al. 2018). A recent review revealed that >130 pathogens, predominantly bacteria (including some serious and life-threatening species), were identified from the gut of houseflies (Khamesipour et al. 2018).
Miller et al. (2004) reported that as early as 1598, Baron Harant de Poljits incriminated flies frequenting the human face as vectors of trachoma. Since then, it has been noted that in endemic areas, a seasonal increase in the prevalence of these infections coincides with an increase in the Musca sorbens fly population (Ramesh et al. 2013). However, the most compelling evidence for the role of mucoid flies as vectors of the trachoma agent comes from a study published by Emerson et al. (1999), in which intervention with the insecticide deltamethrin, led to a 75% reduction in the muscid population (this includes both M. sorbens and M. domestica). This coincided with a 75% fall in the number of new cases of trachoma (Emerson et al. 1999).
Studies in Latin America confirmed that C. trachomatis can be transmitted by synanthropic flies, because childhood prevalence of trachoma was positively associated with fly densities, whereas sanitation measures, such as owning a latrine showed a protective effect (Reilly et al. 2007). Up to this point in time, it was generally accepted that face-seeking flies around the eyes transmit the pathogen through mechanical transmission routes. In their report, Evans et al. (2019) noted that C. trachomatis, “…thrives where water is scarce and hygiene is poor.” Thus, flies in these regions are mainly seeking water; and, the effect that global warming will have on the increase of trachoma remains to be seen.
In 2004, Miller et al. (2004), collected Musca sorbens from the eyes of infected children; and, using PCR for C. trachomatis DNA, examined and found 15 of 103 flies were positive, whereas none of the controls flies tested positive. Based on these results, they reported that the pesky fly vector (i.e., Musca sorbens) was finally found and that transmission was through a mechanical process. Unfortunately, the whole fly was used in the test, including the intestines, which also included the foregut diverticulated crop, with no attempt to uncover where in the gut these microbes might reside. Miller, also, did not consider the common housefly, M. domestica as a potential vector. Of importance, the crop of adult dipterans has been shown to harbor numerous human pathogens and might suggest mechanical transmission may not be the sole method of infection (Stoffolano 2019).
In 2001, Emerson et al. (2001), collected flies from the eyes of children and reported that 90% of them were M. sorbens, but the rest were M. domestica. Because housefly, M. domestica, has also been implicated as a vector of C. trachomatis (Zardi 1964, Darougar et al. 1970, Forsey and Darougar 1981), we decided to re-examine the fate of this pathogen in M. domestica in the laboratory. The primary objective of this research was to determine whether housefly transmission is purely mechanical, or if adults also serve as bioenhanced transmission vectors, as reported for housefly by Kobayashi et al.
Materials and Methods
Cell culture
Cells used in these experiments were grown in minimum essential medium with insulin (IMEMZO; Irvine Scientific, Santa Ana, CA) and supplemented with 5% fetal bovine serum (FBS; Atlanta Biologicals, Norcross, GA). Human foreskin fibroblast (HFF) cells (ATCC SCRC 1041) were grown on 12 mm coverslips in 24-well plates, at 37°C and 5% CO2 to 80% confluence and then infected with materials from the crop, digestive tract, and the regurgitation for 24–36 h to determine chlamydia viability from these anatomical locations of the insect.
Chlamydial strains
We used a human urogenital isolate of C. trachomatis, lymphogranuloma venerum (LGV 434/Bu; ATCC VR-902B, Manassas, VA) because of its more infectious nature, and an ocular strain, serovar Har-36B (ATCC VR-573, Manassas, VA) because of its biological relevance as an ocular strain in these experiments. The results were similar for both strains, although the inclusions from the LGV strain grew faster in tissue culture as expected. In brief, to create a stock solution of C. trachomatous for use in these experiments, LGV434 and Har-36B serovar elementary bodies (EBs) were removed from the −80°C freezer where they are stored in sucrose–phosphate–glutamic acid (SPG). Cryovials were thawed in a 37°C water bath for 3–5 min and the contents placed in flasks containing confluent HFF cells in complete cycloheximide overlay media.
For T75 flasks there was a total volume of 7 mL and for T25 flasks there was a total volume of 4 mL. Flasks were incubated for 4 h and the initial infection media removed and replaced with fresh complete cycloheximide overlay media (100 μg/mL cycloheximide + IMEMZO and 10% FBS) at the same volumes as above and reintubated for at least 60 h. The supernatant was removed and sterile SPG buffer added to the flasks followed by scraping the cells to completely dislodge them from the surface of the culture flask. The cell suspension was then sonicated (∼15–30 s) or vortexed with glass beads to break cells apart and release chlamydial inclusions containing EBs.
The lysed content was centrifuged at 1200 rpm for 10 min and the supernatant containing the viable EBs collected and recentrifuged for 30 min at 4°C and 30,000 g. The supernatant was then aspirated and the pellet containing crude EB preparation was resuspended in SPG and frozen down in 0.5 mL aliquots. Alternatively, infectious EBs were purified by renograffin (Squibb diagnostics, New Brunswick, NJ) density gradient centrifugation as previously described (Caldwell et al. 1981).
Determining Inclusion Forming Units
Inclusion forming units (IFUs) were determined by thawing one of the frozen aliquots and serially diluting it in infection media on confluent HFF cells on 12 mL coverslips in 24-well plates. Approximately 200 μL of diluted (1:10, 1:100, 1:1000, 1:10,000) samples were cultured in triplicates under the culture conditions described previously. After 36 h, the culture supernatant was removed and cells were fixed in 70% cold methanol for 10 min. Coverslips were rinsed with sterile phosphate-buffered saline and immunostained as described hereunder. Inclusions on the 12 mm coverslips were counted in 100 fields at 400 × magnification. The number of inclusions per field was determined by finding the average count over 100 fields and this number was multiplied by 925 (the total number of 400 × fields on a 12 mm coverslip). This gives the approximate number of inclusions in the culture and when multiplied by the dilution factor, one obtains the IFU/mL of the inoculum.
Maintaining flies
Flies were obtained weekly as pupae from Dr. Christopher Geden (USDA) and placed in a 13″ × 13″ × 13″ cubic wire-screened metal cage. The cage was padded with paper towels and included a clear plastic container with the pupae, a water apparatus with an Absorbal
Preparation and infection of flies
Fly pupae within a small plastic cup were placed in a 6.5 by 8.5 inch mesh screened cage with water and sucrose. Twenty-four hours after emerging, sucrose was removed from the cage overnight, leaving the flies for 12 h with only water. Starvation for 12 h ensured that the crop of the fly was empty and increased the likelihood for uptake of the infection and control solutions. The following day, the 48-h-old adult flies were placed in a −20°C freezer for 2–3 min to arrest movement. The flies were removed with tweezers and placed posterior-side down on flypaper to prevent movement and provide access to the proboscis. A Hamilton 1702N, 25 μL syringe (22 s/2″/3) was used for capillary feeding by loading the syringe with C. trachomatis in SPG buffer, supplemented with ∼0.10 g of granulated sugar. The plunger was depressed slightly to create a small visible bubble at the tip of the syringe, which was placed up to the proboscis of each fly. Under a dissecting microscope, the proboscis could be seen extending toward the bubble and consuming its contents. The liquid could also be seen moving into the abdomen of each fly. Each fly was fed until it refused the solution, which on average was ∼3–4 μL. Each group contained 10–15 flies that were fed with C. trachomatis serovar HAR-36B, LGV or SPG solution as a control. In the case of experiments carried out to determine the detection limit of the PCR procedure, we serially diluted the infectious dose used (4.1 × 106/mL) by 10-fold to a final dilution of 1:10,000 and then fed the solution to flies as mentioned previously. Please note that infectious solution was initially fed to flies in a food coloring solution to easily determine that the flies had indeed ingested the material. However, the food coloring inhibited the subsequent PCR reactions and had to be omitted from the protocol. Feeding experiments were repeated between 15 and 20 times for repeatability and protocol refinements.
Dissection of flies and sample preparation
Postfeeding, flies were dissected under a Bausch & Lomb Stereo Zoom 4 dissecting microscope to determine where in the fly the bacteria could be found. The crop, a thin-walled expanded portion of the alimentary canal used for food storage before digestion (Stoffolano 2019) was separated and collected. The remaining portion of the alimentary canal, the midgut and hindguts were also removed and separated. Expelled fecal matter were also collected. The collected crops from five flies, gut components from five flies, and feces from five flies were pooled in separate 2 mL labeled Eppendorf tubes containing 200 μL of SPG buffer. This created two to three sets of each pooled sample for testing. The samples were homogenized using the blunt end of a dissecting pin and vortexed gently. Half of each sample was used for evaluation of C. trachomatis DNA by PCR and the remaining half of the sample was used to determine viability of the chlamydia by cell culture.
Regurgitation
Ten male and female flies (20 flies in total) were fed chlamydia EBs by the capillary method described previously. Flies were placed in a container with a microscope slide containing ∼0.5 g of sugar granules, poured in two separate spots on the slide. The spots of sugar were circled with a marker on the underside of the slide to mark its location. Five flies of each sex were placed in two separate containers with the sugar granules. The hope was to encourage flies to regurgitate the material inside their crops, dissolve, and ingest the sugar granules on the slide. An additional five flies from each sex were placed in two separate containers with a microscope glass slide that contained 0.5 g of sugar and 20 μL of SPG to make a concentrated solution for flies to feed on. The solution was allowed to dry and flies were placed into their respective containers and allowed to feed. The goal here was to test whether flies prefer to regurgitate on a concentrated solution rather than sugar granules. After 24 h, the flies were removed by briefly putting them in the freezer to immobilize them. Then they were washed in a 1.5 mL Eppendorf tube to collect any remnants from regurgitation or defecation. The inside of the containers were scraped using a Cyto-One cell scraper and washed with SPG. The head of the scraper was removed after scrapping the spots from inside the containers and on the microscope slides and placed in 1 mL of SPG in an Eppendorf tube. This same tube was vortexed and briefly centrifuged before 200 μL of this sample was used for DNA isolation followed by PCR amplification. The remaining sample was used for infection in cell culture of HFF cell lines.
Infecting flies
Groups of 30 flies were starved for 12 h before experimental feeding. We initially used a mix of male and female flies, but most of the repeated experiments used female flies only, because they appeared to ingest ∼1–2 μL more of the solution and fed more consistently than male flies. In addition, Emerson et al. (2000) showed that females were caught more commonly than males at the eyes of humans with trachoma. The cage was then placed into the freezer for 2–3 min to temporarily immobilize the flies. Flies were capillary fed by placing them on commercial flypaper, dorsal side down and a dissecting microscope was used with a syringe to feed each fly individually with a solution containing chlamydial EBs in SPG, as described previously. About 0.5 g of sugar was added to the solution of SPG with chlamydia EBs, to further encourage uptake of the bacteria. Each fly ingested ∼4 μL of this solution. Groups of five flies were again placed in six different containers with small holes in the top for oxygen/respiration for the following different times: 0, 3, 6, 12, 24, and 48 h.
Procedure for determining the presence of chlamydia within the fly
To determine the presence of chlamydia within the flies, each fly from every cohort was dissected. The crops and remaining digestive tracts were dissected from each cohort and placed in separate 1.5 μL Eppendorf tubes, crushed with a sharp needle, and vortexed with 200 μL of SPG. One hundred microliters of each cohort was then used for DNA isolation. The remaining 100 μL was used for infecting HFF cells as previously described.
Survival of chlamydia on the body of the fly
Eppendorf tubes were labeled for each time point tested: 0, 3, 6, 12, 24, and 48 h, and 200 μL of SPG was added to each tube. Emerson et al. (2000) showed that female flies were caught more commonly than males at the eyes of humans. Thus, we focused on using females in this study. Ten female flies were collected and kept in vented plastic containers labeled to correspond with each given time point. The flies were picked up with tweezers by the wings and their feet, heads, and bodies were submerged in a tube of chlamydia in SPG solution. The flies were placed back into the plastic containers lined with Parafilm. Once the time point was reached, the flies were removed with tweezers, held in the Eppendorf tubes containing sterile SPG, and the tube was vortexed gently to ensure any material on the body came off in the tube. The tubes were stored at −80°C until all samples had been collected. The samples were then evaluated by PCR and culture as previously described. These experiments were repeated four times to confirm consistency.
DNA isolation and PCR
DNA isolation was performed using the Qiagen QIAmp Blood Mini Kit™ (Qiagen, Germantown, MD). One hundred microliters of the contents of the crop, digestive tract, feces, and regurgitation was used for DNA isolation. OMP1 and OMP2 primers were used to amplify the Momp gene of C. trachomatis resulting in a 1050 bp product size as initially shown by Hoelzle et al. (2000)
Gel electrophoresis
A 2% agarose gel was made and PCR products were loaded into each well with a positive control (C. trachomatis DNA sample) and a negative control (DNA isolated from the guts of 10 different unfed flies and PCR amplified in a similar manner as the positive control). We utilized a 100 base pair ladder (ThermoFisher/Invitrogen, Carlsbad, CA). The gel was run, stained with ethidium bromide, and then imaged using the SmartDoc and an E3000 transilluminator (Accuris™; Edison, NJ).
Immunostaining of Infected Cells
To determine the presence and amount of chlamydia recovered from each sample collected, as well as to determine the IFUs of sample used for feeding experiments, 24-well plates were set up with 12 mm coverslips. Coverslips were seeded with HFF cells in 200 μL of growth media as described previously. Once cells reached 80% confluence, 100 μL of each cohort of crop, digestive tracts, fly washes from the wings, feet, or body were mixed with 100 μL of infection media. The infection was incubated between 24 and 36 h, before being fixed with 100% cold methanol for at least 10 min. Cells were stained with a FITC-conjugated anti-chlamydia antibody (Pathfinder; BioRad, Hercules, CA) for 30 min, followed by washing with sterile phosphate-buffered saline. The coverslips were then mounted on glass slides and examined under 40 × magnification using an epifluorescence microscope and images captured using smartphone camera.
Survival 1of Chlamydia in Ex Vivo Crop
To determine how long C. trachomatis can survive in the crop, M. domestica were infected in groups of five flies (seven groups in total, with each group representing a day) using an IFU of ∼2.5 × 106 IFU/mL. Flies were infected as outlined previously using a syringe. Each fly ingested between 3 and 4 μL of material and were allowed to feed on water for another hour. Flies were then dissected as described previously and the crops carefully removed intact and immediately placed in a 2 mL Eppendorf tube containing 1 mL of SPG. The crops that are innervated immediately contracted to seal their contents inside. Crops were maintained at room temperature for the duration of the 7-day study, with five crops removed each day and the material carefully removed, stored in individual tubes, and immediately frozen at −80°C until time of testing. The crop material was then thawed, diluted in 500 μL of SPG, and used to infect semiconfluent HFF cells on coverslips, in 24-well plates for 36–48 h as outlined previously. Cells were then fixed and immunostained as outlined. IFU counts were also performed as outlined previously.
Results
Chlamydia trachomatis is present on the surface of flies for hours after exposure
Previous research has confirmed that the housefly (M. domestica L.) and blowflies (Chrysomya megacephala) together harbor >600 different bacteria (Junqueira et al. 2017, Nazari et al. 2017). Flies have poor hygiene and can therefore act as shuttles, transferring bacteria from one surface to the next on their legs, feet, and wings (Yap et al. 2008). We wanted to confirm that flies in this study could also transfer C. trachomatis on their outer surfaces in a similar manner to other bacteria and determine the length of time viable organisms could be isolated. Flies were taken up with tweezers by the wings and their feet, heads, and bodies were submerged in a tube of chlamydia in SPG solution. We used both LGV and Ser HAR-36B, C. trachomatis strains in this experiment, to determine if there were any differences in detection. They were then transferred to holding containers for the determined times, after which they were removed, rinsed with sterile SPG, which was frozen at −80°C until DNA extraction and amplification. We amplified C. trachomatis DNA from the outer bodies of the flies up to 24 h after initial exposure (Fig. 1). This was the case for both the ocular and genital strain of C. trachomatis used.
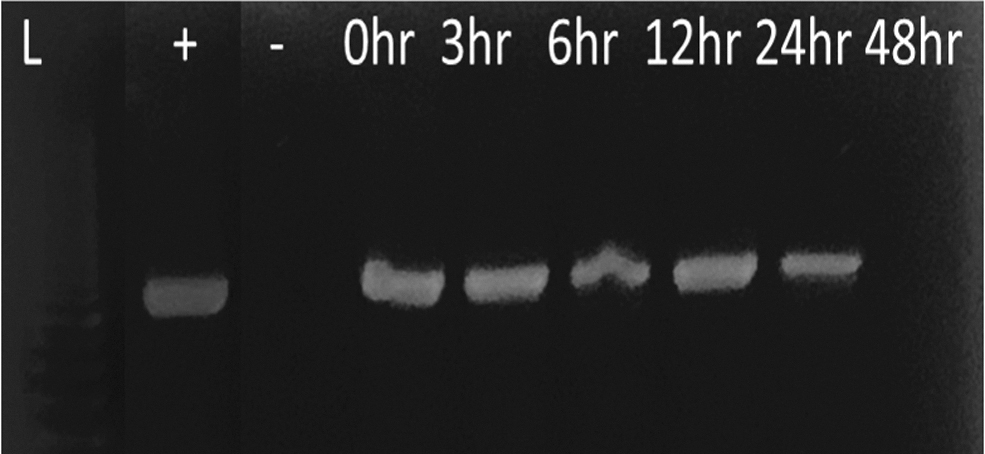
Detection of Chlamydia trachomatis on the body of Musca domestica. PCR amplification of material from rinsed outer body of the housefly (M. domestica) after being submerged in solution containing chlamydia were evaluated at 0, 3, 6, 12, 24, and 48 h) postimmersion. Chlamydia DNA was amplified as detailed in the Materials and Methods section and the PCR products were run on a 20% agarose gel and stained with ethidium bromide. Detected on the body for up to 24 hr.
Musca domestica harbors C. trachomatis in the gut and crop
The primary objective of this study was to determine whether synanthropic flies are purely mechanical vectors in trachoma transmission, or if they have biological roles. To accomplish this, we first fed flies a specific IFU of C. trachomatis HAR-36B or LGV in SPG, through syringe feeding protocol (Fig. 2A) as outlined in the Materials and Methods section. Flies were dissected and the gut and crop carefully removed under a dissecting microscope. DNA was isolated and PCR amplification performed to determine the presence of the organisms at different time points (0, 6, 12, 24, and 48 h postfeeding). Chlamydia trachomatis DNA was amplified from materials obtained from inside the crop, up to 24 h postfeeding and the gut up to 48 h postfeeding (Fig. 3). There were no differences in the presence of C. trachomatis DNA for the two strains used (data not shown). The negative control consisted of materials combined from 10 digestive tracts that were dissected from mock fed female flies.

Fly feeding positions and regurgitation. This image shows M. domestica in specific experimental positions.
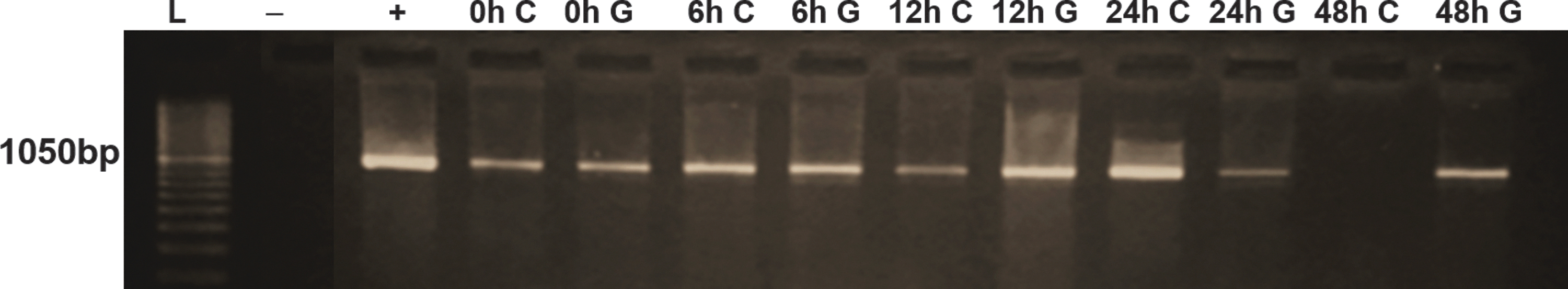
The presence of chlamydial DNA in the crop and gut of houseflies. PCR amplification of material isolated from the crop (C) and gut (G) of female houseflies (M. domestica) fed with C. trachomatis LGV strain. Chlamydia DNA was amplified up to 24 h postfeeding in the gut and 48 h in the crop. We utilized a 100 bp ladder (L) and materials from mock-infected flies (a total of 10 flies) were used as a negative control (−). The positive control DNA (+) was isolated from the same batch of C. trachomatis inoculum used to feed the flies and the products produced a band at 1050 bp. LGV, lymphogranuloma venerum. Color images are available online.
Viability of C. trachomatis isolated from fly gut
With the confirmation that C. trachomatis DNA was present in the gut of houseflies at various times postfeeding, we assessed whether these organisms were still alive and viable. Musca domestica flies were fed viable C. trachomatis EBs and the entire foregut and hindgut removed either immediately after feeding, at 6 h postfeeding, or 24 h postfeeding. The gut samples were homogenized in SPG buffer that keeps EBs viable and used to infect confluent HFF cells on coverslips for ∼36–48 h. The coverslips were then rinsed, fixed, and immunostained, using the Pathfinder C. trachomatis antibody as described in the Materials and Methods section. Figure 4 displays an example of the immunofluorescent images from three different time points (0, 6, and 24 h) using the HAR-36B serovar. Please note that we obtained similar results for LGV (data not shown). The results show that there were several inclusions (shown in green) of chlamydia in infected HFF cells (shown in red), confirming that the ingested chlamydial EBs were indeed viable, retaining their ability to infect human fibroblast cells. Further experiments confirmed that they also infected human epithelial cells, with similar numbers and sizes of inclusions (data not shown).
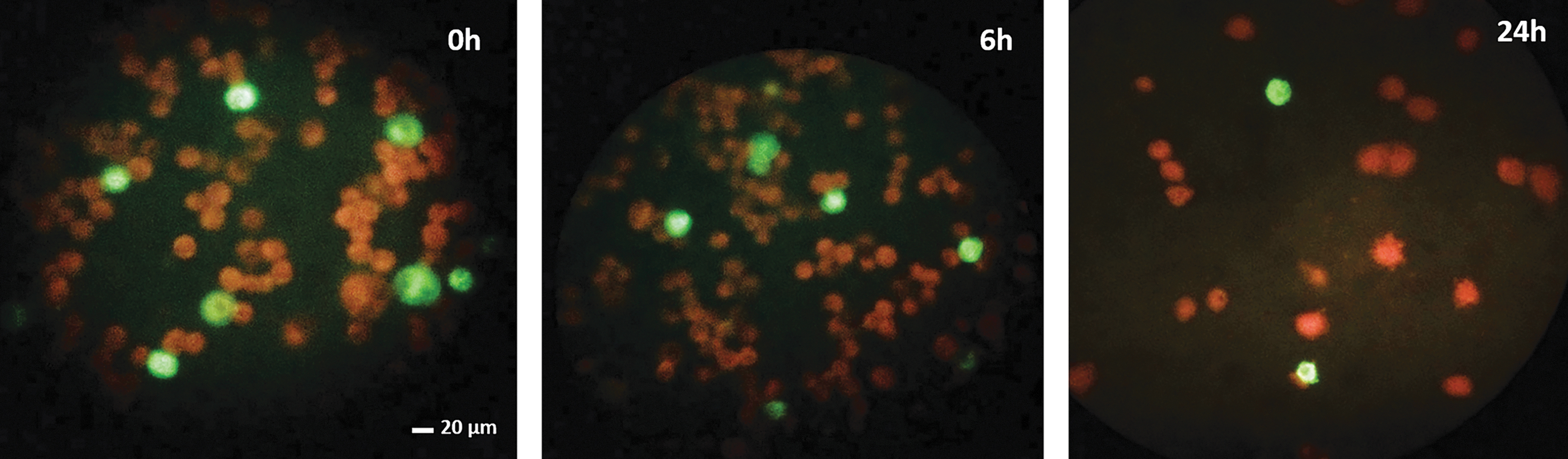
Infectious C. trachomatis from fly gut. Flies were fed C. trachomatis and their entire foregut and hindgut harvested at different time points. The homogenate from fly gut was then used to infect cells in tissue culture and stained with FITC-conjugated anti-chlamydia antibody. This representative image shows C. trachomatis HAR-36B inclusions (green) in samples collected at 0, 6, and 24 h postfeeding. FITC, fluorescein isothiocyanate. Color images are available online.
Presence of chlamydia in fly regurgitation and bubbling
In trachoma-infected areas, face-seeking flies can be seen feeding on the mucus and discharge on human eyes. It has been reported that trachoma infection spreads through personal contact, contaminated clothing, or by flies that have been in contact with discharge from the eyes or nose of an infected person. Domestic, face-seeking flies have therefore been implicated as mechanical vectors, by picking up a pathogen and transferring it to an uninfected host. Recent studies have shown that flies from trachoma-positive households and the faces of children with trachoma test positive for C. trachomatis DNA by PCR (Emerson et al. 2004, Last et al. 2020). However, no published report has examined the ability of face-seeking flies to internalize chlamydia and later transmit it to a susceptible host in the process of feeding and elimination. Musca domestica often regurgitates and defecates during feeding events (Pava-Ripoll et al. 2012). These elimination events suggest that flies could be an important vector in the transmission of important pathogens to humans and their domestic animals. These elimination events are being actively investigated, as they could result in pathogen transfer. We therefore set up regurgitation and defecation experiments as outlined in the Materials and Methods section and collected samples for PCR and culture. Defecation experiments demonstrated that chlamydia DNA, amplified using PCR detection, can be found on spots scraped from containers that had chlamydia-fed male and female flies. Regurgitation material was collected when bubbling on the proboscis of fed flies were observed (Fig. 2B). The liquid was collected with a pipette and examined through PCR amplification and tissue culture. The data show that fly regurgitation and defecation can harbor chlamydia DNA after ingesting a meal that contains the bacteria (Table 1). The positive and negative controls were the same controls used in the time point experiments described previously. Figure 5 shows an example of the cultivation of infectious C. trachomatis from the regurgitated material at 0, 6, and 12 h postfeeding, confirming that when M. domestica regurgitate their meals sporadically, they evacuate viable microbes, making them a good vector for transmission of a disease like trachoma upon subsequent feeding. There was no difference in the ability to detect C. trachomatis DNA in male and female flies, although female flies appeared to ingest 1–2 μL more of material.
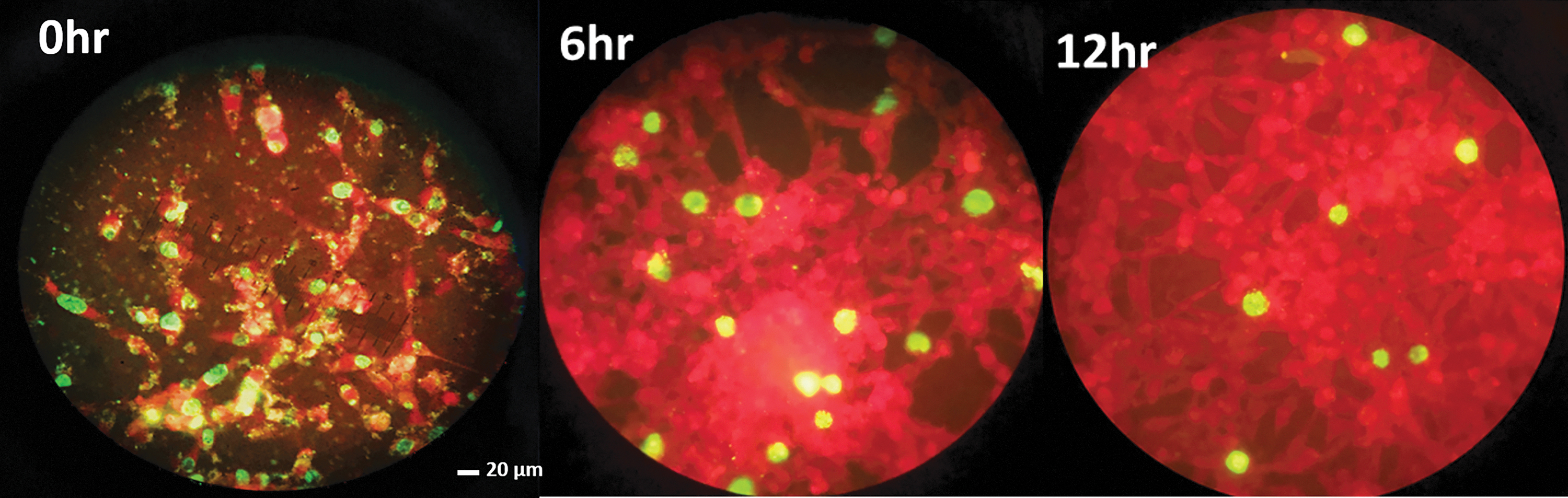
Infectious C. trachomatis from the regurgitation of the housefly. Bubbling seen during dissection of the gut and crop at different time points was collected and used to infect HFF cells and stained with FITC-conjugated anti-chlamydia antibody. Inclusions here are seen at the 0, 6, and 12 h time points. This representative image used the LGV strain of C. trachomatis. HFF, human foreskin fibroblast. Color images are available online.
Flies Were Fed with ∼3 μl of Sucrose–Phosphate–Glutamic Acid Solution Containing Chlamydia trachomatis
Samples were collected from the crop, gut, and evacuated materials (regurgitation and feces) immediately (0 h), at 3, 6, 12, 24, and 48 h from Musca domestica female flies (cohorts of five flies each). Materials were PCR amplified to determine the presence of chlamydial DNA in each body region or evacuated material. The data confirm the presence of the bacteria in the crop up to 24 h and the gut up to 48 h. Regurgitated materials at each time point contained chlamydia DNA, whereas fecal materials only contained chlamydial DNA at the 3- and 6-h time points.
Amount of EBs needed for detection from the crop and gut of houseflies
Our data confirming the presence of C. trachomatis in the crop, gut, and evacuated materials, strongly suggests that M. domestica is more than just a mechanical vector, transmitting trachoma through material on its outer body surface. We hypothesized that houseflies could serve as biological vectors of this serious infection by ingesting and then regurgitating viable microbes while on a second host. A single chlamydial inclusion can contain between 300 and 1000 EBs capable of infecting new cells (Peeling 2012). We used a standard stock solution of C. trachomatis (LGV and HAR36B) and performed serial dilutions before capillary feeding ∼2 μL of the respective diluted solution to different groups of five houseflies each. Samples were collected from the gut and crop, followed by PCR analysis to determine the presence of chlamydial DNA. Table 2 shows the cumulative data, confirming that our PCR analysis was capable of detecting as few as eight EBs from both crop and gut samples.
Chlamydia Was Serially Diluted and Fed to Housefly
The crop and digestive tract (gut) were dissected and the contents used to perform PCR amplification. These findings indicate that chlamydia can be detected in up to 1:100 dilution or ∼8 EBs of the starting material (4.1 × 106 EBs/mL, with each fly capillary fed with 2 μL) in the crop and gut.
EBs, elementary bodies.
How long can viable C. trachomatis be harbored in the crop of M. domestica?
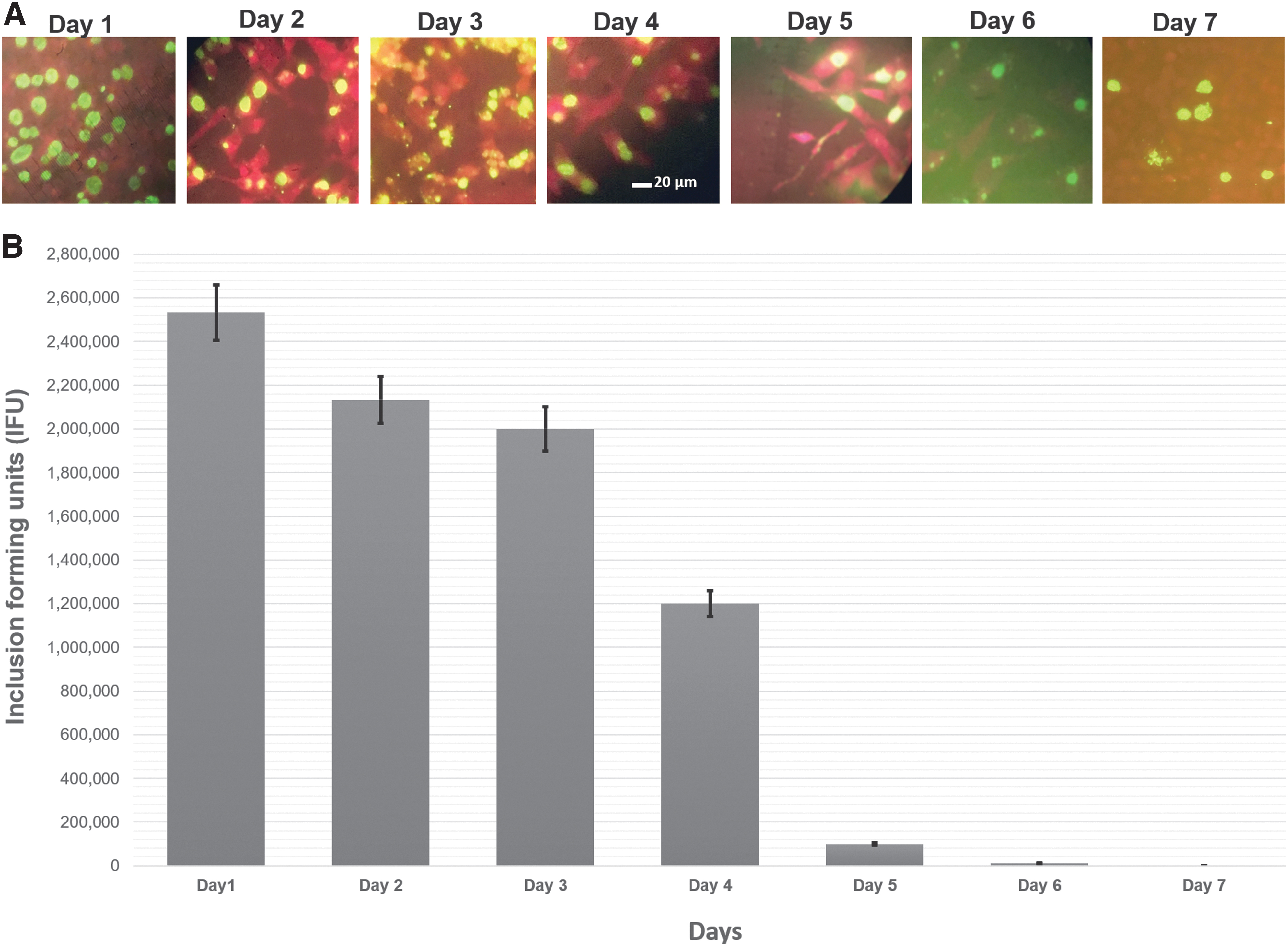
The crops of M. sorbens and other muscids are innervated and spontaneously contract to seal the organ when removed and placed in solution (Stoffolano and Haselton 2013). In an attempt to determine if C. trachomatis can survive in the crop for extended times, flies were fed in groups of five and the crops removed intact and stored in sterile Eppendorf tubes for up to 7 days at room temperature. Crop material was removed each day and frozen at −80°C until tested by PCR and culture for the presence of C. trachomatis. Our data show that chlamydial DNA was present in the crop material all 7 days (Fig. 6). Material from these ex vivo crops was also stored for culture as outlined in the Materials and Methods section. After infection for ∼36 h, coverslips were immunostained and immunofluorescence data confirmed that viable chlamydia was present all 7 days (Fig. 7A). There was, however, a reduction in IFUs after the fifth day (Fig. 7B). We found no significant differences in survival or carriage of C. trachomatis LGV biovar or the HAR-36B serovar associated with insect sex in these experiments (data not shown).
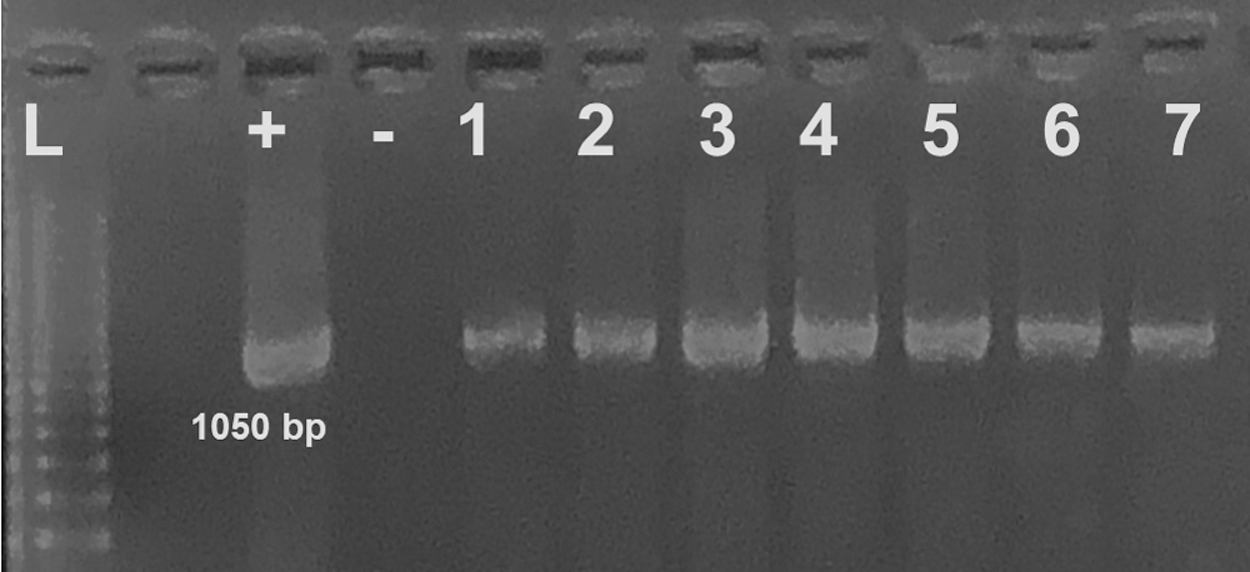
PCR amplification of chlamydia from ex vivo crops. The isolated crops of M. domestica spontaneously contracts when isolated and placed in a solution containing Ca2+. Flies were fed with C. trachomatis in SPG as previously described and the crops isolated and placed in sterile microtubes at room temperature for 1–7 days. At each time point, the crop was broken with a needle and material removed for PCR. The data show that chlamydial DNA was present in the crop material all 7 days of our analysis period.
Discussion
Should M. domestica be considered a bioenhanced vector? In this study, C. trachomatis was found separately in the crops, regurgitant, and guts of houseflies, 24 h postfeeding, whereas bacteria were isolated from feces in flies 6 h after feeding. In another experiment, chlamydia were detected 48 h after feeding in the gut. In this experiment, no chlamydial organisms were isolated in the feces or crop after 48 h postfeeding. Based on our findings, there is evidence that M. domestica is not just a mechanical vector, but may also serve as a bioenhanced transmission vector. If a fly feeds on the eye of an infected individual and goes directly to another individual, it could transmit the pathogen, either mechanically or regurgitate its crop contents, which contains the pathogen. If, however, the fly is unable to find another susceptible host's eye to feed on, or it is replete, it can still (i.e., 24 h later) regurgitate the pathogen from its crop to a susceptible host. Therefore, the importance of the crop in chlamydia transmission should not be underestimated. Previous studies reviewed by Stoffolano (2019) have shown that for E. coli, the crop was the major site of horizontal transmission of resistance; and, although West et al. (2014) found no resistance of chlamydia to azithromycin, most believe it is just a matter of time before it occurs. We therefore propose a revised model of trachoma transmission to involve the biological role played by synanthropic flies.
In this study, we used both male and female flies in several experiments and also separated sexes to see if there were differences in experimental outcomes. We observed no differences in bacterial uptake or in patterns of recovery from regurgitation, defecation, or survival in the gut or crop. The female houseflies, however, ingested an average of 1–2 μL more of the infected inoculum during feeding.
There is no doubt that as global warming continues, face-seeking flies in dry areas will only increase their frequency in an attempt to access the eyes of humans (i.e., especially children) or domestic animals, as a source of water. The report by Ramesh et al. (2013) concluded their study by stating, “Current evidence on the potential role of climate on trachoma distribution is limited, despite a wealth of anecdotal evidence.” At the same time, war, global unrest, and increased migration of individuals from endemic areas (based on 2019 data this represents ∼142 million people), raises concern that individuals having the pathogen, without proper screening, can easily gain entrance to nonendemic areas (West et al. 2015).
Health care workers and researchers concerned with emerging infectious diseases often dismiss the importance of zoonoses when determining epidemiological factors for ocular diseases. Examining various Chlamydiaceae species, Dean et al. (2013) reported that several domestic animals could be a source for C. trachomatis and possibly other new and emerging infections to humans of other chlamydia species. Thus, these animals, which often share close proximity to humans, could serve as new sources of infections and also involve new vector species of flies, other than just M. sorbens and M. domestica.
Since the 1598 report of Baron Harant de Poljits (Duke-Elder 1965), investigations have shown that both M. sorbens and M. domestica are involved in short-term vectoring of the C. trachomatis pathogen. Most reports suggest they do this through mechanical transmission, which mainly involves the legs or proboscis (Graczyk et al. 2001, Emerson et al. 2002, Reilly et al. 2007). Regurgitation has been suggested as one of the major routes of pathogen transfer to the eye of a vertebrate host, as also shown by Mandragora autumnalis, transmitting Moraxella bovis to the eyes of cattle (Glass and Gerhardt 1983). Focus should therefore be on the fly crop as a major site, housing numerous pathogens and its involvement in the process of regurgitation, which empties the crop.
The major objective of the 2004 field study of Miller et al. (2004) was to demonstrate that M. sorbens, when collected from the eyes of trachoma victims, contained pathogen DNA, using PCR. Forsey and Darougar, using adult M. domestica in the laboratory, found C. trachomatis in the intestines up to 6 h and up to 2 h from legs and/or proboscises (Forsey and Darougar 1981). Unfortunately, they did not separate the crop from the rest of the intestine. The identification method used by Darougar et al. (1970) for chlamydial presence was based on the McCoy cell culture technique and probably lacked the sensitivity of the work presented in this study. They also showed, using a guinea pig model, that the fly was able to transmit infection from one animal to another. Zardi (1964), using M. domestica, suggested that the pathogen might be transmitted through feces. Based on feeding and defecation observations, it was reported that lengthy touches of the labellum to food usually resulted in defecation (El-Bassiony et al. 2016). Even so, it seems highly unlikely that the fecal mode of transmission is highly successful. Unless the individual rubs the feces into their eyes, the probability of pathogen transfer appears to be very low. Zardi (1964) also reported that he could retrieve the pathogen between 4 and 15 h after feeding them and these timeframes are in line with our study. A major shortcoming of this study is that we did not perform RT-qPCR on samples from the crop and gut. This could prove much more sensitive, while also confirming viability of the microbe. We instead used a culture technique that while highly accurate in confirming the presence of viable C. trachomatis, is less sensitive then molecular genetics techniques.
Conclusions
Our data suggest that eye-seeking flies such as M. domestica can ingest C. trachomatis during regular feeding and the intake goes mainly to the crop. These data also confirm, for the first time, that ingested chlamydia remains viable inside the flies for 24–48 h postfeeding. Because the housefly is known to be an exceptional regurgitator (El-Bassiony et al. 2016), there is reason to believe that while feeding on an uninfected host, adult flies could regurgitate and transmit the bacteria to a new host, more than 24 h after feeding. Large-scale studies examining the effect of sustainable pesticide control measures are necessary to confirm this new biological role that domestic flies could be playing in trachoma transmission. Fly control will no doubt play a significant role in the WHO's strategy to eliminate blinding trachoma by 2030. We believe that these findings reveal the opportunity for efficient intervention strategies through vector control. In addition, the mechanism(s) involved in fly regurgitation should be fully elucidated.
Footnotes
Authors' Contributions
N.B., M.S.M., M.Z., N.M., and R.V. performed research work that contributed to this article. N.B. and M.M. completed the majority of work and created figures for the article. W.C.W. and J.G.S. conceived of the project and provided overall supervision and support. All authors helped to draft, edit and approve the final version of the article.
Disclaimer
The contents are solely the responsibility of the authors and do not necessarily represent the official views of the USDA or NIFA.
Author Disclosure Statement
No conflicting financial interests exist.
Funding Information
The major part of this work was supported by funding from the National Institute of Food and Agriculture, U.S. Department of Agriculture, the Massachusetts Agricultural Experiment Station and the Department of Microbiology, under HATCH project MAS00400 to W.C.W. The work was also partially supported by USDA/NIFA support to the Stockbridge School of Agriculture at the University of Massachusetts, Amherst, under Hatch project MAS00527 to J.G.S.
