Abstract
Introduction:
Tick-borne encephalitis virus (TBEV) causes one of the most important flavivirus infections of the central nervous system, affecting humans in Europe and Asia. It is mainly transmitted by the bite of an infected tick and circulates among them and their vertebrate hosts. Until now, TBE risk analysis in Germany has been based on the incidence of human cases. Because of an increasing vaccination rate, this approach might be misleading, especially in regions of low virus circulation.
Method:
To test the suitability of rodents as a surrogate marker for virus spread, laboratory-bred Microtus arvalis voles were experimentally infected with TBEV and analyzed over a period of 100 days by real-time (RT)–quantitative polymerase chain reaction. Further, the prevalence of TBEV in rodents trapped in Brandenburg, a rural federal state in northeastern Germany with autochthonous TBE cases, was determined and compared with that in rodents from German TBE risk areas as well as TBE nonrisk areas.
Results:
In experimentally infected M. arvalis voles, TBEV was detectable in different organs for at least 3 months and in blood for 1 month. Ten percent of all rodents investigated were positive for TBEV. However, in TBE risk areas, the infection rate was higher compared with that of areas with only single human cases or of nonrisk areas. TBEV was detected in six rodent species: Apodemus agrarius, Apodemus flavicollis, Apodemus sylvaticus, Microtus agrestis, Microtus arvalis, and Myodes glareolus. M. glareolus showed a high infection rate in all areas investigated.
Discussion and Conclusion:
The infection experiments proved that TBEV can be reliably detected in infected M. arvalis voles. These voles developed a persistent TBE infection without clinical symptoms. Further, the study showed that rodents, especially M. glareolus, are promising sentinels particularly in areas of low TBEV circulation.
Introduction
TBEV is transmitted by the bite of an infected tick or, rarely, through nonpasteurized milk products from infected mammals (Holzmann et al. 2009). The main vectors of TBEV are ticks of the genus Ixodes: Ixodes persulcatus in Asia and eastern Europe and Ixodes ricinus in central, eastern, and western Europe. During the lifecycle of a tick, TBEV can be transmitted transstadially, transovarially, or primarily from infected ticks to other ticks while cofeeding on the same rodent host (Pfeffer and Dobler 2010).
The virus circulates between vector ticks and some of their hosts, mostly deer and small mammals such as rodents and insectivores, whereas only small mammals are presumed to be competent virus reservoir hosts (Mansfield et al. 2009). Larger mammals, such as sheep, cattle, or humans, are accidental hosts, as they develop only a short viremia with low viral titers or no detectable viremia. As a consequence, they are not competent to transmit and spread the virus (Pfeffer and Dobler 2010).
In recent years, the incidence of diagnosed TBE cases has increased throughout Europe. In addition, TBEV infections emerged in new areas, for example, southern Sweden, Denmark, and France, possibly because of socioeconomic or climate changes (Haglund 2002, Bröker and Gniel 2003, Donoso Mantke et al. 2008, Sumilo et al. 2008, Fomsgaard et al. 2009).
In Germany, most of the human TBE cases are reported to occur in the southern federal states. Therefore, certain parts of Bavaria, Baden-Wuerttemberg, and Hesse have been classified as risk areas for TBE (Fig. 1). These risk areas are defined by the incidence of human cases (RKI 2009, 2010). This method of classification might be misleading because of increasing vaccination rates in humans, particularly in risk areas, and a resulting decrease in the number of human TBE cases. Additionally, the exposure of humans to ticks might vary substantially in space and time and might thus also introduce bias to the current risk-mapping approach (Kiffner et al. 2010). Further, humans are accidental hosts rarely becoming infected, and quite often an infection will not be recognized because of unspecific symptoms, mild courses of disease, or unawareness of physicians.
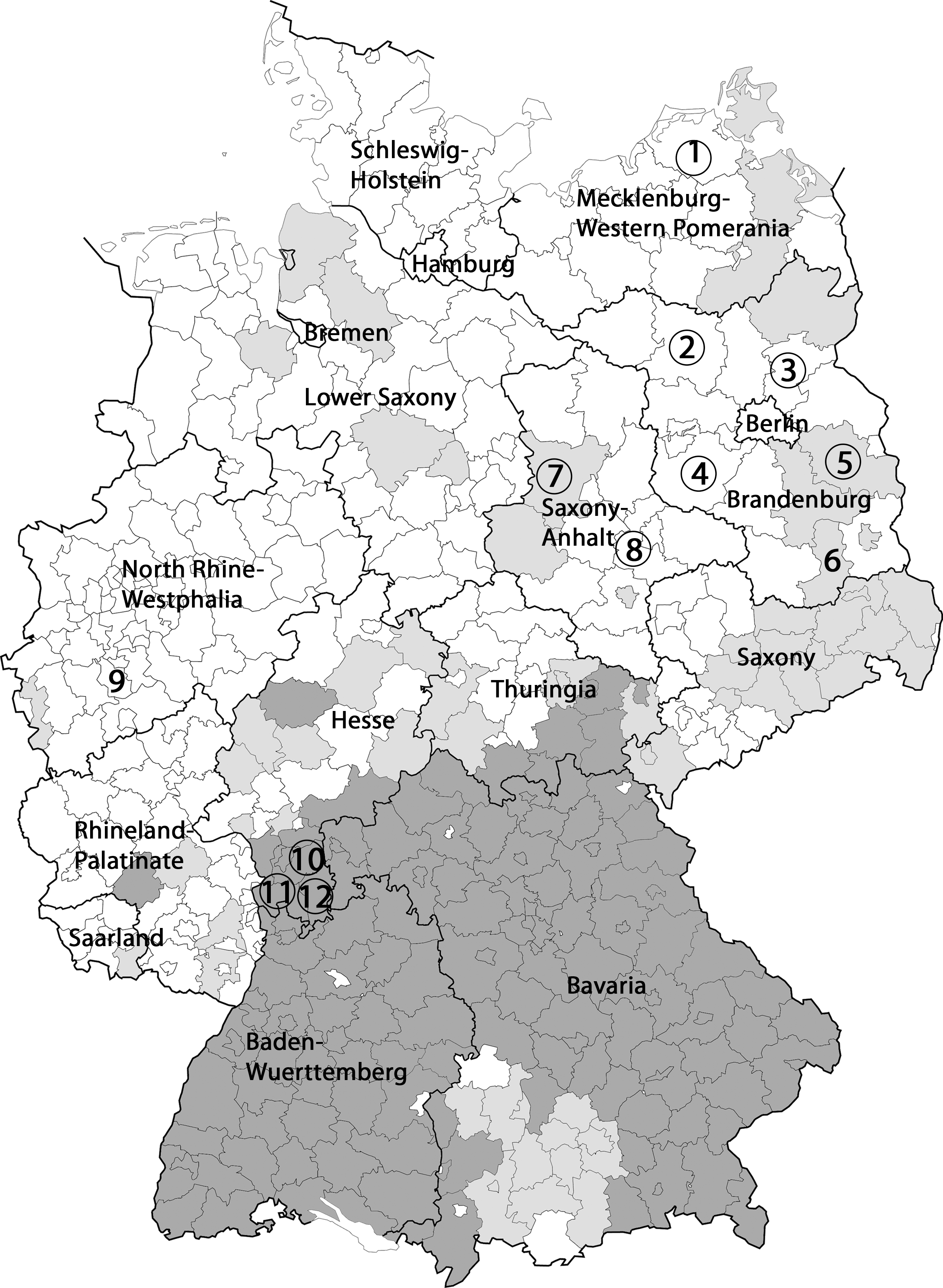
Map of Germany with TBE risk areas and localization of rodent-trapping sites. TBE risk areas (as stated by the Robert Koch Institute) are marked in dark gray, areas with single autochthonous human TBE cases in light gray, and TBE nonrisk areas in white. Rodent trapping sites are indicated through numbers (these numbers correspond to those used in Table 2). Circles indicate trapping sites where TBE virus–positive rodents have been found. 1, Nordvorpommern; 2, Ostprignitz-Ruppin; 3, Barnim; 4, Potsdam-Mittelmark; 5, Oder-Spree; 6, Oberspreewald-Lausitz; 7, Börde; 8, Anhalt-Bitterfeld; 9, Cologne; 10, Darmstadt-Dieburg; 11, Bergstraße; 12, Odenwaldkreis.
Ticks that have frequently been used in prevalence studies are not suitable for TBEV monitoring because infection rates vary greatly between local tick populations. Infections rates are usually below 1% even in highly endemic areas, for example, 0.23% and 0.46% in recent studies from Germany and Switzerland, respectively, implying high costs for collecting and screening (Süss et al. 2004, Gaumann et al. 2010, Kupča et al. 2010).
Alternatively, terrestrial mammals could be used for TBEV monitoring and the definition of risk areas, as virus or virus-specific antibodies have been detected in nearly all species studied so far (Radda 1973, Wurm et al. 2000, Mandl 2005, Mansfield et al. 2009). In line with these findings, TBEV prevalence investigations by detecting TBEV-specific antibodies in game animals such as roe deer (Capreolus capreolus) and red fox (Vulpes vulpes) and other mammals (e.g., dogs, cattle, and goats) revealed reliable information (Radda et al. 1968, Wurm et al. 2000, Klimes et al. 2001, Juceviciene et al. 2005, Lindhe et al. 2009, Sikutova et al. 2009). However, because of their large activity range as well as a lifespan of several years, the exact time and site of an infection cannot be determined.
In contrast, TBE risk analysis based on prevalence studies in small mammals, such as rodents, has advantages. They are widespread in most ecological systems, have a small home range, are easy to trap and monitor, and are heavily parasitized by ticks, which probably results in higher TBE virus prevalence (and thus lower associated costs) than in ticks. Once infected with TBEV, they are supposed to develop a persistent infection (Ernek et al. 1963, Kozuch et al. 1963, Nuttall et al. 1994, Danielova et al. 2002, Süss 2003, Bakhvalova et al. 2006, Gray et al. 2009, Kiffner et al. 2011).
Therefore, in this study, we chose rodents as potential sentinels for distribution analysis studies of TBEV. First, laboratory-bred Microtus arvalis voles were experimentally infected with TBEV, and selected organs and blood were analyzed for viral RNA and, in part, for virus particles over a period of 100 days postinfection (dpi). The results obtained should provide new information about replication, persistence, and organ tropism of TBEV in wild rodents.
To assess the suitability of rodents as sentinels for virus spread in nature, the prevalence of TBEV in rodents trapped in Brandenburg, a rural federal state in northeastern Germany with emerging autochthonous TBE cases, was determined by real-time (RT)–quantitative polymerase chain reaction (RT-qPCR). The prevalence was compared with that in rodents from German TBE risk areas as well as TBE nonrisk areas.
Materials and Methods
Experimental infection of M. arvalis voles and organ sampling
Twelve female, adult, outbred M. arvalis voles obtained from the Department of Zoology, Faculty of Science, University of South Bohemia, Czech Republic, which underwent several generations under specific-pathogen-free conditions, were infected by subcutaneous inoculation of 1000 plaque forming units of TBEV strain Hypr (GenBank accession number: U39292.1) isolated by Pospisil et al. (1954). At 5, 20, 50, and 100 dpi, two or three animals were killed and dissected. Organs (brain, heart, liver, spleen, and kidney) and blood samples were frozen immediately at −80°C for later RNA extraction and virus cultivation.
Collecting and preparation of samples from rodents
Four hundred forty-one wild-living rodents were collected in different federal states of Germany (Fig. 1) between 2002 and 2008 and necropsied according to standard protocols established by the network “Rodent-borne pathogens” (Ulrich et al. 2008). Briefly, the collected rodents were frozen and stored until necropsy. After identifying the species, weight, size, and sex were determined. During necropsy, spleen and brain tissue for TBEV analysis and additional tissues were taken and stored at −80°C until further analysis. The species affiliation of TBEV-positive rodents was confirmed by a PCR-based cytochrome b analysis.
Isolation of viral nucleic acids
Total RNA preparations from whole blood and cell culture supernatant samples were obtained using the peqGOLD blood RNA kit (PeqLab Biotechnology GmbH, Erlangen, Germany) and the Qiagen viral extraction kit (Qiagen, Hilden, Germany), respectively, according to the manufacturers' instructions. Tissue samples were first homogenized with Fast-Prep FP120 (eubio, Vienna, Austria). Afterward, RNA was extracted according to the protocol described by Chomczynski and Sacchi (1987).
Virus detection by RT-qRT-PCR and sequencing
To obtain cDNA, 5 μL of the RNA preparation was reverse-transcribed in a 20 μL final reaction volume by Superscript II kit (Invitrogen, Karlsruhe, Germany). Initially, the RNA was incubated for 10 min at 65°C. The reverse transcription was performed at 37°C for 60 min and at 93°C for 10 min in a Biometra thermoblock cycler (Biometra, Göttingen, Germany). cDNA was tested by a TBEV-specific RT-qPCR as previously described (Achazi et al. 2011). Sequencing of TBEV-positive PCR samples was performed using the BigDye® Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Carlsbad, CA) according to the manufacturer's instructions.
Virus isolation and cultivation
Tissue samples were homogenized in Dulbecco's modified Eagle's medium and the homogenate was clarified by centrifugation. The obtained supernatant was inoculated onto Vero E6 cells (ATCC CRL-1586). Cell culture supernatant was harvested at 4 days after inoculation for later RNA extraction. To assess the presence of viable virus in the sample, the inoculated cell culture was maintained through sequential blind passages over a period of 6 weeks.
Detection of specific anti-TBEV immunoglobulin G antibodies
The detection of specific anti-TBEV immunoglobulin G (IgG) antibodies was done by an immunofluorescence assay from EUROIMMUN (Lübeck, Germany) according to the manufacturer's instructions with the following changes: as secondary antibody, we used the FITC-conjugated goat anti-mouse IgG antibody A90-216F from Bethyl (Montgomery, TX). Samples were diluted 1:10 and 1:100.
Statistical analysis
Laboratory data were handled and analyzed using PASW Statistics 17 (version 17.0.3; SPSS, Inc., Chicago, IL).
Results
As, at present, the knowledge on TBEV replication, persistence, and organ tropism in rodent reservoirs is poor, 11 laboratory-bred M. arvalis voles were experimentally infected with TBEV. Selected organs (brain, heart, liver, spleen, kidney, and blood) were taken at four different days over a period of 100 dpi and analyzed by RT-qPCR and, in part, by a virus isolation approach (Table 1). Throughout the whole period of investigation, no obvious clinical signs of infection were observed in any of the animals. Beginning at 5 dpi, TBEV genome was found in several organs, that is, brain, spleen, and liver, and at 50 dpi, all organs investigated were found to be RT-qPCR positive (in at least one animal). At the final day of investigation, all three animals were found to be TBEV RNA positive in at least one tissue. Mean copy numbers of the different organs and on different dpi ranged from 4×100 to 3×106 genome copies per mg organ or mL blood (Table 1). In the TBEV RNA-positive animals, the highest copy numbers were found in the brain at 5 and 20 dpi, but the initial copy number in the brain of >106 at 5 dpi decreased over time to 101 at 100 dpi.
Samples were analyzed in duplicate.
Isolation of TBEV was performed by incubating the homogenized organ (tested positive for TBEV by RT-qPCR) on VeroE6 cells.
dpi, days postinfection; no., number; nd, not done; RT-qPCR, real-time quantitative polymerase chain reaction; TBEV, tick-borne encephalitis virus.
Blood taken at 5 and 50 dpi (of one animal each) was found positive for TBEV genome by RT-qPCR, indicating a viremia in these two animals on these days. In contrast, all blood samples from the three animals at 100 dpi were negative (Table 1). In the control animal, TBEV-specific RNA was not detectable in brain, spleen, kidney, or blood.
The persistence of infectious TBEV was confirmed by virus cultivation and subsequent RT-qPCR on cell culture supernatants for three RT-qPCR–positive tissue samples, that is, brain samples from animals at 5 and 100 dpi and a liver sample at 100 dpi (Table 1). The samples were chosen because of their high viral load as well as to prove whether viable virus could be recovered from tissue samples at 100 dpi. A typical cytopathic effect was detected for all the brain tissue–inoculated cell cultures at 5 and 8 dpi. In contrast, virus isolation from the liver samples was successful only after two cell passages and an observational period of 28 days until cytopathic effect occurred and the supernatant could be tested positive for TBEV genome (Table 1).
A clear titer of TBEV-specific IgG antibodies of >1:10 could be detected in all samples from 5 dpi on over the whole period of investigation. No specific TBEV-IgG antibodies were detected in the control animal.
To check the suitability of wild-living rodents as sentinels for the spread of TBEV, the prevalence of TBEV was determined in spleen or brain samples from 259 rodents trapped in Brandenburg. This prevalence was compared with that in 137 samples from rodents trapped in TBE risk areas in southern Germany (Hesse) as well as with that in 45 samples from rodents trapped in TBE nonrisk areas in northern and western Germany (Fig. 1).
In 45 of 441 (10%) rodents, TBEV RNA was detected by RT-qPCR in brain or spleen. The infection rates varied between 0% and 20% among the 13 trapping sites (Table 2 and Fig. 1). The average TBEV prevalence in TBE risk areas was about 15%. A statistically significant lower infection rate of about 8% was found in animals from TBE nonrisk areas (χ 2 test, p=0.049).
At least brain or spleen tissue found to be positive in the TBEV RT-qPCR.
MWP, Mecklenburg-Western Pomerania; BB, Brandenburg; SA, Saxony-Anhalt; NRW, North Rhine Westphalia; HE, Hesse; RT-qPCR, real-time quantitative polymerase chain reaction.
Sequencing of amplification products was performed, but because of low copy numbers (mean copy number of rodent spleen and brain samples tested positive on TBEV genome by RT-qPCR=67 copies per mg organ) and the short size of the PCR fragment, the obtained sequence data were of poor quality, so no subsequent subtyping and phylogenetic analysis were possible (data not shown).
TBEV genome was detected in rodents of six species, that is, Apodemus agrarius, Apodemus flavicollis, Apodemus sylvaticus, Microtus agrestis, M. arvalis, and Myodes glareolus (Table 3). Infection rates in these species ranged from 7% to 29%. No viral RNA could be detected in Mus musculus samples, most likely because of a small sample size (n=2). Statistical analysis did not show any significant difference (χ 2 test) in the infection rates of the different species. Interestingly, in the nonrisk areas in Brandenburg, TBEV RNA was detected in five rodent species, with the highest infection rates found in M. glareolus (11%, 8/73), M. arvalis (11%, 2/19), and A. agrarius (11%, 2/19). In the high-risk regions of Hesse and in the nonrisk area of Saxony-Anhalt, three of four investigated rodent species were found to be RT-qPCR positive, with the highest rates in M. glareolus of 18% (11/63) and 9% (2/22), respectively. Remarkably, A. flavicollis showed a higher infection rate in Hesse (13%, 9/69) than in Brandenburg (2%, 1/50).
At least brain or spleen tissue found to be positive in the TBEV RT-qPCR.
Also, there was no statistically significant difference (χ 2 test) in the infection rates of male (11%, 24/216) versus female (8%, 17/207) rodents or in the body mass of animals found positive or negative for TBEV, respectively.
Discussion
The results of the experimental TBEV infectivity study indicate a persistent infection in M. arvalis. Obvious clinical signs of disease were absent despite high virus titers in the brain. At 5 and 50 dpi, virus was detectable in blood, demonstrating a long duration of viremia in some individuals. However, the lack of detection of viral RNA in blood samples drawn at 20 and 100 dpi may suggest an oscillation of the viral load in blood. This assumption might be in line with the previously reported short-term viremia found in other mammalian (including human) hosts (Ernek et al. 1963, Smetana and Malkova 1966, Chunikhin et al. 1981, Schultze et al. 2007, Holzmann et al. 2009). The oscillation and variability in the process of infection in individual voles could be attributed to the variable genetic background of the outbred animals. Experimental TBEV infections in conventional in-bred laboratory M. musculus–derived lines, such as Swiss Albino or BALB/c mice, had a lethal outcome within 1–3 weeks as these mice suffer from a mutation in the Oas 1b gene, which is supposed to be an interferon-induced effector gene (Lindenbach and Rice 2001, Mandl 2005, Růžek et al. 2008).
The presented results prove that TBEV persists for at least 3 months in different organs and for >1 month in the blood of M. arvalis; however, virus titers decreased over time, particularly in the brain. These results support former studies with other rodent species (M. glareolus and A. flavicollis) in which TBEV could be detected in blood only up to 5 dpi and in organs only up to 30 dpi by classical methods (Ernek et al. 1963, Smetana and Malkova 1966, Chunikhin et al. 1981). Besides the different rodent species used in these studies, the prolonged detection of TBEV RNA as well as viable virus in the present study might be related to improved cultivation techniques and the use of highly sensitive RT-qPCR diagnostic methods.
Also, anti-TBEV IgG antibodies could be detected from 5 dpi on over the whole 3 months. These results indicate a normal immune response in the infected animals. It might be interesting to analyze antibody titers over a longer period than 3 months because in a former study with rodents from Finland (Tonteri et al. 2011), antibodies could only be detected in 2 of 16 rodents whose brain tissue tested positive for European TBEV RNA.
The findings of the experimental study and the detection of TBEV RNA in wild-living M. arvalis presented here supports a previous study by Nosek et al. (1970) proposing M. arvalis as a reservoir for TBEV. In line with these findings, related rodent species such as M. agrestis and M. glareolus were also found to contain TBEV RNA. TBEV RNA-positive M. glareolus voles were trapped not only in the TBE risk area in Hesse but also in the nonrisk areas in Brandenburg and Saxony-Anhalt. Further, in all trapping sites, M. glareolus voles showed the highest infection rates. These results suggest that this species might be the most important host for TBEV in Germany.
In Brandenburg, M. arvalis and A. agrarius showed similar infection rates and therefore seem to be important hosts in this federal state. In contrast, in Hesse, A. flavicollis showed the second highest infection rate of about 10% and therefore may substitute the role of A. agrarius and M. arvalis in the western parts of Germany. This is especially interesting because the geographical distribution of A. agrarius is restricted to the eastern part of Germany (Mitchell-Jones et al. 1999).
Because of high variation in sample size and sampling sites of species in this initial study, the significance of the observed differences in infection rates between different rodent species has to be corroborated in future large-scale screening studies. Altogether, previous investigations and our present study suggest TBEV to be rather nonreservoir specific. The reasons and mechanisms for this apparent unspecific virus–host interaction are interesting issues for further investigations (Kozuch et al. 1981).
The detection of TBEV RNA in rodents from four of five districts of the federal state of Brandenburg and both investigated districts of Saxony-Anhalt raises questions on the definition of risk areas, as these regions have not been classified as risk areas so far (RKI 2009, 2010). In future, based on the number or proportion of infected rodent specimens per area, a modified classification would be possible. Taking into account the geographical distribution of the districts (Fig. 1), it becomes evident that the trapping sites with rodents tested positive for TBEV and previously classified as TBE nonrisk areas are located in or near areas with autochthonous TBE cases (light gray). In contrast, rodents trapped in a park in the city center of Cologne (far away from TBE risk areas) are found to be free of TBEV.
In Germany, the overall prevalence of TBEV-positive rodents averaged at 10%. Therefore, Germany seems to be a country of low TBE risk in comparison to a prevalence of 61% in rodents of a high endemic area in Siberia (Bakhvalova et al. 2006). This rating is supported by the low incidence of human TBE cases in Germany compared with other European countries, for example, the Czech Republic or Russia (Donoso Mantke et al. 2008).
In general, the infection rates vary strongly among the different trapping sites. These variations are consistent with the fact that TBEV are concentrated in so-called “natural foci,” habitat sites limited to a few square meters or square kilometers (Pavlovsky 1966, Zeman 1997, Gritsun et al. 2003, Süss 2003, Lindquist and Vapalahti 2008), but could also be led back by seasonal fluctuations, which could not be considered because of missing data.
This investigation provides further direct evidence of TBEV in Brandenburg (northeastern Germany) and is in line with case reports on human TBE patients in Brandenburg (Süss et al. 1996, 2008, Becker et al. 2006, RKI 2009, 2010). Further, the results demonstrate that TBEV is endemic in Brandenburg and Saxony-Anhalt, federal states that have not been previously classified as risk areas. This finding has important implications for public health policy regarding vaccination strategies, awareness training, and the definition of risk areas for TBEV.
We conclude that TBEV detection in rodents represents a promising surrogate marker system for TBE, particularly in areas of low TBEV circulation. Rodents are easy to capture, show a persistent TBEV infection, and are ubiquitous in all ecological systems. Future investigations should assess the value and accuracy of this approach by parallel investigations of ticks, rodents, or other host animals in high and low TBE risk areas in Germany.
Footnotes
Acknowledgments
This study was funded in part by a research grant from the Federal Ministry of Education and Research (BMBF grant 1363120: “Emerging arthropod-borne viral infections in Germany: Pathogenesis, diagnostics, and surveillance”), a grant from the German National Academic Foundation, Czech Science Foundation project No. P302/10/P438, and grants Z60220518 and LC06009 from the Czech Ministry of Education, Youth, and Sports. The support of partners of the network “Rodent-borne pathogens,” namely Kirsten Tackmann (Tornow), Anne Balkema-Buschmann, Daniel Balkema (Riems), and Wolfgang Wegener (Cologne), is kindly acknowledged. Further, the authors thank Aleksandar Radonić, Ravish Paliwal, Hewad Laraway, Peter Hagedorn, Cristina Domingo Carrasco, Nadine Litzba, Patricia Bußmann, Anette Teichmann, Dörte Kaufmann, Heike Kubitza, Roswitha Mattis, Martina Steffen, Claudia Dettmer, Robert Friedrich, Marc Mertens, Robert Klopfleisch, and Konrad Wanka for their technical assistance and support and Michala Zelenková for providing the laboratory-bred voles.
Disclosure Statement
No competing financial interests exist.
