Abstract
Background:
Recent studies have suggested that pathogenic variants of the DICER1 gene could be a driver of alterations in some pediatric thyroid nodules, but data are still limited. The aim of this study was to detect variants in the DICER1 gene in a large cohort of pediatric thyroid nodules and then correlate them with clinicopathological data, with a focus on the disease prognosis in patients with thyroid carcinoma.
Methods:
This retrospective cohort study consisted of 350 pediatric and young adult patients (aged 2–21 years) with thyroid nodules, from whom 275 fresh-frozen thyroid nodule samples and 92 fine-needle aspiration biopsy (FNAB) samples were collected. After an analysis of variants in major genetic alterations of thyroid tumors, variants in the DICER1 gene were identified using next-generation sequencing and multiplex ligation-dependent probe amplification methods. Peripheral blood was analyzed from patients with DICER1-positive tumors. The results of genetic analysis were then correlated with clinicopathological data.
Results:
Variants in the DICER1 gene were detected in a total of 24/350 (6.9%; 95%CI [4.4;10.0]) pediatric and young adult patients, respectively, in 10/119 (8.4%; [4.1;14.9]) patients with benign fresh-frozen tissue, in 8/141 (5.7%; [1.9;9.5]) with papillary thyroid carcinoma (PTC) and in 6/86 (7.0%; [4.1;14.6]) patients with FNAB. No other gene alteration was found in DICER1-positive samples. Germline DICER1 variants were identified in 11/24 (45.8%; [25.6;67.2]) patients. Two somatic (biallelic) variants in the DICER1 gene were found in 9/24 (37.5%; [18.8;59.4]) thyroid nodules. Somatic deletions of at least 3 Mbp long were revealed in 2/24 (8.3%; [1.0;27.0]) cases. DICER1-positive PTCs were significantly associated with the follicular subtype of PTC (p = 0.001), encapsulation (p = 0.006) and were larger in size (p = 0.035), but with no extrathyroidal extension (p = 0.039), and less frequent lymph node metastases (p = 0.003) compared with DICER1-negative PTCs. Patients with DICER1-positive PTC had an excellent response to treatment in 75% of cases.
Conclusions:
Variants of the DICER1 gene are frequently found in the thyroid nodules of pediatric and young adult patients. In our patients, DICER1-positive PTCs showed low invasiveness. Our findings support considering more conservative management for DICER1-positive low-risk PTCs.
Introduction
The human DICER1 gene, located on chromosome 14q32.13 and consisting of 27 exons, encodes the highly conserved RNase III DICER1 enzyme. This enzyme plays an important role in RNA interference pathways, where it is involved in the processing of hairpin-folded pre-miRNA into ∼20-nucleotide-long miRNA. 1
The occurrence of thyroid nodules in pediatric patients has historically been low, but the incidence is increasing. 2 Possible reasons include the increased sensitivity of imaging methods and the more frequent follow-up of a larger number of patients. On the other hand, a higher incidence of larger tumors and more advanced disease have also been reported. 3 Recently, studies have suggested a significant role of DICER1 variants in the development of pediatric thyroid tumors; however, data from larger cohorts are still limited. DICER1-positive thyroid tumors are often reported as being benign, whereas thyroid carcinomas seem to be mostly indolent. 4 –7
When biallelic alterations are identified, DICER1 tumors fulfill the two-hit hypothesis. One alteration (usually germline) is generally a DICER1 loss-of-function (LOF) variant, which coexists with a somatic missense DICER1 variant, the so-called somatic hotspot variant, located on the second allele. Hotspot variants typically occur in the RNase IIIb domain involving residues p.E1705, p.D1709, p.D1713, p.G1809, p.D1810, or p.E1813. 1,8,9
Germline DICER1 variants cause an autosomal dominant tumor predisposition syndrome called DICER1 syndrome. 10 Patients with DICER1 syndrome are at increased risk of developing pleuropulmonary blastoma, pediatric cystic nephroma, Sertoli-Leydig cell tumor, pituitary blastoma, pineoblastoma, and other malignant and benign neoplasms. 11 In the thyroid, DICER1 syndrome is associated with early-onset multinodular goiter, follicular adenoma, and thyroid carcinoma. 5,7,12 –14 Tumors with two somatic pathogenic DICER1 variants, each located on one allele (so-called tumor-specific biallelic variants), have also been described. 15
The aim of this study was to identify variants in the DICER1 gene in a large cohort of pediatric thyroid nodules (using preoperative samples and postoperative fresh-frozen tissues). We also examined association between these variants and clinicopathological features with a focus on the disease prognosis of patients with papillary thyroid carcinoma (PTC).
Materials and Methods
Materials
This retrospective cohort study included 350 pediatric and young adult patients aged 2–21 years. Between 2003 and 2023, fresh-frozen thyroid nodule samples were collected from patients who underwent thyroidectomy at the Motol University Hospital and the University Hospital Kralovske Vinohrady in Prague. Fine-needle aspiration biopsies (FNABs) were obtained at the Institute of Endocrinology in Prague between 2018 and 2023. Informed consent was obtained from all patients or the parents/legal guardians of pediatric patients for the collection of biological material (tissue/peripheral blood), its molecular genetic testing and the use of the results for diagnostic and research purposes. The study was approved by the Ethics Committee of the Institute of Endocrinology (EK-EÚ/F192/14062021). Samples were evaluated by an experienced pathologist and classified according to the 8th edition of the AJCC TNM system. 16 Clinical and pathological data were collected from all patients. Radioiodine treatment was administered based on the size of the carcinoma, the presence of invasion and metastases. Response to treatment (6–24 months after surgery) was determined using the definitions reported in the 2015 American Thyroid Association Guidelines. 17
The first cohort of 264 patients included 275 fresh-frozen thyroid nodule samples: 141 PTCs, four low-risk neoplasms (one noninvasive follicular thyroid neoplasm with papillary-like nuclear features [NIFTP] and three thyroid tumors of uncertain malignant potential), 19 follicular thyroid adenomas (FTAs), 17 specimens with chronic lymphocytic thyroiditis, and 83 benign nodules or goiters. The second cohort of 86 patients included 92 FNAB samples cytologically evaluated as Bethesda II-VI. Peripheral blood samples from patients with a DICER1-positive tumor, and their relatives were analyzed for germline variants when samples were available.
Methods
All molecular genetic analyses were performed at the Department of Molecular Endocrinology of the Institute of Endocrinology in Prague.
DNA extraction
Fresh-frozen thyroid tissues and FNABs were processed with the QIAcube Connect Extraction System (Qiagen, Hilden, Germany) using the AllPrep DNA/RNA/miRNA Universal kit (Qiagen, Germany). DNA from peripheral blood was extracted with a QuickGene-610L (FUJIFILM, Osaka, Japan) using the QuickGene Whole Blood kit L (Kurabo Industries Ltd, Osaka, Japan). The concentrations of all samples were measured using a fluorometer (Qubit 2.0, Invitrogen, Carlsbad, CA, USA). The purity of DNA extracted from fresh-frozen thyroid tissues and blood was measured using a spectrophotometer (QIAxpert, Qiagen).
Molecular analyses
An analysis of variants in major genetic alterations associated with thyroid tumorigenesis (BRAF, HRAS, KRAS, NRAS, and fusion genes) has been performed in our large cohort as described in a previous study. 18 However, our cohort also included pediatric tumors that lacked any driver variant from that analysis set, so other genes, including the DICER1 gene, were tested. The DICER1 gene (exons 1–27 and exon–intron boundaries; NM_177438.3) was sequenced using the Nextera XT DNA Library Prep Kit (Illumina, San Diego, CA, USA) on the MiSeq sequencer (Illumina, USA). Visualization was performed in the Integrative Genomics Viewer (Broad Institute, Cambridge, MA, USA), and variants were evaluated using the VarSome platform (Saphetor SA, Lausanne, Switzerland). DICER1-positive samples were verified on a CEQ8000 Capillary Electrophoretic Genetic Analysis System (Beckman Coulter, Indianapolis, IN, USA) or more recently on the SeqStudio Genetic Analyser (Thermo Fisher Scientific, Cincinnati, OH, USA). The PTEN gene (exon 5–8; NM_000314.8) was analyzed using the same method as the DICER1 gene, but those results are outside the scope of this article.
Multiplex ligation-dependent probe amplification (MLPA) followed by fragment analysis was performed to determine the presence of rare large-scale deletions or duplications in DICER1-positive samples. For this purpose, SALSA MLPA Probemix P482 DICER1 (MRC Holland, Amsterdam, The Netherlands) was used, and fragment analysis was performed on the SeqStudio Genetic Analyser (Thermo Fisher Scientific; Cincinnati, OH, USA). Data were evaluated using Coffalyser v.220513.1739 (MRC Holland, Amsterdam, The Netherlands).
Statistical evaluation
Categorical data are summarized as n (%). Normally distributed data are summarized as the mean (standard deviation). Categorical variables were compared using the Fisher’s exact test, and continuous variables were compared using the t-test. Statistical analyses were performed using the simple interactive statistical analysis 19 and GraphPad tools. 20 If any patient data were missing, these patients were excluded from the statistical evaluation of that category. For the prevalence rate, 95% confidence intervals on the percentage estimates were used: (95% lower confidence limit; 95% upper confidence limit). A p value <0.05 was considered statistically significant.
Results
The detection of DICER1 variants
Variants in the DICER1 gene were identified in 24/350 (6.9%; 95%CI [4.4;10.0]) pediatric patients with thyroid nodules (Fig. 1), including 18/264 (6.8%; [4.1;10.6]) patients with a finding in postoperative material (fresh-frozen thyroid tissues) and 6/86 (7.0%; [4.1;14.6]) patients with a finding in preoperative material (FNABs). Patients whose FNAB samples were analyzed were not included in the postoperative cohort. DICER1 variants were found in 10/119 (8.4%; [4.1;14.9]) benign tissues, 0/4 low-risk neoplasms and in 8/141 (5.7%; [1.9;9.5]) malignant thyroid tumors. Furthermore, DICER1 variants were detected preoperatively in nine nodules from six patients and were cytologically evaluated as Bethesda II-IV.
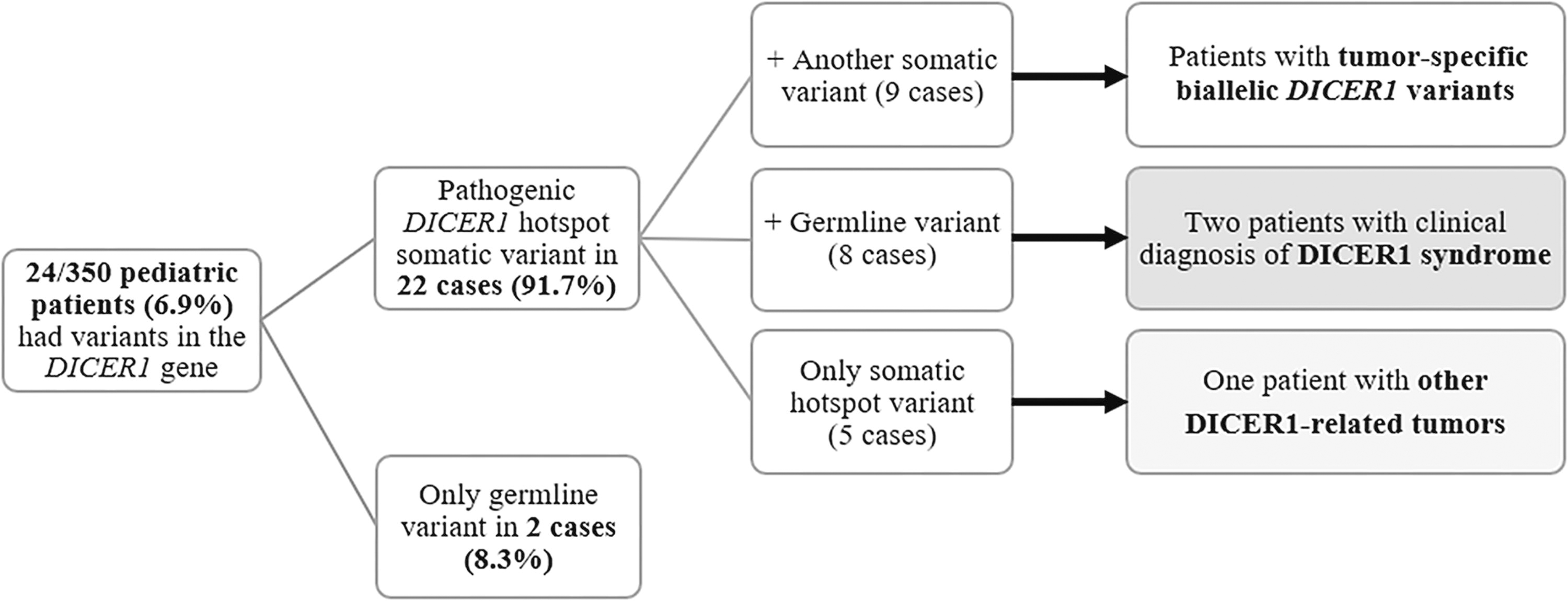
Overview diagram of positive patients and distribution of detected germline and somatic variants in the DICER1 gene.
All pathogenic variants, likely pathogenic variants, and variants of uncertain significance detected in the DICER1 gene are shown in Fig. 2. Hotspot somatic variants were located in codons 1705, 1709, 1810, and 1813. The other identified variants occurred only once and were distributed along the entire gene. Two cases had of a deletion of at least 3 Mbp long (due to the limitation of the MLPA method) involving a complete deletion of the DICER1 gene (one locus). Peripheral blood was not available from one of these patients, so healthy thyroid tissue from the second lobe was used for analysis of the germline/somatic origin of DICER1 variants. Additional information on all detected variants is provided in Supplementary Table S1. Several DICER1 variants of benign/likely benign significance are listed in Supplementary Table S2.
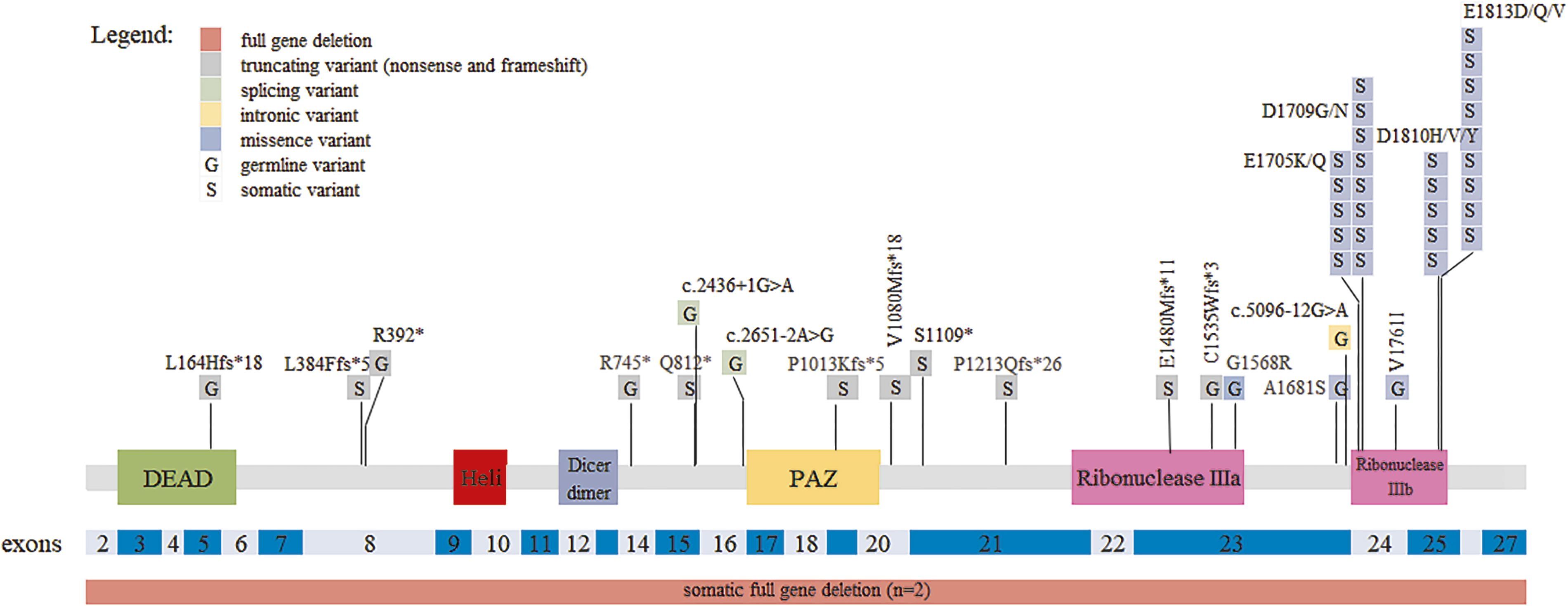
A plot of pathogenic/likely pathogenic/VUS variants detected in the DICER1 gene. Germline and somatic variants have been plotted along the length of the Dicer protein. Exon distribution is shown in shades of blue. Full gene deletions are shown under the plot. DEAD, DEAD/DEAH box helicase; Heli, Helicase conserved C-terminal; Dicer dimer, Dicer dimerisation domain; G, germline; PAZ, Piwi/Argonaute/Zwille domain, S, somatic; VUS, variant of uncertain significance.
The clinicopathological data along with the genetic profile of patients are described in Fig. 3. The mean age of the patients was 15.0 ± 3.7 years, and the cohort of DICER1-positive patients consisted of five males and 19 females. Hemithyroidectomy was performed in seven cases. Four patients who underwent initial hemithyroidectomy subsequently had a follow-up total thyroidectomy due to PTC histology. The other 16 patients underwent total thyroidectomy during one surgery, and one patient did not undergo surgery and continues to be monitored at regular intervals (Case 22).

Tile plot of DICER1-positive nodules in patients and their clinicopathological data. Tile plot is divided into two parts: fresh-frozen tissues and FNABs. Clinical and pathological data as sex, age at diagnosis, histology/cytology, nodule size, encapsulation, lymph node metastases are shown. Extent of radioiodine treatment and follow-up are mentioned. FNABs, fine-needle aspiration biopsies; FTC, follicular thyroid carcinoma; LNM, lymph node metastases; NIFTP, non-invasive follicular thyroid neoplasms with papillary-like nuclear features; PTC, papillary thyroid carcinoma; RAI, radioactive iodine; T, tumor size and extension.
Patients with germline variants and DICER1 syndrome
This cohort was comprised of a total of 11 patients (Fig. 3), including 10 patients with a germline DICER1 variant and one patient (Case 9) who had no germline DICER1 variant detected but who suffered from other DICER1-related tumors—pediatric cystic nephroma (age not available) and pineoblastoma (age 13). In total, 3/11 patients (Case 1, 8, 9) were clinically diagnosed with DICER1 syndrome. Three of the 11 patients were found to have thyroid carcinoma. In addition to Case 9, only Case 1 had additional multiple DICER1-associated tumors—pleuropulmonary blastoma (age 2), pediatric cystic nephroma (age 10), and recently diagnosed ovarian tumor (age 14).
Case 8 was followed by an endocrinologist from early childhood due to a family history of pediatric thyroid nodules (mother and maternal uncle—nodular goiters, grandmother—thyroid carcinoma; Fig. 4). The proband underwent a total thyroidectomy at the age of 10 years. The nodule was histologically evaluated as NIFTP—the follicles were mostly normofollicular, in some areas with a microfollicular, rarely with a macrofollicular pattern. PTC nuclei were without intranuclear grooves. The sporadic presence of true papillae was observed, which rather resembled hyperplastic papillae. Atrophic changes in the parenchyma were observed in a single location. After almost three years, two laterally located lymph node metastases (LNMs) were found, and the primary tumor was reclassified as the classical subtype of PTC. The patient was treated with radioiodine 7.2 GBq and had a stimulated thyroglobulin (Tg) level of 485 μg/L at the time of treatment. The patient was suspected to have additional LNMs on sonography, and there was concern about distant metastases. Five months after treatment, the patient underwent a neck revision for a parapharyngeal abscess. Another hospitalization occurred six months after the first dose of radioiodine. Two viable lymph nodes were seen caudally under the thyroid bed, and the patient received radioactive iodine 7.4 GBq with no evidence of pathological accumulation of I-131 on post-therapy scintigraphy. A high level of stimulated Tg was probably caused by the presence of LNMs. Recently, the patient is in remission with Tg <0.040 µg/L (with concurrent TSH of 0.825 mIU/L). Genetic analysis of the primary tumor revealed a germline variant of the DICER1 gene p.(L164HfsTer18) in exon 5 and a somatic hotspot variant p.(E1705K). Analysis of maternal peripheral blood DNA confirmed the familial origin of DICER1 syndrome. In two other cases (Cases 13 and 20), the familial origin of the germline variant in the DICER1 gene was confirmed.
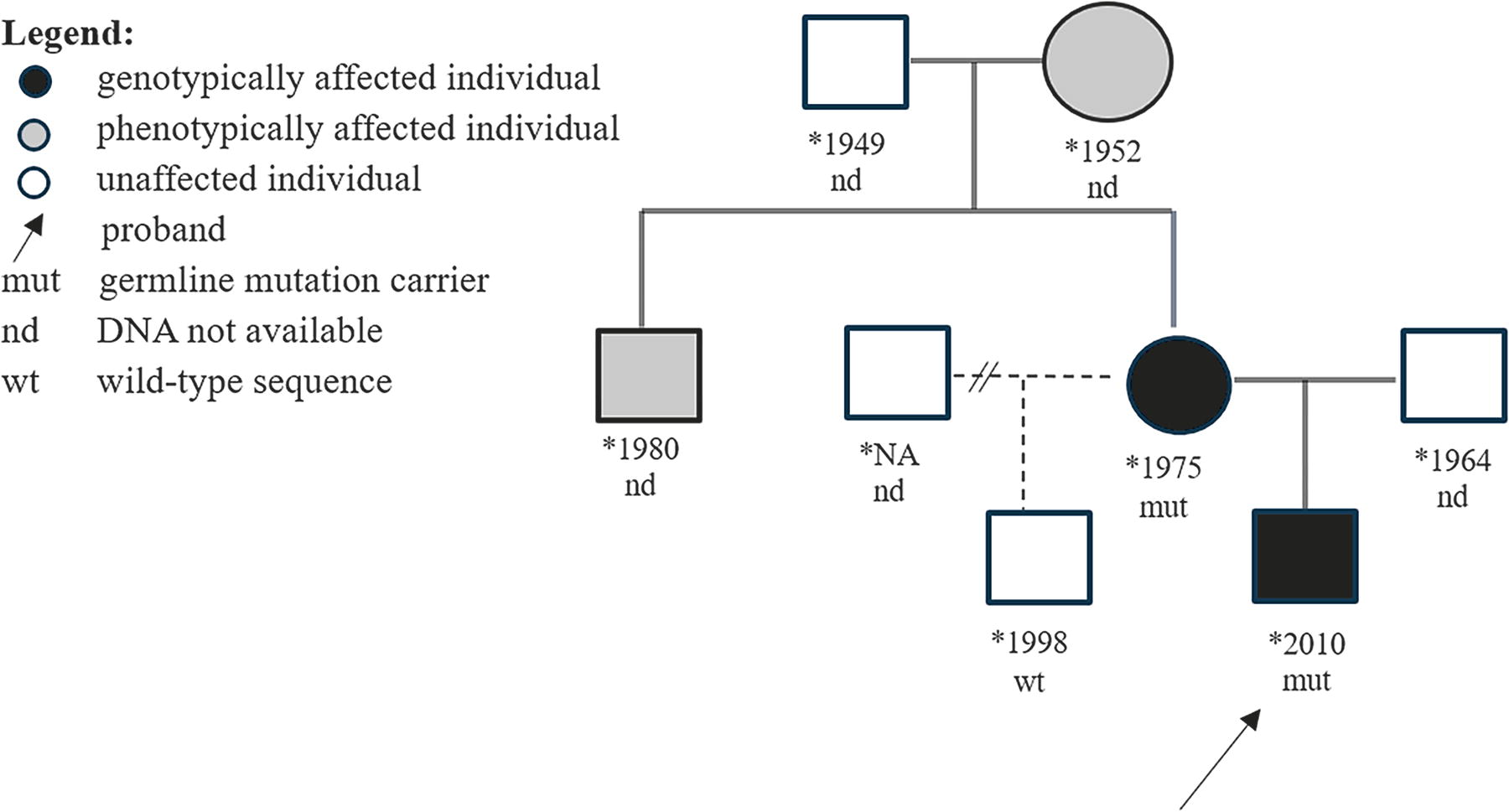
Pedigree of the Family of Case 8.
Patients with tumor-specific DICER1 variants
Two somatic (biallelic) variants in the DICER1 gene were found in a total of 9/24 (37.5%) DICER1-positive samples. In our study, four PTCs were identified with tumor-specific biallelic DICER1 variants. Three of them had a pathogenic hotspot variant in codons 1705 or 1813 followed by a likely pathogenic somatic variant in exons 8, 15, or 21. One patient had a hotspot variant in codon 1813 and a somatic deletion of 3 Mbp.
In five cases (two PTCs, two benign nodules, and one cytologically evaluated FNAB as Bethesda II), only one pathogenic somatic (hotspot) variant was detected. No copy number variants, variants in any exon, or exon–intron boundaries throughout the gene were identified in these samples.
The cohort of patients with DICER1 syndrome/germline variants was compared with patients with tumor-specific DICER1 variants (Table 1). Only the mean age of the patients was statistically significant (p = 0.005); gender, tumor size, and histology did not differ.
Baseline Characteristics of DICER1-Positive Patients and a Comparison of Patients with DICER1 Syndrome/Germline Variant Versus Patients with Somatic DICER1 Variant/s
Values highlighted in bold were statistically significant. SD, standard deviation.
The Comparison of Clinical and Pathological Features of Patients Harboring DICER1-Positive Carcinomas Versus DICER1-Negative Pediatric PTCs
Values highlighted in bold were statistically significant. Tumors were classified according to the 8th edition of the AJCC TNM system, and each response to treatment was determined using definitions reported in the 2015 American Thyroid Association Guidelines.
PTC, papillary thyroid carcinoma.
Pediatric patients with PTCs
The molecular profile of our cohort of pediatric PTCs (n = 141) is shown in Fig. 5. More than half of the pediatric PTCs harbored fusion genes, and one-fifth of the PTCs were positive for BRAF V600E. Variants of the DICER1 gene represented the third most common driver alteration. In 12.8% of pediatric PTCs, the pathogenic genetic change is still unknown.
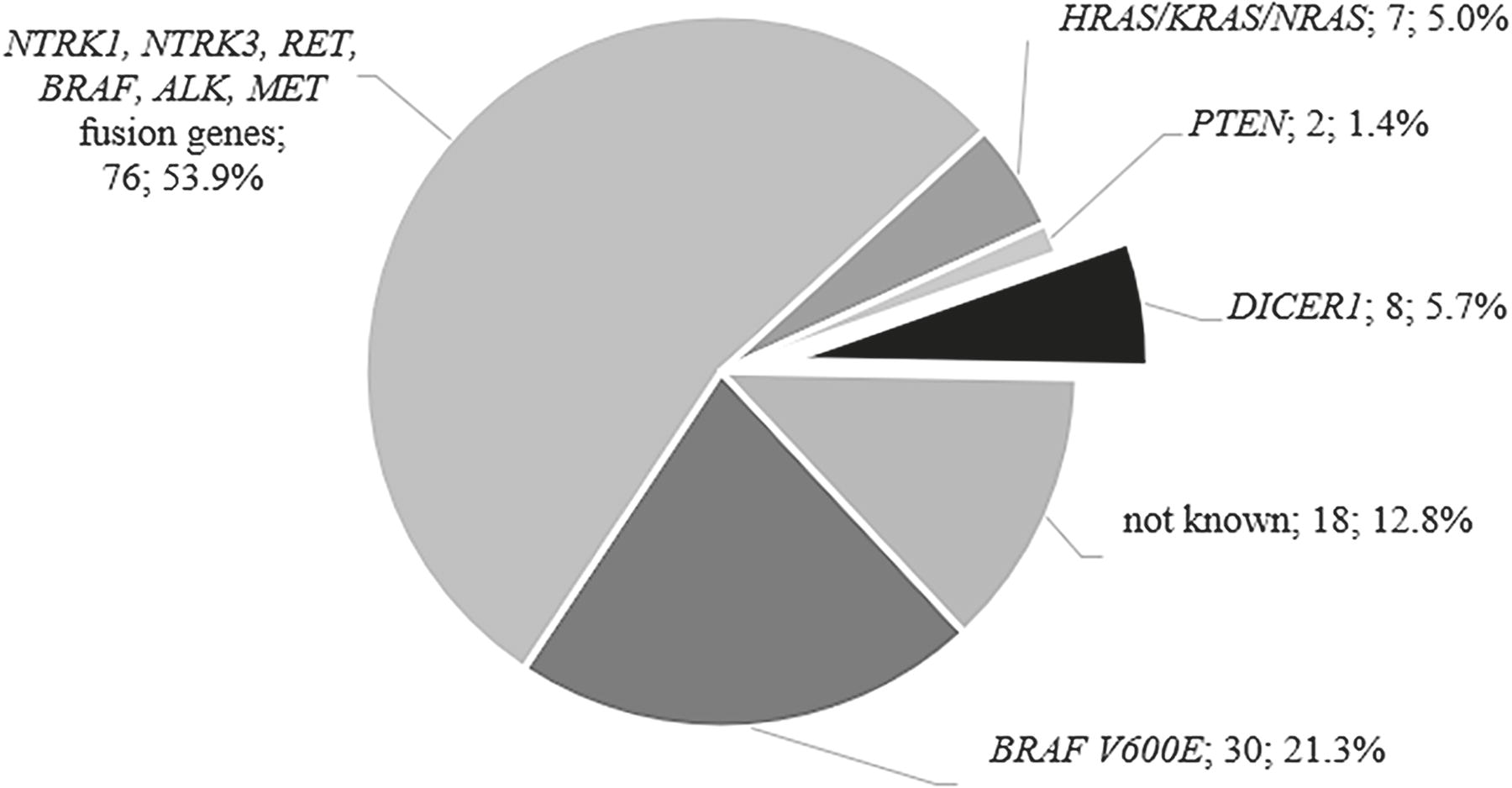
A pie chart of all altered genes detected in a cohort of 141 patients with papillary thyroid carcinoma. Next to the gene name is the number of carcinomas and the percentage.
A total of 8/141 (5.7%) PTCs harbored variants in the DICER1 gene. Four PTCs had biallelic somatic variants, one PTC had only a somatic hotspot variant, one PTC had a somatic hotspot and a germline benign variant, and two PTCs had a somatic hotspot and a pathogenic germline variant (Fig. 3). Most carcinomas (7/8) were a follicular subtype of PTC. Tumors were unifocal (7/8), encapsulated (6/8), and 6/8 (75%) patients had an excellent response to treatment. An exception was in Case 6, where angioinvasion was described and the size of the nodule was 60 mm. This 15-year-old patient received three doses of radioiodine (3.7 + 4.4 + 3.7 GBq) due to persistently elevated levels of stimulated Tg (20 μg/L, no upward trend). The patient is currently in remission with undetectable Tg. Case 8, as mentioned above, received two high doses of radioiodine. The tumors of patients from both cases were radioiodine-avid. A comparison of DICER1-positive carcinomas with the group of DICER1-negative pediatric PTCs is shown in Table 2. DICER1-positive PTCs were statistically significantly larger in size (p = 0.035), but with low invasive pathological features (no extrathyroidal extension [p = 0.039], less frequent LNMs [p = 0.003] and were encapsulated [p = 0.006]). The difference in presence of multifocality only approached the threshold of statistical significance (p = 0.058). Statistically significant differences were also observed for the subtypes of PTCs (follicular subtype p = 0.001; classical subtype p = 0.007).
Preoperatively detected variants in the DICER1 gene
Five of six patients with DICER1-positive nodules tested preoperatively (by FNAB) underwent thyroid surgery. Case 21 was histologically evaluated as an encapsulated follicular carcinoma (FTC) of 9 mm in size, while 4/5 samples were evaluated as benign nodules. One patient (Case 22) with FNAB cytologically evaluated as Bethesda III, and a somatic deletion of at least 3 Mbp long was examined by an endocrinologist several months after sampling. Ultrasound examination showed a regression of nodule size. The patient continues to be monitored at regular intervals.
Discussion
In our cohort variants in the DICER1 gene were detected in 6.9% pediatric and young adult patients, respectively, in 5.7% of patients with PTC. Studies have reported DICER1 variants in up to 22% of pediatric follicular neoplasms (mostly FTAs and FTCs), 7 14% of pediatric PTCs, 4 and 12.5% of pediatric thyroid tumors (PTC and benign nodules). 5 The lower detection rate in our cohort may be explained by the selection of samples (not relying only on follicular morphology), a different upper age threshold, but also by the size of the cohort. To the best of our knowledge, this is the largest cohort of pediatric and young adult thyroid nodules analyzed for variants in the DICER1 gene, and it is also the most comprehensive group of DICER1-positive pediatric and young adult thyroid nodules described to date worldwide. In the literature, the prevalence of DICER1 variants in thyroid tumors in adult patients is significantly lower with a reported 1.4–1.7% detection rate for adult-onset DICER1-positive thyroid nodules. 21,22 There are several differences between adult and pediatric differentiated thyroid carcinomas (DTCs) regarding clinical, pathological, and molecular characteristics, with more frequent NTRK and RET fusion genes in pediatric DTCs. 2
DICER1 variants in our cohort of pediatric PTCs were mutually exclusive with the most common somatic driver alterations and gene rearrangements, which seems to be consistent with other studies. 6,10 Therefore, molecular analysis of the DICER1 gene decreases the percentage of pediatric tumors that lack any driver variant. 5 In preoperatively tested patients with multiple nodules, different nodules from the same individual harbored a distinct hotspot DICER1 variant. These findings have previously been described and suggest that the nodules arose from different clonal events. 4,6,12 Molecular testing of the DICER1 gene in all nodules is therefore desirable. Our findings of DICER1 variants in FNAB samples cytologically evaluated as Bethesda II-IV confirm the results of another study where samples from Bethesda category III-IV prevailed. 23 That study has shown microfollicular patterns, alternatively papillary architecture of these samples. The authors expected benign results in a significant number of thyroid nodules with DICER1 alteration. 23 Our preoperatively tested samples were also histologically evaluated as benign nodules in 4/5 (80%) cases, in contrast to our postoperative cohort where a similar rate of benign and malignant tumors was found.
In our cohort of pediatric DICER1-positive DTCs, follicular-patterned tumors predominated, almost all carcinomas were follicular subtypes of PTC, one FNAB was evaluated postoperatively as minimally invasive FTC, and DICER1 variants were detected in several follicular adenomas as well. This pattern has already been described in the literature. 7
In addition, atrophic changes have recently been described in some DICER1 tumors. 24 We observed atrophic changes only in one location of the parenchyma of a classical PTC (Case 8). Although poorly differentiated thyroid carcinomas (PDTCs) are extremely rare in pediatric and adolescent patients, some cases have been mentioned in the literature. Surprisingly, variants in the DICER1 gene were identified in 5/6 PDTCs with a lack of the molecular alterations characteristic of PDTC in adults. 25 A DICER1 variant coexisted with the TP53 variant in 1 of 2 cases of DICER1-positive PDTCs in another study, 6 however, that study used controversial diagnostic criteria for PDTC (not according to Turin criteria). 25 Pediatric DICER1-positive PDTC were described with widely invasive growth in the thyroid parenchyma but with a lack of vascular invasion. This, along with no recurrence in a 72-month follow-up, suggests better outcomes for the subset of DICER1-positive pediatric PDTCs. 26
DICER1 syndrome is known to have a low penetrance. It was reported that only 5.3% of DICER1 carriers developed a neoplasm by the age of ten years. Later in life, the risk increases in females compared with males, with 26.5% of females and 10.2% of males developing at least one neoplasm by the age of 50. 8 In our study, there are patients with inherited variants of DICER1, who lack a family history of DICER1-related syndromic manifestation. However, DICER1 carriers appear to have a higher penetrance of benign than malignant thyroid nodules. 12,13 The rationale for the inclusion of DICER1 testing in thyroid nodules is also to consider germline testing so that individuals positive for a germline DICER1 variant may initiate follow-up for potentially additional syndromic manifestations.
Although it has been reported that DICER1 carriers have a statistically significant 16- to 24-fold increased risk of developing DTCs compared with the general population, no supporting data are yet available for prophylactic thyroidectomy in individuals with DICER1 syndrome. 12 A recent study showed that hemi-/total thyroidectomy may often be required in cases of DICER1-related thyroid nodules; however, they also stated that radioiodine treatment may be unnecessary given the young age of the patients and the low susceptibility for metastatic disease. 6 When deciding on the treatment of patients with DICER1 variant-positive nodules, it is always important to consider other data such as nodule size, focality, laterality, cytology result, and somatic/germline origin of the DICER1 variant. Active surveillance is reasonable for small unilateral nodules with benign cytology. In the case of large unilateral nodule with Bethesda III-VI cytology, hemithyroidectomy could be considered. If the patient has nodules bilaterally with a germline variant of the DICER1 gene and in addition with Bethesda III-VI cytology, total thyroidectomy may be necessary. Radioiodine treatment may be recommended for patients with DICER1-positive carcinomas with invasive features.
The question arises, if our Case 8 had been immediately evaluated as PTC and had received radioiodine therapy, would the patient later be free of LNMs? The presence of LNMs in pediatric DICER1-positive PTC (respectively one micrometastasis, pN1a) has been mentioned in only one study but unfortunately data regarding radioiodine treatment of this patient are unavailable and only hotspot variants of the DICER1 gene were tested. 4 PTCs positive for DICER1 variants seem to be indolent tumors without extrathyroidal extension and mostly without LNMs compared with DICER1-negative PTCs (where the higher number of LNMs is due to PTCs with fusions). 27 A similar opinion prevails in the literature, 6 although DICER1 variants are rarely found in PDTCs or anaplastic thyroid carcinomas. 22
Case 9 was a typical example of a patient with DICER1 syndrome but without a known germline variant. The patient could have another rare variant (e.g., a deep intronic mutation) not detected by our sequencing assays, or possibly mosaicism. Mosaic DICER1 hotspot mutations have been described with an earlier onset of the first neoplasm and more disease foci than mosaic LOF mutations and autosomal dominant forms. 15,28 In four other cases in our study, the DICER1 hotspot variant was only found in the thyroid nodule. It is hypothesized that it alone may be tumorigenic in some organ/tissue settings. 15,21
Although a low incidence of patients with tumor-specific biallelic DICER1 variants has been described, 5,15,29 our cohort included nine such individuals. It is assumed in the literature that these patients represent sporadic cases of DICER1-related disease and are not at risk for future DICER1-related tumors. 9 Two of our cases were patients with a somatic hotspot variant accompanied by a long-scale somatic deletion (at least 3 Mbp) involving the entire DICER1 gene. Even longer somatic deletions involving the entire DICER1 locus have already been described in two PTCs. 5 Five cases of germline 14q32 deletions associated with DICER1 syndrome have also been described. 30 These findings demonstrate the importance of incorporating molecular techniques capable of revealing long-scale deletions in the screening of the DICER1 gene.
A limitation of our study is the size of the cohort of patients with DICER1 variants. Since the detection of variants in the DICER1 gene is rare, multicenter studies would be appropriate to increase the statistical power and significance of clinicopathological results. Long-term follow-up of probands and family members with germline variants would certainly be of interest to assess the long-term risk associated with DICER1 syndrome.
Conclusion
In summary, in our study 24/350 (6.9%) pediatric and young adult patients had DICER1-positive thyroid nodules. Variants in the DICER1 gene were detected in 8/141 (5.7%) PTCs. Patients with germline variants/DICER1 syndrome were identified in 11/24 cases (45.8%).
Currently, molecular testing of DICER1 variants has become important in assessing pediatric thyroid nodules. Molecular analysis should be performed for at least somatic hotspot variants of the DICER1 gene, and if positive, screening the rest of the gene including exon–intron boundaries (not just the canonical splice site) for the possible detection of other variants should be performed. When analyzing the DICER1 gene, it is important to examine for the presence of large-scale duplications/deletions.
All detected germline mutation carriers are individuals with DICER1 syndrome and should be monitored regularly according to the given surveillance recommendations. 31 However, patients with somatic biallelic DICER1 variants represent sporadic cases of DICER1-related disease without the risk of future DICER1-related tumors.
Most PTCs with DICER1 variants seem to be less invasive in nature and patients have mostly had an excellent response to the treatment. More conservative management of DICER1-positive PTCs with low-risk features may be considered.
Footnotes
Acknowledgment
Acknowledgements to cooperating departments and their specialists.
Authors’ Contributions
K.M.: Conceptualization (equal), methodology, investigation (lead), formal analysis, writing—original draft, and writing—review and editing (equal). B.B.P.: Conceptualization (equal), investigation (equal), formal analysis (supporting), and writing—review and editing (equal). V.K.: Conceptualization (supporting), investigation (equal), and writing—review and editing (equal). E.V.: Investigation (equal). J.C.: Investigation (equal). R.K.: Resources—sample and data collection, and writing—review and editing (supporting). L.F.: Resources—sample and data collection (supporting), and funding acquisition (supporting). P.V.: Resources—clinical data evaluation. D.K. and J.D.: Resources—pathological assessment (equal). M.C.: Resources—sample and data collection. R.M.: Resources—pathological assessment (equal) and writing—review and editing (supporting). P.P. and Z.N.: Resources—sample and data collection. K.P.: Resources—clinical data evaluation (supporting). J.V.: Software, supervision (equal). B.B.: Conceptualization (supporting), supervision (equal), funding acquisition (lead), and writing—review and editing (equal). All authors contributed to this research and reviewed and approved the final version of the article.
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
This work was supported by the Ministry of Health of the Czech Republic AZV
Supplementary Material
Supplementary Table S1
Supplementary Table S2
