Abstract
Introduction:
Thyroid eye disease (TED) is an autoimmune process characterized by extraocular muscle and orbital fat remodeling/expansion resulting in swelling, pain, redness, proptosis, and diplopia. Teprotumumab, an insulin-like growth factor-I receptor inhibitor, demonstrated improvements in TED signs and symptoms in three adequately powered clinical trials of 24 weeks duration. Here we analyze the long-term maintenance of responses with teprotumumab from these trials.
Methods:
A total of 112 patients who received 7 or 8 infusions of teprotumumab in the Phase 2, Phase 3 (OPTIC study), and OPTIC Extension (OPTIC-X) studies were included in this analysis. Responses, including clinical activity score (CAS ≥2-point improvement), the European Group of Graves’ Orbitopathy ophthalmic composite outcome, diplopia (≥1 Gorman grade improvement), proptosis (≥2 mm improvement), Overall (improvement in proptosis + CAS), and disease inactivation (CAS ≤1), were assessed and pooled from study baseline to week 24 (formal study) and up to week 72 (formal follow-up). Graves’ Ophthalmopathy quality-of-life (GO-QoL) scores were also assessed. Outcomes included the percentages of observed patient responses from the study baseline. Additional alternative treatments for TED were assessed as a surrogate of persistent benefit from week 24 through week 120 (extended follow-up). Studies differed in the timing of follow-up visits, and data from some visits were unavailable.
Results:
At week 72, 52/57 (91.2%), 51/57 (89.5%), 35/48 (72.9%), 38/56 (67.9%), and 37/56 (66.1%) of patients were responders for CAS, composite outcome, diplopia, proptosis, and Overall response, respectively. The mean reduction in proptosis was 2.68 mm (SD 1.92, n = 56), mean GO-QoL improvement was 15.22 (SE 2.82, n = 56), and disease inactivation (CAS ≤1) was detected in 40/57 (70.2%). Over 99 weeks following teprotumumab therapy, 19/106 (17.9%) patients reported additional TED therapy during formal and extended follow-up.
Conclusion:
The long-term response to teprotumumab as observed 51 weeks after therapy was similar to week 24 results in the controlled clinical trials. Inflammatory and ophthalmic composite outcome improvements were seen in 90% of patients with nearly 70% reporting improvement in diplopia and proptosis. Further, 82% of patients in this analysis did not report additional TED treatment (including surgery) over 99 weeks following the final teprotumumab dose.
Introduction
Thyroid eye disease (TED) is an autoimmune process most frequently affecting individuals with Graves’ disease (GD). 1 –3 It is characterized by remodeling and expansion of fat, connective tissue, and extraocular muscles. TED is associated with significant quality-of-life disutility. 3,4 Major disease manifestations include strabismus, diplopia, and proptosis. 3 The inflammatory findings historically have been described as waning 1–3 years after initial disease development; 5 however, recent studies indicate that patients with moderate-severe disease often experience symptoms chronically. 6,7 Sequelae of proptosis and diplopia can profoundly impact patients’ daily living, mental health, and quality of life. 6 –13
While steroids have been prescribed historically to address predominately inflammation-related symptoms/signs of TED, they may have a limited effect on proptosis and diplopia. 14,15 Recently, an American Thyroid Association/European Thyroid Association Consensus Statement recommended first-line use of teprotumumab, a fully human monoclonal insulin-like growth factor-I receptor (IGF-IR) inhibitory antibody, for patients with moderate-severe disease presenting with proptosis and/or diplopia. 2 These recommendations were based on results from three major trials: the randomized, masked, placebo-controlled Phase 2, 16 Phase 3 (Treatment of Graves' Orbitopathy [Thyroid Eye Disease] to Reduce Proptosis with Teprotumumab Infusions in a Randomized, Placebo-Controlled, Clinical Study [OPTIC]) 17 with expanded pooled/exploratory results from these two studies, 18 and the OPTIC open-label, retreatment/extension study (OPTIC-X). 19 The placebo-controlled Phase 2 and OPTIC trials included follow-up 51 weeks after last teprotumumab treatment and 48 weeks after the initial trial completion. OPTIC placebo and teprotumumab nonresponders who received teprotumumab in OPTIC-X were evaluated 27 weeks after final teprotumumab dose. 19 The follow-up data from these trials are assessed here for response from pretreatment baseline. We pooled the formal follow-up findings from the Phase 2, OPTIC, and OPTIC-X studies and reported additional TED therapy up to 99 weeks after teprotumumab. These are the longest follow-up data on teprotumumab efficacy available to date.
Materials and Methods
Patients and studies
Patients were assessed for outcome responses as previously defined in the Phase 2, 16 OPTIC, 17 and OPTIC-X 19 studies during formal follow-up. Additionally, patients requiring supplemental TED treatments throughout formal and extended follow-up were identified. The trial designs have been described in detail. 16 –19 Briefly, they were conducted at 28 US and European specialty academic referral centers. They included 121 patients assigned to teprotumumab; Phase 2 and OPTIC patients had similar demographics and study criteria.
Adult patients with GD aged 18–75 years in the Phase 2 trial 16 or 18–80 in OPTIC 17 with recent onset (≤9 month duration) active TED, as defined by clinical activity score (CAS) ≥4, were included. Placebo and teprotumumab non responders from OPTIC could enter the open-label OPTIC-X trial immediately. The current analysis also included the OPTIC placebo-non-responding patients who received initial teprotumumab treatment in the open-label OPTIC-X study and therefore had a longer TED duration. 19 Patients with previous medical and orbital radiation or surgical treatment for TED were excluded. All patients received eight teprotumumab infusions (10 mg/kg for the first infusion, 20 mg/kg for subsequent infusions over a total of 21 weeks) except one who received seven, with the final study visit at Week 24 (3 weeks after the final dose).
Only patients who received one course of teprotumumab and entered the follow-up were included, and outcome responses were assessed from original study baseline (Fig. 1). In the formal follow-up, patients from the Phase 2 and OPTIC trials were assessed out to study Week 72 (51 weeks after the final dose). Assessments for the non-responding OPTIC placebo patients receiving their initial teprotumumab course in OPTIC-X were completed at Week 48 (27 weeks after the final teprotumumab dose). Patient data obtained in the follow-up periods from Phase 2, OPTIC, OPTIC-X studies were pooled at common time points.
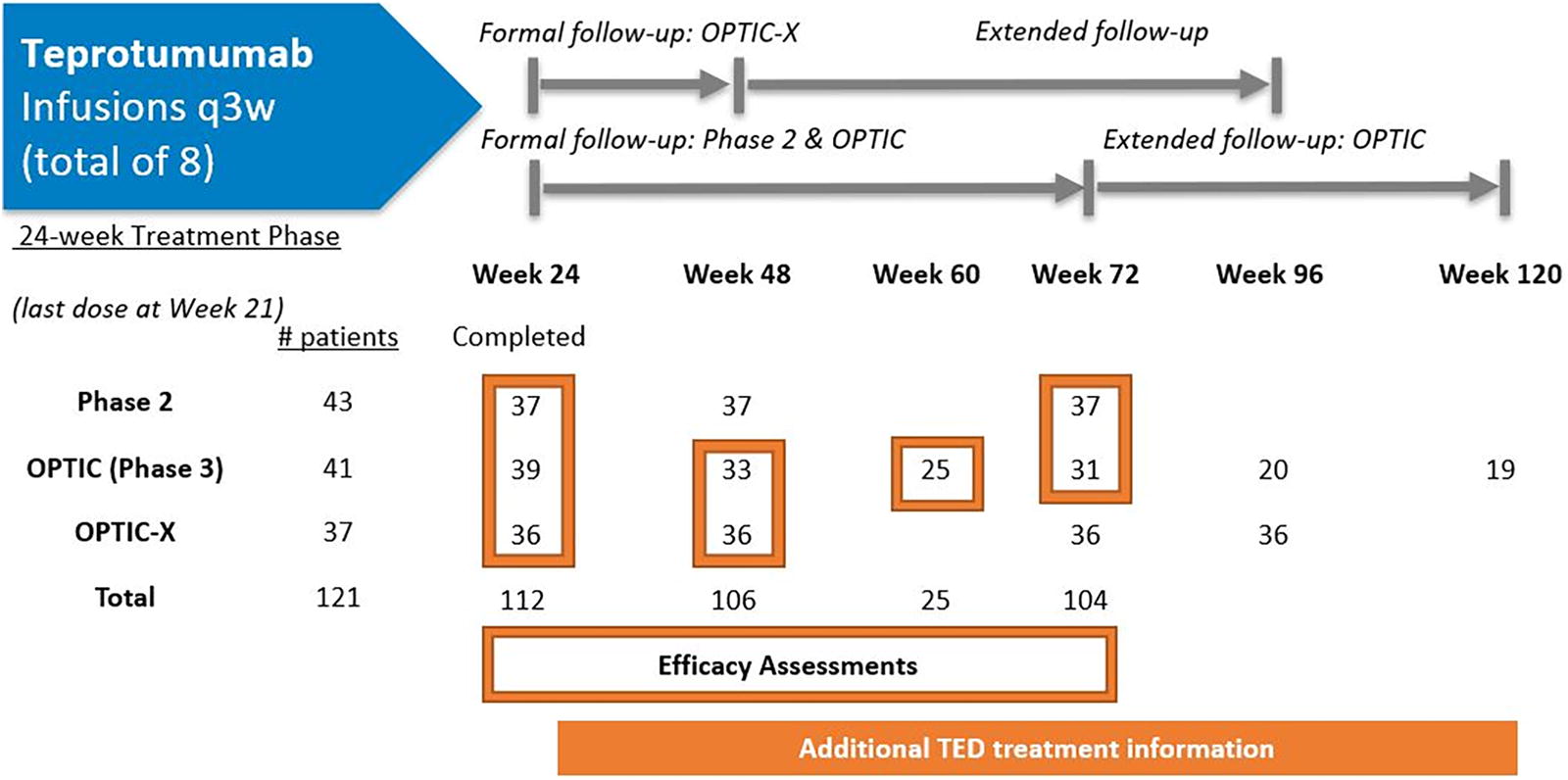
Teprotumumab-treated patients receiving a full course and assessments during follow-up times from three clinical trials. Teprotumumab-treated patients receiving one full course includes 1 patient who received 7/8 infusions and completed the treatment period. Efficacy assessments: at Week 48: 37 patients from Phase 2 (GO-QoL only), 33 from OPTIC and 35 from OPTIC-X (1 additional OPTIC-X patient had only additional therapy data) and for outcomes other than GO-QoL, n = 68; at Week 60: 25 from OPTIC; at Week 72: 37 from Phase 2 (3 missing some outcomes, so n = 36), 21 from OPTIC, and none from OPTIC-X, a total n of 57. Additional treatments: at Week 48: 106 patients; Week 72: 104; Week 96: 56; Week 120: 19. Formal follow-up: Through Week 72 for Phase 2 (all patients followed up) and OPTIC (proptosis responders); Through Week 48 for OPTIC-X. Extended follow-up: included contact at Week 96 and 120 for OPTIC and Week 72 and 96 for OPTIC-X.
An extended follow-up period was completed to determine if patients had received additional medications or surgery for TED after the formal follow-up. OPTIC responders who lost proptosis response from baseline in the follow-up and received another course of teprotumumab in OPTIC-X were considered to be receiving additional therapy. 19 OPTIC patients completing a Week 72 visit were contacted at Weeks 96 and 120, and OPTIC-X patients who completed the Week 48 visit were contacted at Weeks 72 and 96.
Outcomes
All outcomes that were collected in the trials were assessed and pooled (where possible) at each time point during formal follow-up (up to Week 72) from baseline and included: the proportion of patients with CAS response (≥2 points reduction from baseline) and mean change from baseline and from Week 24 to Week 72, disease inactivation (CAS < 2), the European Group on Graves’ Orbitopathy (EUGOGO)-recommended ophthalmic composite response (i.e., improvement of at least two of the following without respective deterioration of at least two others in either eye: proptosis, lid-width, eye muscle motility, and the five-item CAS excluding the subjective patient-reported spontaneous or gaze-evoked pain), 1 diplopia response (diplopia reduction of ≥1 Gorman grade [range, 0–3]) among patients with baseline diplopia 20 and mean diplopia change from Week 24 to Week 72, proptosis response (≥2 mm decrease in proptosis in the study eye without a corresponding increase ≥2 mm in the fellow [contralateral] eye) and mean change in proptosis (mm) from baseline and from Week 24 to Week 72, and Overall response (reduction ≥2 mm proptosis plus CAS reduction ≥2 points).
The mean change from baseline for the total and the visual and appearance subscales in the Graves’ Ophthalmopathy quality of life questionnaire (GO-QoL) scores 21 was also assessed. Positive changes of 10 to 20 points were noted and are considered clinically relevant, moderate, and large, respectively. 22 Week 24 responders for CAS, diplopia, proptosis, and GO-QoL (≥10-point improvement) were assessed for maintenance of response from Week 24 to Week 72 and for worsening in CAS (≥2 points), proptosis (≥2 mm), diplopia (≥1 grade), and GO-QoL (≥10 points) from Week 24. Outcome responder rates were assessed without and with patients receiving additional TED therapies during formal follow-up (up to Week 72).
Additional medications and surgeries for TED were assessed, during the formal and extended follow-up period. Extended follow-up was available for OPTIC (Weeks 96 and 120) and OPTIC-X (Weeks 72 and 96). Additional therapies included intravenous (IV) and high-dose oral steroids, an additional course of teprotumumab if the patient initially responded to the first course and lost their proptosis response, specific TED therapy resulting in study withdrawal, and TED surgical procedures (including strabismus, orbital decompression, tarsoraphy). Additional courses of teprotumumab were administered to OPTIC teprotumumab-treated patients meeting very broad flare criteria (in OPTIC-X). Flare was adjudicated if CAS worsened ≥2 points with a total CAS score of ≥4 (in the study eye or both) and/or study-eye proptosis worsened ≥2 mm from Week 24. In this analysis, patients from OPTIC who were treated with an additional course of teprotumumab in OPTIC-X were not counted as needing additional treatment if their proptosis reduction from OPTIC baseline was ≥2 mm.
Safety in the follow-up is reported, including adverse events of special interest and whether events are resolved. Adverse event severity was graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.03. 23 All patients provided written informed consent. The studies were registered: Phase 2 (NCT01868997, EUDRACT 2014-000113-31), Phase 3-OPTIC (NCT03298867, EUDRACT 2017–002763-18), and OPTIC-X (NCT03461211).
Results
A total of 121 patients were assessed (43 from Phase 2, 41 from OPTIC, and 37 from OPTIC-X) with 112 (92.6%) receiving 7 or 8 teprotumumab infusions and 106 (95%) entering the follow-up (Fig. 1). The total exposure for those in the follow-up was approximately 132 patient-years. 24 TED duration was longer in OPTIC-X patients compared to Phase 2 and OPTIC with OPTIC-X patients having slightly lower CAS. Other baseline characteristics were generally similar (Table 1).
Characteristics and Demographics at Study Baseline of Pooled Trial Patients Receiving One Full Course of Teprotumumab (n = 112)
For free T3, normal range 3.5–6.5 pmol/L, n = 111.
For free T4, normal range is 11.5–22.7 pmol/L, n = 111.
CAS, Clinical Activity Score; GO-QoL, Graves’ orbitopathy quality of life questionnaire.
At Week 48, patients were assessed for responses and GO-QoL in OPTIC (n = 33) and OPTIC-X (n = 35) trials. Only GO-QoL data were assessed at Week 48 for Phase 2, while only OPTIC patients underwent Week 60 assessments (n =25). At Week 72, outcomes were available for both Phase 2 (n = 37 with three Phase 2 patients missing some outcomes) and OPTIC (n = 21) trials. By Week 48, 106 patients (37 from Phase 2, 33 from OPTIC, and 36 from OPTIC-X) were included in the analysis of additional TED therapy. Details for these analyses can be found in Supplementary Figures S1, S2, and S3.
Clinical activity score
Pooled CAS response at Week 24 was observed in 97/112 (86.6%), 54/68 (79.4%) at Week 48, and 21/25 (84.0%) at Week 60 (Fig. 2A). At Week 72, 52/57 (91.2%; CI [0.81, 0.97]) patients were CAS responders. Mean (SD) CAS reduction from baseline throughout follow-up was similar: 3.4 (1.58) at Week 24 (n = 112), 3.2 (1.67) at Week 48 (n = 68), 3.6 (1.60) at Week 60 (n = 25) and 3.9 (SD 1.64) at Week 72 (n = 57) with a mean (SD) change from Week 24 to Week 72 of 0.00 (1.63), range −4.0 to 5.0. Disease inactivation (i.e., CAS ≤ 1) was observed in 78/112 (69.6%) at Week 24, 49/68 (72.1%) at Week 48, and 14/25 (56.0%) at Week 60 (Fig. 2B). At Week 72, 40/57 (70.2%; CI [0.57, 0.82]) patients with disease inactivation were observed.
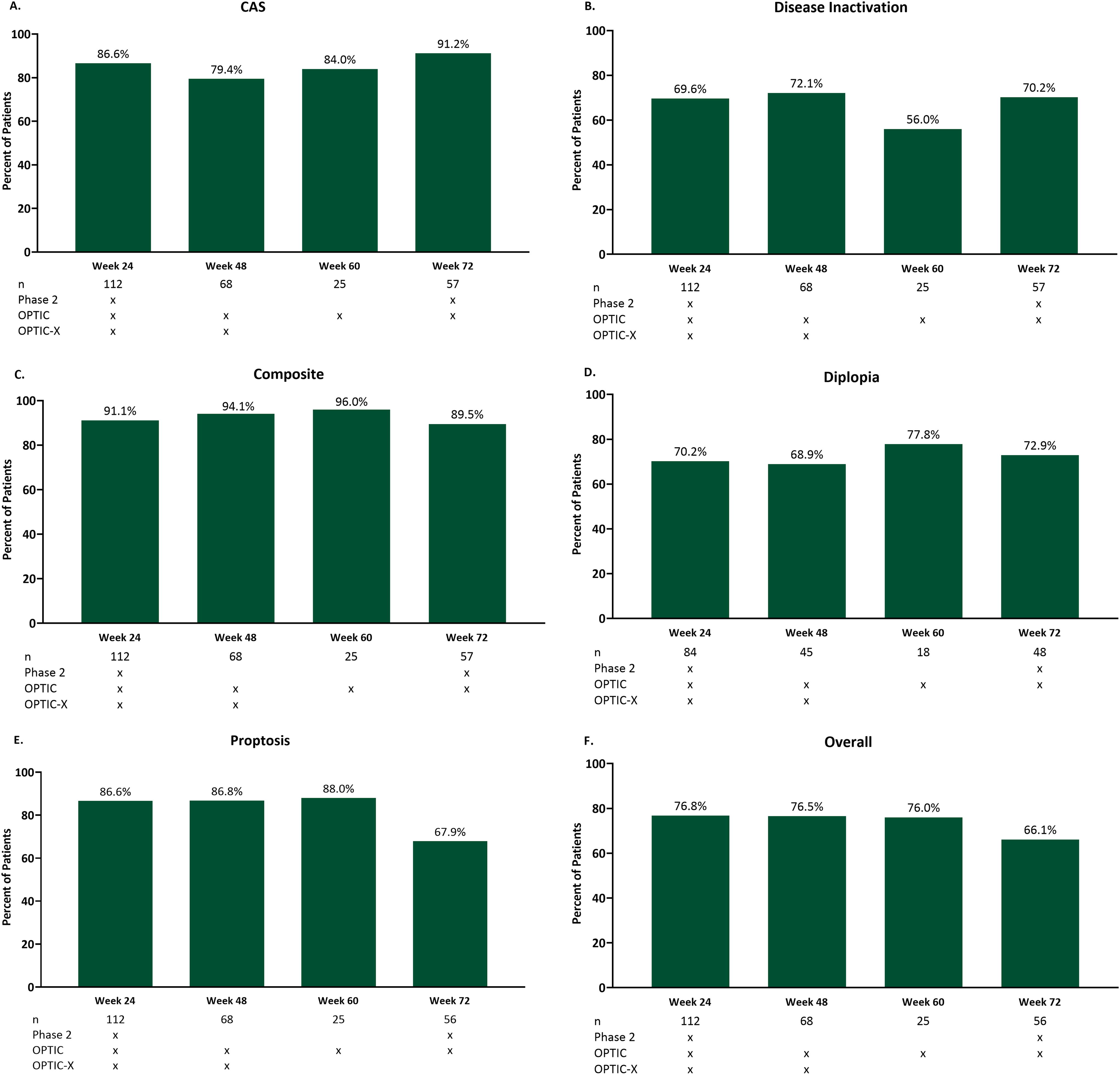
Follow-up response outcomes in pooled teprotumumab-treated trial patients. Outcomes are reported including patients who received additional TED treatments. CAS, Clinical Activity Score.
Composite responders
For the EUGOGO-defined composite outcome, which included proptosis, lid-width, eye muscle motility, and five-item CAS improvement, 102/112 (91.1%) had a response at Week 24, which was similar throughout the follow-up (Fig. 2C). At Week 48, 64/68 (94.1%) patients responded, and 24/25 (96.0%) patients responded at Week 60. At Week 72, 51/57 (89.5%; CI [0.78, 0.96]) patients were responders. No major baseline differences were observed in long-term responders versus nonresponders at Week 72 (Supplementary Table S1). Further, the difference in serum thyroid hormone levels from baseline to Week 72 between these responders and nonresponders was not significantly different.
Diplopia
Diplopia response was sustained throughout follow-up among patients with diplopia at baseline: 59/84 (70.2%) at Week 24, 31/45 (68.9%) at Week 48, and 14/18 (77.8%) at Week 60 (Fig. 2D). At Week 72, 35/48 (72.9%; CI [0.58, 0.85]) diplopia responders were observed. Mean (SD) diplopia score change from Week 24 to 72 was 0.05 (0.961), range −3.0 to 3.0, median 0.00 (n =56).
Proptosis
At Week 24, 97/112 (86.6%) of the pooled teprotumumab-treated patients were proptosis responders from baseline (mean [SD] proptosis reduction 3.33 [1.60] mm, n = 112) (Fig. 2E). At Week 48, 59/68 (86.8%) patients responded (mean [SD] reduction 3.22 [1.86] mm, n = 68) from baseline. At Week 60, 22/25 (88.0%) were proptosis responders (mean [SD] reduction 3.62 [1.76] mm, n = 25). At Week 72, 38/56 (67.9%; CI [0.54, 0.80]) responders were observed (mean [SD] reduction 2.68 [1.92] mm; CI [2.17, 3.19], n = 56). The mean (SD) proptosis change from Week 24 to Week 72 was 0.54 (1.810), range −3.0 to 5.0 mm (n = 56).
Overall responders (2-point CAS and 2-mm proptosis improvement)
Overall responder proportions were similar throughout the follow-up: 86/112 (76.8%) responded at Week 24, 52/68 (76.5%) responded at Week 48, and 19/25 (76.0%) responded at Week 60 (Fig. 2F). At Week 72, 37/56 (66.1%; CI [0.52, 0.78]) patients were Overall responders.
GO-QoL
Improvement in GO-QoL scores was similar throughout follow-up (Fig. 3). Mean (SE) change from baseline was 17.73 (1.92) for the total score, 18.24 (2.52) for the visual function subscale, and 17.30 (2.06) for the appearance subscale at Week 24 (n = 112). Mean (SE) change from baseline was 17.87 (2.00) for the total score, 18.03 (2.65) for visual function, and 17.80 (2.18) for appearance at Week 48 (n = 105). GO-QoL mean change from baseline was 15.22 (SE 2.82; CI [9.573, 20.867]) for total, 15.73 (SE 3.58; CI [8.548, 22.912]) for visual function, and 14.96 (SE 3.07; CI [8.815, 21.105]) for the appearance subscale at Week 72 (n = 56). Each subscale improvement was considered a moderate-large change. 22 Mean change from Week 24 to Week 72 was −3.90 (median: 0.00, range: −50.0, 87.5) for the total GO-QoL, −4.11 (median 0.00, range: −68.8, 93.8) for visual function, and −3.57 (median: 0.00, range −50.0, 81.3) for appearance.
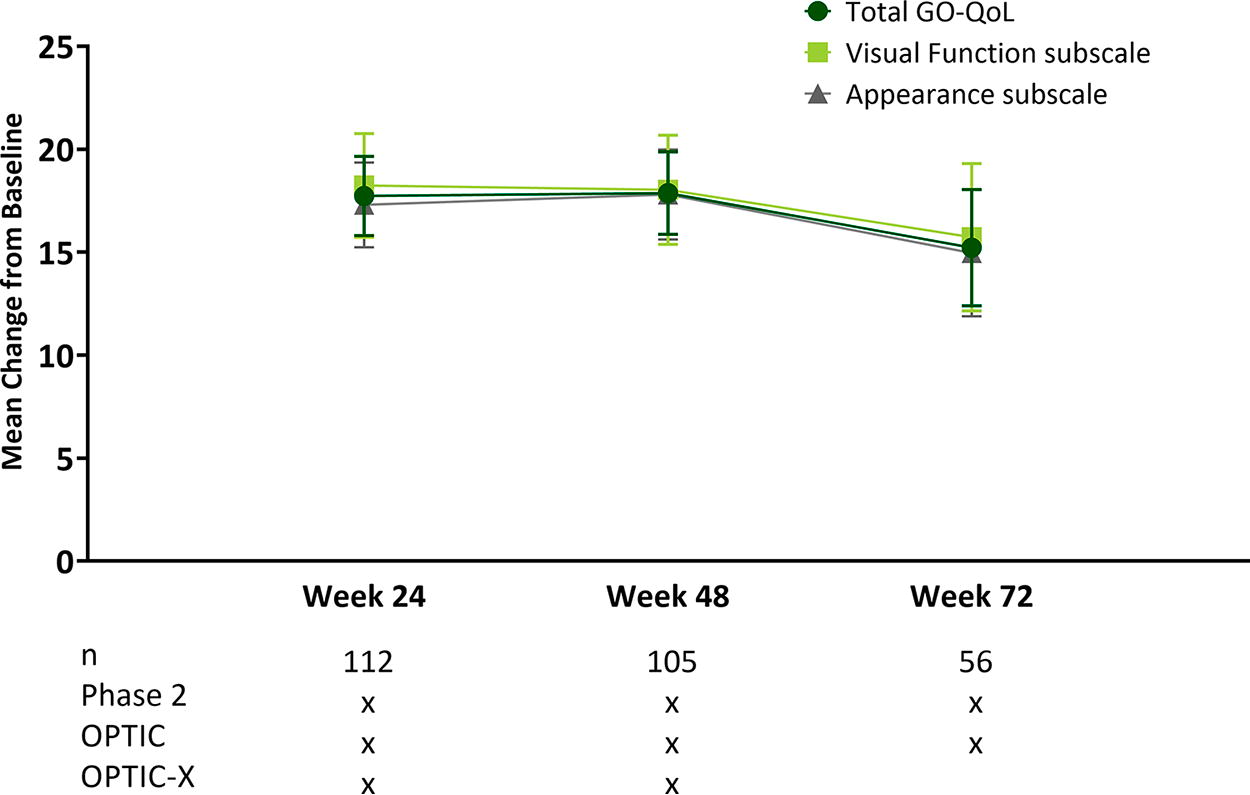
Follow-up mean change from baseline GO-QoL scores in pooled teprotumumab-treated trial patients. Error bars are standard error. GO-QoL, Graves’ orbitopathy quality of life questionnaire.
Maintenance of response
Among CAS responders at Week 24, 45/54 (90.7%; CI [79.70, 96.92]) maintained response at Week 72. Of diplopia responders at Week 24, 31/35 (88.6%; CI [73.26, 96.80]) maintained response at Week 72. Among proptosis responders at Week 24, 35/50 (70%; CI [55.39, 82.14]) maintained response at Week 72. Of patients with ≥10-point GO-QoL improvement from baseline at Week 24, 21/33 (63.6%; CI [45.12, 79.60]) maintained this response at Week 72. Worsening occurred in 7/54 (12.9%) of CAS, 7/35 (20%) of diplopia, 12/50 (24%) of proptosis, and 14/33 (42.4%) of GO-QoL responders at Week 72. Further, three new patients became responders by Week 72, each for CAS and proptosis.
Additional treatments for TED during follow-up periods
Few teprotumumab-treated patients received additional therapies during the formal follow-up periods (Table 2). By Week 48, 3/106 (2.8%) had received supplemental therapy. In addition to collecting TED treatment information during formal follow-up, OPTIC-X patients were contacted regarding additional therapies at Week 72 and Week 96 and patients from OPTIC at Weeks 96 and 120. The Phase 2 study did not include extended contact.
Additional Therapies for TED After 24-Week Treatment Period
This includes therapies for TED any time after the 24-week Treatment Period.
From the 112 teprotumumab patients who completed one full course, five OPTIC nonresponders withdrew from follow-up for OPTIC-X and one OPTIC patient withdrew for radioiodine treatment.
CAS, Clinical Activity Score; TED, thyroid eye disease. “Strabismus” refers to corrective surgery.
By Week 72 (51 weeks after the last teprotumumab dose), 18/106 (17.0%; CI [0.10, 0.26]) had received additional TED therapy. By Week 120, one other patient had received additional therapy from Week 72; thus, 19/106 (17.9%; CI [0.11, 0.27]) underwent additional therapy out to 99 weeks after the final dose of teprotumumab. Post-teprotumumab treatment, 3 patients in OPTIC follow-up met the criteria of this analysis, not maintaining a 2 mm reduction from baseline, and received additional teprotumumab treatment in OPTIC-X. Otherwise, 15% (16/106) received systemic steroids and/or remedial periocular surgery (6 patients receiving steroids only, 7 undergoing surgical procedures only and 3 receiving steroids) as of 99 weeks post-therapy. Only three of 106 patients (2.8%) underwent orbital decompressions. Similar responses for all responder outcomes were seen in those without or with additional therapy out to Week 72 (Supplementary Table S2).
Safety
Adverse events (AEs) were collected in the formal follow-up (Week 72 for Phase 2 and OPTIC and Week 48 for OPTIC-X) with 113 teprotumumab-treated patients included in the follow-up period safety analysis, including one who did not complete therapy. No new safety concerns were identified from the follow-up period with most AEs being mild-to-moderate (Supplementary Table S3). 18,19 Two serious and severe AEs (intercostal neuralgia and optic neuropathy) were considered unrelated to teprotumumab. 18 The only other severe AE occurring in the follow-up, an event of hypothyroidism, was not serious and was considered unrelated to teprotumumab. 18 The rest of AEs in the follow-up were mild or moderate. There were no deaths.
Of the four hyperglycemia-related events newly reported during the follow-up, two were hyperglycemia that resolved without medication, one case of glucose intolerance resolved, and one diabetes event occurring in a patient with prediabetes was treated with metformin and was ongoing when the patient discontinued from the study due to a flare. 18 There were two additional hyperglycemia-related events of type 2 diabetes persisting from the treatment period in patients who were prediabetic at baseline. 19 Any other treatment-emergent hyperglycemia-related events resolved during the treatment period. 18,19,25
Among the three hearing-related AEs newly reported in the follow-up, one case of hypoacusis (mild intensity) considered unrelated by the investigator to study drug was ongoing at last visit, and a case of unilateral deafness in a patient with previous eustachian tube dysfunction was improved on study day 497 compared to study day 274. 18 The third one was a case of neurosensory deafness that was mild and considered unrelated to teprotumumab by the investigator for which the patient was administered a hearing aid. Four additional hearing-related AEs were ongoing from the treatment period: hypoacusis in a patient during a second teprotumumab course who had recovered from a deafness AE, recurring autophony in a patient during a second teprotumumab course, one hearing event that was improving but was lost to follow-up, and a recurring tinnitus event. 18,19 No patients discontinued because of these treatment-emergent hyperglycemia or hearing events.
Discussion
A dearth of literature exists concerning TED recrudescence, primarily because, until recently, few pharmacotherapies had an appreciable effect on proptosis and diplopia, the main hallmarks of TED progression and severity. 15,26 Previous publications, predominantly with steroid therapy, have reported remission of inflammation, which generally quiesces over time. 27,28 Previous studies assessing the efficacy of TED therapies have rarely examined the long-term impact on proptosis and diplopia. Inflammatory relapse rates with IV steroid therapy can approach 25–30%, but limited data are available regarding relapse rates for proptosis and diplopia, likely the consequence of an inadequate number of patients demonstrating an initial response. 15,26 As is becoming increasingly evident, TED is a chronic life-long disease, which can be symptomatic and life-altering over decades, especially in those with moderate to severe disease. 6 –8,29,30 A Dutch follow-up study of 154 patients treated with steroids and/or radiotherapy and/or orbital surgery reported the continued presence of proptosis (60%) and diplopia (51%) out to a median follow-up of nearly 12 years. 31
Teprotumumab is the first pharmacotherapy to meaningfully impact either proptosis or diplopia in three 6-month trials. 18,19 These results are superior to those of the current EUGOGO-recommended IV steroid dosing. 15 A proptosis relapse rate with teprotumumab has previously been cited as 37% at 72 weeks; 2 however, it is important to note that many patients in the teprotumumab clinical trials maintained large proptosis reductions and remained proptosis responders while meeting this definition of relapse during follow-up. The same issue applies to patients considered to have flared during any follow-up period after teprotumumab therapy and simultaneously who remained study responders upon follow-up. As an example, 6/9 patients entering the OPTIC-X study for retreatment with teprotumumab, according to this flare definition, remained proptosis study responders. 19
The current analysis details findings regarding inflammatory and progressive outcomes from these trials in patients followed up to 51 weeks after therapy conclusion, as well as identifies additional therapies received up to 99 weeks as available (Supplementary Fig. S4). These follow-up data indicate that inflammatory outcome responses (CAS improved by ≥2) were maintained in >90% of reporting patients, nearly equivalent to the pooled population findings at formal study end (Week 24) from the three clinical trials. Diplopia improvement and proptosis response were reported in 73% and 68%, respectively. Among study responders who worsened in the follow-up, outcomes were still often clinically meaningful and better than baseline, as exemplified among patients with proptosis, diplopia, and GO-QoL clinical worsening where 25%, 43%, and 29%, respectively, remained responders by Week 72. These data, taken collectively, indicate a maintenance of study response status from the formal study end in both inflammatory and progressive outcomes up to nearly one year after teprotumumab cessation. No major trends in baseline characteristics were observed among long-term responders versus nonresponders. Few patients received additional TED therapy during the 72-week follow-up, with no impact on outcome responses observed between those with or without additional therapy. In all, up to 99 weeks post-teprotumumab therapy, 82% of patients did not report need for additional medical or surgical therapy for TED.
An earlier study of the most frequently recommended and utilized 12-week IV steroid regimen reported minimal proptosis (20% improved, 13% worsened with a net 0.4 mm reduction) and motility improvement (26% improved while 34% worsened). 27 Only 35% of the patients had overall ophthalmical improvement, while 21% of these worsened during posttreatment follow-up at Week 24. Finally, rehabilitative surgery (orbital decompression and/or strabismus, and/or eyelid surgery) was completed during the follow-up in 15/49 (31%) patients with long-term follow-up.
In regard to other TED therapies used, in a very small, double-masked study comparing different doses of rituximab with a fixed high-dose IV steroid treatment, disease reactivation, defined by inflammatory flare, was seen in 5/11 CAS responders (45%) in the steroid-treated group with a proptosis response rate of 1/16 (6.25%) and Gorman diplopia response rate 1/15 (6.7%) over the follow-up period of 52 weeks. 32 Rituximab results indicated initial diplopia (3/15, 20%) and proptosis (0/15, 0%) response at Week 24 were reported to improve to 8/15 (54%) at Week 72 for diplopia and 5/15 (33%) for proptosis at Week 52 during the follow-up, indicating spontaneous improvement in about 33% of non-responding patients well after treatment cessation. 32 TED-related surgical procedures were completed on 10/16 (63%) of IV steroid-treated patients versus 3/15 (20%) with rituximab during the follow-up. Another 24-week, placebo-controlled trial of rituximab indicated that proptosis was reduced minimally (<1 mm), and diplopia worsened from baseline to Week 52 in both groups. 33 Additionally, in a 12-week, placebo-controlled trial with tocilizumab in a small group with steroid-resistant TED, no significant differences were identified versus placebo with regard to diplopia or proptosis response at Week 40 follow-up (28 weeks after treatment). 34 Finally, although head-to-head controlled study comparisons between teprotumumab and IV steroids have yet to be performed, the data reported here and meta-analyses of existing data suggest the superiority of the IGF-IR blockade in patients with moderate-to-severe TED, especially those with significant proptosis and/or diplopia. 1,2,15 Two recent single-center, retrospective follow-up analyses reported similar short-term efficacy to that reported in the clinical trials but reported lower follow-up maintenance rates than we report here in some of the outcomes. 35,36 The clinical practice patients reviewed in these two reports are very different from the clinical trial patients. With our population being newer-onset TED, naïve to pharmacotherapy/surgery or failure to previous therapies, they are apparently unlike the patients in these two reports. It is intriguing to speculate that earlier use of teprotumumab in newly diagnosed TED patients may produce better long-term results, but additional analyses are required to confirm this.
Here we report a retrospective analysis of observed prospectively collected data, thus not all data points were available for every patient. Although this study presents the longest follow-up to date of up to 18 months after treatment, longer follow-up periods for this therapy are warranted for this chronic disease. The data presented here included patients with relatively short TED duration; thus, future studies including greater numbers of patients with longer TED duration and less clinically active disease are also warranted. Additionally, a control group for background disease improvement over time would add additional context to this long-term observed benefit of teprotumumab, as spontaneous recrudescence occurs in up to 15% of patients. 37 This analysis is limited to follow-up in patients receiving a single course of teprotumumab. Outcomes of those undergoing retreatment with teprotumumab in OPTIC-X were not included because of limited follow-up data, although short-term results have been reported previously. 19
In conclusion, TED is a chronically symptomatic autoimmune disease with therapeutic options that can alter the natural course and progression of the disease, especially with early intervention. Evidence from these analyses indicates that in aggregate, patients treated with teprotumumab maintain long-term effects on inflammatory signs, symptoms, and progressive outcomes in approximately 80% of the patients up to 99 weeks after therapy completion.
Footnotes
Acknowledgment
Statistical support was provided by Qianhong Fu, MS, an employee of Amgen (formerly Horizon).
Authors’ Contributions
G.J.K.: Data curation, writing—reviewing and editing, P.S.: Data curation, writing—reviewing and editing. E.C.: Data curation, writing—original draft preparation. R.J.H.: Conceptualization, methodology, writing–reviewing and editing. T.J.S.: Data curation, writing—reviewing and editing.
Authors Disclosure Statement
Horizon (now Amgen) sponsored the study, contributed to data collection, analysis, and interpretation, and to writing, reviewing and approval of the final version. G.J.K.’s institution received research-associated funding from Horizon (now Amgen) for study conduct, and he is a consultant for Amgen (formerly Horizon); P.S.S.’s institution received research funding from Horizon (now Amgen), and he is a consultant for ACELYRIN, GenSight, Horizon (now Amgen), Tourmaline Bio, Viridian Therapeutics. E.C. and R.J.H. are employees and hold stock in Amgen (formerly Horizon). T.J.S. was issued US patents covering the use of IGF-IR inhibitors in TED, which are held by UCLA and the Lundquist Institute, and he is a consultant for Amgen (formerly Horizon), Viridian, Minghui, and Lundbeck.
Funding Information
This study was sponsored by Horizon Therapeutics (now Amgen). Preliminary findings of this study were presented at the 2022 annual meeting of the North American Neuro-Ophthalmology Society (NANOS), February 12–17, 2022.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
