Abstract
Importance:
There is uncertainty as to whether treatment of subclinical hypothyroidism (SCH) is associated with cardiovascular outcomes.
Objectives:
To determine whether levothyroxine replacement therapy decreases the risk of major adverse cardiovascular events (MACE) among individuals with SCH defined as having a thyrotropin (TSH) level between 5 and 10 mU/L.
Design:
We conducted a population-based cohort study using a prevalent new-user design.
Setting:
The study utilized data from the United Kingdom Clinical Practice Research Datalink.
Participants:
We identified a base cohort of individuals aged ≥18 years with incident SCH defined as having at least two TSH levels between 5 and 10 mU/L within one year between 1998 and 2018. We matched 76,946 levothyroxine treated to 76,946 untreated individuals based on age, sex, calendar time, duration of SCH, and time-conditional propensity score. We compared individuals with SCH treated with levothyroxine with individuals with no treatment.
Exposure:
Levothyroxine treatment versus no treatment.
Main Outcome Measures:
The primary outcome, MACE, was defined as a composite of nonfatal myocardial infarction, nonfatal ischemic stroke, and cardiovascular-related mortality.
Results:
The mean age of the study cohort was 62.8 years, and 76.5% were women. During a median follow-up time of 1.6 years (interquartile range: 0.5–4.2), the incidence rate for MACE among individuals treated with levothyroxine was 12.8 per 1000 person-years; confidence interval (CI): 12.2–13.3 and 13.9 per 1000 person-years; CI: 13.4–14.3 among nontreated individuals. Levothyroxine treatment was associated with a small decreased risk of MACE (hazard ratio: 0.88; CI: 0.83–0.93).
Conclusions:
Levothyroxine treatment of SCH was associated with a small decreased risk of MACE. However, given the observational nature of the study, residual confounding should be considered in the interpretation of this finding.
Introduction
Subclinical hypothyroidism (SCH) affects approximately 5% to 10% of the population globally. 1,2 SCH affects cardiovascular risk factors, such as increasing cholesterol levels, arterial blood pressure, and body weight, 3 –5 and alters cardiomyocytes and endothelial function. 6
The American Thyroid Association (ATA) and United Kingdom (UK) National Institute for Health and Care Excellence (NICE) guidelines suggest treating SCH when the thyrotropin (TSH) level is above 10 mU/L. 7,8 However, ∼80% of SCH are mild, defined by a TSH level that is above normal range but <10 mU/L. 4 In these cases, the ATA, American Association for Clinical Endocrinology, and NICE suggest initiation of levothyroxine only if patients are symptomatic, have evidence of an autoimmune condition, 7 –9 and to consider if there are cardiovascular risk factors, as treatment of SCH has been shown to lower atherogenic lipid levels and improve carotid media thickness. 7,9
Recently, two randomized controlled trials (RCTs) showed that levothyroxine treatment among participants (aged ≥65 years) with SCH did not affect the risk of cardiovascular events and all-cause mortality, but had insufficient power to assess these outcomes. 10,11 Given this evidence, a recent clinical practice guideline recommends treatment of SCH when the TSH is >20 mU/L. 12 However, this guideline acknowledges that there is a lack of evidence to support these recommendations. As such, the National Heart, Lung and Blood Institute emphasized the need for further studies to determine whether non-treatment of SCH is associated with cardiovascular outcomes. 13 Thus, we conducted a population-based cohort study to assess whether levothyroxine treatment versus no treatment among individuals with SCH is associated with major adverse cardiovascular events (MACE).
Methods
Data source
We used the Clinical Practice Research Datalink (CPRD) Aurum, which is a large primary care database of electronic medical records, containing information on demographic characteristics, diagnoses, laboratory test results, procedures, prescriptions, and administrative information, documented by general practitioners on over 40 million individuals enrolled in over 1700 general practices in the UK. 14 Diagnoses and nonprescription data were documented in the CPRD using a combination of SNOMED CT, Read Version 2, and local EMIS® Web software-specific codes. 15 Prescriptions were identified by the Dictionary of Medicines and Devices codes in the CPRD. 15 The CPRD was linked to the Hospital Episode Statistics (HES) database, which contains full hospitalization records and is restricted to 85% of English practices in the CPRD. Finally, the CPRD was also linked to the Office of National Statistics (ONS), which provides the electronic death certificate with the cause of death for all citizens in the UK. Clinical diagnoses recorded in the CPRD have been shown to be highly valid with a median rate of 89% of cases having a confirmed diagnosis. 16,17
Study population
We assembled a base cohort of individuals aged ≥18 years with newly-diagnosed SCH who were linkable to HES and ONS. SCH was defined as having two TSH measurements between 5 and 10 mU/L on different dates within one year of each other (i.e., not necessarily consecutive) between April 1, 1998 and December 31, 2018. This definition for SCH was used given that the recommended TSH threshold to start levothyroxine treatment for SCH was 10 mU/L as per ATA and NICE guidelines. 8,9 Base cohort entry was defined by the date of the second observed TSH value between 5 and 10 mU/L.
Inclusion was then restricted to patients who had ≥12 months of recorded medical history in the CPRD prior to cohort entry. Individuals who received prescriptions for thionamides (i.e., methimazole or propylthiouracil), levothyroxine, and amiodarone during the 12 months prior to or on base cohort entry were excluded. We excluded women who were pregnant 12 months prior to or on base cohort entry given that thyroid dysfunction arising postpartum is likely secondary to thyroiditis, which is usually self-limited. 18 Finally, patients with a history of a pituitary disorder at any time before base cohort entry were excluded since the TSH measurement is not a reliable indicator of thyroid function in such patients. 7
Study design
We used a prevalent new-user design with time-conditional propensity scores (TCPS). 19,20 All patients who initiated levothyroxine from the base cohort were used to create time-based exposure sets, including all non-users up to this point in time with a general practitioner visit within 30 days of the levothyroxine user’s prescription (calendar time), same age (±3 years), same sex, and a similar duration of SCH (±30 days) (Supplementary Fig. S1). A general practitioner visit was defined by the date when a patient had both a medical code and a prescription code on the same day. We used conditional logistic regression (with the models conditioned on the exposure set number) to estimate the TCPS. We then used TCPS to match individuals who initiated levothyroxine to individuals who had the opportunity to initiate levothyroxine, but remained untreated. 19,20 The positivity assumption was assessed, and those with violations were excluded. Using nearest neighbor matching without replacement, we then matched chronologically each exposed patient to an unexposed patient (1:1) in the exposure set. Identified covariates (described below) were included in the model to predict the probability of exposure to levothyroxine.
Study cohort individuals were followed until the occurrence of the outcome (detailed below), non-cardiovascular death, end of registration with the general practice, end of the study period (December 31, 2019), a TSH level >10 mU/L on two consecutive tests, initiation of methimazole, propylthiouracil, or amiodarone, pregnancy, or diagnosis of a pituitary disorder, whichever occurred first.
Exposure
The primary exposure definition used an as-treated approach in which individuals in the study cohort were followed until the earliest of MACE outcome, crossover (either discontinuation of levothyroxine [defined by a treatment gap of >30 days between successive prescriptions] or the initiation of levothyroxine among unexposed patients), or another censoring event (described above).
Outcome
Our primary outcome was MACE defined as a composite endpoint of hospitalization for non-fatal myocardial infarction (MI), non-fatal ischemic stroke, and cardiovascular-related mortality. Events were identified as a primary diagnosis of the outcome of interest using International Classification of Diseases (ICD)-10 codes in HES. ICD-9 (prior to 2001) and -10 codes (2001 and afterwards) were used to identify mortality-related outcomes using the ONS vital statistics data (Supplementary Table S1). The event date was defined as the date of admission for HES-defined events and the date of death for ONS-defined events. Secondary outcomes included heart failure (HF) hospitalization and all-cause mortality. HF was identified using ICD-10 codes in the HES, and all-cause mortality was defined using any recorded death in any of the linked data sources.
Covariates
Propensity score estimation included all the following potential confounders. The following variables were measured at study cohort entry: baseline TSH level (average of the two recorded TSH values prior to and on base cohort entry) and year of study cohort entry. The following variables were measured in the five years before or at study cohort entry: body mass index (BMI), excessive alcohol use (i.e., alcohol abuse or dependence), smoking, Charlson comorbidity index (CCI), 21 history of diabetes, renal insufficiency (i.e., acute kidney injury, chronic renal failure, or dialysis-related procedures), cancer, chronic obstructive pulmonary disease, dementia, liver disease, obstructive sleep apnea, and rheumatoid disease. The following variables were measured at any time before or at cohort entry: MI, unstable angina, chronic stable angina, coronary artery disease, peripheral arterial disease, atrial fibrillation, HF, and cerebrovascular accident. The following variables were measured within one year of cohort entry: number of physician visits, use of oral contraceptives, hormonal replacement therapy, beta-adrenoceptor blocking drugs, nitrates, antihypertensive, lipid-lowering, anticoagulants, antiplatelet, diuretic, antidiabetics, antipsychotic, antiarrhythmic, antidepressant, nonsteroidal anti-inflammatory, and opioid medications. We also adjusted for quintiles of the Index of Multiple Deprivation 2010 22 as a proxy for socioeconomic status. Variables with missing values were entered into the propensity score model as categorical variables with a missing category.
Statistical analysis
Descriptive statistics with standardized mean differences were estimated to summarize patient characteristics of each exposure group before and after TCPS matching. To provide a description of the distribution of covariates prior to TCPS matching, we created a comparison cohort. The comparison cohort consisted of levothyroxine treated individuals matched with one randomly selected nonuser from each time-based exposure set. The incidence rates for each outcome were estimated along with their confidence interval (CI) based on the Poisson distribution. Cox proportional hazards models were used to estimate the hazard ratio (HR) and CI of MACE associated with levothyroxine use versus no treatment among matched patients with SCH. Cumulative incidence curves were plotted for the primary outcome and its individual components by exposure group. Age stratified Cox proportional hazards models (<70 and ≥70 years) were conducted to account for the differences in the baseline hazard of MACE.
Four secondary analyses were conducted. First, we assessed the association between levothyroxine treatment and the risk of individual outcomes of MACE. Second, we assessed the association between levothyroxine treatment and the risk of all-cause mortality. Third, we assessed the association between levothyroxine treatment and the risk of HF hospitalization. Finally, we repeated the primary analysis stratified by age (<70 versus ≥70 years), sex, and history of renal insufficiency and cardiovascular disease (i.e., MI, unstable angina, chronic stable angina, coronary artery disease, peripheral arterial disease, and cerebrovascular accident).
To assess the robustness of our findings, we conducted 12 sensitivity analyses (Supplementary Data S1). Two exploratory analyses were conducted. First, we assessed the risk of MACE and all-cause mortality associated with SCH stratified by TSH levels of 5–8 mU/L and 8.1–10 mU/L at base cohort entry. Second, to assess whether patients given levothyroxine were adherent to treatment, we determined the mean TSH and thyroxine levels at the end of follow-up for levothyroxine users and nonusers. The reporting of the study findings followed the STROBE guideline recommendations. 23
The study protocol was approved by the Independent Scientific Advisory Committee of the CPRD (Protocol number: 20_055) and the Biomedical Research Ethics Committee of the Jewish General Hospital, Montreal, Canada (#2021–2190). Given that the study was a secondary data analysis and did not include patients as study participants, patient consent was not required by the Research Data Governance of the CPRD and the Research Ethics Board.
Results
After applying all eligibility criteria, we identified a base cohort of 91,299 individuals with SCH. The median time between the two TSH readings to identify SCH was 110 days (25th quantile: 56 days, 75th quantile: 210 days). Before TCPS matching, individuals treated with levothyroxine had a slightly higher baseline TSH level, were less likely to have diabetes, had a lower CCI, fewer physician visits, and less likely to be on antidiabetic, antihypertensive, antiarrhythmic, antipsychotic, antidepressant, nonsteroidal anti-inflammatory, and opioid medications compared with untreated individuals. After TCPS matching, the study cohort included 76,946 individuals with SCH treated with levothyroxine matched to 76,946 untreated individuals with SCH (Fig. 1, Table 1). Covariates were well balanced after matching with standardized mean difference of less than 0.1, except for baseline TSH, which had a standardized mean difference of 0.15. The mean age was 62.8 years, and 76.5% were women.
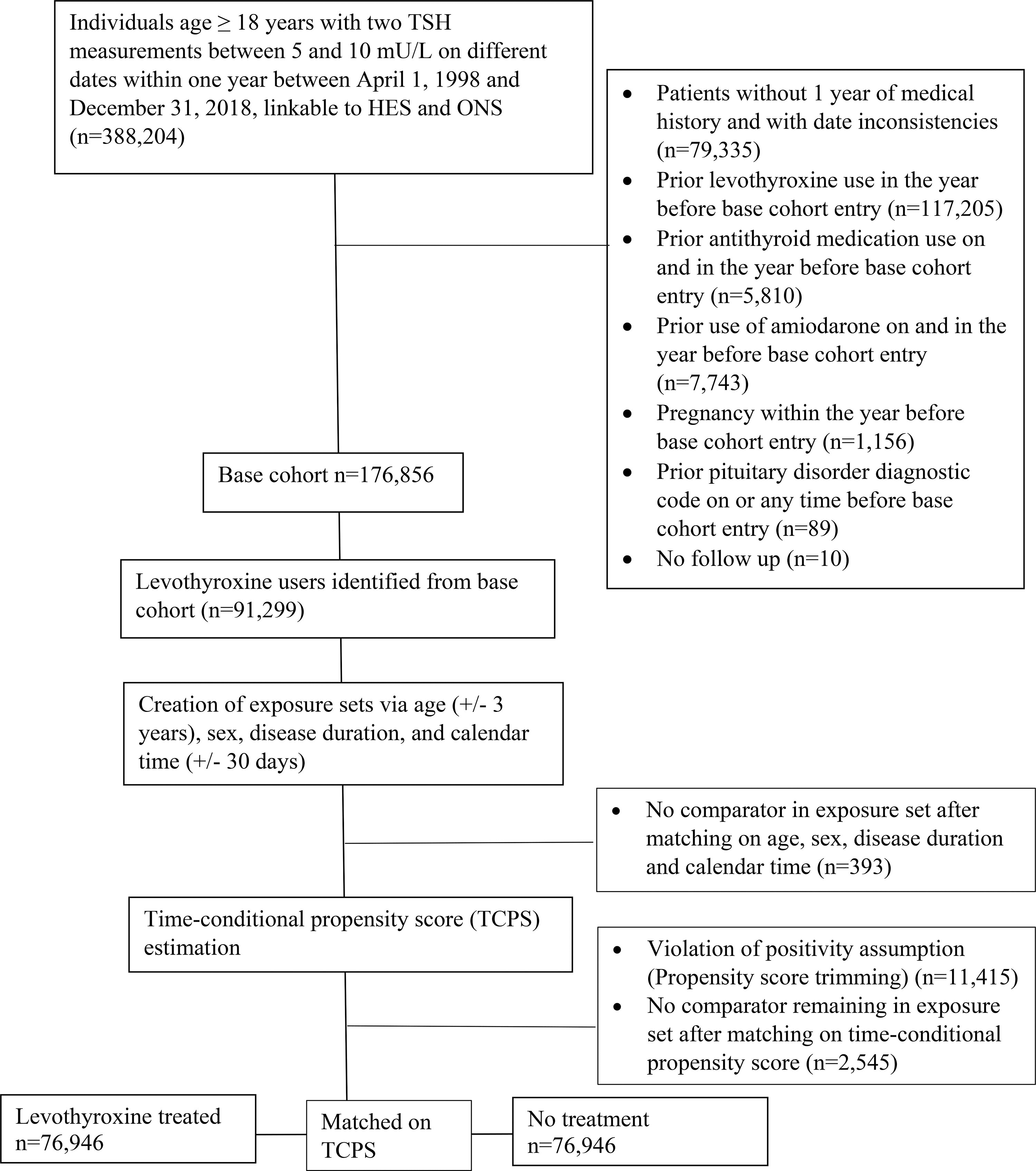
Flowchart showing assembly of study cohort of individuals with subclinical hypothyroidism.
Baseline Characteristics of Levothyroxine Treated and Untreated Individuals with Subclinical Hypothyroidism Before and After Time-Conditional Propensity Scores Matching
Comparison cohort before time-conditional propensity score matching was created by randomly selecting 1 untreated individual from each time-based exposure set matched on age (±3 years), sex, calendar time (±30 days), and duration of subclinical hypothyroidism (±30 days).
Matched cohort was created after matching for age (±3 years), sex, calendar time (±30 days), duration of subclinical hypothyroidism (±30 days), and time-conditional propensity scores. Individuals with no comparators in exposure set, violation of positivity, or had no more comparators when matching with time-conditional propensity score were excluded in the matched cohort.
Index of Multiple Deprivation is the measure of deprivation. It is a composite measure derived from the following domains of deprivation: income, employment, education and skills, health, housing, crime, access to services, and living environment. The Index of Multiple Deprivation is calculated as a weighted sum of these domain measures.
Body Mass Index definitions: underweight (<18.5 kg/m2), normal (18.5–24.9 kg/m2), overweight (25–29.9 kg/m2), obese (≥30 kg/m2).
Charlson Comorbidity Index is the measure of comorbidity using a score developed by Khan et al. 21
COPD, chronic obstructive pulmonary disease; CVA, cerebral vascular accident; DPP, dipeptidyl-peptidase; GLP, glucagon-like peptide; NSAIDs, nonsteroidal anti-inflammatory drugs; SD, standard deviation; SGLT, sodium-glucose cotransporter; TIA, transient ischemic attack; TSH, thyrotropin.
The median follow-up overall was 1.6 years (interquartile range [IQR]: 0.5–4.2), 1.0 year (IQR: 0.4–2.7), and 2.5 years (IQR: 0.9–5.7) among those treated and not treated with levothyroxine, respectively. During follow-up, 2028 MACE occurred among levothyroxine treated individuals (incidence rate: 12.8 per 1000 person-years; CI: 12.2–13.3) and 4068 MACE occurred among individuals not treated (incidence rate: 13.9 per 1000 person-years; CI: 13.4–14.3). Baseline TSH levels for individuals treated with levothyroxine was 6.6 mU/L (standard deviation [SD]: 1.0), and 6.7 mU/L (SD: 1.0) for untreated individuals. Levothyroxine treatment was associated with a mild decreased risk of MACE among individuals with SCH (HR: 0.88; CI: 0.83–0.93) (Table 2, Fig. 2). When assessing the individual outcomes of MACE, levothyroxine treatment of SCH was associated with a decreased risk of nonfatal MI and cardiovascular-related mortality (Table 2, Fig. 2). However, levothyroxine treatment of SCH was not associated with the risk of nonfatal ischemic stroke or HF hospitalization (Table 2, Fig. 2). Levothyroxine treatment of SCH was associated with a decreased risk in all-cause mortality (Table 2). There was no difference in the risk of MACE in stratified analyses by age, sex, history of renal insufficiency, and history of cardiovascular disease (Table 3).
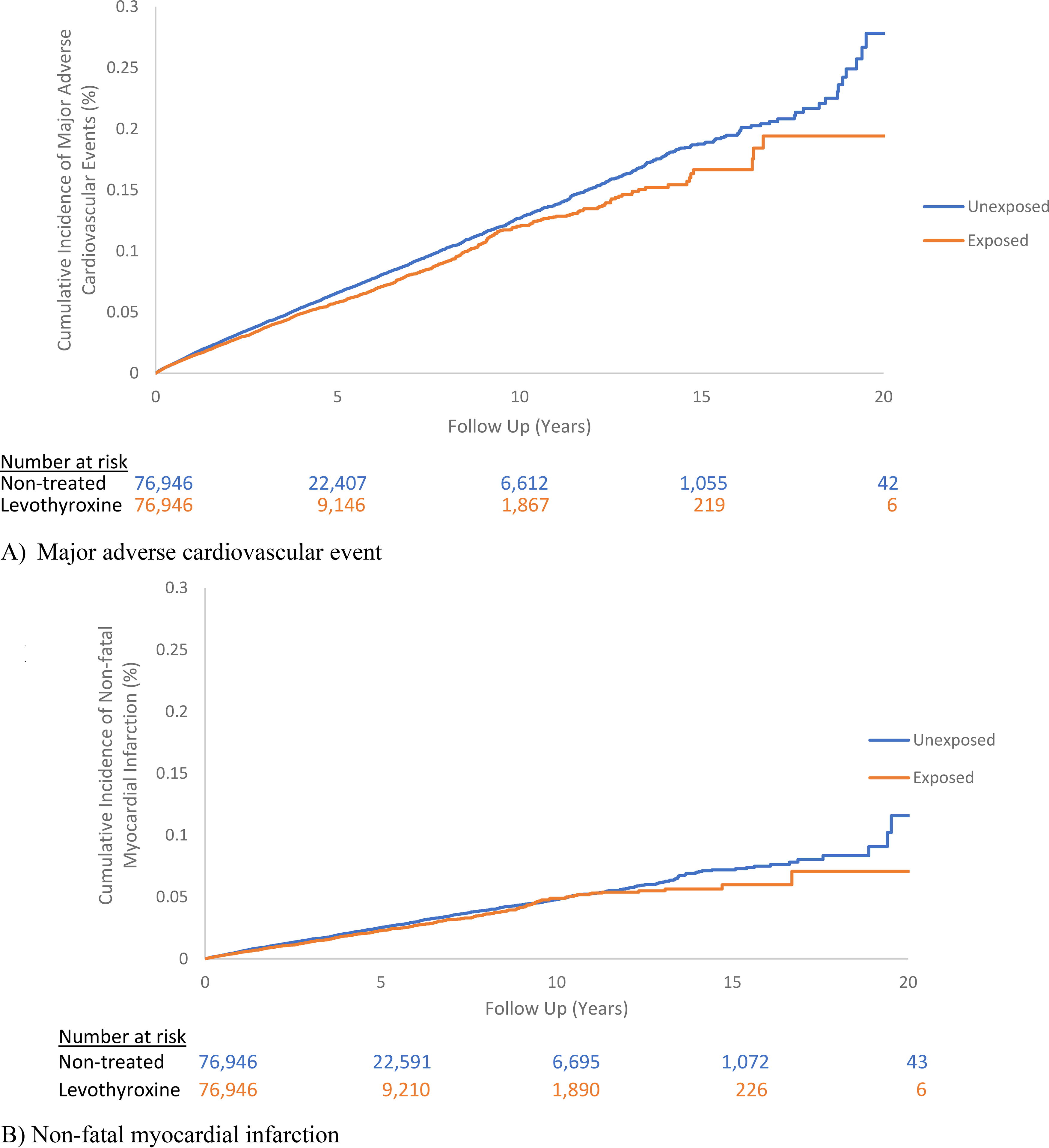
Cumulative incidence curves of major adverse cardiovascular events
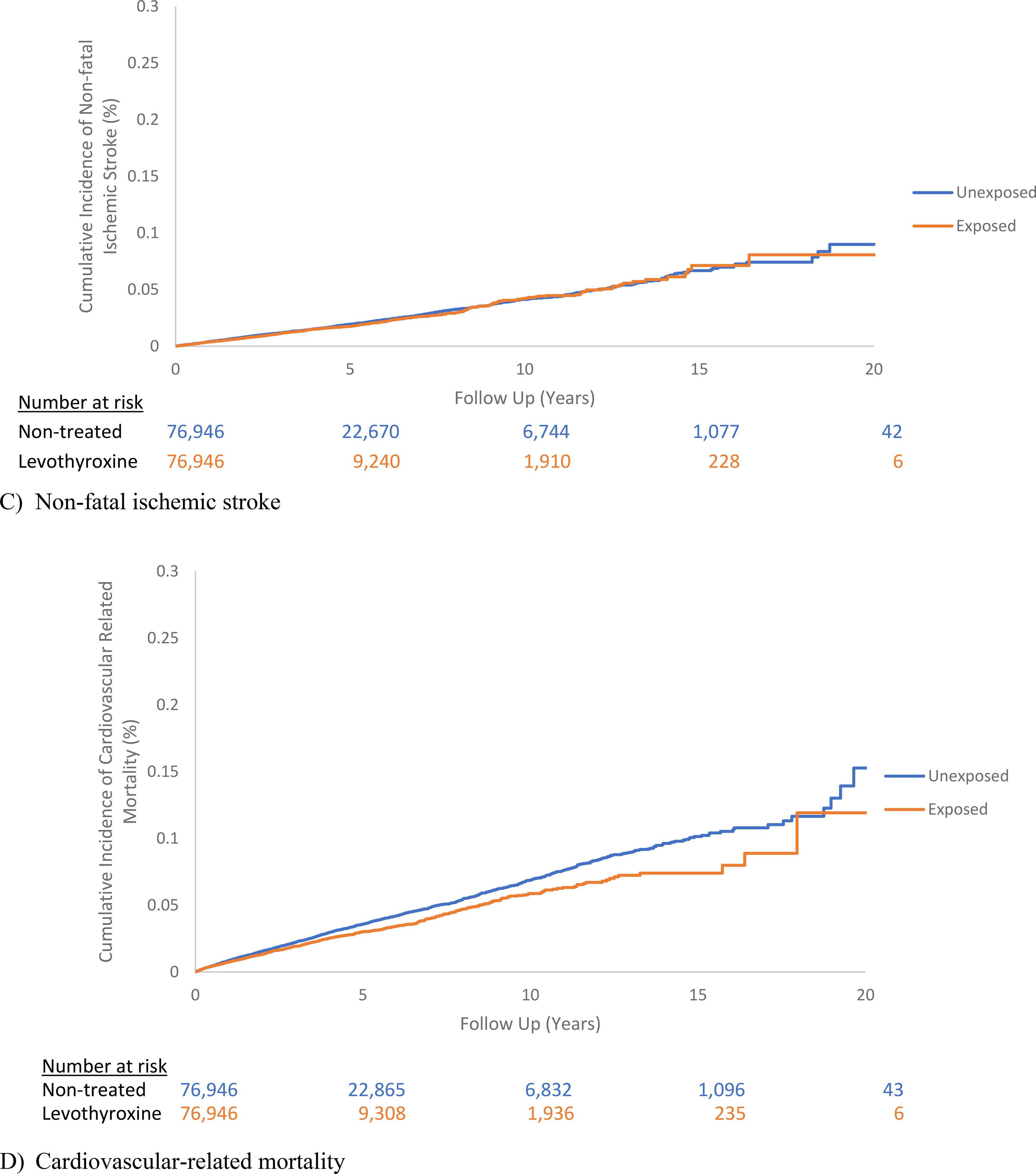
(Continued).
Risk of Major Adverse Cardiovascular Events and Individual Outcomes of Myocardial Infarction, Ischemic Stroke and Cardiovascular Mortality, Heart Failure, and All-Cause Mortality Among Individuals with Subclinical Hypothyroidism Treated with Levothyroxine Versus No Treatment
per 1,000 Person-Years.
Individuals were matched on calendar time, subclinical hypothyroidism duration, sex, age, and time-conditional propensity scores. To account for differences of baseline hazard of major adverse cardiovascular outcomes, age stratified Cox models were run (<70 versus ≥70 years).
In addition to matching on calendar time, subclinical hypothyroidism duration, sex, age, and time-conditional propensity scores, analyses were adjusted for time-conditional propensity scores. To account for differences of baseline hazard of major adverse cardiovascular outcomes, age stratified Cox models were run (<70 versus ≥70 years).
CI, confidence interval.
Risk of Major Adverse Cardiovascular Events Among Individuals with Subclinical Hypothyroidism Treated with Levothyroxine Versus No Treatment Stratified by Age, Sex, History of Renal Insufficiency, and History of Cardiovascular Disease
per 1,000 person-years.
Individuals were matched on calendar time, subclinical hypothyroidism duration, sex, age, and time-conditional propensity scores. To account for differences of baseline hazard of major adverse cardiovascular outcomes, age stratified Cox models were run (<70 versus ≥70 years).
In addition to matching on calendar time, subclinical hypothyroidism duration, sex, age, and time-conditional propensity scores, analyses were adjusted for time-conditional propensity scores. To account for differences of baseline hazard of major adverse cardiovascular outcomes, age stratified Cox models were run (<70 versus ≥70 years).
CI, confidence interval.
In the sensitivity analyses, most results were consistent with those in the primary analysis (Supplementary Tables S2–S13). Namely, the findings remained consistent when the TSH levels used to define SCH were at least 3 months apart (Supplementary Table S11) and in the stratified analysis using a cutoff age of 65 years (Supplementary Table S12). However, the association between levothyroxine treatment and the risk of MACE in the modified as-treated and intention-to-treat (ITT) analyses was no longer statistically significant. Exploratory analyses showed that the association between levothyroxine treatment and MACE among individuals with TSH levels between 5 and 8 mU/L compared with those with TSH levels between 8.1 mU/L and 10 mU/L was similar (Supplementary Table S14). The mean TSH and thyroxine levels last measured during follow-up were 4.25 mU/L and 15.30 pmol/L among individuals treated with levothyroxine and 6.88 mU/L and 13.23 pmol/L among untreated individuals (Supplementary Tables S15 and S16).
Discussion
In this population-based cohort study of individuals with SCH, levothyroxine treatment was associated with a mild decreased risk of MACE compared with no treatment. Levothyroxine treatment was associated with a decreased risk for nonfatal MI and cardiovascular-related mortality, but was not associated with the risk of nonfatal ischemic stroke and HF hospitalization. Although there was a decrease in all-cause mortality associated with levothyroxine treatment, this finding is likely due to residual confounding. There was no significant difference in the association between levothyroxine treatment and risk of MACE in stratified analyses by age, sex, history of renal insufficiency, and cardiovascular disease.
Few studies assessed the association between SCH and cardiovascular outcomes. 24,25 One retrospective cohort study using the CPRD found that levothyroxine treatment was associated with a decreased risk of ischemic heart disease (IHD) and all-cause mortality, but not associated with the risk of cerebrovascular disease among individuals aged 40 to 70 years with SCH (TSH 5.01 to 10mU/L). 26 There was no significant association between levothyroxine treatment and the risk of IHD (HR: 0.99; CI, 0.59–1.33), all-cause mortality (HR: 0.71; CI: 0.56–1.08), and cerebrovascular disease (HR: 0.81; CI: 0.31–2.12) among individuals with SCH aged >70 years. However, the CIs were wide, and thus, the findings were inconclusive. The differences with our findings may arise from the way individuals with SCH were identified, the study design, primary outcome definition, and sample size.
Recently, two RCTs assessed whether levothyroxine treatment for SCH (TSH 4.66 mU/L to 19.99 mU/L) improved the quality of life at 12 months after randomization among individuals aged ≥65 years (Thyroid Hormone Replacement for Untreated Older Adults with Subclinical Hypothyroidism [TRUST] study) 10 and aged ≥80 years (Institute for Evidence-Based Medicine in Old Age [IEMO]). 11 In both RCTs, levothyroxine treatment did not improve the quality of life and did not affect cardiovascular or all-cause mortality outcomes. However, they did not have sufficient statistical power to address these outcomes.
Our study included adults <65 years and had sufficient statistical power to address the risk of MACE associated with levothyroxine treatment among people with SCH with similar duration of mean follow-up as the TRUST and IEMO trials. Indeed, TSH levels tend to increase as people age irrespective of the presence of underlying thyroid disease, and thus, the findings from our study suggest that there may be a small decreased risk of MACE associated with levothyroxine treatment among older individuals with SCH. 27
This study has several strengths such as matching on age, sex, calendar time, duration of SCH, and TCPS, which included a large number of potential confounders, including variables associated with lifestyle (i.e., BMI and smoking) and frailty. The study also used the HES and ONS to identify cardiovascular outcomes, which reduce the risk of outcome misclassification. Finally, the CPRD contains biochemical data allowing the study cohort assembly of individuals with SCH.
This study also has limitations. First, despite adjusting for multiple comorbidities, the overall health of individuals may not be fully captured using these covariates alone, and the possibility of residual confounding should be considered in the interpretation of the results. Given that SCH rarely causes symptoms, it is possible that people given treatment are healthier compared with those not given treatment. This factor likely led to the finding of a significant decreased risk of all-cause mortality associated with levothyroxine treatment. However, this residual confounding is less likely to entirely account for the association observed between levothyroxine use and MACE and its individual outcomes given that the large number of covariates were well-balanced between groups after TCPS matching and there was a lack of divergence of cumulative incidence curves seen during the beginning of follow-up. Moreover, in our negative control sensitivity analysis, levothyroxine treatment was not associated with a decreased risk of gastroesophageal reflux disease. Second, the follow-up time in this study was relatively short, with a median of 1.6 years. In addition, the follow-up time varied between the levothyroxine treated and untreated group due to discontinuation of treatment among those that were exposed to levothyroxine. However, we conducted sensitivity analyses to address this issue. The modified as-treated and ITT analyses resulted in a point estimate similar to that obtained in our primary analysis, but with nonstatistical significance, likely due to decreased number of events captured when the follow-up was limited to one year in both groups. Third, there may be misclassification of exposure. However, our results remained consistent in sensitivity analyses with varying grace periods. Similarly, there may also be misclassification of covariates in the CPRD. However, the clinical diagnoses in the CPRD have been shown to be accurate with high validity. 16,17 Fourth, our study did not assess the potential harms (i.e., cardiac arrhythmias, osteoporosis, and fragility fractures) that can occur with long-term levothyroxine treatment. Finally, we were unable to identify individuals with underlying thyroid autoimmune disease.
Conclusion
In this population-based cohort study, levothyroxine treatment was associated with a 12% reduction in the risk of MACE. The magnitude of effect in MACE outcomes is small, and its clinical significance is uncertain. Given the observational nature of the study, potential residual confounding should be considered in the interpretation of the results.
Footnotes
Acknowledgments
R.W.P. holds the inaugural Albert Boehringer I Chair in Pharmacoepidemiology at McGill University. K.B.F. is supported by a Senior Scientist salary support award from the Fonds de recherche du Québec-Santé (FRQS) and a William Dawson Scholar award from McGill University.
Authors’ Contributions
O.H.Y.Y.: developed the study protocol (lead), methodology (equal), wrote the first draft of the article, and review and editing (equal). C.F.: conducted statistical analyses (lead) and writing—review and editing (equal). K.B.F.: contributed to study design and writing—review and editing (equal). R.W.P.: contributed to study design and writing—review and editing (equal). R.G.: contributed to writing—review and editing (equal). C.R.: contributed to study design, methodology (equal), and writing—review and editing. O.H.Y.Y.: and C.F.: had full access to all the data in the study. O.H.Y.Y.: takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors provided final approval of the revised article and agreed to be accountable for all aspects of the work, ensuring that issues related to the accuracy or integrity of the work submitted are appropriately investigated and resolved.
Author Disclosure Statement
R.W.P. has received consulting fees from Biogen, Boehringer Ingelheim, Merck, Nant Pharma, Pfizer, and Vanda Pharmaceuticals for work unrelated to this project. O.H.Y.Y. received fees for a Novo Nordisk Advisory Board conducted in December 2021 for work unrelated to this project. The other authors have no relationships to disclose.
Funding Information
This study was funded by a Project Grant from the Canadian Institutes of Health Research [Project Grant Reference #169011 (
Supplementary Material
Supplementary Data S1
Supplementary Figure S1
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Table S9
Supplementary Table S10
Supplementary Table S11
Supplementary Table S12
Supplementary Table S13
Supplementary Table S14
Supplementary Table S15
Supplementary Table S16
