Abstract
Background:
Remote patient monitoring (RPM) has potential in hypertension management, but limited studies have focused on maternal hypertension, especially among vulnerable populations. The objective of this study was to integrate RPM into perinatal care for pregnant patients at elevated risk of hypertensive disorders to show feasibility, acceptability, and safety.
Methods:
A prospective pilot cohort study was conducted at the University of Mississippi Medical Center 2021–2023. Participants' blood pressure readings were remotely captured and monitored until 8-week postpartum, with timely assessment and intervention.
Results:
Out of 98 enrollees, 77 utilized RPM, and no maternal or neonatal deaths occurred within 60-day postpartum. High program satisfaction was reported at discharge.
Conclusion:
This study demonstrates the feasibility and acceptability of RPM for perinatal care in a vulnerable population. Positive outcomes were observed, including high patient satisfaction and no maternal or neonatal deaths. Further research should address patient engagement barriers and develop tailored protocols for improved clinical outcomes.
Introduction
Hypertensive disorders of pregnancy (HDP) pose significant risks for maternal and fetal complications. 1 In the United States, preeclampsia affects ∼4% of pregnancies, disproportionately impacting Black women with a threefold higher case fatality rate compared to White women. 2 This disparity can be attributed to multiple factors, such as lack of access to health care, systemic racism, and inequitable care, influencing Black maternal health experiences. 2 In Mississippi, severe maternal hypertension from preeclampsia and cardiovascular disease is the leading cause of pregnancy-related deaths, with up to 80% of fatalities occurring among Black mothers. 3 Those in rural areas further encounter challenges in accessing adequate health care services, exacerbating existing health inequities. 4 Findings from both maternal mortality review committees underscore the urgent need for improving health care practices and a more patient-centered approach. 3,5
Blood pressure (BP) monitoring is vital in prenatal care, with early interventions prompted by home BP monitoring showing promise for women at risk of HDP. 1,6,7 This is particularly relevant for pregnant women in Mississippi facing barriers to accessing care. Therefore, utilizing telehealth and mobile technologies becomes crucial in enhancing pregnancy care and addressing barriers to timely interventions. Although there have been studies on home BP monitoring in Mississippi's general population and postpartum care, this study seeks to fill a research gap by utilizing home BP monitoring in pregnant women at risk of HDP from a predominantly Black, rural population. 8 –12
This study aims to show the feasibility, acceptability, and safety of integrating home BP monitoring for early intervention during pregnancy. The innovative nature and unique implementation of remote patient monitoring (RPM) for maternal BP management hold the potential to address the limited access to care, especially in underserved areas. The findings of this study could inform future strategies for improving pregnancy care, reducing maternal complications, and ensuring equitable access to health care services in underserved areas like Mississippi.
Methods
This prospective cohort study was conducted at the University of Mississippi Medical Center (UMMC) from July 19, 2021 to February 23, 2023, following Institutional Review Board approval.
PARTICIPANTS AND SETTINGS
Adult patients with an intrauterine pregnancy, receiving prenatal care at UMMC and affiliated practices, proficient in English, and having reliable internet access were recruited. Study sites include Jackson Medical Mall, Grants Ferry, and Women's Specialty Care at Mirror Lake. Trained clinic staff confirmed patients' interest and their contact information. Eligible patients were then sent a telemonitoring kit within 3–5 days that included an iPad tablet equipped with a cellular chip and a Bluetooth-enabled BP cuff for enabling direct transmission of measurements to the UMMC's Electronic Medical Record (EMR) system and monitoring by study nurses.
Upon receiving the telemonitoring kit, patients were contacted by a nurse coordinator through secured audio/video connections to confirm informed consent. After obtaining informed consent, the nurse coordinator provided instructions to nurses on proper BP measurement techniques, emphasizing the importance of cuff fit. Before measuring BP, participants were instructed to sit quietly for 5 min with feet flat on the floor, supported back and arms, and to avoid caffeine, exercise, and having a full bladder within the last 30 min.
During pregnancy, participants were asked to record at least one BP measurement weekly in the antenatal period, daily in the postpartum period for 1 week, and weekly thereafter until 8-week postpartum. The tablet system prompted medical guidance if BP readings exceeded 140 mmHg systolic or 90 mmHg diastolic with symptoms, such as headache, visual changes, shortness of breath, chest pain, and swelling. Alerts were also triggered if BP readings exceeded 155 mmHg systolic or 90 mmHg diastolic on two measurements 15 min apart. Each BP measurement was reviewed daily by a nurse, with elevated readings prompting contact, timely assessment, intervention recommendations, messaging maternal fetal providers, and triage instructions if needed. Participants were monitored. Satisfaction surveys were conducted upon discharge.
DATA COLLECTION AND STATISTICAL ANALYSIS
All patients' medical records at UMMC and data obtained through the telemonitoring kits were integrated within the UMMC EMR system. Study data containing protected health information were extracted by an honest broker and securely stored in Research Electronic Data Capture, a designated platform for managing research data. Additionally, survey responses from participants were collected by the project manager and stored within REDCap.
Study enrollees were divided into four groups based on their adherence to the protocol during the prenatal period: fully adhered, mostly adhered, partially adhered, and not adhered. The classification was determined by the frequency of BP measurements before delivery. Fully adhered participants measured BP weekly, mostly adhered participants measured BP in more than half of the weeks, partially adhered participants measured BP in less than half of the weeks, and the not adhered participants did not measure BP at all.
Baseline characteristics, engagement activities, and pregnancy outcomes were summarized for all enrollees and by groups. Shapiro–Wilk tests were employed to assess the normality of continuous variables. Mean (standard deviation) and Median (Min, Q1, Q3, Max) were utilized to summarize normal and nonnormal continuous variables, respectively, while numbers (%) were utilized to summarize categorical variables. Analysis of variance and Kruskal–Wallis tests were utilized to compare the mean or median for normal and nonnormal continuous variables across all groups. Pearson's Chi-squared test and Fisher's exact test were utilized to assess differences in frequency distributions of categorical variables across groups. If a significant difference was observed, pairwise comparisons were performed with p value adjustment. The Scheffe method was used for normally distributed variables, Dunn's test for nonnormally distributed variables, and Hochberg was used for categorical variables. Bar charts were used to intuitively visualize the participant satisfactions. An alpha level of 0.05 was used to determine statistical significance. All analyses were conducted using SAS statistical software (version 9.4; SAS Institute Inc., Cary, NC, USA).
Results
A total of 100 patients were referred to the program, with 98 patients consenting to enroll (95 in person and 3 virtually). Seventy-seven (N = 77) patients actively participated after receiving kits and instructions. Among them, 21 (21.43%) fully adhered to the prenatal protocol, 36 (36.73%) mostly adhered, and 21 (21.43%) partially adhered. The remaining 21 (21.43%) patients did not use RPM, with reasons including one patient not signing for the telemonitoring kit package for collection, three opting out, and 17 being unreachable (Fig. 1).
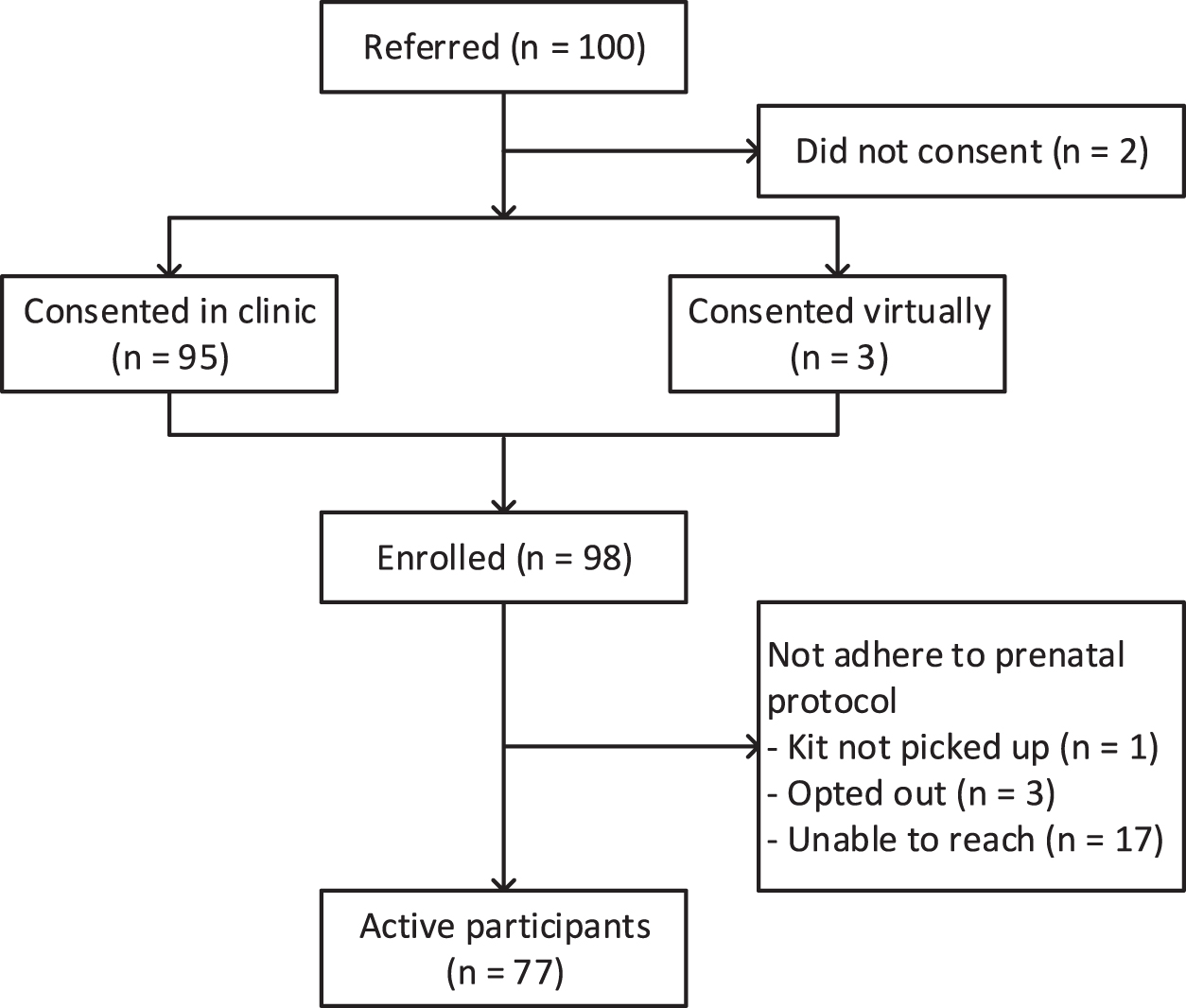
Study flowchart.
At baseline, the enrolled patients had an average age of 30.53 ± 5.95 years and an average gestational age of 23.57 ± 6.91 weeks. The fully adhered group had a significantly higher average gestational age of 27.63 ± 6.92 weeks compared to the mostly adhered group (22.27 ± 5.20 weeks) and the partially adhered group (21.05 ± 7.22 weeks). The majority identified as Black/African American (86.60%), and 78.72% were classified as obese. Among patients in the partially adhered group, all reported being obese. Furthermore, 90.82% of the patients were covered by Medicaid, and 58.16% resided in rural areas (Table 1).
Baseline Characteristics for All Enrollees and by Adherence to the Prenatal Protocol
p Value <0.05.
Fully adhere group; bMostly adhere group; cPartially adhere group; dNot adhere group; ab* The comparison between the fully adhere group and mostly adhere group has a p value <0.05; ac* The comparison between the fully adhere group and partially adhere group has a p value <0.05; bc* The comparison between the mostly adhere group and the partially adhere group has a p value <0.05.
BMI, body mass index; Max, maximum; Min, minimum; SAB, spontaneous abortion; SD, standard deviation; TAB, tubal abortion.
During pregnancy, 21 (21.43%) patients were admitted to study sites, with 7 from the fully adhered group, 6 from the mostly adhered group, 6 from the partially adhered group, and 2 from the nonadhered group. Among them, 6 were admitted for gestational hypertension, with 3 from the fully adhered group, 1 from the mostly adhered group, 1 from the partially adhered group, and 1 from the nonadhered group. No statistical significance was observed in these numbers across the different adherence groups (Supplementary Table S1).
Table 2 displays the postpartum complications and clinical outcomes across groups. Significant differences in average birthweight were observed across the four groups: fully adhered (2.75 ± 0.95 kg), mostly adhered (2.57 ± 0.80 kg), partially adhered (3.23 ± 0.88 kg), and nonadhered (2.46 ± 0.69 kg). Pairwise group comparisons did not reveal significant differences. No maternal or neonatal deaths occurred within 60 days after delivery at any study sites.
Pregnancy Outcomes By Adherence to the Prenatal Protocol
Fully adhere group; bMostly adhere group; cPartially adhere group; dNot adhere group.
AROM, artificial rupture of membranes; EASI, extra-amniotic saline infusion; ED, emergency department; NA, not applicable; SMM, severe maternal morbidity.
Although no patient strictly adhered to the postpartum BP measurement protocol, significant associations (p < 0.001) were found between prenatal adherence groups and telemonitoring utilization at two time points: within the first 7-day postpartum and between 7-day and 8-week postpartum. Specifically, within the first 7-day postpartum, 14 (66.67%) patients from the fully adhered, 17 (48.57%) from the majorly adhered, 3 (14.29%) from the partially adhered, and none from the nonadhered measured their BP. Between 7-day postpartum and 2 months after, 18 (85.71%) patients from the fully adhered, 20 (57.14%) from the majorly adhered, 3 (14.29%) from the partially adhered, and none from the nonadhered measured their BP. After delivery, 69 patients completed in-person postpartum visits at study sites. The rate of postpartum visit completion was significantly higher in the fully adhered group (95.24%) compared to the partially adhered group (52.38%, adjusted p value = 0.018) and the nonadhered group (52.38%, adjusted p value = 0.018) (Supplementary Table S2).
Thirty-eight patients completed the satisfaction survey at discharge, and most expressed high satisfaction with various aspects of the program, including the telemonitoring kit, the education provided, communication with the nurse, and the quality of care. The comments received further reflected positive feedback and satisfaction, emphasizing the value of the program in improving maternal health and well-being, and recommending its participation to others (Fig. 2; Supplementary Table S3).
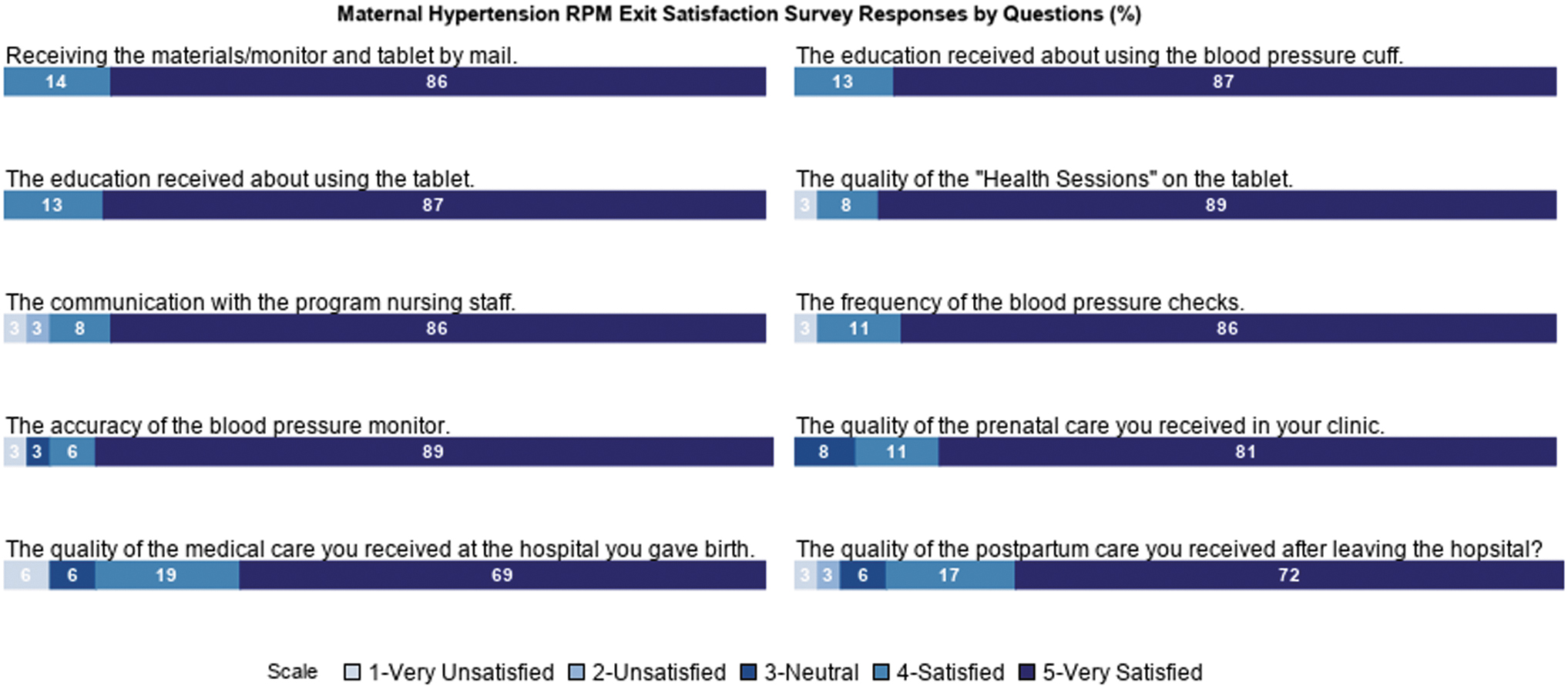
Survey response (n = 38). RPM, remote patient monitoring.
Discussion
The findings of this study highlight several important aspects of program implementation and its potential impact on perinatal care. The high enrollment rate indicates a strong interest within the study population. However, nonadherence issues emerged, warranting strategies for patient engagement, such as comprehensive patient education and patient-family advisory councils in future work. 13 The demographic profile reflects the challenges faced by vulnerable mothers, particularly those who are Black/African American, obese, covered by Medicaid, and residing in rural areas, contributing to health care disparities. The prevalence of previous pregnancy experiences, including preterm births and spontaneous abortions, further underscores the importance of effective management strategies for this population.
Hospital utilization was similar across adherence groups, but the timely triage assisted patients in avoiding unnecessary visits and prompting necessary visits. Moreover, the fully adhered group displayed higher postpartum visit completion and telemonitoring use, indicating the potential benefits of consistent adherence in promoting patient engagement and facilitating timely monitoring and detection in postpartum care. Importantly, no maternal or neonatal deaths were identified within 60-day postpartum, highlighting the overall safety and positive outcomes associated with this program. The patient satisfaction survey responses echoed a strong contentment, reinforcing the value and relevance of the program in improving maternal health and well-being among the vulnerable population.
While the findings of this study show feasibility, acceptability, and safety of the RPM program for pregnancy care, its generalizability may be limited due to its single-institution scope. Nonetheless, it still offers a foundation for future research. Adaptation of the study design and implementation can facilitate the exploration of RPM programs for prenatal care in various settings.
There are several limitations to be considered. First, the small sample size of the pilot study may affect statistical power and limit the detection smaller effects or differences. Additionally, selection bias may exist, given patients who enrolled in the study may have had different characteristics compared to those who did not enroll. Moreover, all data used in this study were obtained from UMMC Epic, which may limit our access to health care utilization data from other institutions and may restrict the overall comprehensiveness of the results. Furthermore, the study focused on short-term outcomes and did not assess long-term effects or sustainability of the program. Future research with larger sample sizes, more rigorously designed control group, such as randomized controlled trials, and longer follow-up periods is needed for more comprehensive insights into the effectiveness and sustainability of RPM for pregnancy care.
Conclusion
In conclusion, this study provides valuable insights into the implementation and outcomes of a telemonitoring program for prenatal care. Despite some challenges with adherence, the program shows promise in improving maternal health management and receiving positive feedback from participants. Overall, this study highlights the potential of RPM programs in enhancing perinatal care for vulnerable mother populations and their newborns. Future research should consider addressing barriers to patient engagement and further investigating the clinical and economic impact of telemonitoring. Additionally, it is important to assess the program sustainability to optimize its long-term clinical effectiveness and its value to patients, the health care system, and society as a whole. 14
Footnotes
Acknowledgments
The authors express gratitude to the Center for Telehealth and the Maternal Fetal Medicine team at the UMMC for their dedicated effort in implementing the program.
Authors' Contributions
Y.Z.: Conceptualization, methodology, software, validation, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, visualization. Y.-Y.L.: Conceptualization, software, investigation, writing—review and editing, visualization. L.L. and J.M.S.: Conceptualization, investigation, writing—review and editing. T.T.: Conceptualization, validation, resources, writing—review and editing, project administration. D.M.I.: Validation, resources, writing—review and editing, project administration. Y.Z.: Software, resources, data curation, writing—review and editing. S.C. and C.C.: Conceptualization, methodology, validation, investigation, resources, writing—review and editing, supervision, project administration, funding acquisition.
Disclosure Statement
L.L. is an employee of ConcertAI.
Funding Information
This work is supported by the Office for the Advancement of Telehealth, Health Resources and Services Administration, U.S. Department of Health and Human Services under cooperative agreement award no. 5U66RH31459-05-00. The information, conclusions, and opinions expressed are those of the authors, and no endorsement is intended or should be inferred.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
