Abstract
Twelve years after the Kikwit Ebola outbreak in 1995, Ebola virus reemerged in the Occidental Kasaï province of the Democratic Republic of Congo (DRC) between May and November 2007, affecting more than 260 humans and causing 186 deaths. During this latter outbreak we conducted several epidemiological investigations to identify the underlying ecological conditions and animal sources. Qualitative social and environmental data were collected through interviews with villagers and by direct observation. The local populations reported no unusual morbidity or mortality among wild or domestic animals, but they described a massive annual fruit bat migration toward the southeast, up the Lulua River. Migrating bats settled in the outbreak area for several weeks, between April and May, nestling in the numerous fruit trees in Ndongo and Koumelele islands as well as in palm trees of a largely abandoned plantation. They were massively hunted by villagers, for whom they represented a major source of protein. By tracing back the initial human-human transmission events, we were able to show that, in May, the putative first human victim bought freshly killed bats from hunters to eat. We were able to reconstruct the likely initial human-human transmission events that preceded the outbreak. This study provides the most likely sequence of events linking a human Ebola outbreak to exposure to fruit bats, a putative virus reservoir. These findings support the suspected role of bats in the natural cycle of Ebola virus and indicate that the massive seasonal fruit bat migrations should be taken into account in operational Ebola risk maps and seasonal alerts in the DRC.
Introduction
After two simultaneous outbreaks in 1976—one involving EBOV-Z (Johnson 1978) in the northern Democratic Republic of Congo (DRC) and the other due to EBOV-S in the southwest of Sudan (Smith 1978)—as well as an isolated case of EBOV-Z infection in Tandala, DRC, in 1977 (Heymann et al. 1980) and an EBOV-S outbreak in 1979 at the same location in Sudan (Baron et al. 1983), no other outbreaks were reported until 1994 (Table 1). Since 1994 there have been 13 human outbreaks of Ebola in Africa. An isolated case of EBOV-IC infection occurred in 1994 in Ivory Coast (Formenty et al. 1999). An EBOV-S outbreak occurred in Uganda in 2000 (World Health Organization 2001) and another in Sudan in 2004 (World Health Organization 2004a). Between 1994 and 2005, eight EBOV-Z outbreaks occurred in central Africa: one in southern DRC in 1995 (Khan et al. 1999) and seven in the same regions of northeastern Gabon and northwestern Republic of Congo (RC) (Georges et al. 1999, World Health Organization 2004b, Pourrut et al. 2005). During this period, EBOV-Z simultaneously infected wild nonhuman primates in Gabon and RC, probably killing thousands of gorillas and chimpanzees in these regions (Walsh et al. 2003, Leroy et al. 2004a). Finally, EBOV-B was responsible for a large hemorrhagic fever outbreak in western Uganda, causing 30 deaths of 116 confirmed cases in November and December 2007. This new virus is distantly related to the EBOV-IC and is characterized by a lower case-fatality rate (26%) than EBOV-Z and EBOV-S (Towner et al. 2008).
DRC, Democratic Republic of Congo; RC, Republic of Congo; EBOV-Z, Ebolavirus Zaire; EBOV-S, Ebolavirus Sudan; EBOV-IC, Ebolavirus Ivory Coast; EBOV-B, Ebolavirus Bundibugyo.
The source of the primary human case(s) was not identified in seven (Yambuku, 1976; Nzara, 1976; Tandala, 1977; Nzara, 1979; Kikwit, 1995; Gulu, 2000; Yambio, 2004; and Etoumbi, 2005) of the 17 outbreaks and isolated cases that occurred since 1976. In contrast, seven outbreaks (Cote d'Ivoire, 1994; Mekouka, 1994; Mayibout, 1996; Booué, 1996; Mekambo, 2001; Kelle, 2003; and Mbomo, 2003) were clearly linked to exposure to animals found dead in the forest (chimpanzees, gorillas, monkeys, and duikers). The isolated case in Ivory Coast in 1994 occurred following autopsy of an infected chimpanzee, and the 1996 Mayibout outbreak in Gabon started among children who had found and butchered a chimpanzee carcass in the forest (Georges et al. 1999). The outbreaks between 2001 and 2003 occurred after hunters had handled animal carcasses (mainly gorillas, chimpanzees, and duikers) found in the forest (Leroy et al. 2004a).
Recently, several studies have shown the presence of specific IgG and Ebolavirus nucleotide sequences (Leroy et al. 2005, Pourrut et al. 2007) in the liver and spleen of three African fruit bat species (Hypsignathus monstrosus, Epomops franqueti, and Myonycteris torquata). Marburgvirus sequences (Swanepoel et al. 2007, Towner et al. 2007) have been found in one fruit bat species (Roussettus aegypti) and two insectivorous bat species (Rhinolophus eloquens and Miniopterus inflatus), raising the possibility that bats may be natural hosts (reservoir species) or intermediate hosts for the two filoviruses. In the 1976 Ebola outbreak in Sudan, the first six patients worked in a cotton factory in Nzara, where insectivorous bats were present (Arata and Johnson 1978). However, there had been no evidence that humans can be directly infected by bats. In this study, we obtained the first evidence linking a human Ebola outbreak to exposure to the putative fruit bat reservoir by collecting social and environmental information through interviews with villagers during the EBOV-Z outbreak that occurred in DRC from May to November 2007.
Materials and Methods
We helped with investigations and outbreak control as part of an international WHO team in October 2007. The investigation team formed by the DRC Ministry of Health (MoH), Médecins-Sans-Frontières (MSF)-F, and WHO was sent to Mweka and Luebo to carry out prospective and retrospective analysis of the Ebola outbreak and to try and identify its source. As in previous Ebola outbreaks, three case-notification categories were used: suspected, probable, and confirmed. Cases were identified from the medical records of healthcare centers and through both passive and active surveillance. Cases were passively identified by healthcare and hospital facilities that acted as sentinel surveillance sites. Active case finding was organized by the investigating team that travelled from village to village in areas where cases had been reported. Several villages reporting suspected cases were visited, and healthcare staff were trained in Ebola disease surveillance and control when appropriate. Epidemiological and clinical information on each case was collected by interviewing survivors, family members, and key informants, and by holding focus group discussions. Data were also collected by comprehensive review of available patient files from the Kampungu MSF clinic or from healthcare centers, as appropriate. Data on each patient were recorded on a standardized Ebola investigation form that included the patient's identity, clinical manifestations, treatment, the likely mode of infection, and any laboratory results. With the patient's verbal consent the geographic position of the house was recorded using a GPS device.
We also interviewed villagers about the different mammalian species present in the area and the number found dead or sick in the forest. We recorded dietary habits and hunting protocols with the villagers during focus group discussions. Pictures of animals known to be possible carriers of Ebola virus (rodents, monkeys, etc.) were used to facilitate their identification by the native population.
This study was part of an Ebola outbreak control operation coordinated by MoH, and no ethics committee approval was considered necessary, as usual in this kind of emergency situation. The family members and surviving patients were informed that this report would be published, and that their privacy would be preserved. Given the situation of Occidental Kasaï in DRC, we had no way of sending them a hard copy of the manuscript for approval, but the MoH authorities (a cosignatory of the paper) gave their approval.
Results
The outbreak
An Ebola outbreak occurred in the Occidental Kasaï province of DRC between May and November 2007 (Fig. 1). The epicenter was an agglomeration of 10 villages situated on the north-south road linking Luebo and Mweka, about 200 km from the location of the 1995 outbreak in Kikwit, a town located on the river Kwilu (Fig. 1). Epidemiological investigations conducted by the DRC MoH and international teams showed that this outbreak might have caused 264 suspected cases including 186 deaths. The first case probably occurred in May. However, the extreme inaccessibility of the affected area delayed the detection of the outbreak and the mobilization of control teams. The virus was detected on 10 September 2007 in samples that had been collected on 22 August and sent to both the Centre International de Recherches Médicales de Franceville (CIRMF) in Gabon and the Centers for Disease Control and Prevention(CDC) in Atlanta, USA. The international response was launched in mid-September, and the epidemiological surveys were mainly retrospective. An isolation ward was set up by MSF in a hospital building in Kampungu, the northernmost village in the outbreak epicenter. Joint laboratory analyses by CDC and CIRMF identified the outbreak species as EBOV-Z, as in other outbreaks occurring in central Africa (DRC, RC, and Gabon).
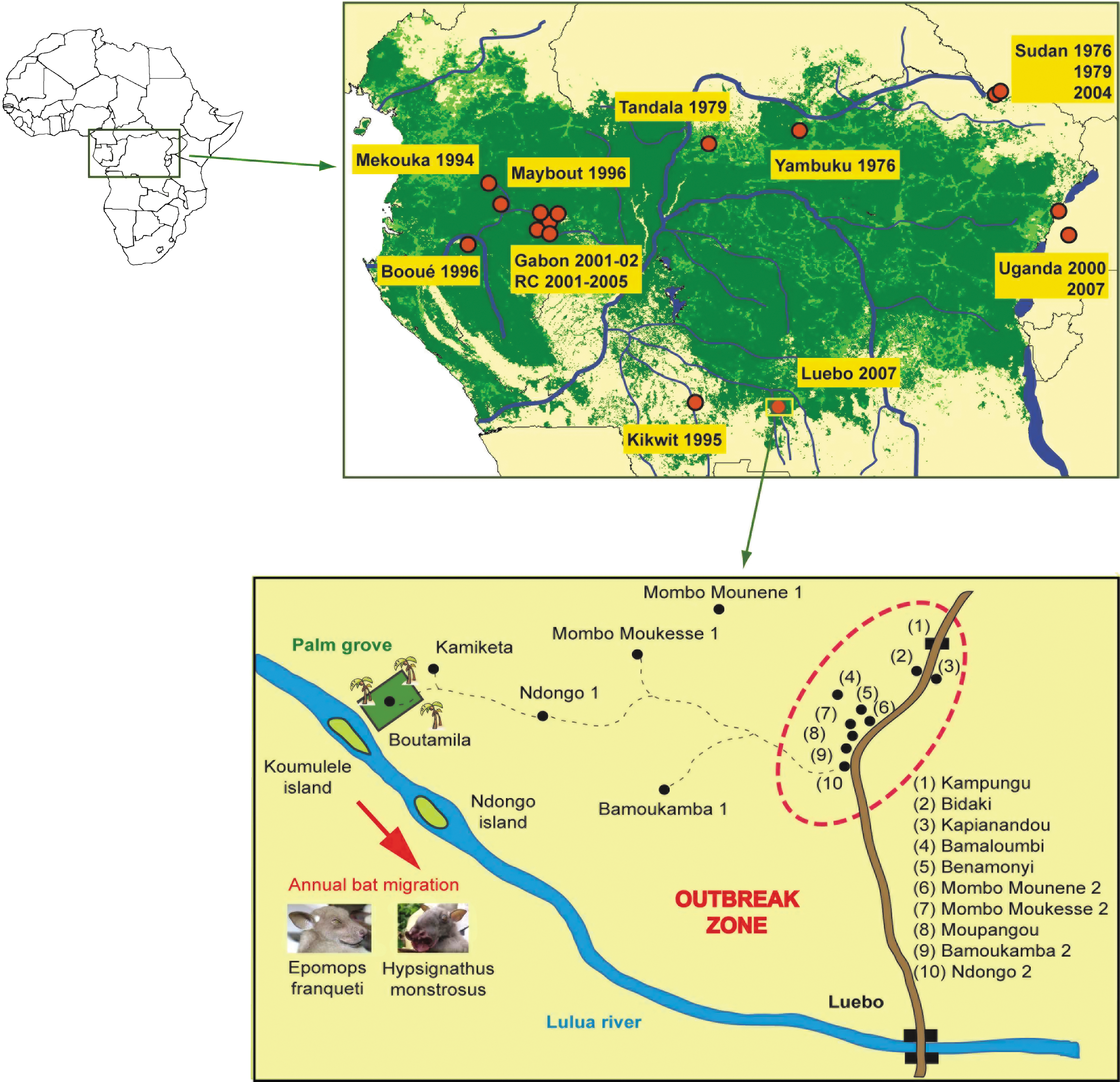
Location of the 2007 Ebola outbreak in DRC. The Kampungu agglomeration is situated along the main road between Luebo and Mweka villages. The old villages are located in the forested section between the road and Lulua River. Lulua River, oriented north-south, corresponds to the bat migration flyway, including the islands where they settled for a few weeks before and during the human outbreak.
Context of the outbreak
By tracing back the transmission chains of cases confirmed in September, investigators found that the likely index case of this outbreak, patient A, was a 55-year-old woman who lived in Ndongo 2 village. She had developed a typical Ebola hemorrhagic fever (high fever, vomiting, diarrhoea, hemorrhages) and died on 3 July 2007. Then, 11 contacts (mainly family members) who had taken care of her became ill 5–10 days after the patient was affected; they developed a typical viral hemorrhagic fever and died a few days later. No samples were collected from patient A, and so laboratory confirmation was not possible. She had fallen ill around 25 June 2007. In early October, retrospective epidemiological studies failed to link patient A to any other cases. We therefore extended the investigation to neighboring villages. We learned that the villages in the outbreak epicenter had sprung up along the road in the 1960–1970s. Indeed, shortly after gaining independence, the DRC authorities had demanded that all remote villages move close to main roads to ensure better access to healthcare, schooling, and public services, and also for more efficient political management. The villages of the Kampungu agglomeration had previously been located in a forest area mostly belonging to the forest/savannah transition zone between the Luebo-Mweka main road and the river Lulua (Fig. 1). We explored this zone, visiting the villages, systematically recording GPS coordinates, interviewing the inhabitants at length, and closely examining the environment and fauna. Villages in this zone are accessible only by motorcycle or, in some cases, only on foot, and all are located 3–4 hours on foot from the main road. Far from being abandoned, the old villages are thriving and are surrounded by fields and plantations. Each of the 10 villages in the Kampungu agglomeration has its “twin village” in the forest zone, as if each village in fact has two components, one situated in the forest (village “1”), and the other near the trunk road (village “2”). Villages “2” house public services (healthcare, education, administration, police, religion, marriage, burials, etc.), shops, and markets. In contrast, villages “1,” situated in the bush, supply agricultural products and hunted/gathered products to villages “2,” where they are consumed by the families or sold in the markets. The main market is held every Monday in Mombo Muonene 2.
The local populations informed us of a massive annual bat migration, southeast along the river Lulua (Fig. 1). Each year in April, bats arrive in their thousands or tens of thousands and settle briefly on the islands of Ndongo and Koumulele (Fig. 1). They resume their journey by mid-May, flying up river Lulua in a southerly direction. During their stay on these islands, the bats eat the fruit of trees belonging to the Rubiaceae, Ficus, and Moraceae (Great Blue Toraco, Musanga cecropioides), which are highly abundant. A massive palm oil plantation (about 2 km2) was developed close by river Lulua in 1925 and cultivated in such a way that production peaked in April of each year. This plantation was no doubt an important food source for bats for more than half a century, eventually becoming a stopover on their migratory path. As a result, palm nuts were widely disseminated throughout the neighboring environment, and the landscape in this region is remarkable for the large numbers of scattered palm trees. The plantation, now largely neglected, still represents a place where bats can feed undisturbed. Field investigations showed that the migratory bats included two of the three species suspected of being the reservoir of EBOV-Z, namely, H. monstrosus and E. franqueti (Fig. 1).
During the migratory period, when the bats roost in massive numbers on tree branches on Ndongo and Koumulele islands, hunters shoot them on a daily basis, a single shotgun cartridge killing up to several dozen animals. Outside of this period, relatively small numbers of bats are killed, individually with machetes, catapults, or by hand, often close to the villages. Some hunters in three villages—Ndongo 1, Mombo Munene 1, and Bamaloumbi—that are relatively close to the river specialize in hunting bats. During the annual migrations of bats these hunters supply their own villages “1” and “2,” and the villages of their friends and families. They also supply the weekly market at Mombo Muonene 2, where freshly killed bats are sold. During this period, the bats represent an important food source, mainly for men, postmenopausal women, and children. Indeed, women of child-bearing age are not allowed to eat bats, but it is often they who butcher, prepare, and cook them. Villagers informed us that the migratory bat population had been particularly massive in April 2007 and, consequently, in April and May 2007, the 10 villages were literally inundated with bats killed by shotgun, a method that makes the animals bleed from multiple wounds. No precautions were taken during their manipulation, clearly resulting in thousands of direct contacts with blood-covered bats (Fig. 2).
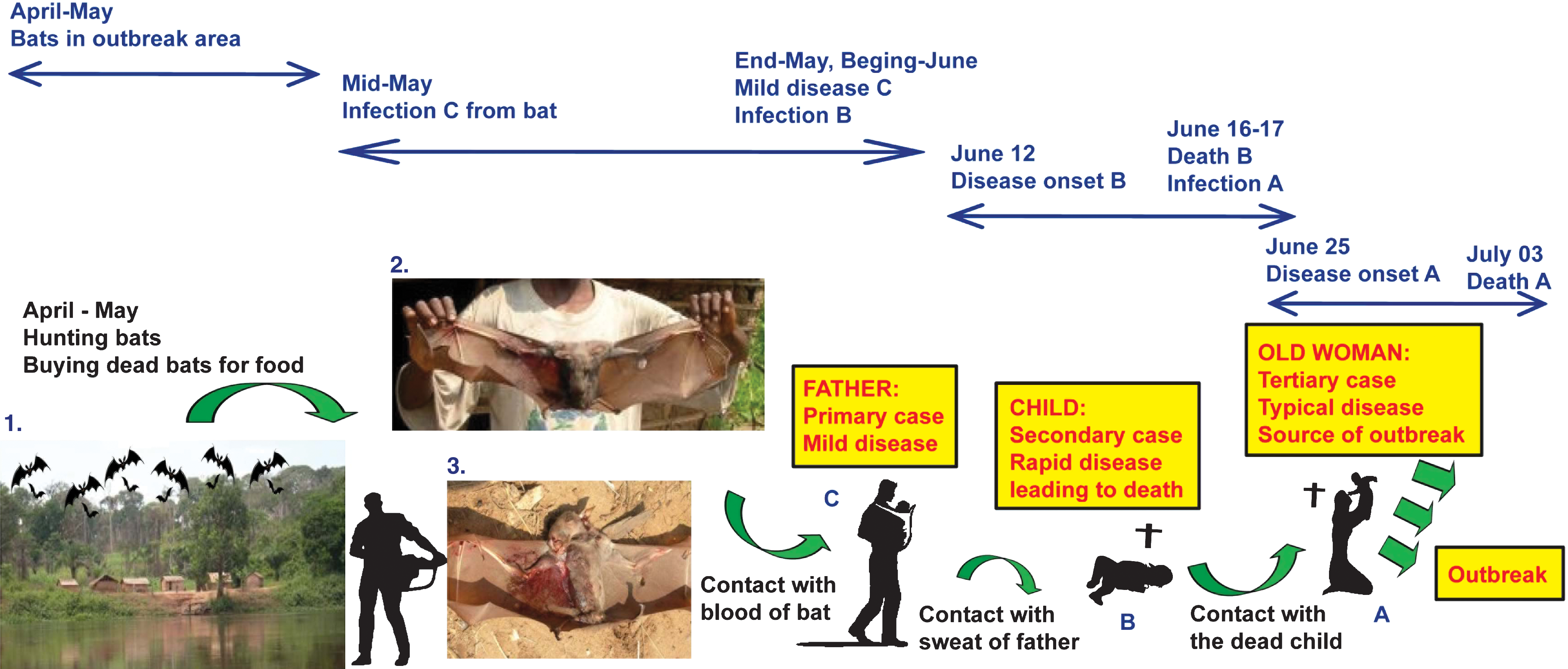
Schematic diagram of the putative chain of Ebola virus transmission from bats to the initial human cases in the 2007 outbreak in Luebo, DRC. The first picture shows a village near the islands where migrating bats settle during April and May. The second picture shows the main migratory bat species (H. monstrosus) hunted by villagers during April to May. The third picture shows how villagers handle the bats they kill and eat, which underlines the direct contact with blood and other potentially infected bat body fluids.
We found no signs of unusual morbidity or mortality, past or present, in bats or other animals. Villagers reported no evidence of a decline or an increase in any particular animal species, and the demographics of the animal fauna had remained stable. Chimpanzees and gorillas, which are susceptible to the virus and had been the sources of several outbreaks in Gabon and Congo between 1994 and 2005, are not present in this part of DRC. Bonobos—other great apes present in DRC—are not found in the outbreak epicenter, as the southern limit of their distribution is the river Sankuru, 100 km north of Kampungu.
Taken together, the information collected during the different surveys showed a strong spatial and temporal association between the annual bat migration and the Ebola outbreak. However, there was still no evidence of a direct link between these animals and any of the human victims.
Human transmission and first transmission events
Patient A died on 3 July 2007 after falling sick around 25 June 2007. The maximum incubation period of Ebola is 21 days, meaning that patient A would have been infected at the earliest around 4 June 2007, about a month after the bats had departed. To find a link between the bats and patient A, we tried to determine how the patient was infected, and then to seek a link between this source and the bats.
Our investigations revealed the following sequence of events. An inhabitant of Bamoukamba 2 village (patient C; Fig. 2), who was not a hunter, had regularly bought bats at Mombo Mounene 2 market, as he did every year. He had been in direct contact with the blood of the bats he had bought. He reported having mild symptoms (mainly fever and headache) sometimes during this period. Patient C was the father of a 4-year-old girl (patient B), who suddenly fell ill on 12 June and died on 16 June 2007, having had vomiting, diarrhoea, and high fever. No hemorrhagic signs were reported. Like all the inhabitants of the agglomeration of 10 villages, patient C and his wife, accompanied by their daughter, had made regular stays in their bush village, Bamoukamba 1. It is noteworthy that, on the trail between villages “1” and “2,” children other than infants are usually carried by their older siblings or by men, suggesting that patient B might have been infected through sweat or skin swabs of her father. Indeed, a recent study has shown the presence of Ebola virus in body fluids such as saliva, tears, and skin swabs (Bausch et al. 2007).
Assuming that patients B and C had Ebola, the small girl (patient B) might have been infected during prolonged contact with her father's sweat on the trail between the two villages. The girl would probably have been infected toward the end of May, which is compatible with an incubation period of about 2 weeks. Patient C and his wife confirmed that they went to their bush village several times at the end of May. In keeping with local traditions, the small girl's dead body was washed by a close friend of her parents (patient A), assisted by the girl's mother and grandmother. The body was washed on 17 June, and patient A fell sick on or around 25 June, about 8 days later, which is again compatible with the Ebola incubation period (Fig. 2). Except for patient A, there were no deaths among the young girl's contacts. With the exception of patient B, patient A had no proven contact with any other persons who fell sick or died during the 21 days before her symptoms occurred.
Discussion
We report the first evidence connecting a human Ebola outbreak to exposure to the putative fruit bat reservoir. We also describe the initial human-to-human transmission events that may have initiated the outbreak. Each year, thousands or tens of thousands of fruit-eating bats (H. monstrosus and E. franqueti) migrate southeast up the river Lulua. The migration probably begins at the river Congo, continues along the river Kasai, and follows the river Lulua upstream toward Angola and/or Zambia. During their migration, the bats settle for about a month, between April and May, on the islands of Ndongo and Koumulele. Each year, village hunters kill large numbers of bats and sell them fresh to inhabitants of the Kampungu agglomeration. It is very likely that, in 2007, when the migratory bat population was especially large, an inhabitant of Bamoukamba village was infected by bat blood, developed a mild form of Ebola, and then infected his daughter, who died. Patient A was likely infected when washing the child's body, and was the source of the subsequent outbreak.
These observations raise several questions.
First, why was there only one bat-to-human transmission event? The outbreak only really started after the third human case had occurred. Transmission from the father (patient C) to his daughter (patient B), then from the girl's corpse to patient A, took place in very particular circumstances and went unnoticed. In fact, it is highly likely that several other persons were infected by bats but the circumstances required for subsequent human-to-human transmission were not present.
Second, why was only one secondary case (patient B) observed among all the people who came into contact with patient C? The mild illness (isolated fever) in the primary case suggests a low viral load. This would imply a low risk of transmission to another person, except in certain conditions (e.g., close and long-lasting contact, with a young child, for example).
Third, why was the first secondary case (patient B) so rapidly fatal (4 days)? The answer is probably that patient B, being a young child, succumbed rapidly to untreated dehydration due to vomiting and diarrhoea.
Finally, why was there only one tertiary case (patient A)? Patient B might have died before developing a high viral load. A low viral load, combined with the short symptomatic period without bleeding, might explain the absence of secondary cases in the family of patient B. In addition, the person who was in closest contact with the girl was her father, who had already been infected and had probably developed specific protective immunity. Thus, virus transmission may have occurred when patient A prepared the corpse for burial ceremony. When interviewed, the two other preparers, the girl's mother and grandmother, reported they did not have direct contact with the corpse and they did not develop any clinical sign of infection in the four following weeks.
These findings strongly suggest that Ebola virus can be contracted by humans through direct exposure to fruit bats, and the massive fruit bat migrations should therefore be taken into account in operational Ebola risk maps and seasonal alerts. Specific studies of the routes and dates of fruit bat migration are needed. However, in the short term, it may be difficult or even impossible to halt human consumption of fruit bats, which represent a readily available and abundant source of protein, especially as many game species are protected or becoming rare.
Footnotes
Acknowledgments
We are very grateful to the MoH; Mr. Victor Makwenge Kaput from DRC; and Dr. Jean-Baptiste Roungou, WHO representative in DRC, for their constant support throughout the study. We are very thankful to all our colleagues from MoH, WHO, and MSF who supported our work in Luebo and Mweka.
This work was supported by the WHO Department of Epidemic and Pandemic Alert and Response at the regional office for Africa and at headquarters.
Disclosure Statement
No competing financial interests exist.
