Abstract
Background:
Thyroid hormones (TH) play a key role in fetal brain development. While severe thyroid dysfunction, has been shown to cause neurodevelopmental and reproductive disorders, the rising levels of TH-disruptors in the environment in the past few decades have increased the need to assess effects of subclinical (mild) TH insufficiency during gestation. Since embryos do not produce their own TH before mid-gestation, early development processes rely on maternal production. Notably, the reproductive network governed by gonadotropin-releasing hormone (GnRH) neurons develops during this critical period.
Methods:
The risk of mild maternal hypothyroidism on the development of GnRH neurons and long-term effect on neuroendocrine function in the offspring was investigated using a mouse model of gestational hypothyroidism induced by methimazole (MMI) treatment.
Results:
MMI treatment during gestation led to reduced litter size, consistent with increased miscarriages due to hypothyroidism. E12/13 embryos, collected from MMI-treated dams, had a decreased number of GnRH neurons, but the migration of the remaining GnRH neurons was normal. Cell proliferation was reduced in the vomeronasal organ (VNO), correlating with the reduced number of GnRH neurons detected in this region. Using a GnRH cell line confirmed attenuated proliferation in the absence of T3. Pups born from hypothyroid mothers had normal postweaning growth and estrus cycles, yet adult offspring had significantly more cells expressing estrogen receptor alpha in the arcuate nucleus. Notably, by adulthood, GnRH cell number and distribution was comparable with nontreated controls indicating that compensatory mechanisms occurred after E13.
Conclusion:
Overall, our work shows that mild TH disruption during gestation transiently affects proliferation of the pool of GnRH neurons within the VNO and has a long-term impact on neuroendocrine systems.
Introduction
Since the 19th century and the discovery that congenital hypothyroidism can cause cretinism, 1 thyroid hormones (TH) have been shown to play a key role in fetal brain development by regulating neurological developmental processes including neurogenesis, gliogenesis, neuronal migration, differentiation and synaptogenesis. 2,3 However, during gestation, the fetus does not start secreting its own TH before mid-gestation for humans 4 and slightly later for rodents. 5,6 As such, early neurodevelopmental processes are dependent on maternal TH production. 2,7 This time window is also critical for the development of gonadotropin-releasing hormone (GnRH) neurons, key players in the neuroendocrine system controlling reproduction. Between embryonic day 10.5 (E10.5) and E16 in mice, ∼CS16 and GW12 in humans, GnRH neurons are born in the olfactory placodes and migrate from the developing vomeronasal organ (VNO) to the hypothalamus. 8 During this critical period, changes in hormonal signaling have been shown to alter the development of this neuronal network, possibly leading to life-long reproductive dysfunctions (e.g., advanced/delayed puberty onset, hypogonadism, polycystic ovarian syndrome (PCOS)). 9 –11
During pregnancy, due to estrogen and human chorionic gonadotropin (hCG) level changes, the production of maternal triiodothyronine (T3) and thyroxine (T4) naturally varies to cope with the energetic demand of fetal development. 7 If TH levels are not monitored during gestation, mild hypothyroidism, often remains undetected because symptoms such as fatigue or weight changes, are common during pregnancy. However, deficits of TH during gestation, even mild, have been shown to increase the risk of miscarriage or preterm birth and subclinical hypothyroidism can lead to complications for both mother and baby, including offspring cognitive impairment. 12 –14 Currently, there are growing concerns about the impact of hormonal disruption during gestation, considering the rising levels of endocrine disrupting chemicals (EDCs) in the environment in the past few decades. EDCs have been shown to affect TH production and have direct effect on GnRH neuronal systems, pinpointing the importance of evaluating the transgenerational effects of maternal TH disruption, notably for the reproductive system development. 15 –18 Considering the role of TH for neurological development, we hypothesized that proliferation and/or migration of GnRH neurons, occurring during the period of maternal T3 dependance, might be affected by gestational hypothyroidism. We used a mouse model of gestational hypothyroidism to tackle this question and investigated the possible long-term consequences on reproductive function in the offspring.
Material and Methods
Animals and treatment
All procedures were approved by
For postnatal studies, dams and pups were housed under standard conditions (2–4 mice per cage). Weight was monitored after weaning and in females, vaginal opening and estrus cycles were assessed as previously described. 19 At 2 months of age, offspring mice were anaesthetized with isoflurane and euthanized by an intraperitoneal overdose of ketamine (0.2 mL/mouse). Testes were dissected and weighed. Perfusions and brain collection, postfixation and embedding were done as described above. This protocol was repeated three times for embryos and postnatal brain collection, including a total of eight control dams and nine MMI-treated dams.
Measurement of serum TH
Blood collected from dams was centrifugated at 4°C, and the serum was collected and stored at −20°C until use. Dosing of T3 and T4 was conducted in duplicate using Calbiotech ELISA kit for T3 (Al Cajon, CA; T3043T-100) or T4 (T4044T-100) using 25 or 10 µL of serum respectively.
Tissue preparation
Embryonic sections (14–16 µm) were cut in three series with a cryostat (Leica, Wetzlar, Germany), mounted on Superfrost slides (Daigger, Vernon Hills, IL), and stored at −80°C until use. Adult brains sections (35 µm) were cut in four series with a microtome and stored at −80°C in cryoprotectant solution until use. Sections were washed in PBS, mounted on Superfrost slides, and air-dried the day before staining.
Immunohistochemistry
Staining was performed using standard avidin–biotin horseradish peroxidase (HRP)/3,3-diaminobenzidine (DAB) detection procedures. Briefly, sections were postfixed with 4% formaldehyde (10 minutes) and rinsed with PBS. Antigen retrieval (pH 6.0 citrate buffer, 1 hour, 90°C) was performed for adult brain sections. Sections were then incubated in 0.3% H2O2 and 30% methanol solution (15 minutes) to suppress endogenous peroxidase activity, incubated in 10% normal horse serum, 0.3% Tx (1 hour) and placed in rabbit anti-GnRH primary antibody (SW-1; RRID:AB_2629221; 1:10000 in PBS, 1% bovine serum albumin (BSA), 0.1% NaAzide, 2 nights, 4°C) followed by donkey anti-rabbit biotinylated secondary antibody (Jackson Immunoresearch, West Grove, PA; 711-065-152; 1:500 in PBS/0.3% Tx, 1 hour). Sections were incubated with avidin-biotin-HRP solution (Vector Laboratories ABC-HRP kit PK-6100;1h) and reacted with DAB enhanced with nickel (blue)/glucose oxidase. Sections were co-stained for either phospho-histidine (rat anti-pHIS, 1:1000, Novus Biologicals, Centennial, CO), RF-amide related peptide-3 (RFRP-3; rabbit anti-GnIH, 1:10000, gift from G. Bentley, UC Berkeley) or ERα (rabbit anti-ERα, 1:5000, Millipore C1355, Burlington MA) following the same protocol, and reacted with DAB (brown). Slides were airdried overnight, dipped in xylene and coverslipped with permount (Thermofisher Scientific, Hampton, NH).
Migration assay in explants
Explants containing primary GnRH cells were made as previously described 20 and grown in serum-free medium (SFM) in a 37°C, 5% CO2 incubator for 3 days before incubation with fresh SFM containing or not 30 nM T3 (Sigma-Aldrich T6397) for 24 hours. During the last hour of this treatment, migration assay was performed by live recording of GnRH neuronal movement to assess their migration rate (details in methods supplements). At the end of the recording, explants were fixed (4% formalin, 1 hour), and stained for GnRH neurons as described above.
Microarray data on single GnRH cells and PCR
Single GnRH cells from explants were used to generate poly(A) amplified cDNA libraries. 21 Expression of TRα1, TRα2, TRβ, integrin αV, and β3 in single GnRH neurons, and Gn11 cell culture was verified by PCR (methods supplements).
Cultures of Gn11 cells 22 for pHIS staining
See Supplementary Data
Analysis
For all experiments, the number of embryos/explants/animals used in each condition is shown in Table S1 and details of immunostaining analysis are described in Supplementary Data.
Dosage of Thyroid Hormone Levels and Litter Size in Control and Methimazole-Treated Dams
Statistical tests were conducted using GraphPad Prism, using unpaired Student t-test, and multiple t-test for the comparison of two groups. A p value <0.5 was considered statistically significant. *p < 0.5; ***p < 0.01
Results
TH receptors are expressed in GnRH neurons
Analysis of microarray data obtained from GnRH neurons maintained in nasal explants cultures for 4div or 10div, showed expression of TH receptors (TR) (mean RMA value: TRα 9.2–10.4; TRβ 3.7–3.9; integrin αV/β3 11.9–11.4/4.6–4.1;), TH transporters (MCT8 5.7–8.2; MCT10 5.0–8.8; OATP1C1 3.0–3.1), and deiodinases (Dio2 3.6–3.4; Dio3 3.9–6,2) (Fig. 1A). cDNA from single GnRH neurons and Gn11 cells showed robust expression of TRα1 and TRα2 and the two subunits of the integrin αVβ3 membrane receptor known to mediate non-genomic action of TH. 23 GnRH neurons did not express TRβ and only 1/3 neurons expressed the Dio2 (Fig. 1B).
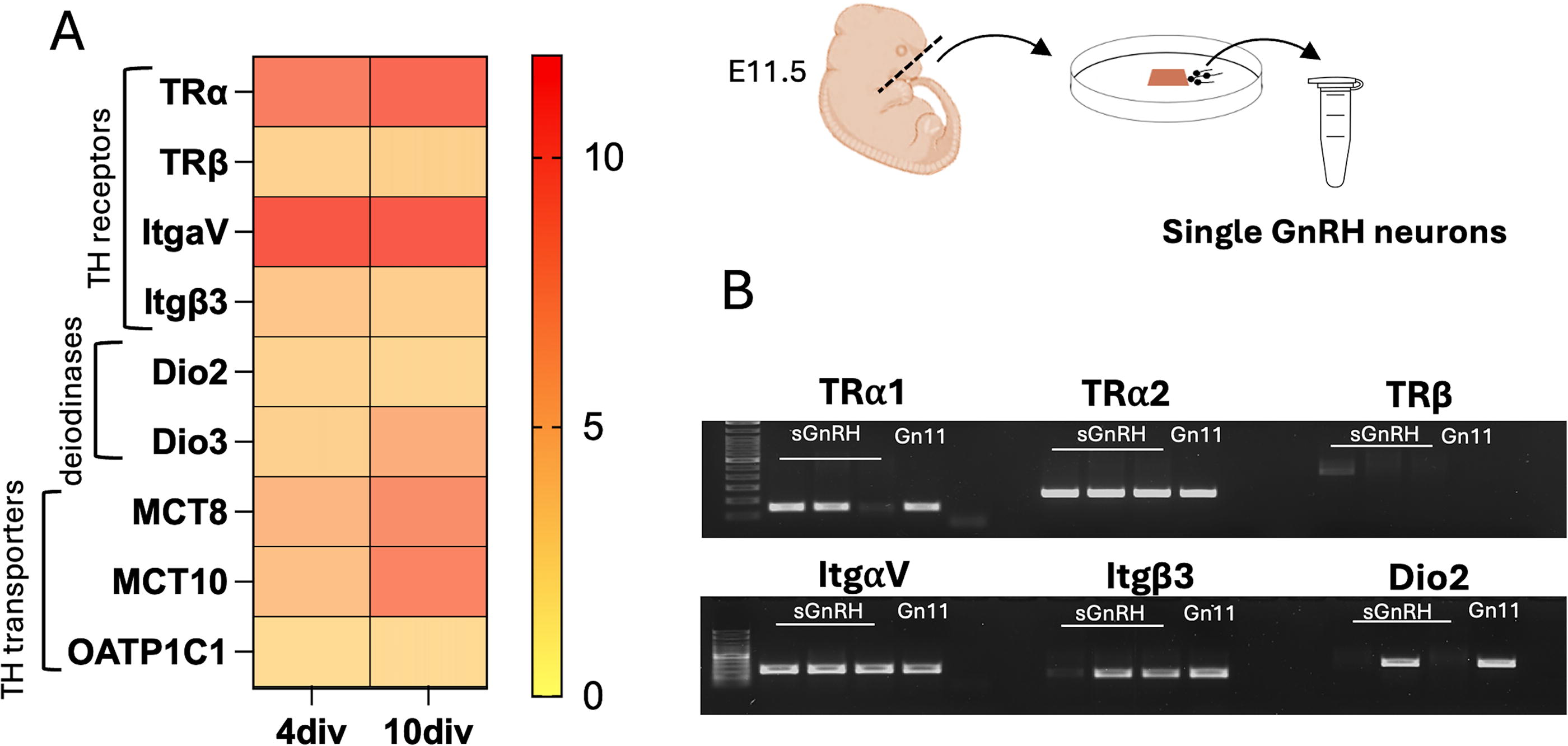
Expression of thyroid hormone (TH) signaling pathway elements in gonadotropin-releasing hormone (GnRH) neurons. Nasal region of E11.5 embryos was dissected, cultured and subsequently used to generate microarray data from single GnRH cell cDNA libraries.
MMI-induced hypothyroidism during gestation is associated with reduced litter size
To study the effect of mild hypothyroidism during gestation on the development of reproductive function, dams were treated with MMI, an inhibitor of the thyroid peroxidase (TPO), from GD6 to collection of embryos (GD 12/13) or birth (Fig. 2A). Dosage of TH in serum showed a significant decrease in T3 in dams receiving MMI, consistent with mild hypothyroidism (Fig. 2B, Table 1). MMI-treated females were found to have significatively reduced litter size, indicative of an increase in resorbed embryos in the uterus (Fig. 2C, Table 1).
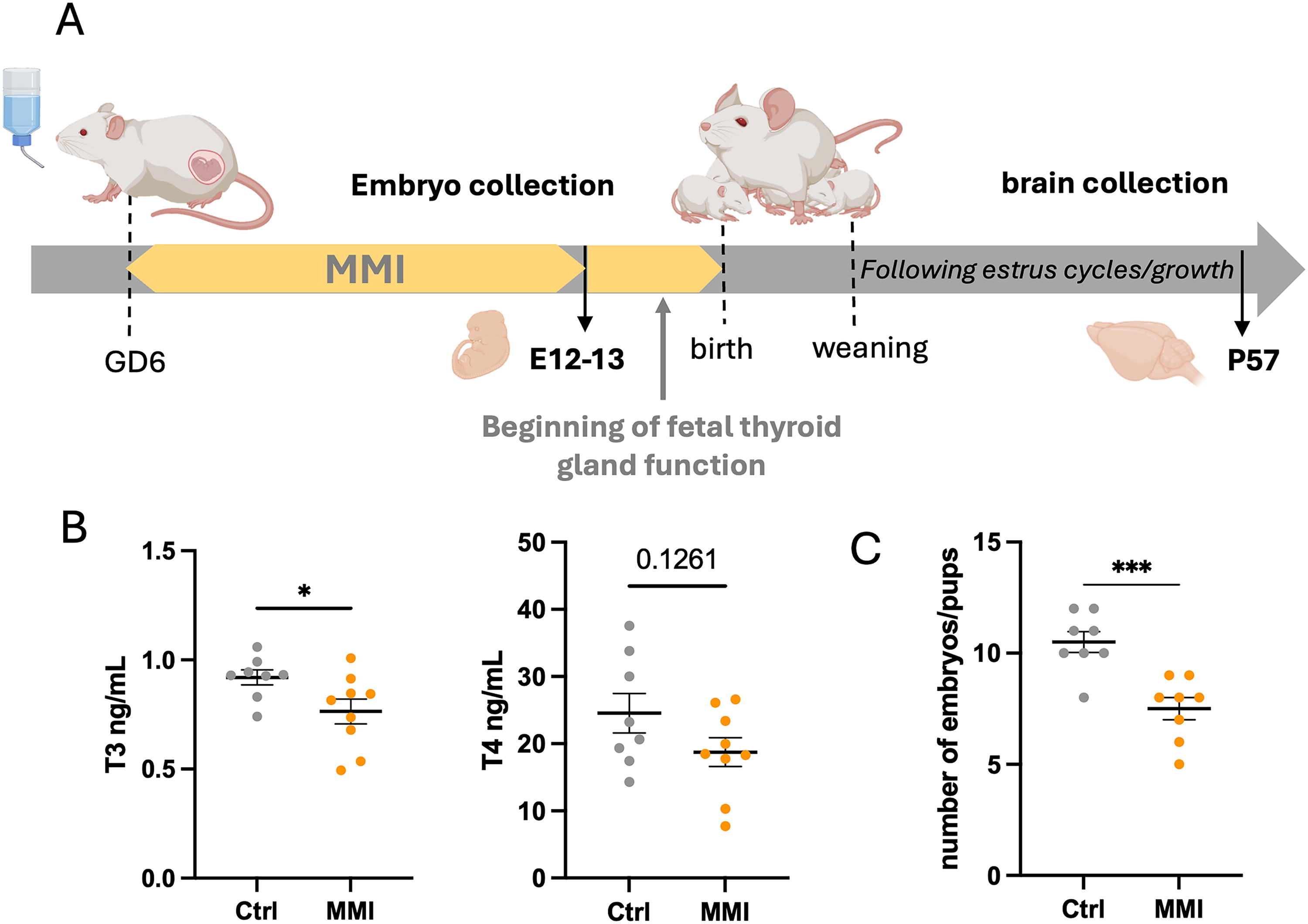
Inducing hypothyroidism during gestation.
Maternal mild hypothyroidism decreases GnRH neurons number without affecting their migration
To assess the effect of hypothyroidism during gestation on the development of GnRH neurons, embryos from control and MMI-treated mice were collected at E12 or E13, coinciding with the period in which GnRH neurons migrate along tracts from the developing VNO to the junction between the nasal region and forebrain and finally enter the brain (Fig. 3A). GnRH neurons in these areas were counted. At both E12 and E13, the total number of GnRH neurons was reduced in embryos from MMI-treated mothers (Fig. 3B). At E12, most GnRH neurons in both groups were in the VNO. However, the number of GnRH neurons in this region was significantly lower in embryos from MMI-treated dams (Fig. 3C, left). No differences were detected in the number of neurons on tracks or at the nasal forebrain junction (NFJ) at this stage. At E13, most GnRH neurons in both groups were detected on tracks between the VNO and the NFJ (Fig. 3C, right). A significant decrease in the number of GnRH neurons in this region as well as within the olfactory pit was found in the embryos from MMI-treated dams at this stage.
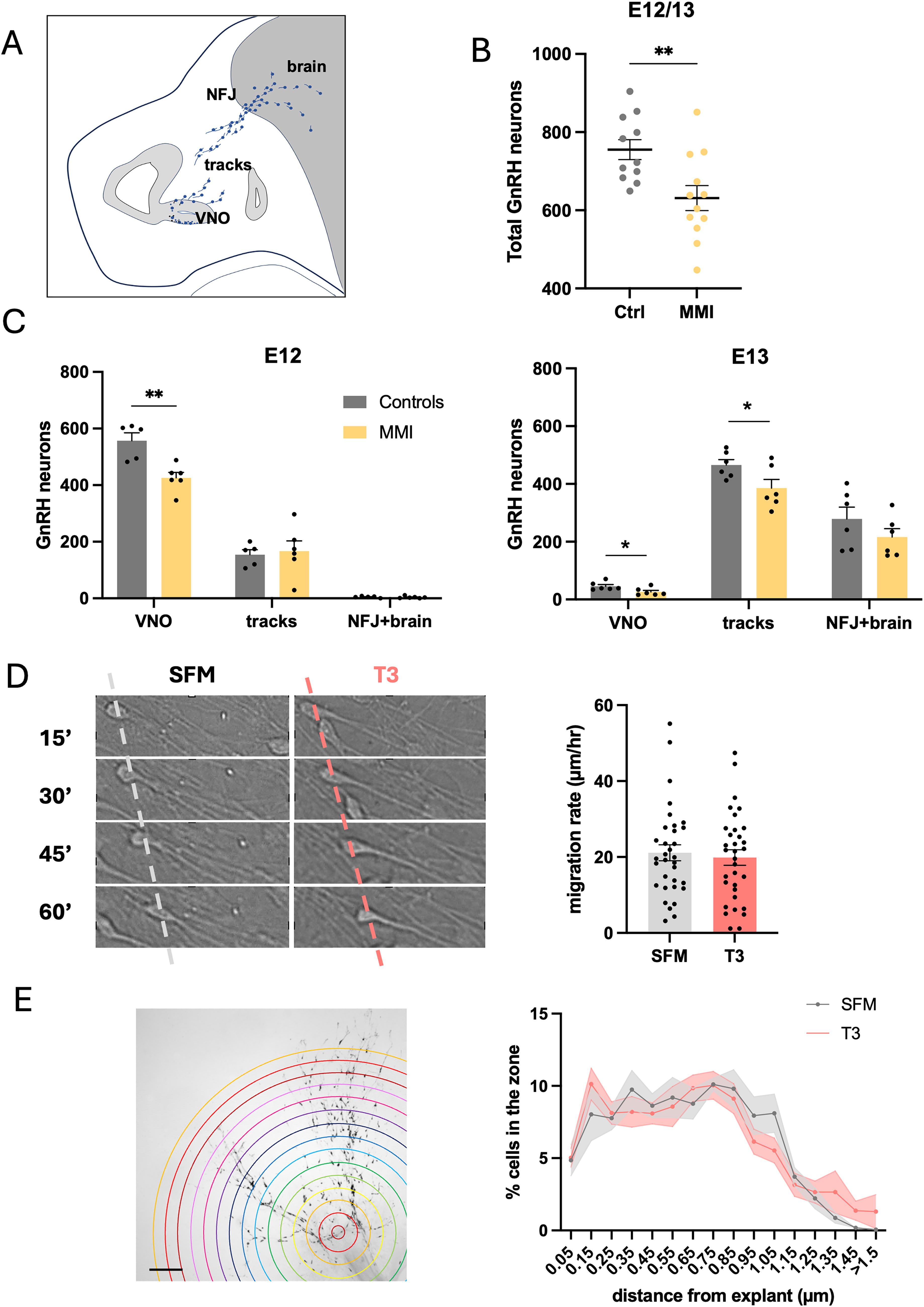
Effect of gestational hypothyroidism on GnRH neuronal number and migration.
Although the number of GnRH neurons was reduced in the embryos, no change in migration was found, with the distribution of neurons shifting from the VNO to along the tracks and at the NFJ being similar between embryos from controls and MMI-treated dams (Supplementary Figure S1). To examine this issue in a model devoid of brain cues, we used nasal explants in which GnRH neuronal migration has been well documented. 24 Nasal explants were treated for 24 hours with or without T3 (30 nM) in SFM culture medium and GnRH neurons were recorded with live imaging for 1 hour to assess their migration rate and then stained for GnRH using immunohistochemistry to assess whether a change occurred in the distance the overall GnRH neuronal population migrated after treatment. Treatment of GnRH neurons in vitro with T3 did not change their acute migration rate (Fig. 3D) nor the overall distance that the GnRH neuronal population migrated (Fig. 3E), consistent with our ex vivo data.
Maternal T3 is important for proliferation of GnRH neurons precursors during embryonic development
To assess whether the change in number of GnRH neurons found in embryos collected from hypothyroidic dams was due to a lack of cell proliferation, E12 sections were stained with the proliferation marker pHIS (Fig. 4A). pHIS+ cells were counted in the VNO as well as along the lateral ganglionic eminence (LGE) and median ganglionic eminence (MGE), areas known to be proliferating at this embryonic stage. 25 In all three zones, the number of cells expressing pHIS was significantly reduced in embryos from MMI-treated dams (Fig. 4A) and the number of pHIS+ cells in the developing VNO correlated with the number of GnRH positive cells counted (Fig. 4B).
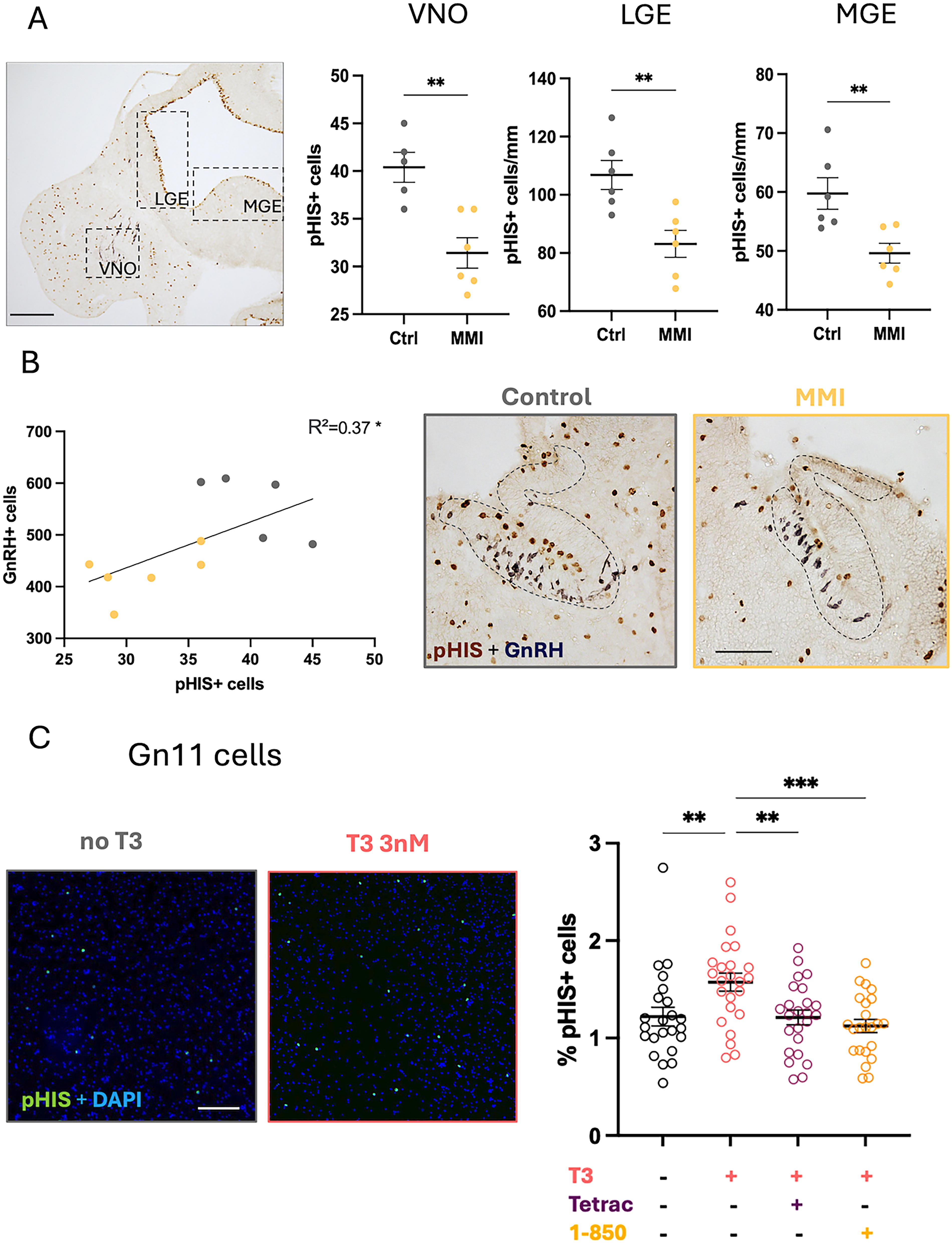
Effect of gestational hypothyroidism on GnRH cell proliferation.
To directly investigate the role of TH on GnRH cells proliferation, a GnRH cell line (Gn11) was treated with T3 (3 nM) for 24 hours. In the presence of 10% fetal bovin serum (FBS) in the medium, T3 treatment induced a significant increase in proliferation of GnRH cells, seen by an increase of the percentage of cells expressing pHIS (Fig. 4C). In the absence of FBS, a condition promoting cell survival but no proliferation, the number of GnRH cells was reduced and remained so after T3 treatment, showing no effect of T3 on cell survival (Supplementary Figure S2). The use of both an integrin αVβ3 receptor antagonist (Tetrac, 100 nM) and a TH nuclear receptor antagonist (1–850, 1 µM) prevented the proliferative effect of T3 on GnRH neurons (Fig. 4C).
Adult offspring born from hypothyroidic dams has a normal number of GnRH neurons and subventricular zone proliferation but increased estrogen receptors
To study the long-term consequences of mild hypothyroidism during gestation, a variety of parameters were assessed in pups born from dams treated with MMI until birth (Fig. 2). No difference in weight gain, from weaning (postnatal day 21, PN21) to sacrifice (PN57), was found in either sex between the control and treatment groups (Fig. 5A, left). The vaginal opening was monitored in six females from each group. Both groups reached 80% on PN34. All control mice had vaginal opening by PN35, whereas treated mice reached 100% after 2 days, PN37 (Fig. 5A, right). Estrous cycles were monitored daily after weaning in females and no change in the distribution of time spent in each estrous stage was found between groups (Fig. 5B, left). There was also no change in the testis/body weight ratio in males at PN57 (Fig. 5B, middle).
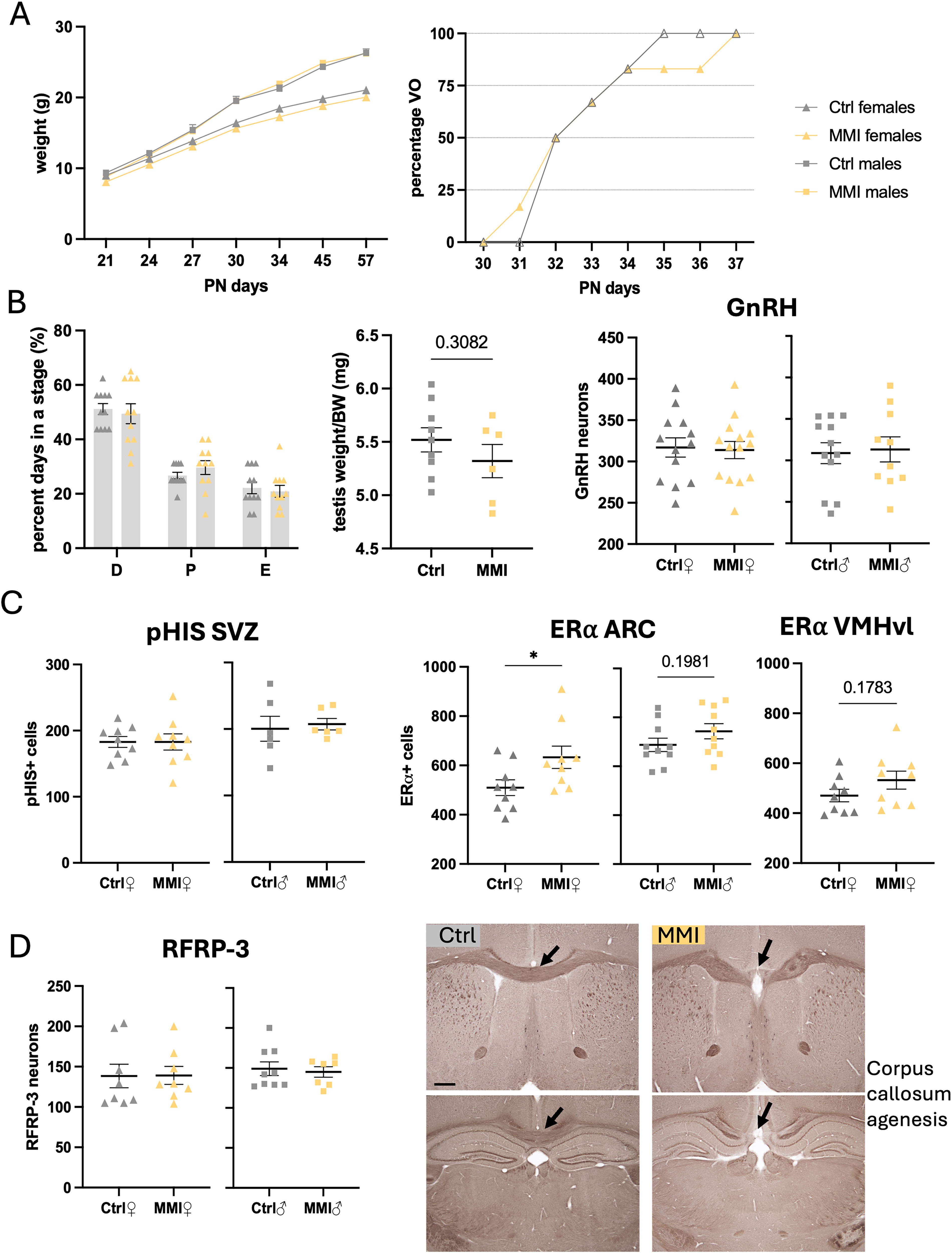
Long-term effect of gestational hypothyroidism on offspring reproductive functions, cell proliferation, and hypothalamic neuroendocrine regulation. (
In the offspring, no difference was found in the number of GnRH neurons (Fig. 5B, right) and the distribution of GnRH neurons along the rostro-caudal axis was similar in the brains of male and female mice from both groups (Supplementary Figure S3). Considering the effect of gestational hypothyroidism on cell proliferation during embryonic development, the number of pHIS+ cells in the subventricular zone (SVZ) was also assessed. No difference in cell proliferation in this zone was found between the two groups in either sex (Fig. 5C, left). In contrast, female adult mice born from MMI-treated dams had a significant increase in the number of cells expressing the ERα receptor in the arcuate nucleus (ARC). Although nonsignificant, a similar trend was also observed in the number of cells expressing the ERα in the ARC of males and in the ventromedial hypothalamus ventrolateral (VMHvl) of females (Fig. 5C, right). RFRP-3 neurons are upstream regulators of GnRH neurons located in the dorso- and ventro-medial hypothalamus, which have been previously shown to be involved in the delayed pubertal onset caused by prepubertal hypothyroidism. 26 No effect of gestational hypothyroidism on the number of RFRP-3 neurons was found in the offspring (Fig. 5D, left). Photos of immunostainings for GnRH, pHIS in the SVZ, RFRP-3, and ERα are shown in Supplementary Figure S4. Finally, 7 of the 24 mice (29%) born from MMI-treated dams (4/14 females + 3/10 males) had corpus callosum agenesis, a malformation previously associated with developmental hypothyroidism in rats and humans. 27 –30 Only 1/25 (4%) from the control group had a similar condition (Fig. 5D, right).
Discussion
It is well established that alteration of maternal TH production during gestation can negatively impact neurodevelopment and have long-lasting effect in the offspring (Bernal, 2005; Escobar et al., 2004). Most studies have focused on severe thyroid disorders and as such, there is a critical gap in assessing the effects of subtler forms of TH disruption, which are often undetected due to mild symptoms. However, subclinical hypothyroidism have been shown to also alter neurodevelopmental processes. 31 Thus, this study investigated the effect of mild gestational hypothyroidism on the development of the reproductive axis, analyzing the proliferation and migration of GnRH neurons prenatally and evaluating the long-term effects of maternal hypothyroidism on neuroendocrine systems in the offspring (Fig. 6). We first showed that, young GnRH neurons express TH nuclear receptors (TRα1 and TRα2) and both subunits of the integrin αVβ3 membrane receptor. We found that female mice treated with MMI to induce mild hypothyroidism during gestation give birth to significantly smaller litters. This result is consistent with previous reports that subclinical hypothyroidism during pregnancy increases the risk of miscarriage in humans and rodents. 32,33 The loss of embryos in the MMI-treated group is important to keep in mind when assessing the effect of maternal hypothyroidism on the next generation, as the most severely affected mice may have been resorbed. Exclusion of such mice could dampen the significance of the observed effects and as such trends are noted.
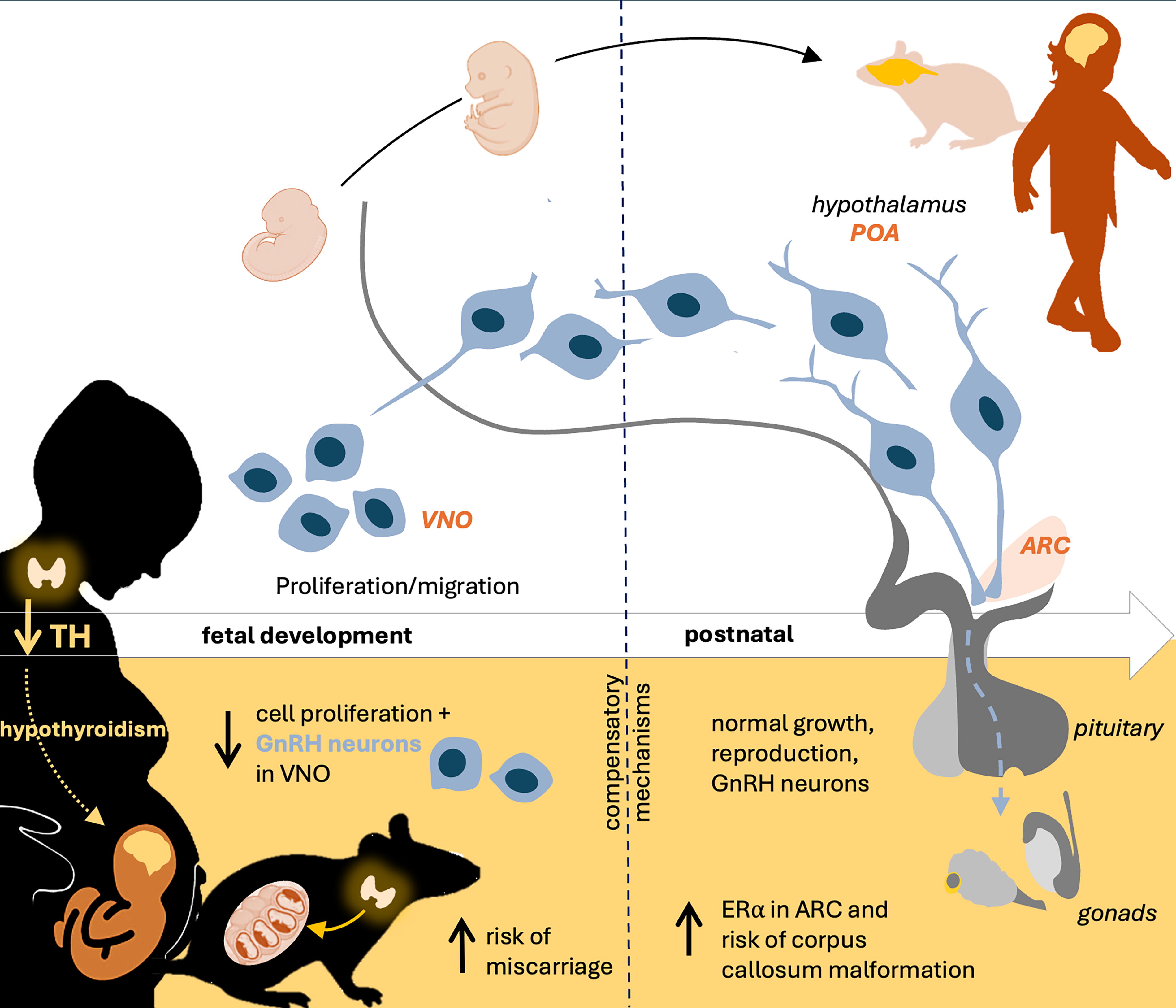
Graphical summary of the developmental and long-term effect of maternal hypothyroidism during gestation in offspring.
Decreased number of GnRH neurons due to altered embryonic cell proliferation
Embryos collected from hypothyroid females had fewer GnRH neurons, with a marked decrease in newly differentiated GnRH cells in the olfactory pit. Staining with a proliferation marker showed that cell proliferation is reduced in these embryos in the VNO as well as in the LGE and MGE, brain regions normally exhibiting robust cell proliferation. Consistent with TH altering generation of GnRH neurons in embryos, the addition of T3 to the culture medium of immortalized GnRH cells in vitro increased proliferation and this effect was blocked by TR antagonists. Of note, during gestation, maternal T4 is the main TH reaching the fetal brain where it is locally converted to T3 by the Dio2. 34 A small amount of T4 is present in our culture medium (approx. 20 nM) and although Dio2 is not expressed in all GnRH neurons from explants, its presence in some of these cells and in Gn11 cells suggests that T4 might contribute to the proliferative effect observed in vitro. The importance of TH to regulate cell proliferation, especially during fetal development, has been extensively studied 35 –37 and developmental hypothyroidism has been previously shown to decrease SVZ and cortical proliferation. 38,39 Here we show for the first time that hypothyroidism also decreases proliferation in the VNO, reducing the embryonic pool of GnRH neurons. Vancamp and collaborators have shown a long-term effect of gestational severe hypothyroidism on olfactory memory in the offspring, which they attribute to changes in the SVZ. 39 A major difference in the behavioral test to male urine was noted in this paper, unfortunately this study did not examine the glomeruli of the accessory olfactory bulb, known to be involved in pheromonal processing. 40 The impaired proliferation in the VNO during embryonic development noted in the present study could have contributed to the altered function Vancamp et al. noted in adults. TH have also been reported to be involved in neuronal migration. 36,41 –43 However, in the present study, both hypothyroidism and treatment of nasal explants in vitro with T3 did not change migration of GnRH neurons. These results suggest that while TH are needed for proliferation of GnRH progenitors, they have a limited effect on their differentiation and migration.
Possible compensatory mechanisms during offspring development
Although the number of GnRH neurons was reduced during embryonic development, the number of GnRH neurons in adult offsprings was not different between animals born from control and MMI-treated females, indicating that compensatory mechanisms occurred. Indeed, while the proliferation of GnRH neurons progenitors in mice stops around E14.5, 44 the fetal thyroid gland starts functioning around E16.5-E17; 5 therefore, endogenous local TH production may allow proliferation to resume. Alternatively, but not exclusively, GnRH neuronal survival may be altered. A total mean of 790 GnRH neurons was counted in control E13 embryos, while adult controls had an average of 626 neurons, suggesting that a small portion of GnRH neurons may die or remain stuck in nasal areas before reaching their final destination (approximately 21%). In embryos from hypothyroidic mothers, the reduced amount of GnRH neurons (16.5% decrease) could favor successful migration into the hypothalamus. This hypothesis is supported by the fact that the distribution of GnRH neurons along the migratory path and in adults is not affected by TH disruption. A recent clinical study showed that boys born from hypothyroidic mothers have decreased gonadotropins and testosterone levels during the first 6 months of life, 45 but another clinical study found no effect of maternal hypothyroidism on the timing of puberty onset in both sexes. 46 These studies suggest that in humans, maternal thyroid dysfunction could alter minipuberty, the early postnatal period of transient activation of the reproductive system, but that prepubertal compensatory mechanisms might occur resulting in unaltered puberty. Other embryonic and perinatal development stages should be evaluated to better understand possible compensatory mechanisms.
Effect of maternal hypothyroidism on other neurons and interaction with estrogen receptors
In seasonal species, the reproductive axis is tightly regulated by TH to ensure the appropriate timing of sexual maturation needed for reproductive success and offspring survival. 47 The TH-driven seasonal control of reproduction seems to involve an indirect regulation of GnRH as the number of GnRH neurons and Gnrh transcription levels remain relatively stable through the year. RFRP-3 (gonadotropin-inhibitory hormones) and Kisspeptin neurons have been proposed to mediate the effect of T3 on GnRH neurons. 48 In addition, postpubertal treatment of rats to induce gestational hypothyroidism has been reported to have no effect on the GnRH mRNA expression but modified the distribution of its receptor in the pituitary. 32 Altogether, these studies show the potential effect of maternal hypothyroidism on different neuronal populations, including upstream and downstream regulators of GnRH neurons. In fact, RFRP-3 has been reported to regulate GnRH neuronal activity and is increased with hypothyroidism, causing delayed puberty. 26 In the present study, in MMI-treated offsprings, no changes in RFRP-3 number were found in either sex. However, Kiyohara and collaborators 26 induced hypothyroidism just before puberty, whereas in our study, hypothyroidism was indirectly induced via the dam during embryonic development, as such the endogenous TH production in the offspring should be intact prior to puberty. In contrast to our findings, proliferation in the SVZ in adult offspring was reported to be altered by gestational hypothyroidism. 39 Again, the difference in results might be due to the timing of treatment, which continued after birth until PN21 in the Vancamp study. 39 Altogether, these results show that disruption in TH production during the first few weeks after birth can be critical and result in long-term effects while maternal hypothyroidism is detrimental for fetal development but some of the impaired processes are compensated for, after the fetus starts producing its own TH.
Although MMI treatment during gestation, did not alter weight gain, testis weight and estrus cycles in offspring, developmental hypothyroidism led to an increased number of cells expressing ERα in the ARC. TH and estrogen bind to nuclear receptors, and crosstalk exists between TRs and estrogen receptors (ERs). Indeed, TR and ER have similar binding sites resulting in possible competitive binding between the two receptors which may have antagonistic or compensatory effects. 49 –51 In addition, using PROMO tool, we found TR binding sites in the promoter sequence of ERα. A direct effect of TH on the expression of the receptor is therefore possible, although a study in pituitary cells reported a transcription-independent effect of TH of ERα levels by preventing the degradation of the receptor. 52 Various clinical and experimental studies have highlighted the interaction between these two hormonal systems in adults, with a mutual effect of estrogen treatment altering TH production and hypothyroidism leading to high levels of estrogen. 53 –56 A clinical study showed that children born from women exposed to high levels of estrogen during early pregnancy have increased risks of thyroid disfunction. 57 In addition, studies using EDCs including TH disruptors, such as bisphenol, have shown transgenerational effects of these EDCs on estrogen and ER levels in the offspring. 58,59 The present study shows long-term effects of maternal alteration of TH on hypothalamic ERα in the offspring. Considering the role of the ARC in reproductive and metabolic functions, 60 developmental hypothyroidism could alter the normal functioning of these cell populations by modifying their sensitivity to estrogen. Kisspeptin neurons located in the ARC express ERα receptors and have a major role in controlling the GnRH pulse generation. 61 An increase in estrogen feedback to kisspeptin neurons could result in an overstimulation of GnRH neurons, possibly leading to endocrine disorders such as PCOS. 62 Many clinical studies have correlated PCOS with TH disorders but the mechanistic link between both disorders remains largely unknown. 63 –65 In our study using a mild hypothyroidism model, reproductive function was not impaired, but additional hormonal dosages and evaluation of ovarian tissues in females could be evaluated to assess other potential effect(s) of increased ERα expression.
Increased risk of the agenesis of the corpus callosum, evidence in favor of universal thyroid screening during pregnancy
The corpus callosum is a structure connecting both hemispheres in the brain. Agenesis of the corpus callosum (ACC) is characterized by the absence of corpus callosum crossing and is often associated with mutations in axon guidance receptors and periventricular heterotopia. 66 We observed an increased incidence of ACC and heterotopia in adult mice born from hypothyroid dams. Such malformations due to developmental hypothyroidism have been previously reported in both animal models and humans. 27 –30 Symptoms associated with this malformation are variable ranging from no noticeable effects to severe cognitive and/or physical dysfunctions. 67,68 In rodent studies, different reports using TPO inhibitors have shown that the frequency of these malformations depends on the timing and dose of exposure. 69 In particular, the percentage of pups affected increases with prenatal treatment, highlighting the importance of this critical window and timing of treatment. 28,70 Heterotopia resulting from developmental hypothyroidism appears less frequent in mice than rats, 71 nonetheless, various TPO inhibitors have been shown to induce malformation, 69,72,73 supporting the fact that this permanent effect is triggered by TH deficiency during development rather than a direct action of antithyroid drugs. These findings are crucial in the debate for universal screening of TH during pregnancy, as increased risks of heterotopia have now been associated with subclinical/mild gestational hypothyroidism in multiple studies.
Although universal screening for TH during pregnancy has been adopted in several countries such as Spain and India, many others are only screening women at risk as recommended by the American Thyroid Association guidelines. 74,75 However, several studies in humans and animal models show detrimental effects of maternal subclinical (mild) hypothyroidism on fetal development can have long-lasting cognitive and physiological effects in the offspring. 2,7,12,13,30,31,39 In this study we demonstrated that mild maternal hypothyroidism decreases cell proliferation during embryonic development, including GnRH progenitors in the olfactory pit. While there seems to be a compensatory mechanism regulating the number of GnRH in the adult offspring, we found a long-term effect of gestational hypothyroidism on the estrogen system. An increase in hypothalamic ERα may lead to hormonal imbalance and aberrant stimulation of the reproductive axis in the adults. Given the significant increase in environmental EDCs these past few decades, more than ever, experimental and clinical studies are needed to evaluate the neurodevelopmental risks associated with maternal thyroid disruption and their consequences on pregnancy and long-term physiological outcomes in the offspring.
Footnotes
Acknowledgments
The authors thank the NINDS animal facility personnel and IT department for their support. The authors also thank Zaria Wilson, Ndubuisi Ugochuku, Jacob Short and Drs. Amy Moritz, Naira Mansano, and Maleeha Akram for their technical help and valuable insights on the paper.
Ethic Statement
The animal study was reviewed and approved by the
Authors’ Contributions
C.Q. and S.W. conceptualized and designed experiments. C.Q., A.B., J.K., and H.B. performed experiments. C.Q. and S.W. analyzed data. C.Q. and S.W. wrote and reviewed the article. All authors contributed to the article and approved the submitted version.
Author Disclosure Statement
The authors declare no competing interests.
Funding Information
This work was supported by the Intramural Research Program of the National Institutes of Health, National Institute of Neurological Disorders and Stroke (ZIA NS002824-34).
Supplementary Material
Supplementary Data
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
