Abstract
Background:
Eastern equine encephalitis virus (EEEV) is a mosquito-borne virus that is primarily found in North America and the Caribbean. Over the past decade there has been an increase in virus activity, including large outbreaks in human and horse populations. Predicted climate change is expected to affect the range of mosquitoes including vectors of EEEV, which may alter disease risk posing a public health concern.
Methods:
A scoping review (ScR) was conducted to identify and characterize the global evidence on EEEV. A thorough search was conducted in relevant bibliographic databases and government websites. Two reviewers screened titles and abstracts for relevance and the characteristics of relevant articles were extracted using a uniformly implemented data collection form. The study protocol was developed a priori and described the methods and tools used and this article follows the PRISMA-ScR guidelines for reporting ScRs.
Results:
The ScR included 718 relevant research articles. The majority of the articles originated from North America (97%) between 1933 and 2019. EEEV has been identified in 35 species of mosquitoes, over 200 species of birds, various domestic animals, wild mammals, reptiles, and amphibians. Articles identified in this ScR primarily covered three topic areas: epidemiology of hosts and vectors (344 articles) including surveillance results (138), pathogenesis of EEEV in hosts (193), and in vitro studies characterizing EEEV (111). Fewer articles evaluated the accuracy of diagnostic tests (63), the efficacy of mitigation strategies (62), transmission dynamics (56), treatment of EEEV in hosts (10), societal knowledge, attitudes, and perceptions (4), and economic burden (2).
Conclusion:
With the projected impact of climate change on mosquito populations, it is expected that the risk of EEEV could change resulting in higher disease burden or spread into previously unaffected areas. Future research efforts should focus on closing some of the important knowledge gaps identified in this ScR.
Introduction
Eastern equine encephalitis virus (EEEV) is a mosquito-borne virus belonging to the Alphavirus genus, Togaviridae family and is closely related to Western equine encephalitis virus and Venezuelan equine encephalitis virus. EEEV was first recorded in North American horses in 1831. In 1938, the first human case was identified in Massachusetts, United States, during an Eastern equine encephalitis (EEE) outbreak involving 38 human and 248 horse cases. EEEV is considered an endemic virus in North America (mainly Atlantic/Gulf Coast States and Great Lake Regions), and has been reported on islands in the Caribbean. Historically there have been four EEEV lineages: Group I is responsible for most human cases and includes EEEV strains from North America and the Caribbean, whereas Groups II, III, and IV primarily cause illness in horses and include EEEV strains from Central and South America (Brault et al. 1999). Due to genetic divergence and differences in ecology and pathogenesis, strains from lineages II–IV were reclassified in 2010 as a distinct virus now known as Madariaga virus (MADV) (Arrigo et al. 2010). Although EEEV and MADV are distinct species, recent phylogenetic analyses show that there is overlap in their geographic regions (Hoyos et al. 2015, Burgueno et al. 2018).
EEEV is transmitted through the bite of an infected mosquito and most spillover transmission occurs in low-lying areas with hardwood trees and swamps conducive of mosquito larvae development. Culiseta melanura is the primary vector transmitting EEEV, which feeds mainly on birds (Molaei et al. 2015). Thus, C. melanura is considered to circulate EEEV among wild birds in an enzootic cycle. Bridge vectors transmit the virus from viremic birds to other susceptible hosts outside the enzootic cycle (Armstrong and Andreadis 2010). Susceptible hosts are usually dead-end hosts, such as equine species (horses, zebras, donkeys, etc.), humans, other birds (emus, ostriches, owls, etc.), or other livestock species (e.g., swine and cattle).
The neuroinvasive disease associated with severe cases of EEEV makes it one of North America's most severe arboviral encephalitidies. Over the past decade there has been an increase in virus activity, including a large outbreak in the summer of 2019 (ProMED 2019). Environmental changes associated with climate change may alter disease risk by changing the abundance and distribution of the primary mosquito vector, by lengthening the virus-transmission season, and by extending the transmission range to areas previously unsuitable (Ng et al. 2017, Khan et al. 2020).
Vector-borne diseases likely to emerge or expand in range due to climate change are a public health concern. A scoping review (ScR) was conducted to collect and characterize the existing global evidence on EEEV and identify knowledge gaps that may assist in decision-making on this public health issue.
Methods
Review topic and scope
An ScR uses reproducible and rigorous methodology to identify and map the scope and volume of literature available on a topic (Peters et al. 2015, 2017, Tricco et al. 2016, Munn et al. 2018). In addition to synthesizing the knowledge on a particular topic, an ScR can also identify areas of research saturation or gaps to ultimately guide future research questions and decision-making (Peters et al. 2015, 2017, Tricco et al. 2016, Munn et al. 2018). This ScR adheres to the Joanna Briggs Institute methodology for ScRs, which is similar to the structure and rigor of Cochrane Collaboration systematic reviews (Higgins et al. 2020, Peters et al. 2015, 2017).
The objective of this ScR was to summarize the characteristics of the global evidence on EEEV. For the purpose of this ScR, EEEV is defined as genotype I, commonly occurring in North America and the Caribbean. The other EEEV genotypes (II, III, and IV), have been reclassified as MADV and are outside the scope of the review. All articles that reported on observational studies outside of North America and the Caribbean were excluded.
Review question and inclusion criteria
The research question for this ScR was “What are the characteristics of the global evidence on EEEV?” The following inclusion criteria was applied: Publication date: All Country: All (Studies from any country were eligible as long as the topic was EEEV—Group I) Language: English and French Document type: All primary research (e.g., journal articles, government reports, predictive models using primary research, theses describing primary research). All other documents were excluded (secondary research, literature reviews, books, etc.) Agent/disease: EEEV—Group I Study design: All
Review protocol and team
An ScR protocol was developed a priori to ensure the methods were transparent, consistent, and reproducible (Supplementary Document S1). The protocol includes the search strategy and algorithm, inclusion and exclusion criteria for relevance screening, and the data characterization form for data extraction. A multidisciplinary team with expertise in knowledge synthesis, epidemiology, vector-borne diseases, and public health created the protocol and conducted the ScR. The reporting of this ScR follows the guidelines of PRISMA-ScR (Tricco et al. 2018) (Supplementary Document S2). A critical appraisal of the evidence was not conducted in this ScR.
Search strategy and verification
The search algorithm (“eastern equine encephalitis” OR “eastern equine encephalomyelitis”) OR (EEE and virus) was applied to five databases that were appropriate to the ScR: Scopus, PubMed/MEDLINE, Embase, Cochrane Central Register of Controlled Trials, and ProQuest. The Cochrane library was searched for any relevant trials in the trial registry and ProQuest was used to search for relevant theses and dissertations. The initial search was conducted in May 2017 and was updated in May 2019.
A complementary search for gray literature was conducted by hand searching state health department websites for published primary reports, news bulletins, and surveillance reports that reported EEEV cases that were not captured by the original electronic search. The Centers for Disease Control and Prevention (CDC) Stacks, Public Health Agency of Canada, Caribbean Public Health Agency (CARPHA), Public Health Ontario, and Institute National De santé Publique Du Québec were also searched for reports on cases of EEEV (confirmed and probable). In addition, each United States state website that had a confirmed EEEV case in any reservoir, vector, or incidental host was searched for annual communicable disease reports. The gray literature search was conducted in May 2017 and was updated in May 2019.
To confirm that all relevant primary research was found during the search, the reference lists of 12 literature reviews/key articles focused on EEEV identified by the ScR team were hand searched for additional articles (Sellers 1989, Freier 1993, Calisher 1994, Deresiewicz et al. 1997, Olsen et al. 1997, Armstrong and Andreadis 2010, 2013, Zacks and Paessler 2010, Silverman et al. 2013, Arechiga-Ceballos and Aguilar-Setien 2015, Kumar and Patil 2017, Chapman et al. 2018). At the point of saturation, when no new references were identified, the ScR team stopped evaluating reference lists. Eleven references that had been omitted by the electronic and gray literature search were added to the ScR from the search verification process.
Review management
All potentially relevant citations that were found by the search strategy were imported into the reference management software EndNote (EndNote X7, Clarivate Analytics) and duplicate citations were removed. The unique citations were then imported into the web-based systematic review management software DistillerSR (Evidence Partners, Ottawa, Canada) where additional duplicates were removed. All subsequent stages of this ScR, including relevance screening and data characterization, were performed within this software.
Relevance screening and data characterization
A relevance screening form was developed a priori to incorporate the inclusion and exclusion criteria of the ScR (Supplementary Document S1). Two reviewers independently used this form to screen citation titles, abstracts, and keywords for relevance to the ScR topic. Due to limited translation resources, citations in languages other than English and French were excluded. Full-text articles were then procured and independently reviewed by two individuals using a data characterization form developed a priori (Supplementary Document S1). The data characterization form aimed to categorize the research on EEEV to understand where there are areas of knowledge saturation and gaps. Each article was categorized into one or more research focus areas: epidemiology, surveillance, pathogenesis, virus study, diagnostic tests, mitigation, treatment, societal knowledge, and economic analysis. Additional pertinent information from each relevant article such as study design, population, and key outcomes were then extracted. Definitions for what was included in each focus area, study designs, and information extracted are outlined in detail in the protocol (Supplementary Document S1). During both stages, reviewers resolved conflicts by consensus or a third party reviewer where necessary.
Data analysis
The final dataset was exported into MS Excel (Excel 2010; Microsoft Corporation, Redmond, WA), cleaned, and tabulated for use in the descriptive analysis and summarization of results. A repository of relevant articles including bibliographic details and the full dataset from the completed ScR are available in Supplementary Dataset S3.
Results
ScR descriptive statistics
There were 1330 unique citations screened for relevance, of which 971 were potentially relevant and full articles were procured. Articles in languages other than English and French were excluded from this ScR due to a lack in translational resources. The articles excluded because of language were in Russian (n = 36), German (12), Spanish (9), Polish (4), and Chinese (1). Since EEEV typically occurs in North America and the Caribbean, we believe there is a low potential for language bias in this ScR. Data characterization was conducted for 718 relevant articles included in this ScR (Fig. 1).
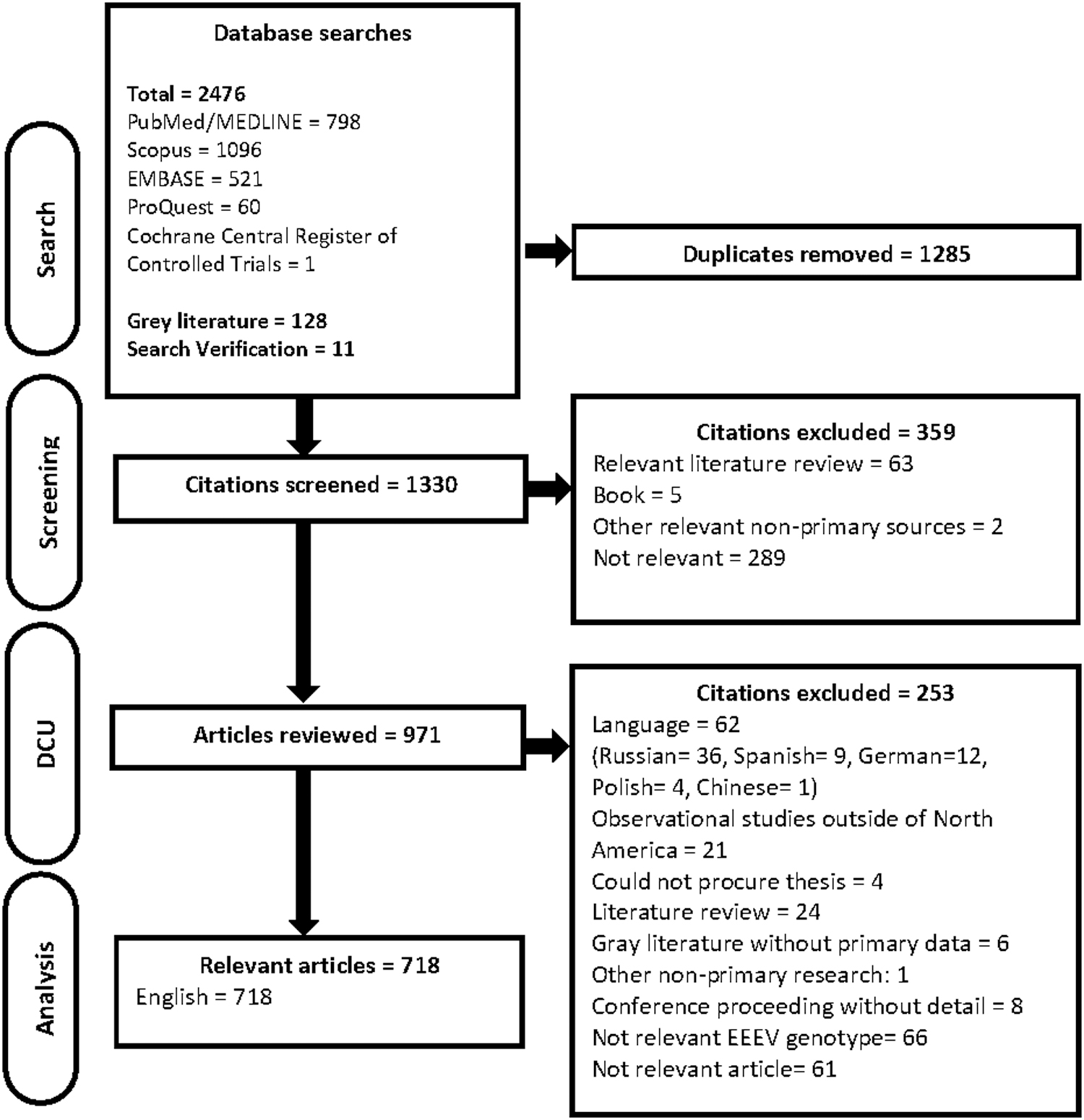
PRISMA flow diagram of articles through the scoping review process. DC, data characterization.
Articles were published between 1933 and 2019 with 28.4% (204/718) published since 2011. The majority of the articles originated from North America (696/718) and were primary research (580/718) (Table 1). North America includes the United States, Canada, Mexico, and the Caribbean.
General Characteristics of 718 Included Primary Research Publications on Eastern Equine Encephalitis Virus
Total number sums to >718 as some studies were conducted on more than one continent, had more than one type of study design, or sampled more than one population
Includes arthropod and mosquito species.
Other includes: experimental methods for cultivating and purifying EEEV.
Other includes: investigating a system for deriving EEEV antigens from infected chick embryos and content analysis (mixed methods).
EEEV, eastern equine encephalitis virus.
Articles primarily covered three EEEV topic areas as shown in Fig. 2: epidemiology of hosts and vectors (344/718) including surveillance (138/718), pathogenesis of EEEV in hosts (193/718), and in vitro studies characterizing EEEV (111/718) (Fig. 2). Other EEEV topics such as the accuracy of diagnostic testing (63/718), efficacy of mitigation strategies (62/718), EEEV transmission dynamics (56/718), treatment of EEEV in hosts (10/718), societal knowledge, attitudes, and perceptions (4/718), and economic burden (2/718) were captured to a lesser extent. No predictive models on the impact of climate change on EEEV were identified. Some articles did not fit into the predetermined topic areas (5/718) and included articles on studies analyzing experimental methods for cultivating and purifying EEEV and the precipitating effect of methanol on EEEV. Observational study designs were the most widely used (415/718), of which the majority were estimates of the burden of EEEV in vectors or hosts through surveillance activities (194/718), provided prevalence estimates in a defined population (84/718), or presented cases series or reports (88/718).
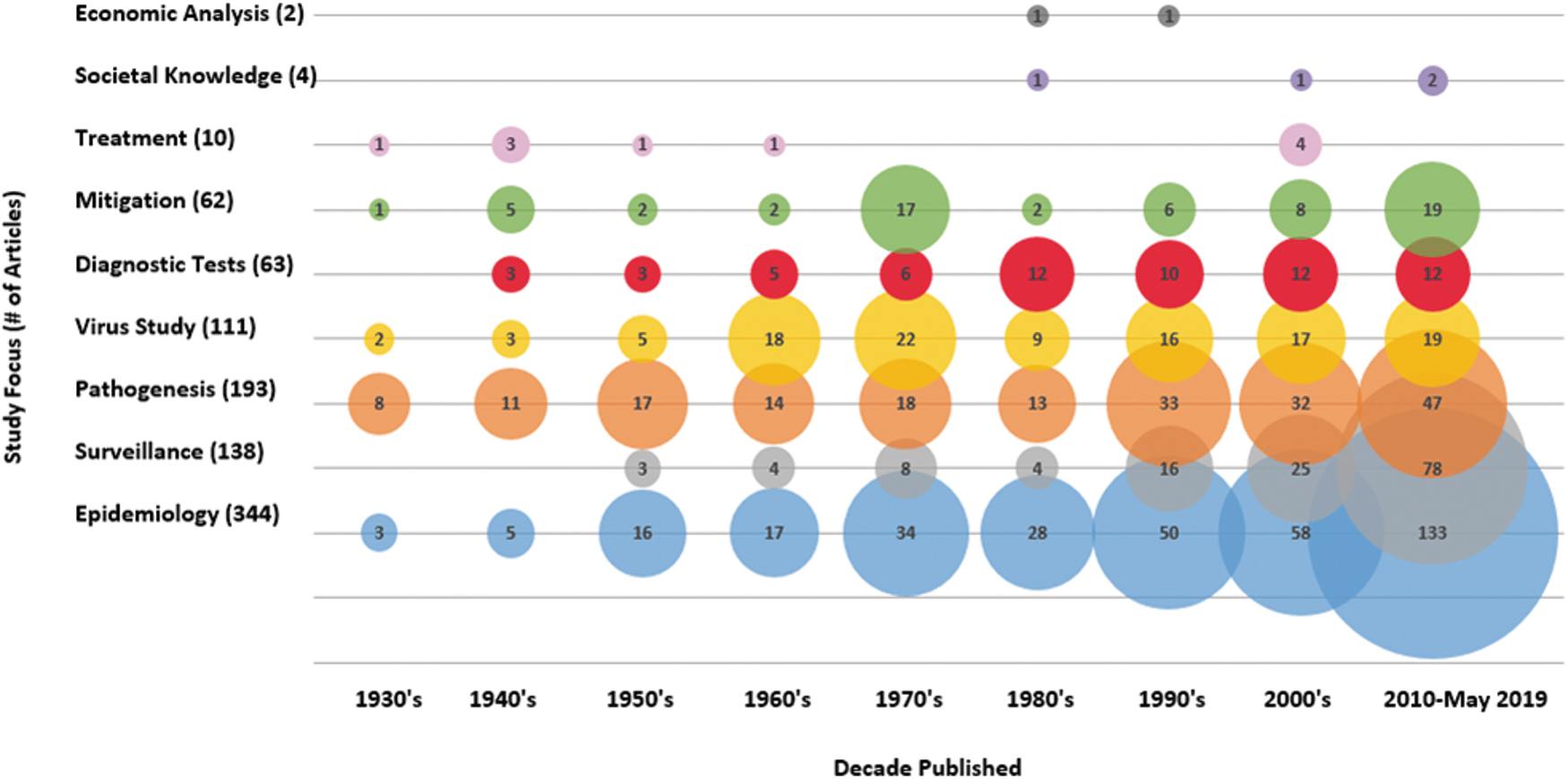
Bubble chart of the number of different article foci published in the years 1933–2019.
EEE virus
In vitro methods were used (134/719 studies) to characterize the virus, evaluate the impact of an intervention, or described methods for cultivating EEEV. Studies that focused specifically on the virus were reported in 111 of 134 articles. EEEV has a single-stranded, positive sense RNA genome; the virus is spherical in shape with a diameter of 60–65 nm (Aguilar et al. 2007). Many of the in vitro studies focused on the structure and function of selected part of the virus. Categorization of these articles included those that reported on the pathogenic attributes of the virus (68/111), transmission characteristics (3/111), and the molecular characterization of EEEV (51/111). Of the articles that reported molecular characterization of EEEV, there was a phylogenetic analysis in approximately half of the studies: 8 of 51 were molecular epidemiology studies, 17 of 51 reported phylogenetic trees, and 6 of 51 reported whole genome sequencing results.
Other research conducted in vitro reported on the effectiveness of antiviral treatments under experimental conditions in 5 of 134 articles (Katz et al. 1975, Amaya et al. 2015, Rico 2016, Lundberg et al. 2018, Jonsson et al. 2019). Possible mitigation options evaluated in vitro (12/134 articles) examined possible formulas for vaccination (7/12) and the use of biological and chemical agents as virucides (5/12). Four of these articles focused on laboratory methods for working with EEEV. These included articles on experimental methods for cultivating and purifying EEEV (Palmer et al. 1968), deriving EEEV antigens from chick embryos (Soret and Sanders 1954), the precipitation effect of methanol on EEEV (Pollard et al. 1949), and evaluation methods to screen EEEV for purity and contamination (Gollapudi et al. 2017).
Accuracy of diagnostic tests
The accuracy of diagnostic tests to diagnose EEEV was studied in 63 of 718 articles. Several techniques to isolate and/or identify EEEV or identify exposure to EEEV were evaluated including virus isolation (16/63), molecular tests (22/63), immunoassays (41/63), and clinical diagnosis (1/63). The performance of these tests were evaluated in different types of hosts including humans (10/63), naturally exposed animals (20/63), experimentally infected animals (14/63), mosquitoes (16/63), and well-characterized sample libraries (23/63). Information regarding sensitivity, specificity, test agreement, or raw data were provided in 26 of 63 of these articles. The citations for each of these categories can be identified in Supplementary Dataset S3.
EEEV in vectors
Research on vectors of EEEV was reported in 209 of 718 articles. While the majority of these articles focused on epidemiology (155/209), there was also research on EEEV transmission dynamics (43/209) and mitigation strategies (3/209).
Over 150 mosquito and arthropod species have been tested for naturally occurring exposure or infection with EEEV in 155 of 209 articles. Arthropod species included midges, mites, fleas, flies (sand flies, horse flies, deer flies, stable flies, louse flies, and house flies), bedbugs, mealworms, lice, and ticks. Vector populations were frequently trapped, identified, and tested for arboviruses as part of prescribed surveillance activities. Evidence of EEEV in wild mosquito and arthropod species was reported in 127 of 155 articles, mostly from 24 states in the United States (122/127) and to a lesser extent in the Caribbean (3/127) and Canada (3/127) (Table 2).
Mosquitoes and Arthropod Vectors Naturally Infected with Eastern Equine Encephalitis Virus Reported in 127 Articles Across North America (Including the Caribbean) Between 1947 and 2018
Mosquito species that have been used in transmission and/or competence studies. Other species including Aedes aegypti, Aedes atropalpus, and Culex taeniopus have been used in transmission and/or competence studies, but there have been no articles reporting natural infection of EEEV in these mosquitoes.
Between 1948 and 2018, 35 specific mosquito species tested positive for EEEV (Table 2). EEEV was most commonly identified in C. melanura (74/127), Coquillettidia perturbans (29/127), Aedes canadensis (18/127), Aedes vexans (15/127), and Culiseta morsitans (14/127). The minimum infection rate was reported in 22/127 articles. In addition to mosquitoes, arthropod species (Culicoides, Dermanyssus gallinae, and Menacanthus stramineus) tested positive for EEEV in two articles from the United States between 1947 and 1959 (Table 2). A detailed list of vectors naturally infected with EEEV along with the location and dates sampled is given in Supplementary Table S1.
Risk factors that were associated with exposure to EEEV or acquiring EEEV in mosquito populations were reported in 5 of 209 articles (Crans et al. 1994, Takeda et al. 2003, Hachiya et al. 2007, Kelen et al. 2012, Skaff et al. 2017). In vectors, the method of identifying EEEV was described in 92 of 155 articles, the most common diagnostic method used was virus isolation (60/92) followed by molecular tests (37/92), or immunoassays (37/92). Many of these articles (40/92) used multiple diagnostic methods to identify EEEV in vectors.
Three articles investigated mitigation strategies to prevent EEEV infection in mosquitoes. Aerial spraying with 95% malathion was conducted in a study from Massachusetts; however, the effectiveness of this intervention was not evaluated (Grady et al. 1978). Mosquitoes were used in two studies on developing EEEV vaccine candidates. To assess the replication competence of a recombinant EEEV that was attenuated, Aedes albopictus mosquitoes were challenged with the virus (Pandya et al. 2012). This ensured the virus would only replicate in vertebrate cells, which is an important safety feature for live vaccines against mosquito-borne viruses (Pandya et al. 2012). A second study demonstrated that two chimeric vaccine candidates for EEEV were successful in reducing mosquito infectivity when tested on Aedes sollicitans and Aedes taeniorhynchus mosquitoes (Arrigo 2010).
Over 30 mosquito and arthropod species were used to study transmission dynamics and vector suitability (competence and behavior) in 44 of 209 articles. Mosquito transmission dynamics were reported in 31 of 44 studies and specific species are listed in Table 2. Transmission studies reported on vector-to-host transmission (13/31), host-to-vector transmission (24/31), and transovarial transmission (5/31). Mosquito competence, such as viral replication, dissemination, and transmission was investigated in 29 of 30 articles, whereas 2 of 30 articles studied mosquito behavior such as feeding patterns and temporal preferences. In addition, there were a few studies (6/44) that reported on EEEV transmission dynamics and competence of the following arthropod species: Haemogamasus liponyssoides, Ornithonyssus bacoti, Ixodes ricinus, Dermacentor marginatus, D. gallinae, Stomoxys calcitrans, Culicoides variipennis, and Culicoides crepuscularis. Some articles on the vector competence of arthropods have demonstrated an ability to ingest and maintain EEEV (Kissling et al. 1954, Rehacek 1958, 1960, Scanlon 1960, Clark et al. 1966), even throughout winter (Rehacek 1960). Only one article reported successful transmission of EEEV from D. gallinae to chickens under laboratory conditions (Durden et al. 1993). However, the importance of potential arthropod vectors in the EEEV transmission has not recently or extensively been investigated.
EEE and human hosts
EEEV in human populations was studied in 206 of 718 articles. Targeted observational studies were conducted in 19 of 206 articles where sample populations were evaluated for exposure to EEEV (i.e., serological antibodies) or infection with EEEV (i.e., confirmed case), of these 12 of 19 identified EEEV exposure in a proportion of the sample population between 1948 and 2013 (Table 3). Most of the human EEEV data were identified in surveillance reports (104/206) and provided data such as incidence (104/104), case fatality rates (39/104), proportion hospitalized (15/104), and prevalence of long-term sequelae (3/104). Risk factors associated with human exposure to EEEV or acquiring EEEV were reported in 2 of 206 articles (Letson et al. 1993, Hachiya et al. 2007).
Observational Studies That Identified Eastern Equine Encephalitis Virus in Targeted Human Populations (n = 12 Articles) Between 1948 and 2013 That Reported Positive Results for Exposure to Eastern Equine Encephalitis Virus
One Canadian province (Ontario) and 29 states in the United States have reported human EEEV cases through surveillance, case, or outbreak reports (Table 4, Supplementary Tables S2 and S3, and Fig. 3). The United States states include Alabama, Arkansas, Connecticut, Delaware, Florida, Georgia, Illinois, Indiana, Louisiana, Maine, Maryland, Massachusetts, Michigan, Mississippi, Missouri, Montana, New Hampshire, New Jersey, New York, North Carolina, Oklahoma, Pennsylvania, Rhode Island, South Carolina, Tennessee, Texas, Vermont, Virginia, and Wisconsin.
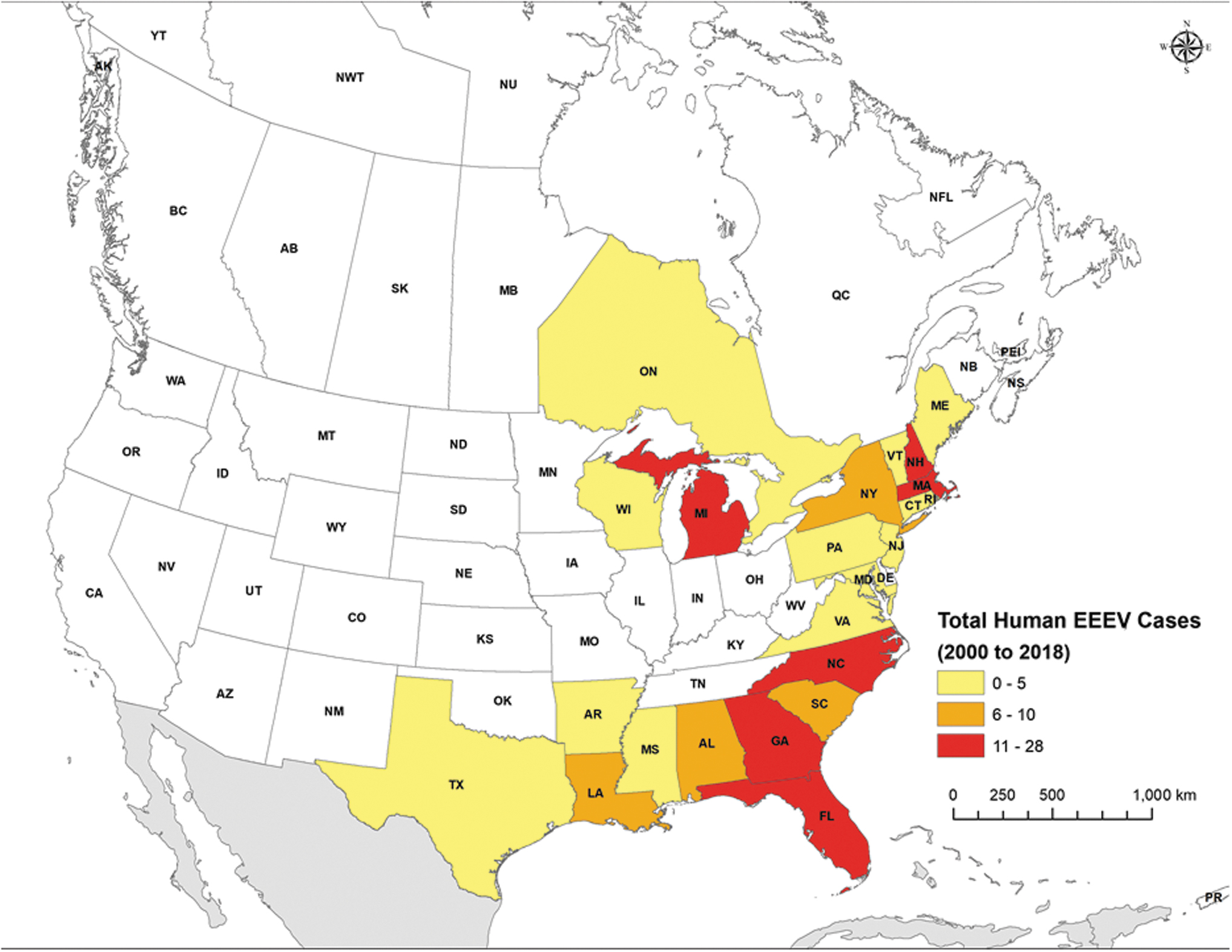
Human cases of EEEV reported in North America between 2000 and 2018. EEEV, eastern equine encephalitis virus.
Sixty-Five Outbreaks Involving Humans and Animals Reported in 54 Articles Between 1933 and 2018
Case fatalities reported.
Details of 145 sporadic human EEEV cases were described in 53 of 206 surveillance and case report studies during 1955–2019, Supplementary Table S2. Of these documented sporadic cases, 47 of 53 articles reported hospitalizations and 30 of 53 reported case-fatality rates. EEEV exposure or infection was identified in both men and women between the ages of 3 months and 87 years old. However, of the 71 of 145 cases that reported age, 49.3% (35/71) were <16 years, 35.2% (25/71) were between 16 and 59 years, and 15.5% (11/71) were 60 years of age or older. In cases where gender was reported, 71.7% were male (38/53) and 28.3% (15/53) were female. Among the articles that documented sporadic cases of EEEV, 47 of 53 articles reported signs and symptoms of infection such as fever, chills, vomiting, myalgia, arthralgia, malaise, and encephalitis. In addition, these articles also reported pathology of the disease (44/53), postmortem investigations (18/53), and sequelae after infection (25/53). One article (1/206) examined the economic burden of EEEV human cases (1/206) (Villari et al. 1995).
Sixteen outbreaks involving human cases were reported in 20 articles between 1938 and 2006 (Table 4). Most outbreaks occurred in the United States (14/16), and one each in Jamaica (1/16) and Dominican Republic (1/16). Reported outbreaks ranged in size from 1 to 38 human cases and outbreaks in horses commonly preceded the identification of human cases of EEEV during the outbreak period. In the 20 articles describing outbreaks involving humans, sequelae (4/20), pathology (5/20), signs and symptoms (8/20), and postmortem (4/20) results were reported. Additional outbreaks may have occurred, however only the outbreaks that were cited in the literature as an outbreak were recorded in this table.
In articles detecting EEEV in humans through epidemiological studies, case, outbreak, and surveillance reports, 73 of 206 provided testing details; the majority used an immunoassay (72/73), virus isolation (25/73), or molecular test (8/73) for laboratory diagnosis.
The efficacy of mitigation strategies, specifically vaccination on human volunteers was assessed in 7 of 206 articles (Bartelloni et al. 1970, Darwish 1972, DeMeio et al. 1979, Strizki 1994, Strizki and Repik 1995, Pittman et al. 2009, Reisler et al. 2012). The majority of these articles (6/7) reported successful results of vaccination attempts and suggested further evaluation in human populations. Research was also conducted on societal knowledge and attitudes about EEEV (2/206) and transmission dynamics (1/206) (Liao 1955, Rodriguez 2008, Ackerson and Viswanath 2010).
EEE in nonhuman hosts and animal models
EEEV was studied in both nonhuman hosts where the animals were naturally exposed to EEEV (280/718 articles) and animal models where the animals were experimentally challenged with EEEV (170/718). Some articles (13/718) reported on both nonhuman hosts and animal models.
The majority of the articles on nonhuman hosts (280/718) focused on the epidemiology of EEEV (257/280), which included case reports, outbreak investigations, surveillance, and prevalence studies. Of these articles, 242 of 257 reported finding EEEV in nonhuman host populations including results on sentinel surveillance using hosts (e.g., chickens, quail) monitored for exposure to EEEV (42/242).
Of the articles that reported on how EEEV was identified in animal populations, the most common diagnostic tests used were immunoassays (153/167), followed by virus isolations (87/167), and molecular tests (27/167). EEEV was reported in >155 different species of passerine birds and >65 species of nonpasserine birds (Supplementary Table S4). The virus has also been documented in various domestic and farm animals such as equine, pheasants, dogs, and swine and in other mammals such as bats, white-tailed deer, and opossums. These are listed by species, date, location, and type of evidence in Supplementary Table S4. Over 15 species of reptiles and amphibians were also shown to be naturally exposed to EEEV (Supplementary Table S4). Risk factors associated with exposure to EEEV or acquiring EEEV in nonhuman host populations were reported in 15 of 280 articles (Main 1979, Emord and Morris 1984, Centers for Disease Control and Prevention 1985, Crans et al. 1994, Spalding et al. 1994, Ross and Kaneene 1995, Elvinger et al. 1996, Dunbar et al. 1998, Garvin et al. 2004, Burkett-Cadena 2010, Mutebi et al. 2011, Vander Kelen et al. 2012, Estep et al. 2013, Elias et al. 2017, Heberlein-Larson et al. 2019).
Sixty-five outbreaks involving nonhuman hosts were reported in 54 articles between 1933 and 2018 (Table 3). The majority of the outbreaks occurred in the United States (60/65), and to a lesser extent in Canada (1/65), Mexico (1/65), and the Caribbean (3/65). Case fatalities in nonhuman hosts were reported in 50.7% (33/65) of the outbreaks. Additional outbreaks may have occurred, however only the outbreaks that were cited in the literature as an outbreak were recorded in this table.
In nonhuman hosts, studies on pathogenesis of EEEV (47/280), efficacy of mitigation strategies (8/280), and transmission dynamics (7/280) were also captured. In addition, one article (1/280) reported on survey results about management practices of horses in Florida in addition to the economic impact of EEEV to the equine industry (Wilson et al. 1986). The pathogenesis of EEEV in nonhuman hosts was reported in 47 of 280 articles, 40 of which described the signs and symptoms of EEEV infection in nonhuman hosts including those that develop clinical disease: equine (horse, mule, emus, etc.), birds (pheasants, partridges, chickens, quail), camelids (llamas, alpacas), white-tailed deer, dogs, and swine. Results of pathology (24/47) and postmortem investigations (35/47), viremic (1/47) and incubation periods (1/47), and infection mechanism/immune response (2/47) were also reported in these articles. Mitigation strategies, specifically the efficacy of vaccinations were explored in 8 of 280 articles using nonhuman hosts such as horses, pheasants, and emus. Transmission dynamics were explored in 7 of 280 articles between nonhuman host to nonhuman host (2/7), mosquito to nonhuman host (3/7), nonhuman host to mosquito (2/7), and nonhuman host to human (1/7).
Of the 170 articles that employed animal models, 159 of 170 were experimental involving challenge trials (147/159), control trials (6/159), and quasi-experiments (4/159). Fourteen articles used animal models to evaluate diagnostic testing methods. Mice were the most common animal used in these experiments (86/170) followed by domestic birds (42/170), wild birds (17/170), guinea pigs (17/170), horses (11/170), hamsters (8/170), and to a lesser extent, other animals such as monkeys and rabbits.
The pathogenesis of EEEV in animal models was reported in 93 of 170 studies, of which 26 of 93 articles included studies on infection and immune responses in these animals. Outcomes such as signs and symptoms (46/93), pathology (35/93), and postmortem investigations (46/93) were reported in these articles. Other characteristics of EEEV infection in animal models were reported including the time between exposure and viremic period (35/93), viremic period (34/93), and intrinsic incubation period (15/93).
Transmission dynamics using animal models were reported in 37 of 170 articles. These articles looked at transmission dynamics from animal model to mosquito (23/37), mosquito to animal model (14/37), animal model to animal model (7/37), and a single article reported on differences in transmission dynamics when comparing different strains of EEEV (1/37) (Arrigo et al. 2009). The efficacy of mitigation strategies, in particular vaccination, was tested using animal models in 43 of 170 articles. The majority of these articles (38/43) reported on the effectiveness of vaccine candidates. Animal models were also used to explore EEEV treatment options such as antivirals in 6 of 170 articles.
Discussion
EEEV is a vector-borne disease of public health concern that is likely to emerge or expand in range due to climate change. To prepare for the anticipated risk of increase in EEEV cases, we summarized the characteristics of the global evidence on EEEV and identified gaps in research. Our intention is that the information synthesized and knowledge gaps identified will support decision-making and further research on this priority issue. Although we attempted to capture all relevant literature on this topic, it is possible that some research was not captured due to failure to publish or an absence of citation indexing in the bibliographic databases examined. As discussed in the results, we expect language bias will have a minimal impact on this ScR as EEEV is a North American virus.
In 2010 EEEV underwent a reclassification and three of the four lineages were classified as a distinct species due to genetic diversity and differences in the ecology and pathogenesis of the strains, lineages II–IV are now known as MADV (Arrigo et al. 2010). EEEV generally circulates in North America and the Caribbean and causes the most human disease cases. MADV, formerly known as South American EEEV circulates in Central and South America and mainly causes illness in horses and mammals. Recent phylogenetic analyses show that there is overlap in the geographic regions of the two viruses. For example, MADV was reported in a cohort of children in Haiti in 2015, and the phylogenetic analysis suggests the virus was introduced into Haiti from Panama between 2012 and 2015 (Lednicky et al. 2019). Two studies found pools of mosquitoes infected with EEEV, Culex pipiens mosquitoes in Uruguay (Burgueno et al. 2018) and mosquitoes of the Culex genera in Colombia (Hoyos et al. 2015). This raises questions about the potential spread of MADV and EEEV into new geographic areas and warrants additional epidemiological research. Because of the reclassification of EEEV in 2010 and the potential spread of MADV and EEEV into new geographic areas, it is possible that some of the studies captured before 2010 were misclassified as EEEV. This is a potential limitation to our ScR.
According to the CDC, there is an average of seven clinical EEEV cases diagnosed per year in the United States, which is estimated to represent only 4–5% of human EEEV infections that have actually occurred (Centers for Disease Control and Prevention 2019b). Historically, human cases have been relatively infrequent, as it is atypical for humans to reside in low-lying swampy areas, where the main vectors of EEEV reside. In 2019, a record number of human cases (38 confirmed) were reported to the CDC (Centers for Disease Control and Prevention 2019a). The trend over the past decade has also noted EEEV activity in new areas where its circulation was previously unknown or rare such as Montana, Arkansas, Connecticut, and Pennsylvania. Canada also reported its first human case in Southwestern Ontario in 2016 (Public Health Ontario 2017). The increased activity may represent another mosquito-borne disease that requires public health awareness and monitoring. It is also possible that the rise in cases is due to increased awareness of the disease and improved diagnostics rather than an actual increase in human exposure to EEEV. Continued surveillance and large epidemiological studies would be beneficial to address these uncertainties.
Although EEEV has a high-case fatality rate (∼33%) in humans and many patients who survive develop severe sequelae as a result of EEEV infection, there is no approved human vaccine or antiviral treatment (Centers for Disease Control and Prevention 2019a). It has been estimated that the average lifetime cost to a person who suffers sequelae as a result of EEEV is ∼$3 million USD (Villari et al. 1995). This is a very large burden and impacts on the affected case's quality of life. General public health programs about mosquito bite prevention and mosquito-borne illness are recommended in affected areas to promote protective behaviors in the general population. Horses are also particularly sensitive to EEEV; 70–90% of horses infected with EEEV die and the ones that survive have permanent brain damage (Public Health Ontario 2014). Although there is a vaccine for horses, the impact to equine and industry can be financially damaging. In 1986, it was estimated that the cost of EEEV to the state of Florida equine industry was >$1 million USD/year (Wilson et al. 1986). In conducting this ScR, outbreaks in some types of poultry such as pheasants, turkeys, ratites, and quail were identified. These birds are also susceptible to EEEV that can be financially damaging for poultry farmers income. There is a clear need for additional research on viable treatment options and updated economic analyses on burden of this disease in both human and animal populations.
Given there is no human vaccine or effective antiviral therapies, appropriate mitigation strategies should be evaluated and applied. These include mosquito control, typically in the form of insecticides to large areas by fogging. Larvacides may also be a viable option for EEEV vector control. Personal protective measures and mosquito-borne disease knowledge and awareness programs should be undertaken by public health to prevent as many cases as possible (Region of Peel 2016) There is little research on determinants of public personal protective behavior against mosquito bites and what types, intensity, and duration of program is most effective at resulting in behavioral change. This is an important area for future research to guide decision-making on preventative actions to mitigate EEEV.
EEEV has been identified in 35 species of mosquitoes, over 200 species of birds, various domestic and farm animals, wild mammals, reptiles, and amphibians. Although there are a large number of mosquitoes and animals susceptible to EEEV, there is minimal research on the transmission dynamics between species and mosquito competence. Thus, additional research on drivers of transmission and the relative importance of different species may be important in predicting and preparing for the spread of EEEV into new areas.
This ScR did not identify any predictive models examining the impact of climate change on EEEV. Environmental changes associated with climate change may alter disease risk by increasing the abundance and distribution of the primary mosquito vector, by lengthening the virus-transmission season, and by extending transmission range. Transmission of EEEV is highly seasonal, with cases occurring in late spring and early fall. Climate change could lead to warmer and later winters, producing habitats that could sustain mosquito populations and increase the window for EEEV exposure (Shah and Cherabuddi 2016). Available epidemiological and climate information could be used to create predictive models to forecast the spread of EEEV with predicted climate change.
The results of this ScR characterize the 718 global research articles on EEEV. Several knowledge gaps were highlighted in this discussion. Most troubling is the impact of EEEV on affected cases and the lack of effective treatment options. The next was an apparent knowledge gap in the dynamics EEEV spread and an explanation for the increasing number of human cases over the past decade. Whether this is due to the impacts of climate change or new competent vectors, increasing cases of EEEV is a public health concern and additional research is warranted to close some of these knowledge gaps. For public health in affected areas and areas close to those infected area, mosquito surveillance and public health mosquito bite prevention programs are important mitigation strategies in the prevention of EEEV and other mosquito-borne diseases. The summary of research on EEEV presented in this ScR provides a foundation for evidence-based policy and decision making on this important public health issue.
Footnotes
Acknowledgments
The authors acknowledge Nassim Haghighi, Shannon Harding, Nicole Pachal, Carl Uhland, Barb Wilhelm, Ian Young, and Kaitlin Young for assisting with relevance screening and data characterization. Also, Ian Young and Barb Wilhelm for their input into the conceptualization of this project and the Public Health Agency of Canada library for help with article procurement.
Author Disclosure Statement
All authors declare that no competing financial interests exist.
Funding Information
No funding was received for this work.
Supplementary Material
Supplementary Document S1
Supplementary Document S2
Supplementary Dataset S3
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
