Abstract
Aim:
The study aimed to analyze the long-term outcomes of [177Lu]Lu-DOTAGA.FAPi dimer therapy in individuals diagnosed with radioiodine-resistant (RAI-R) follicular cell-derived thyroid cancer.
Materials and Methods:
In this retrospective study, 73 patients with RAI-R follicular thyroid carcinoma who had undergone multiple lines of previous treatments were included. Following [68Ga]Ga-DOTA.SA.FAPi positron emission tomography–computed tomography scan, among the 73 patients, 65 received [177Lu]Lu-DOTAGA.FAPi dimer monotherapy with a median activity of 5.5 GBq per cycle at 8-week intervals. The remaining eight patients underwent tandem [177Lu]Lu/[225Ac]Ac-DOTAGA.FAPi dimer therapy, consisting of a median of two cycles of [177Lu]Lu-DOTAGA.FAPi dimer followed by one cycle of [225Ac]Ac-DOTAGA.FAPi dimer, also at 8-week intervals. The primary endpoint included progression-free survival (PFS) and overall survival (OS). Secondary endpoints included PERCIST criteria response assessment and safety assessment according to Common Terminology Criteria for Adverse Events (V5.0).
Results:
We enrolled 37 female and 36 male patients, with a mean age of 54.3 years (range: 27 − 80 years). The patients received a median cumulative activity of 22.2 GBq (range, 4 GBq–55.5 GBq) of [177Lu]Lu-DOTAGA-FAPi dimer over one to nine cycles, with a median of three cycles. Among 73 patients, 20 died and 16 deaths were due to thyroid cancer. Nineteen patients experienced disease progression, with an estimated median PFS of 29 months [CI 14–34 months]. The estimated median OS was 32 months [CI 21–40 months]. Four patients (5.4%) encountered grade III anemia, primarily linked to bone metastasis in three cases and neck tumor mass bleed in one. Grade III thrombocytopenia occurred in three patients (4%). No grade III renal or hepatotoxicity was observed.
Conclusion:
In this study, [177Lu]Lu-DOTAGA.FAPi dimer therapy showed promising safety and efficacy in aggressive, radioiodine-resistant thyroid cancer, achieving a median PFS and OS of 29 and 32 months, respectively, with manageable adverse events. Confirmation of our findings is needed from prospective clinical trials comparing [177Lu]Lu-DOTAGA.FAPi dimer therapy to other treatments.
Introduction
Follicular cell-derived thyroid cancer (FCTC) encompasses various histological types, from slow-growing to aggressive tumors. Differentiated thyroid cancer (DTC), including papillary and follicular types, accounts for over 90% of cases with a favorable 10-year survival rate of 80–95%. Poorly differentiated thyroid carcinoma (PDTC) and anaplastic thyroid cancer (ATC) are rare but aggressive, comprising 5% and 1% of cases, respectively, with ATC known for rapid progression. 1 –3 Radioiodine-refractory disease in differentiated thyroid cancer (RR-DTC) is linked to an increased risk of death, with a 10-year survival rate of only 10%, often necessitating multimodal treatment incorporating systemic therapies. 4 –7
The therapeutic approach to radioiodine-refractory thyroid cancer has advanced with tyrosine kinase inhibitors (TKIs) such as sorafenib and lenvatinib, supported by the DECISION trial and SELECT study results. 8,9 However, their potential risks, including significant side effects, require cautious consideration, particularly in patients with comorbidities such as cardiac issues, hypertension, stroke, and kidney dysfunction, who may not be optimal candidates for TKI therapy. Recently, [177Lu]Lu-fibroblast activation protein (FAP) inhibitor therapy marks a significant advancement in the field of thyroid cancer theranostics. In thyroid cancer, FAP in cancer-associated fibroblasts (CAFs) influence the tumor microenvironment (TME), angiogenesis, and chemotherapy resistance. CAFs interact with cancer cells, fostering tumor growth, desmoplasia, and aggressiveness, with CAF presence correlating with genetic mutations such as BRAFV600E, highlighting their significance as therapeutic targets and in elucidating tumor progression mechanisms. 10
Radiolabeled FAP inhibitors (FAPIs), such as [68Ga]Ga-FAPI-04, -46, 2286, and DOTA.SA.FAPi, have shown promising results in imaging various malignancies. 11 –17 Initial studies on [68Ga]Ga-DOTA.SA.FAPi in 54 patients with 14 cancer types revealed high detection rates for primary tumors and metastases, particularly brain metastases. In medullary and FCTC, [68Ga]Ga-DOTA.SA.FAPi outperformed [18F]F-FDG and [68Ga]Ga-DOTANOC positron emission tomography–computed tomography (PET/CT), indicating its potential for diagnosing and treating RR-DTC. 14 –16 Significant progress has been achieved in therapeutics with [177Lu]Lu-labeled FAP inhibitors, addressing a key challenge of rapid clearance seen in initial small molecule FAPIs. Moon et al. 18 and Martin et al. 19 developed [177Lu]Lu-labeled FAP inhibitors such as DOTAGA.(SA.FAPi)2, showing enhanced tumor residence time and uptake compared to DOTA.SA.FAPi. These radiolabeled FAP inhibitors hold promise for diagnostic and therapeutic interventions in radioiodine-refractory differentiated thyroid cancer (RAI-R DTC). Specifically, [177Lu]Lu-DOTAGA.FAPi dimer therapy has advanced significantly in treating various thyroid cancer types, demonstrating higher tumor absorbed doses and efficacy in patients with RAI-R DTC, as evidenced by improved performance scores, reduced serum thyroglobulin levels, and a disease control rate of 88% in a pilot study. 20,21
Based on promising initial observations, our study aimed to evaluate the long-term outcomes regarding the safety and efficacy of [177Lu]Lu-DOTAGA.FAPi dimer therapy across diverse radioiodine resistant FCTC (RAI-R FCTC). Our objective is to evaluate survival outcomes and identifying factors that influence the therapeutic effectiveness of [177Lu]Lu-DOTAGA.FAPi dimer treatment.
Material and Methods
The study received ethical approval from the Institute Ethics Committee of the All India Institute of Medical Sciences (AIIMS), India (Ref No: IEC 483, dated June 5, 2020). Patient enrolment occurred between November 2020 and March 2024. The research was carried out at the Department of Nuclear Medicine in collaboration with the Department of Medical Oncology at AIIMS and the Department of Chemistry at Johannes Gutenberg University, Mainz, Germany, which supplied the DOTA.SA.FAPi and DOTAGA.FAPi dimer compounds. The study adhered to the principles of the Declaration of Helsinki, and all patients provided written informed consent prior to treatment. The local interdisciplinary tumor board assessed patient eligibility for [177Lu]Lu/[225Ac]Ac-DOTAGA.FAPi dimer therapy, and patients were informed of the investigational nature of the treatment before administration. The study is based on real-world observations and reports a single-center experience in real-life clinical scenarios. Adherence to STROBE was followed to ensure transparent and accurate reporting of the study’s observational data.
Patient selection
Patients with RAI-R FCTC, including those with DTC, high-grade DTC, and high-grade follicular cell-derived poorly differentiated thyroid cancer (HG-FC-PDTC), were included in the study. Specifically, patients with locally advanced or metastatic stage disease who underwent [68Ga]Ga-DOTA.SA.FAPi PET/CT scans were considered for this retrospective analysis. Inclusion criteria comprised individuals who were over 18 years old and did not have any known inflammatory conditions, dual malignancies, or pregnancy. Histopathological evaluation was conducted on all patients, including those with primary tumors and suspicious lymph nodes. Exclusion criteria included impaired bone marrow function (defined as hemoglobin concentration <7 g/dL, platelet count <50 × 103/L, or white blood cell count <2 × 109/L), compromised renal function (glomerular filtration rate <30 mL/min/1.73 m2), impaired liver function (albumin concentration <2.5 g/L), or uptake lower than twice the physiological uptake in the normal pancreas on [68Ga]Ga-DOTA.SA.FAPi PET/CT imaging.
Out of 105 thyroid cancer patients treated with [177Lu]Lu-DOTAGA-FAPI dimer at our institution, including 23 with medullary thyroid cancer and nine with ATC, 73 patients had FCTCs and all met the inclusion criteria for this study (Fig. 1).
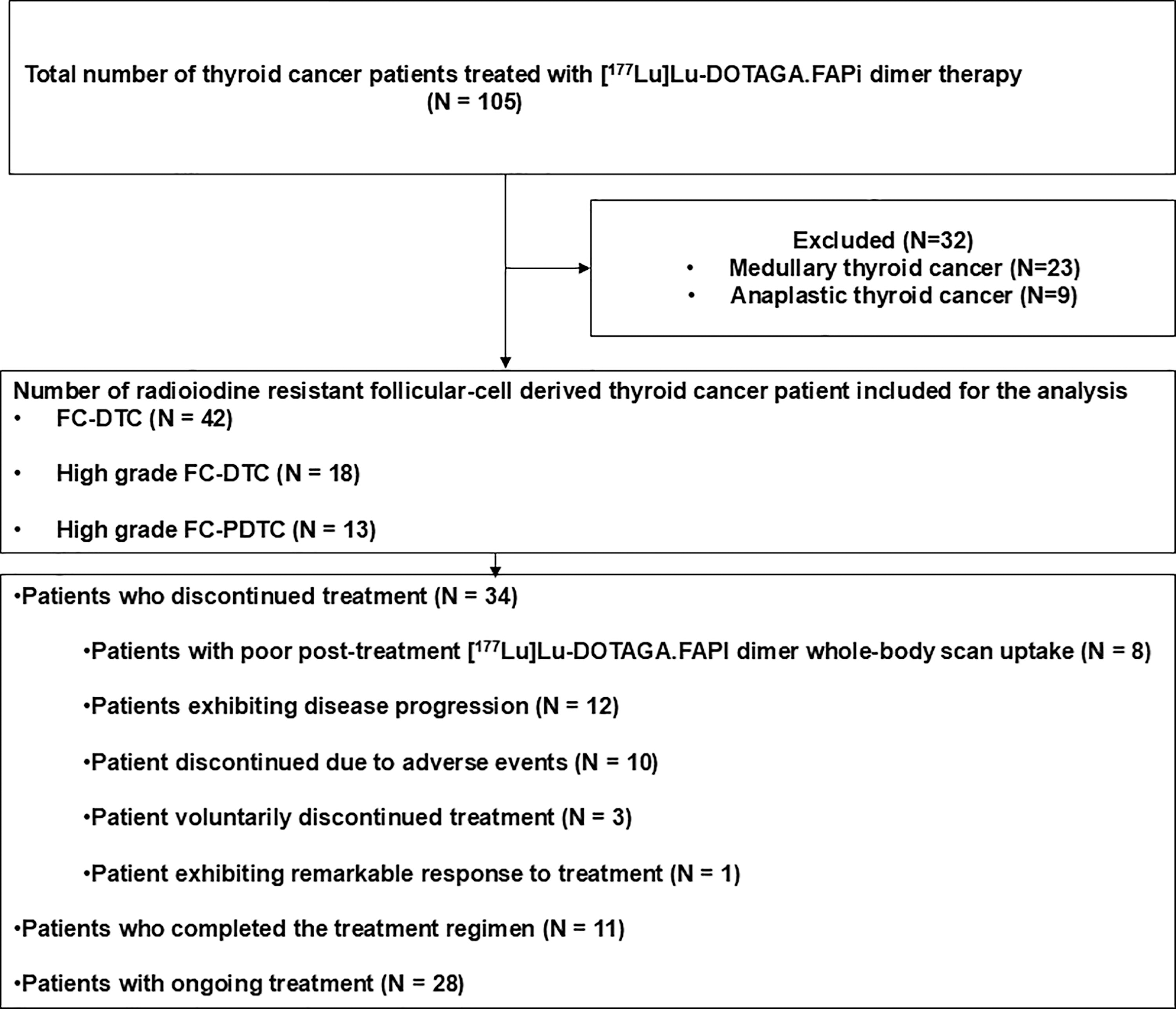
Participant flow chart for study on FAPI therapy in thyroid cancer. FAPI, FAP inhibitor; FC-DTC, follicular cell-derived differentiated thyroid cancer; FCTC, follicular cell-derived thyroid cancer, N, number of patients.
Triiodothyronine (T3), thyroxine (T4), and thyrotropin (TSH) tests were conducted using radioimmunoassay techniques. TSH and thyroglobulin tests were performed using enzyme-linked immunosorbent assay. All patients remained on a suppressive dose of thyroxine according to the American Thyroid Association (ATA) guidelines throughout the study, with no interruptions in thyroid hormone therapy.
Definition of RR-DTC
The definition of RR-DTC was adopted as per ATA 2015 guidelines, as structural progressive disease according to RECIST1.1 criteria, 22 with no further indication for radioiodine treatment, or because of partial or complete lack of RAI uptake, or evidence of disease progression despite RAI avidity at the time of treatment or after receiving cumulative RAI activity of 22.2 GBq. 5
[177Lu]Lu-DOTAGA.FAPi dimer treatment protocol
Following the [68Ga]Ga-DOTA.SA.FAPi PET/CT scan, the treatment protocol for [177Lu]Lu-DOTAGA.FAPi dimer involved intravenous administration of the radiopharmaceutical solution and 30 mL of 0.9% normal saline over 10 minutes, followed by a saline flush. Patients were closely monitored for adverse reactions post-administration before discharge. The median activity per injection was 5.5 GBq per cycle, administered at 8-week intervals. Additionally, eight patients received tandem [177Lu]Lu/[225Ac]Ac-DOTAGA.FAPi dimer therapy. In the tandem [177Lu]Lu/[225Ac]Ac-DOTAGA.FAPi dimer therapy regimen, patients received a median of two cycles of [177Lu]Lu-DOTAGA.FAPi dimer followed by one cycle of [225Ac]Ac-DOTAGA.FAPi dimer, repeated in succession. The interval between each treatment cycle was consistent with the standard approach, maintaining an 8-week interval between treatments. Post-treatment [177Lu]Lu-planar whole-body scans were performed at 24 hours using a dual-headed gamma camera equipped with high-energy general-purpose collimators (GE, Discovery NM/CT 670).
Follow-up
Patient medical records were reviewed to identify adverse events related to hematological, gastrointestinal, renal, and hepatic systems. Follow-up care, including laboratory testing at 2-, 4-, and 8-week intervals, was provided to all patients. Interim [68Ga]Ga-DOTA.SA.FAPi PET/CT scans were conducted in some patients 6 − 8 weeks after the three or four cycles of therapy. Tumor response imaging evaluations were performed every 3 − 6 months during the post-treatment regimen follow-up phase. Information regarding patients lost to follow-up or still alive at the time of the primary analysis was censored based on their latest known status date. Treatment was postponed for patients with abnormal profiles, and if toxicity persisted, treatment was permanently discontinued.
Efficacy
Endpoints.
Primary endpoint
Overall survival (OS) was calculated from the start of treatment until death from any cause. Progression-free survival (PFS) was defined as the time from treatment initiation to disease progression, which included clinical, biochemical, and molecular/metabolic progression according to the PERCIST criteria, or death from any cause.
Secondary endpoints
Functional imaging response assessment was conducted using [68Ga]Ga-DOTA.SA.FAPi/[18F]F-FDG PET/CT scans according to the PERCIST criteria. 23 Complete molecular response (CMR) is defined as the complete resolution of [68Ga]Ga-DOTA.SA.FAPi/[18F]F-FDG uptake in the measurable lesions, such that uptake is less than the mean liver activity and indistinguishable from surrounding background blood-pool levels. The findings were correlated with morphological imaging (CT) for confirmation. Partial molecular response (PMR) is defined as a minimum reduction of 30% in the target measurable tumor [18F]F-FDG/[68Ga]Ga-DOTA.SA.FAPi standardized uptake value (SUVpeak), with no appearance of new lesions. Stable disease is defined as not meeting the criteria for CMR, PMR, or progressive molecular disease (PMD). PMD is defined as a 30% increase in [18F]F-FDG/[68Ga]Ga-DOTA.SA.FAPi SUVpeak. Safety evaluations were conducted periodically throughout the study. Adverse events were assessed in accordance with the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. 24
Statistical analysis
The study utilized descriptive statistics to depict categorical variables as counts and percentages, while the D’Agostino–Pearson test assessed normality for continuous data. Normally distributed continuous variables were expressed as mean with confidence intervals (CI), standard deviation, and range, while skewed data were summarized using median and interquartile range. Pre- and post-therapy parameters were compared using either paired-sample t-tests (parametric) or Wilcoxon signed-rank tests (nonparametric). Survival analysis for OS and PFS was conducted, and Kaplan−Meier survival plots were generated. The median survival time and its CI were calculated using the Brookmeyer and Crowley method, 25 a nonparametric procedure for comparing median survival times across k-independent samples of censored data. Statistical analyses were conducted using MedCalc software, with significance set at a p-value <0.05. The comparison of baseline [18F]F-FDG and [68Ga]Ga-DOTA.SA.FAPi SUV values, as well as the investigation of the association between prior TKI treatment and FAPI expression across different lesion sites, were secondary analyses that were not pre-specified. Therefore, these analyses are considered post hoc.
Results
Patient characteristics
The detailed patient demographics and clinical details are listed in Table 1. The study included 73 patients with follicular-cell derived thyroid cancer, with a mean age of 54.3 years. For the resection of the primary thyroid tumor, 63 patients (86.3%) underwent total thyroidectomy, 2 patients (2.7%) underwent hemithyroidectomy, and for the remaining 8 patients (11%), the primary thyroid tumor was deemed inoperable. Additionally, neck dissection was performed in 41 cases (56%). Histopathological analysis showed 42 patients with follicular cell-derived differentiated thyroid cancer (FC-DTC), 18 with high-grade FC-DTC, and 13 with HG-FC-PDTC. Radioiodine treatment was administered to 52 patients, with various reasons for discontinuation. TKIs were prescribed to 46 patients, with different treatment regimens. The median duration of TKI treatment was 7 months. The median interval between discontinuation of TKI therapy and the start of [177Lu]Lu-DOTAGA.FAPi dimer treatment was 3 months. External beam radiation therapy was administered to 4 patients targeting the primary tumor and to 31 patients for metastatic sites. The median baseline thyroglobulin level was 213 ng/mL.
Patient Demographic and Clinical Characteristics
FC-DTC, follicular cell-differentiated thyroid cancer; FC-PDTC, follicular cell poorly differentiated thyroid cancer; GBq, gigabecquerel; mCi, millicurie; MRND/CCND, modified radical neck dissection/central compartment neck dissection; PET/CT, positron emission tomography–computed tomography; RAI, radioiodine; SD, standard deviation.
Dosage information for [177Lu]Lu-DOTAGA.FAPi dimer treatment
The patients received a median cumulative activity of 22.2 GBq (range, 4–55.5 GBq) of [177Lu]Lu-DOTAGA-FAPi dimer over one−nine cycles, with a median of three cycles. The median interval between treatment cycles was 8 weeks (range, 6–12 weeks). Four patients completed the treatment regimen, and 40 patients received a cumulative dosage exceeding 14.8 GBq. In the study cohort, a total of 235 treatment cycles were administered. Specifically, 7 patients underwent one cycle, 26 received two cycles, 15 were treated with three cycles, 12 underwent four cycles, and 3 received five cycles. Six patients completed six cycles of treatment, while four patients received seven, eight, or nine cycles each. Patient follow-up durations ranged from 6 to 40 months, with a median follow-up period of 3 years. The median number of cycles administered across all patients was four, ranging from one to nine cycles. Additionally, eight patients underwent tandem therapy involving both [177Lu]Lu and [225Ac]Ac-DOTAGA.FAPi dimer treatment, comprising a median of three cycles of [177Lu]Lu and two cycles of [225Ac]Ac-DOTAGA.FAPi dimer therapy. A total of 19 cycles of [225Ac]Ac-DOTAGA.FAPi were administered. The median cumulative activity for [225Ac]Ac-DOTAGA.FAPi dimer was 14.8 MBq (range: 7.4 − 25.9 MBq) (400 µCi [range: 200 − 700 µCi]), with a median of 7.7 MBq per cycle (210 µCi) over a median of two cycles. Among the seven patients who underwent only one cycle of treatment, treatment was discontinued in four patients due to the absence of promising uptake in the post-treatment scan with [177Lu]Lu-DOTAGA.FAPi dimer. The remaining three patients exhibited biochemical disease progression, leading to treatment cessation. Additionally, three patients received palliative radiotherapy for painful skeletal lesions, after completing three cycles of [177Lu]Lu-DOTAGA-FAPi dimer.
Extent of disease on [68Ga]Ga-DOTA.SA.FAPi PET/CT scan
[68Ga]Ga-DOTA.SA.FAPi PET/CT scans identified residual or remnant disease in the thyroid bed for 40 patients and lymph node involvement in 45 patients. Metastases were found in the following sites: lung involvement in 49 patients, bone in 36 patients, liver in 14 patients, and brain in 4 patients. Three patients had muscle and one patient had adrenal gland metastases (Table 1).
Progression-free survival
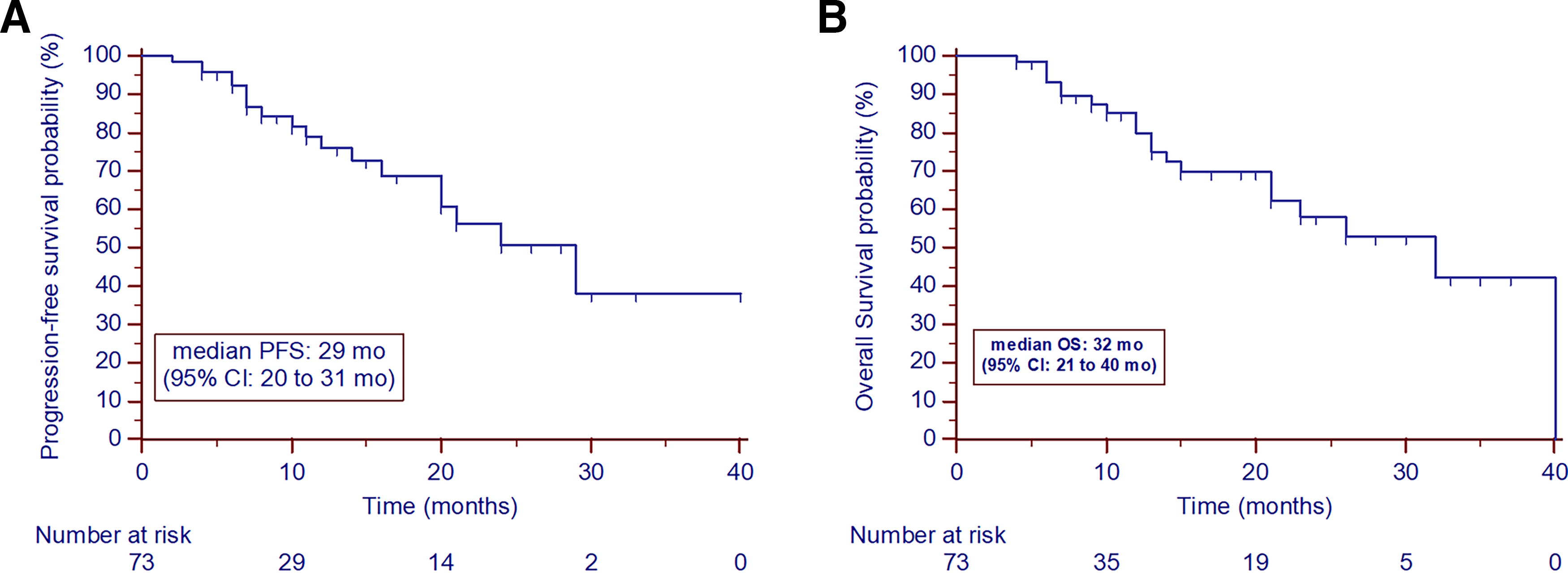
Nineteen patients experienced disease progression, with an estimated median overall PFS of 29 months [CI 14–31 months] (Fig. 2A). There were no statistically significant differences observed in the estimated median PFS among patients based on gender (hazards ratio [HR]: 1.118 [0.443–2.822]; p = 0.809) or age ≥55 years (HR: 1.699 [CI 0.706 − 4.090]; p = 0.231). Similarly, there was no significant variation found in the estimated median PFS between patients who underwent primary thyroid resection and those who did not (HR: 1.247 [0.201 − 7.719]; p = 0.824), nor between those diagnosed with DTC and HG-FC-PDTC (HR: 1.297 [CI 0.485 − 3.467); p = 0.576). No statistically significant difference was noted in PFS between patients who had undergone prior TKI treatment and those who were TKI treatment-naive ([median PFS: 24 months vs. not attained] HR: 1.887 [CI 0.716 − 4.969]; p = 0.480). Regarding the time interval between TKI discontinuation and initiation of FAPI therapy, there was no statistical difference (HR: 1.533 [CI 0.635 − 3.699]; p = 0.343). The dosage of [177Lu]Lu-DOTAGA.FAPi dimer also did not impact PFS. Additionally, the median PFS duration in the tandem therapy group was not attained compared with 24 months in the monotherapy group (Table 2).
Univariate and Multivariable Analysis of Prognostic Factors Associated with Overall and Progression-Free Survival
FC-DTC, follicular cell-derived differentiated thyroid cancer; HG-FC-DTC, high-grade follicular cell-derived thyroid cancer; mo, months; OS, overall survival; PFS, progression-free survival.
Overall survival
During analysis, 20 patients died, with 16 deaths directly due to thyroid cancer. The estimated OS was 32 months [CI 21–40 months] (Fig. 2B). No statistically significant differences were found in median OS based on gender (HR: 1.039 [CI 0.421–2.503]; p = 0.928) or age ≥ 55 years (HR: 1.332 [CI 0.553 − 3.207]; p = 0.507). Similarly, there was no significant variation in median OS between patients who underwent primary thyroid resection and those who did not (HR: 0.418 [CI 0.074 − 2.365]; p = 0.142), nor between those diagnosed with DTC and HG-FC-PDTC (HR: 0.512 [CI 0.1963 − 1.340]; p = 0.116). Moreover, no statistically significant differences were noted in OS between patients who had prior TKI treatment and TKI treatment-naive patients (HR: 2.184 [CI 0.873 − 5.465]; p = 0.126), or regarding the duration between TKI treatment discontinuation and initiation of FAPI therapy (HR: 2.010 [CI 0.736 − 5.489]; p = 0.151). In univariate analysis, disease progression on [177Lu]Lu-DOTAGA.FAPi dimer therapy showed an association with poorer OS (median OS: 23 months vs. 40 months; HR: 1.961 [CI 0.796 − 4.829]; p = 0.116), although not statistically significant. Patients who received tandem beta−alpha particle therapy with [177Lu]Lu-[225Ac]Ac-DOTAGA.FAPi dimer showed a trend toward higher OS (not attained versus 32 months, p = 0.200) compared with those who received [177Lu]Lu-DOTAGA.FAPi dimer monotherapy (Table 2).
Comparison of baseline [18F]F-FDG and [68Ga]Ga-DOTA.SA.FAPi SUV values
Secondary analyses comparing the SUVmax values between [68Ga]Ga-DOTA.SA.FAPi and [18F]F-FDG across different primary tumor sites and metastases revealed several findings of interest (Table 3). For primary tumors, the mean SUVmax was 7.5 for [68Ga]Ga-DOTA.SA.FAPi and 8.6 for [18F]F-FDG, with no significant difference (p = 0.476). Similarly, in lymph nodes, the mean SUVmax values were comparable at 7.7 for [68Ga]Ga-DOTA.SA.FAPi and 7.8 for [18F]F-FDG (p = 0.975). In the lung, both tracers showed an identical mean SUVmax of 8.5 (p = 0.962). However, significant differences were observed in the liver, bone, and brain. The liver showed a higher mean SUVmax for [68Ga]Ga-DOTA.SA.FAPi at 10.1 compared with 5.5 for [18F]F-FDG (p = 0.003). Conversely, in bone, [18F]F-FDG had a significantly higher mean SUVmax of 15.9 compared with 9.8 for [68Ga]Ga-DOTA.SA.FAPi (p = 0.017). In the brain, [68Ga]Ga-DOTA.SA.FAPi demonstrated a much higher mean SUVmax of 16.6 versus 6.1 for [18F]F-FDG (p = 0.025). For muscle, the mean SUVmax values were 10.0 for [68Ga]Ga-DOTA.SA.FAPi and 8.7 for [18F]F-FDG, with no significant difference (p = 0.499). These results suggest that [68Ga]Ga-DOTA.SA.FAPi may be superior in detecting liver and brain lesions, while [18F]F-FDG may be more effective for bone lesions.
SUVmax Values in Various Sites of Disease on [68Ga]Ga-DOTA.SA.FAPi and [18F]F-FDG PET/CT Scans
SUV, standardized uptake value.
Association between previous TKI treatment on FAPI expression in different lesion sites: Insights from [68Ga]Ga-DOTA.SA.FAPi PET/CT scan
In secondary analyses, we compared FAPI expression detected by [68Ga]Ga-DOTA.SA.FAPi PET/CT scans in patients with and without prior TKI treatment across different lesion sites. No significant differences in mean SUV were found for primary lesions (p = 0.101) or lymph node lesions (p = 0.330) between treated and untreated patients. In liver metastases, a significant difference was observed (p < 0.0001), while no significant difference was noted in bone metastases (p = 0.939). Significant differences were seen in brain (TKI treatment naive group:13.2 ± 3.2 vs. TKI group: 8.7 ± 3.6) and muscle metastases (TKI treatment naive group: 9.4 ± 4.2 vs. TKI group: 7.3 ± 3.2 (p < 0.0001). Overall, TKI treatment did not notably impact FAPI expression across lesion sites (Table 4).
Comparison of Standardized Uptake Values in Various Stites of Disease Based on Prior TKI Treatment
SUV, standardized uptake value.
Assessment of response using PERCIST criteria
A secondary molecular response assessment was conducted in patients with available data. The molecular response assessment conducted in 36 patients demonstrated partial response in 50% of cases (18 out of 36) (Fig. 3), disease progression in 25% of cases (9 out of 36) (Fig. 4), and stable disease in 25% of cases (9 out of 36). Complete resolution of FAPI-expressing lesions was not observed in any of the patients.
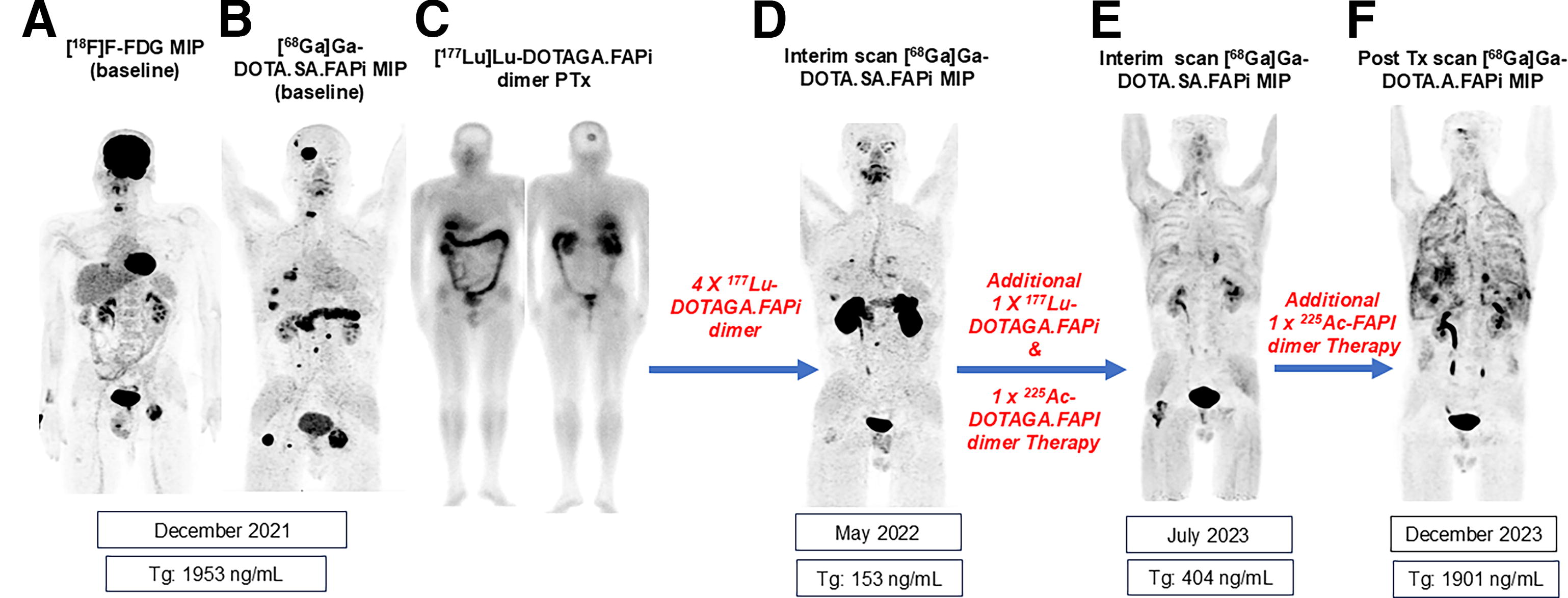
Case report: Neo-adjuvant FAPI therapy in a 37-year-old male with poorly differentiated thyroid cancer. A 37-year-old male was diagnosed with inoperable poorly differentiated thyroid cancer presenting with de novo radioiodine refractory disease after surgery (mixed radioiodine uptake in the lesions on the diagnostic whole body radioiodine scan), precluding the use of radioiodine treatment. Lenvatinib was not a viable option due to adverse events, specifically proteinuria exceeding 1 gm/24 h in urine, rendering tyrosine kinase inhibitors contraindicated. The patient declined local radiation therapy due to concerns regarding its side effects. Baseline [18F]F-FDG and [68Ga]Ga-DOTA.SA.FAPi PET/CT scans revealed disease involvement with hypermetabolic and intensely FAP-expressing large enhancing necrotic mass lesions replacing the right lobe of the thyroid adjoining the isthmus, as well as hypermetabolic and intensely FAP-expressing bilateral cervical lymph nodes and in the upper mediastinal region. After exhausting conventional treatment options, the patient underwent eight cycles of FAPI therapy, comprising five cycles of [177Lu]Lu-DOTAGA.FAPi dimer and three cycles of [225Ac]Ac-DOTAGA.FAPi dimer treatment. Evaluation post four cycles of [177Lu]Lu-DOTAGA.FAPi dimer therapy demonstrated a partial treatment response
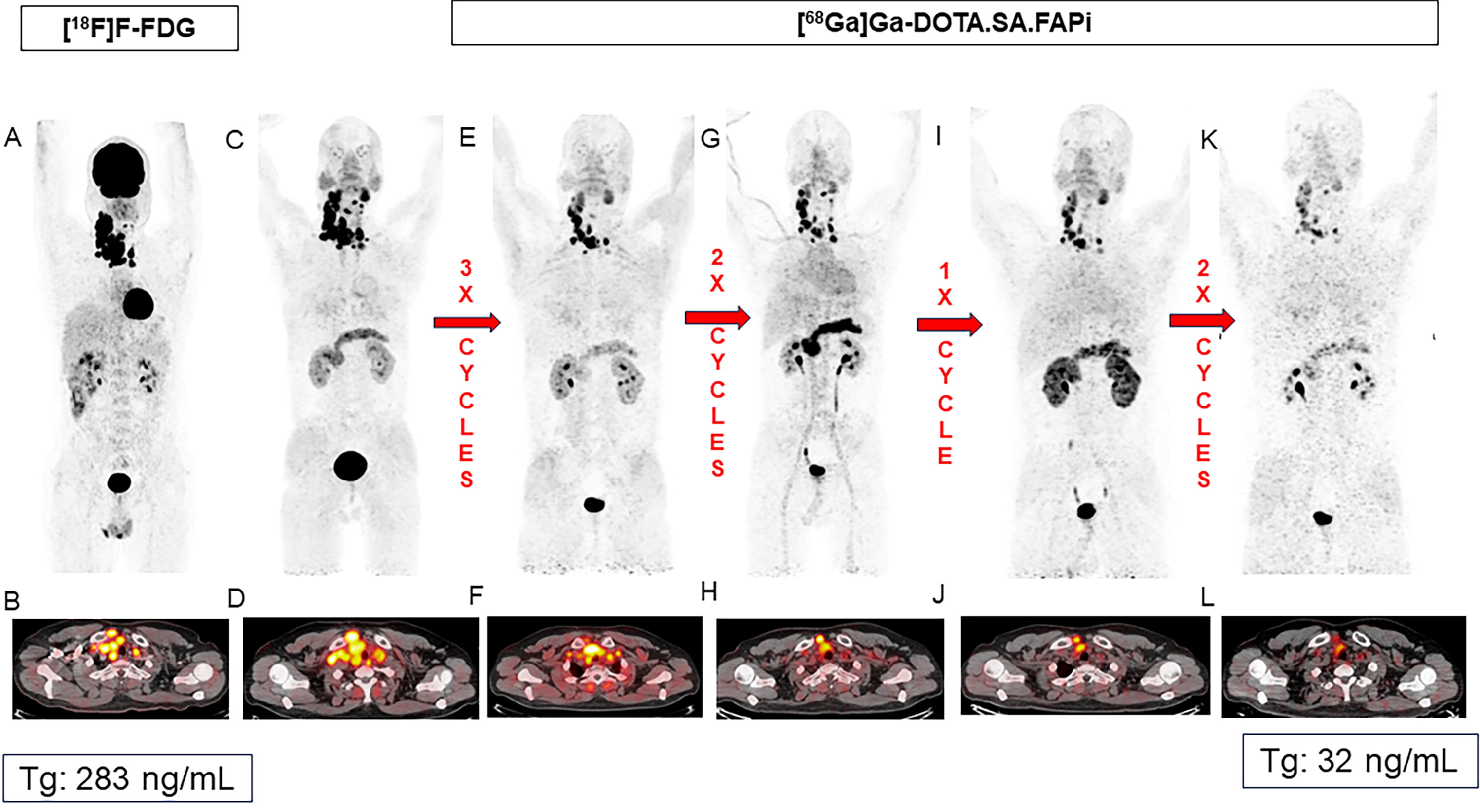
Case report: FAPi therapy outcomes in a 35-year-old male with refractory poorly differentiated thyroid cancer. A 35-year-old male was diagnosed with poorly differentiated thyroid cancer underwent total thyroidectomy and right modified radical neck dissection. Following treatment with over 37 GBq of radioactive iodine therapy, the patient developed radioiodine refractory disease. Stereotactic radiosurgery was performed on the C6 vertebrae and right and left ischium. Additionally, the patient underwent further treatments with TKIs. Despite these interventions, disease persistence was observed, evident in a baseline [18F]F-FDG PET/CT
Safety
The administration of [177Lu]Lu-DOTAGA.FAPi dimer was well tolerated, with no immediate toxicity observed in any patient. Three patients withdrew from the study due to hematotoxicity. Four patients experienced grade III anemia, three of whom had bone metastasis, and one had a neck tumor mass bleed. Grade III thrombocytopenia was observed in three patients. There were no instances of grade III renal or hepatotoxicity noted in the patient population. Additionally, three patients developed pleural effusion and only underwent two treatment cycles.
Discussion
In a pilot study by our group, 21 [177Lu]Lu-FAPi therapy displayed promising efficacy in 15 patients with RR-DTC, leading to significant reductions in serum thyroglobulin levels, partial response in four patients, and stable disease in three. Notably, patients experienced significant improvements in performance scores without severe adverse events. No grade III/IV hematological, renal, or hepatotoxicity events were observed. 21 This study reports the long-term outcomes of [177Lu]Lu-DOTAGA.FAPi dimer therapy in 73 patients, indicating its safety and potential effectiveness in treating aggressive RAI-R FCTC after standard treatments. These results introduce a novel therapeutic approach for this challenging patient group. Lenvatinib, in a phase 3 study, displayed significant efficacy in iodine-131 refractory thyroid cancer, boasting a median PFS of 18.3 months and a response rate of 64.8%. In contrast, the DECISION trial revealed sorafenib’s superiority over placebo in median PFS (10.8 vs. 5.8 months) for radioactive iodine-refractory DTC albeit lacking OS data. 9 Compared with the outcomes of the DECISION trial, [177Lu]Lu-DOTAGA.FAPi dimer therapy for aggressive radioiodine-refractory thyroid cancers showed prolonged median PFS and OS (29 and 32 months), potentially serving as a viable, cost-effective alternative, especially for TKI-ineligible patients. [177Lu]Lu-DOTAGA.FAPi dimer therapy exhibits promise in enhancing both PFS and OS with potentially fewer adverse events, necessitating further comparative studies for the optimal management of progressive thyroid cancer.
The imaging uptake value findings in our study suggest that prior TKI treatment may not significantly affect FAPI expression across various lesion sites, suggesting a consistent FAPI expression pattern regardless of prior therapy. Interestingly, patients not exposed to TKIs exhibited higher PFS and OS compared with those treated with TKIs, although these differences were not statistically significant. This suggests that factors beyond TKI treatment may influence PFS and OS outcomes. Other tumor microenvironment factors or mutations may influence FAPI expression and treatment response, which is beyond the scope of this study and warrants further investigation. Understanding these additional factors could provide insights into optimizing treatment strategies for patients with thyroid cancer. The tandem beta–alpha therapy group showed trends towards enhanced OS, and PFS, with median survival durations not reached at analysis, compared to the monotherapy group treated with [177Lu]Lu-DOTAGA.FAPi dimer (32 and 24 months). However, the difference lacked statistical significance in the context of the small size of the tandem therapy group (eight patients). Further research with a larger cohort is needed for definitive insights into the potential benefits of tandem therapy in this patient population. In contrast to the common side effects of lenvatinib, [177Lu]Lu-DOTAGA.FAPi dimer treatment was notably well-tolerated, with minimal immediate toxicity observed. Withdrawals due to hematotoxicity were few, with only three patients discontinuing treatment. Grade III anemia was observed in four patients, whereas grade III thrombocytopenia was reported in three patients. Importantly, no grade III renal or hepatotoxicity was observed. Three patients had a pleural effusion, indicating radiological disease progression, and received just two treatment cycles. The high costs of currently approved TKIs pose significant challenges, especially in low-to-medium income countries, making radionuclide therapy a cost-effective alternative. Studies by authors such as Spada et al. 26 demonstrate that radionuclide therapies, with spaced-out administrations and limited cycles, alleviate financial burdens while maintaining efficacy and safety, as seen with [177Lu]Lu-DOTATATE showing lower costs per progression-free month compared with sunitinib in pancreatic NET patients (€2989 vs. €5337).
Limitations
The primary limitation of this study is its nonrandomized, retrospective design and the absence of a comparison group. The study demonstrates that a significant number of patients exhibit therapeutically relevant uptake of FAPI-based ligands, that there are no major safety concerns, especially when compared with established second-line therapies that may have significant toxicity, and that it is feasible to administer this therapy on a large scale. A broad spectrum of patients with diverse inclusion criteria were included in the study. The selection of patients for tandem therapy was not randomized; it was based on the disease burden and the patient’s consent to undergo the combination therapy approach. Additionally, only a small number of patients received tandem therapy which may account for the lack of significance in the univariate and multivariate analyses. Another important limitation noted in our study was the small sample size in histological subgroups of RAI-R FCDTC. To improve study quality, increasing the sample size and using immunohistochemistry markers for FAPI expression will enhance statistical power and provide reliable, generalizable results. Stratified analyses will offer comprehensive insights into response variations and progression among subgroups yielding higher quality and more conclusive findings. This study demonstrated the feasibility and directional approach of [177Lu]Lu-DOTAGA.FAPi dimer treatment. Future phase II/III trials on thyroid cancer should incorporate systematic inclusion criteria, standardized dosages, and a direct comparison between a TKI treatment group and a [177Lu]Lu-DOTAGA.FAPi group.
Conclusion
In conclusion, the study highlights the promising safety and efficacy of [177Lu]Lu-DOTAGA.FAPi dimer therapy in aggressive radioiodine resistant FC-DTC, including refractory cases. Notably, the treatment demonstrated median OS and PFS durations of 32 and 29 months, respectively, with a 50% partial response rate. Adverse events, primarily hematotoxicity were manageable. Overall, no factors were associated with either PFS or OS, emphasizing the therapy’s potential as a promising option for this challenging patient population. Confirmation of our findings is needed from prospective clinical trials comparing [177Lu]Lu-DOTAGA.FAPi dimer therapy to other treatments.
Footnotes
Authors’ Contributions
F.R., M.M., and E.S.M.: Synthesized FAP precursor, DOTA.SA.FAPi, and DOTAGA.FAPi dimers. S.B., M.M., and E.S.M.: Radiolabeling. S.B., M.P.Y., F.R., and C.B.: Conceptualization. S.B., C.B., S.B., S.R., M.M., and F.R.: Methodology. S.B., C.B., M.Y., S.S., and K.R.C.: Investigation. S.A.: Pathology review. S.S., K.C., and C.B.: Data interpretation. C.B., F.R., and S.B.: Validation. S.B. and C.B.: Formal analysis. S.B. and S.S.: Data curation. S.B., M.P.Y., and F.R.: Writing and original draft preparation. C.B., F.R., S.B., S.S., and K.R.C.: Reviewing and editing. C.B. and S.R.: Supervision. C.B., F.R., and S.B.: Project administration. All authors reviewed the final version of the article and are accountable for all aspects of the work.
Author Disclosure Statement
The authors have nothing to disclose.
Funding Information
No funding was received to conduct this study.
Supplementary Material
Supplementary Data S1
Supplementary Data S2
