Abstract
Background:
Although lenvatinib is the preferred treatment for unresectable radioactive iodine-refractory differentiated thyroid cancer (RR-DTC), this agent exerts considerable toxicities, which can lead to frequent dose interruptions and modifications. The adoption of planned drug holidays has been recently suggested as one means of minimizing or avoiding these severe adverse events. Our retrospective study demonstrated that planned drug holidays appear to be a promising strategy for continuing of lenvatinib. However, the benefits of planned drug holidays in a prospective study have yet to be clarified. Here, we investigated the impact of planned drug holidays on clinical outcomes in patients treated with lenvatinib in the COLLECT study.
Methods:
In COLLECT, a prospective observational study, patients with RR-DTC were treated with lenvatinib in a real-world clinical setting. Lenvatinib was administered orally at a dose of 24 mg daily. Dose modification for toxicities was permitted. Furthermore, planned drug holidays were allowed to avoid severe or intolerable toxicities. The present post hoc analysis focused on evaluating the impact of planned drug holidays on clinical outcomes, including overall survival (OS), time to treatment failure (TTF), time to failure strategy (TFS), and progression-free survival (PFS), in patients in the COLLECT study who were treated with lenvatinib.
Results:
In total, 262 patients were included. Of the 253 patients evaluable for efficacy, 73 undertook a planned drug holiday at the discretion of the attending physician. OS, TTF, TFS, and PFS were significantly longer in patients who used a planned drug holiday than in those who did not. The planned drug holiday group demonstrated notable clinical outcomes, with a 1-year OS of 95.8% and a 1-year PFS of 94.5%. Moreover, planned drug holidays demonstrated a clinically meaningful advantage in clinical outcomes. The planned drug holiday group had a significantly longer duration of administration at a dose of ≥10 mg.
Conclusions:
Planned drug holidays for lenvatinib were associated with significantly improved clinical outcomes compared to daily oral administration. Further investigation of the optimal treatment schedule for lenvatinib is warranted.
Clinical Trial Registration:
UMIN000022243.
Introduction
Prognosis for patients with locally recurrent or metastatic, progressive, radioiodine-refractory differentiated thyroid cancer (RR-DTC) remains poor, with a 10-year overall survival (OS) rate of 19%. 1,2
Lenvatinib, an oral multikinase inhibitor, exerts its effect through a number of targets, including vascular endothelial growth factor (VEGF) receptors 1, 2, and 3; fibroblast growth factor receptors 1 through 4; platelet-derived growth factor receptor α (PDFGRα); RET proto-oncogene (RET); and stem cell growth factor receptor (KIT). 3,4
In the randomized phase 3 SELECT trial, lenvatinib in primary analysis significantly improved progression-free survival (PFS) versus placebo (18.3 vs. 3.6 months; hazard ratio [HR], 0.21; 99% confidence interval [CI], 0.14–0.31; p value < 0.001) in patients with progressive RR-DTC, 5 and based on this result was subsequently licensed for the treatment of RR-DTC. Additional support came from the 2022 National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology, which classified lenvatinib as a “preferred regimen” for progressive and/or symptomatic RR-DTC. 6
Despite the efficacy of lenvatinib, toxicities are considerable and require frequent dose interruptions and modifications. Furthermore, subgroup analysis of Japanese patients in SELECT (lenvatinib, n = 30; placebo, n = 10) showed that dose reduction was more frequent among the Japanese patients than overall (90.0% vs. 67.8%) and occurred earlier in the Japanese patients (Japanese, 0.9 months; overall, 3.0 months). 7 Moreover, 51 Japanese patients with thyroid cancer, including 25 with RR-DTC, 9 with medullary thyroid cancer, and 17 with anaplastic thyroid cancer, received lenvatinib in a phase II study. 8,9
However, the number of Japanese patients who have received lenvatinib in clinical trials is small, with the result that Japanese data on safety, efficacy, and prognostic factors remain limited. Therefore, we conducted a prospective observational study (COhort study of Lenvatinib [LEnvima] in differentiated Cancer of Thyroid: COLLECT) to collect safety and efficacy data in patients receiving lenvatinib for unresectable RR-DTC. 10
Recently, planned drug holidays have been proposed as a means of avoiding severe adverse events due to lenvatinib. 11 Briefly, if severe or intolerable adverse events begin to occur at day X after the start of lenvatinib, the next drug administration should be interrupted before day X, and the planned drug holiday should start (Fig. 1). Planned drug holidays can thereby prevent frequent severe or intolerable adverse events, and in turn, avoid dose reductions.
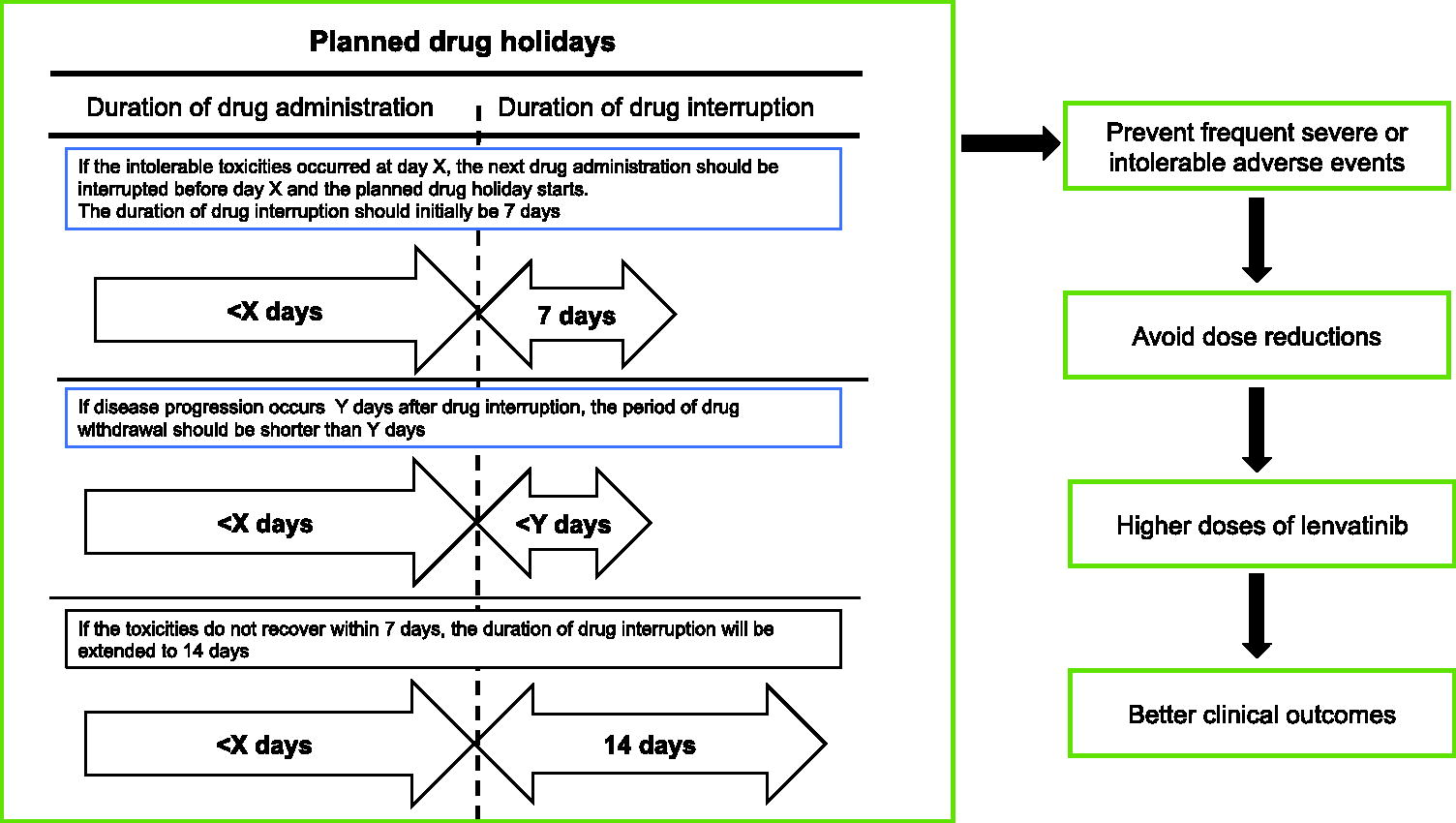
Planned drug holiday strategy.
Previously, we retrospectively evaluated the impact of planned drug holidays in RR-DTC patients receiving lenvatinib. 12 The study population included 25 patients who used planned drug holidays and 21 patients who did not (daily group). The planned drug holiday group demonstrated significantly longer time to treatment failure (TTF), PFS, and OS than the daily group (median not reached [NR] vs. 14.9 months, HR: 0.25, 95% CI: 0.11–0.58; p < 0.001; NR vs. 15.1 months, HR: 0.31; 95% CI: 0.14–0.68; p = 0.002; and NR vs. 26.6 months, HR: 0.20; 95% CI, 0.073–0.58; p = 0.001, respectively). Lenvatinib at a dose of ≥10 mg was associated with a significantly longer duration of administration in the planned holiday group (NR vs. 6.5 months, HR: 0.22; 95% CI: 0.10–0.49, p < 0.001), and a lower frequency of drug interruption due to adverse events (68% vs. 95%, p = 0.027). Thus, planned drug holidays appear to be a promising strategy for the continuation of lenvatinib. However, the benefits of planned drug holidays in a prospective study have yet to be clarified.
Here, we evaluated the impact of planned drug holidays on clinical outcomes, including OS, TTF, time to failure strategy (TFS), and PFS, in patients from the COLLECT study who were treated with lenvatinib.
Materials and Methods
Patients and study design
The present study population consisted of patients receiving lenvatinib in the COLLECT study, a prospective observational study of lenvatinib for unresectable DTC. 10 Inclusion criteria included unresectable, radioiodine-refractory, and progressive differentiated thyroid cancer (DTC) suitable for treatment with lenvatinib; initiation of lenvatinib treatment after this study was approved by the study site committee (e.g., institutional review board or ethical review board); and informed consent to participate was obtained. Exclusion criteria included a decision by the attending physician that the patient was unsuitable for the evaluation of toxicities or drug adherence in this study.
The institutional ethics committee at each participating center approved the study protocol, and the study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients prior to study enrollment. This trial was registered at the UMIN Clinical Trials Registry as UMIN000022243.
The aim of the present post-hoc analysis was to evaluate the impact of planned drug holidays on clinical outcomes, including OS, TTF, TFS, and PFS, in patients in the COLLECT study who were treated with lenvatinib.
Study treatment and assessments
Lenvatinib was administered orally at a dose of 24 mg daily. Dose modifications for toxicities were permitted. Furthermore, planned drug holidays were allowed to avoid severe or intolerable toxicity (Fig. 1). Drug holidays were established in accordance with the timing of adverse events. 11 Briefly, when a severe or intolerable adverse event first arose after the start of lenvatinib, the next drug administration was interrupted before the severe or intolerable stage was reached. Typically, the duration of drug interruption should initially be 7 days. If tumor regrowth occurs faster and leads to a worsening of quality of life (QOL), the period of drug withdrawal should be shorter. Drug continuation after disease progression during drug interruption was allowed because this disease progression was associated with drug interruption, not with drug resistance. If the toxicities do not resolve within 7 days, the duration of drug interruption could be extended to 14 days. However, the restart of lenvatinib within 2 weeks is preferable given concerns about tumor regrowth. If intolerable toxicities occur, the next dose of drug should be reduced. If intolerable toxicities do not occur during the last cycle, dose reduction is not needed. Once the use of planned drug holidays was initiated, it was continuous, but the schedule was adapted in accordance with the occurrence of adverse events. The lenvatinib dose and treatment schedule of planned drug holidays in our institute is shown in Supplementary Figure S1. Lenvatinib was continued until disease progression or the development of unacceptable toxicity. Response was evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST).
Statistical analysis
In the COLLECT study, the primary endpoint was OS. Therefore, the study was designed to estimate the 95% CIs of the 1-year and 2-year OS rates with a two-sided width of <8–10%. The planned sample size (n = 300 or more) was set to assess the 1-year and 2-year OS (1-year, n = 240–370; 2-year, 380–590) based on data from SELECT (1-year, 81.6%; 2-year, 58.2%) with the normal approximation of a binomial distribution.
Secondary endpoints included TTF, TFS, PFS, response rate, QOL survey, safety, and patient reports. PFS was defined as the interval from the initiation of treatment to the time at which any of the following events first occurred: disease progression by imaging scan or clinical progressive disease (cPD), meeting the following criteria: (1) progressive symptoms related to disease progression, (2) worsening performance status related to disease progression, (3) multiple disease progression including disease progression of lymph node or skin metastasis by visual examination, (4) life-threatening organ metastasis, or (5) elevation of thyroglobulin. 8 Determination of cPD was made after comprehensive evaluation. TTF was defined as the interval from the initiation of treatment to whichever of the following events occurred first: cPD, treatment discontinuation due to toxicity, loss to follow-up, or death. TFS was defined as the interval from the initiation of treatment to the time at which any of the following events first occurred: addition of any agent not in the primary strategy, which means the subsequent therapy after becoming refractory to lenvatinib, progression on full therapy, progression and no subsequent therapy, or death. Patients who used planned drug holidays were defined as those who interrupted lenvatinib before developing severe or intolerable adverse events, restart lenvatinib after drug interruption, and continued to receive lenvatinib according to the treatment schedules for planned drug holidays, which were determined in accordance with the occurrence of adverse events in the individual patients. The initial treatment schedules for planned drug holiday were collected.
PFS, TTF, TFS, and OS rates were estimated and plotted using Kaplan–Meier methods. The HR and CIs were estimated using a stratified Cox proportional hazards model. The current analysis did not include any confounders related with any group and excluded missing data. All statistical analyses were performed using SAS® version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient characteristics
A total of 262 patients were included (Fig. 2). Of the 253 patients evaluable for efficacy, 73 used planned drug holidays at the discretion of the attending physician, in all cases after the development of severe or intolerable adverse events. The treatment schedules for planned drug holidays were determined in accordance with the occurrence of adverse events in the individual patient. The median duration of the initial planned drug holiday was 2.86 weeks (95% CI: 2.14–3.29). The most common initial treatment schedule was a 14-day-on/7-day-off cycle (Table 1). Adverse events related to drug interruption just before starting the planned drug holiday are summarized in Table 2. Patient characteristics are summarized in Table 3. Compared with the patients who did not used planned drug holidays (nonplanned drug holiday group), those using planned drug holidays (planned drug holiday group) had a higher proportion of females (69.9% vs. 56.7%) and no histology of poorly DTC (0% vs. 2.2%). In contrast, the two groups were similar with regard to age; histology, including papillary thyroid cancer and follicular thyroid cancer, and previous therapy, including surgery, radioactive iodine therapy, and molecular targeted drugs.
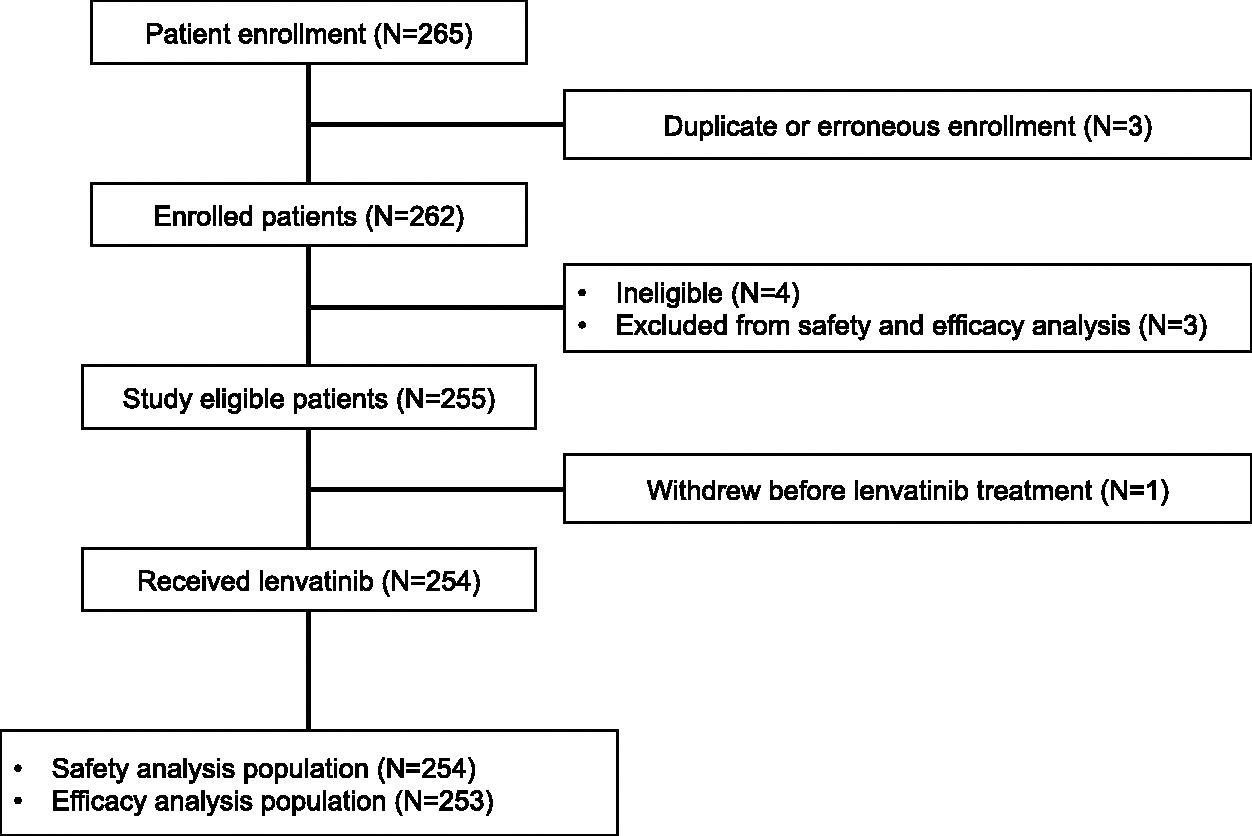
Participant flow diagram.
Initial Planned Drug Holiday Cycle(n = 73)
±3 days.
Adverse Events Related to Drug Interruption Just Before Starting the Planned Drug Holiday
Some patients developed multiple adverse events.
ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Patients Characteristics (n = 253)
DTC, differentiated thyroid cancer.
Efficacy
Overall response according to RECIST was complete response in 3 (1.2%) patients and partial response in 151 (61.9%). Objective responses in the planned and nonplanned drug holiday groups are compared in Supplementary Table S1. No difference in objective response was observed between the groups (66.7% vs. 61.6%).
Of the 241 patients with disease progression, progressive disease by imaging scan occurred in 210 patients (87.1%); cPD in 23 (9.5%), including progressive symptoms in 18, multiple disease progression in 3, worsening performance status in 1, and life-threatening organ metastasis in 1; elevation of thyroglobulin in 6 (2.5%); and other reasons in 2 (0.8%). However, none discontinued lenvatinib due to the elevation of thyroglobulin only. Subsequent therapy after becoming refractory to lenvatinib is summarized in the Supplementary Table S2.
At data cutoff on August 21, 2020, the 1-year OS rate in the total population was 85.6% (95% CI: 80.6–89.4). More importantly, OS, TTF, TFS, and PFS were significantly longer in patients who used planned drug holidays than those who did not: respective differences were 95.8% (95% CI: 87.6–98.6) vs. 81.5% (95% CI: 74.9–86.5) in 1-year OS (HR: 0.311; 95% CI: 0.132–0.732; p = 0.0047; Fig. 3A); 87.6% (95% CI: 77.6–93.4) vs. 69.8% (95% CI: 62.4–75.9) in 1-year TTF (HR: 0.421; 95% CI: 0.226–0.784; p = 0.0049; Fig. 3B); 84.9% (95% CI: 74.4–91.3) vs. 65.0% (95% CI: 57.5–71.5) in 1-year TFS (HR: 0.441; 95% CI: 0.260–0.747; p = 0.0017; Fig. 3C); and 94.5% (95% CI: 86.1–97.9) vs. 83.5% (95% CI: 77.2–88.3) in 1-year PFS (HR: 0.307; 95% CI: 0.108–0.873; p = 0.0190; Fig. 3D).
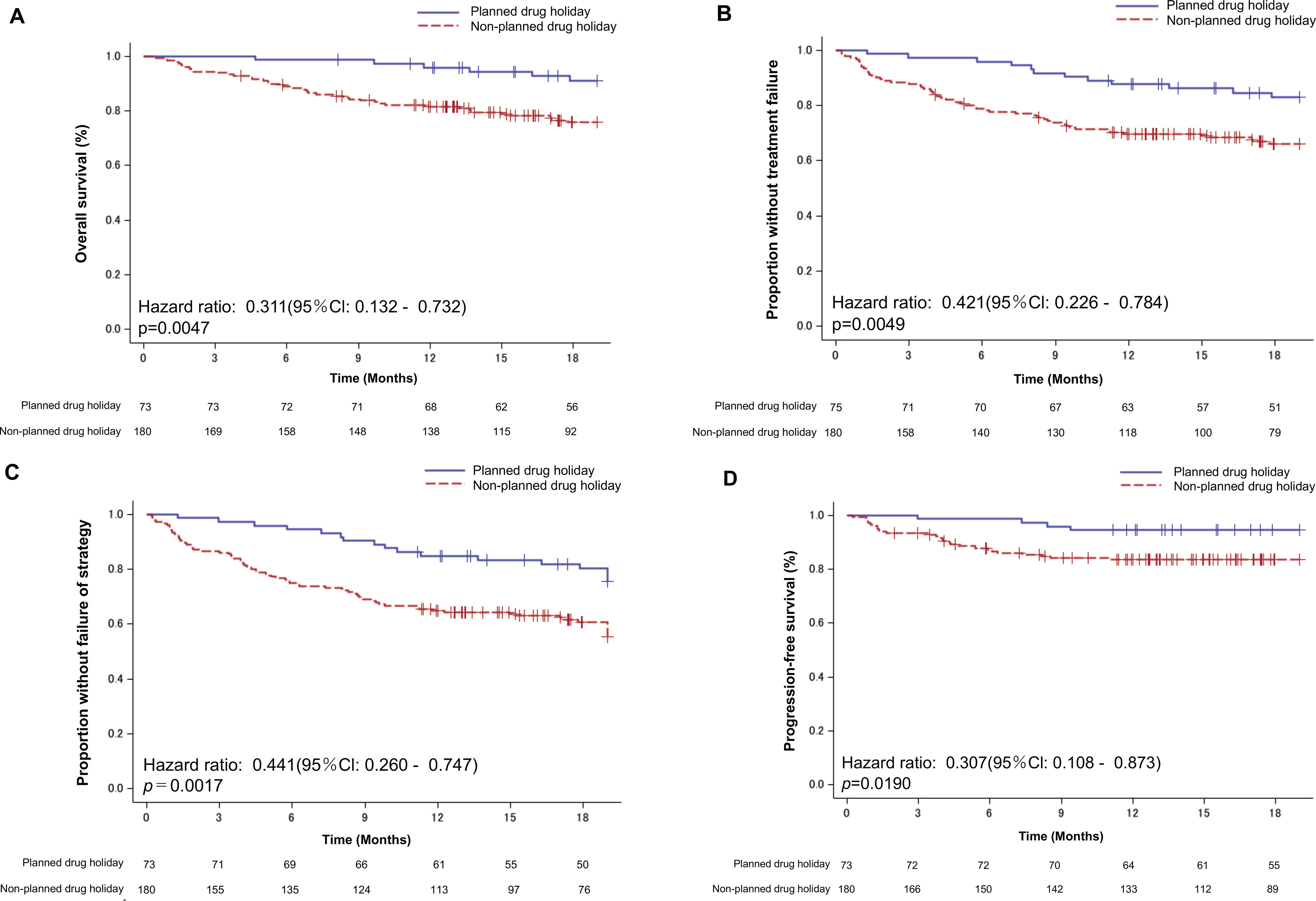
Efficacy according to the use of planned drug holidays.
Safety and treatment compliance
Adverse events are shown in Table 4. In the total population, the most common grade ≥3 toxicities were hypertension (61.4%), proteinuria (33.5%), hand–foot syndrome (10.2%), fatigue (9.1%), and anorexia (8.3%). Grade 5 toxicities occurred in 4 patients (fistula, hypoxia, respiratory failure, trachea stenosis) in the nonplanned drug holiday group. The planned and nonplanned drug holiday groups were similar in terms of grade ≥3 hypertension (64.4% vs. 60.0%) and grade ≥3 hand–foot syndrome (11.0% vs. 10.0%). In contrast, the planned drug holiday group had a higher incidence of grade ≥3 proteinuria (45.2% vs. 28.9%), grade ≥3 diarrhea (11.0% vs. 2.2%), grade ≥3 nausea (6.8% vs. 1.1%) and grade ≥3 vomiting (2.7% vs. 0.6%) than the nonplanned drug holiday group. However, no patient developed grade ≥3 fistula in the planned drug holiday group, while 2.8% did so in the nonplanned drug holiday group.
Adverse Events (n = 254)
GI, gastrointestinal tract.
Treatment discontinuation is shown in Table 5. Out of 141 patients (55.9%) with continuing treatment, more patients in the planned drug holiday group continued therapy than patients in the nonplanned drug holiday group (68.5% vs. 50.6%). The most common reason for treatment discontinuation included disease progression (43.4%), adverse events (33.6%), patient’s refusal (9.7%), and death (9.7%). More patients in the nonplanned drug holiday group discontinued treatment due to death (11.2% vs. 4.3%).
Treatment Discontinuation (n = 254)
When we compared the duration of administration at a dose of ≥10 mg between the planned and nonplanned drug holiday groups (Fig. 4A), the planned drug holiday group had a significantly longer duration of administration at this dose (HR: 0.599, 95% CI: 0.369–0.973, p = 0.0363). When we compared the duration of administration at a dose of ≥14 mg (Fig. 4B), the planned drug holiday group had a numerically longer duration of administration, but the difference was not significant (HR: 0.767; 95% CI: 0.554–1.067, p = 0.1072). These findings indicate that planned drug holidays result in higher dosages of lenvatinib.
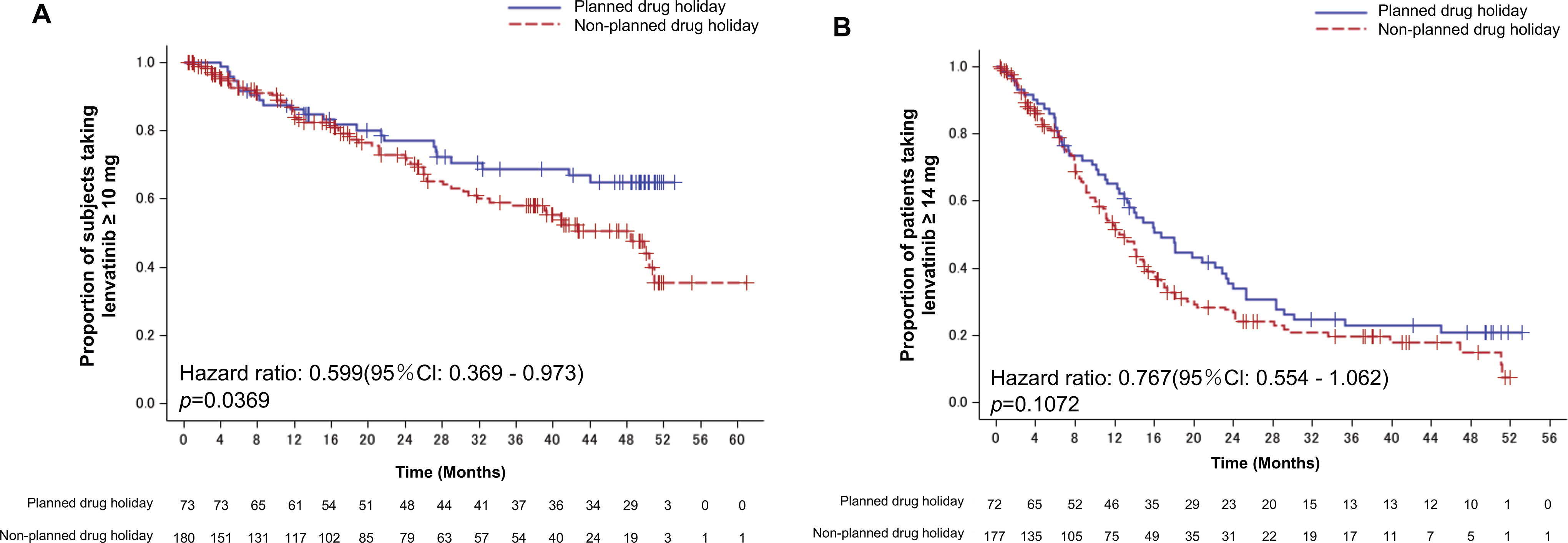
Duration of lenvatinib administration.
Discussion
This study is the first, to the best of our knowledge, to evaluate the impact of planned drug holidays on clinical outcomes in patients in prospective studies who were treated with lenvatinib. Although the planned sample size (n = 300 or more) was not reached due to poor patient enrollment, the study population included 253 patients evaluable for efficacy and 73 who used planned drug holidays, which were considered sufficient to allow evaluation of the impact of planned drug holidays on clinical outcomes. Most patient characteristics were comparable between the planned and nonplanned drug holiday groups.
In this prospective study, OS, TTF, TFS, and PFS were significantly longer in patients who used a planned drug holiday than those who did not. The planned drug holiday group demonstrated a notable survival rate, with a 1-year OS of 95.8% (95% CI: 87.6–98.6) and a clinically meaningful survival advantage over the nonplanned drug holiday group. Furthermore, the planned drug holiday group demonstrated a clinically meaningful advantage over the nonplanned group in both TTF and TFS. Moreover, the planned group demonstrated notable PFS, with a 1-year PFS with cPD of 94.5% (95% CI: 86.1–97.9) and a clinically meaningful advantage in PFS over the nonplanned group.
Among other findings, the planned drug holiday group had a significantly longer duration of administration at a dose of ≥10 mg and a numerically longer duration of administration at a dose of ≥14 mg. In other words, planned drug holidays result in higher doses of lenvatinib, which might have been associated with better clinical outcomes in patients who used planned drug holidays.
Objective response in the total population was 63.1%, which was comparable to that in the SELECT study (64.8%). No remarkable difference in objective response was observed between the planned and nonplanned drug holiday groups (66.7% vs. 61.6%), indicating that planned drug holidays do not increase response.
The planned drug holiday group had a higher incidence of grade ≥3 adverse events than the nonplanned drug holiday group, including proteinuria, diarrhea, nausea, and vomiting. These high incidence rates may be associated with the longer duration of administration at a dose of ≥10 mg and survivorship bias. Although 2.8% in the nonplanned drug holiday group developed grade ≥3 fistula, none did so in the planned group, indicating that planned drug holidays could avoid life-threatening toxicities.
There are several limitations in the present study. First, it was not a randomized clinical trial of the use versus nonuse of planned drug holidays in patients receiving lenvatinib. Therefore, we cannot make a definitive conclusion about the role of planned drug holidays for patients receiving lenvatinib. Further randomized clinical investigation of planned drug holidays is warranted.
Second, a standard approach to planned drug holidays has not yet been established. The treatment schedules for planned drug holidays were determined in accordance with the occurrence of adverse events in the individual patients. Planned drug holidays were introduced to all 73 patients who developed severe or intolerable adverse events. Based on the current promising results, however, the most suitable introduction would be from the beginning of treatment. The 2 weeks-on/1 week-off cycle (2/1 schedule) of lenvatinib from the beginning of treatment was evaluated in a phase I dose escalation study. 13 Dose limiting toxicities included grade 3 aspartate aminotransferase (AST)/alanine aminotransferase (ALT) increase in one patient at 16 mg bid and a grade 3 platelet count decrease in two patients at 20 mg bid. A maximum tolerated dose of 13 mg bid was established. Based on these results, the administration of 24 mg once daily with a 2/1 schedule with 24 mg by single daily administration of lenvatinib would be a reasonable initial treatment schedule for planned drug holidays. In fact, this 2/1 schedule was the most common initial treatment schedule in the present study.
Sunitinib, an oral tyrosine kinase inhibitor that targets VEGF receptor and PDFGR, is the standard first-line treatment for metastatic renal cell carcinoma treatment. 14 The standard schedule is a 4 weeks-on/2-weeks-off cycle (4/2 schedule). Continuous daily dosing was compared with the standard 4/2 schedule in a randomized phase II trial, which demonstrated no difference in survival, drug tolerance, or QOL but showed a strong trend suggesting continuous daily dosing was inferior to the 4/2 schedule in terms of time to progression. This standard 4/2 schedule can cause clinically significant adverse events and dose delay (38–49%) or dose reduction (50–51%), thereby potentially affecting dose intensity and decreasing drug exposure. Moreover, approximately 20% of patients eventually discontinue sunitinib due to adverse events. Therefore, alternative sunitinib treatment regimens have been investigated. 15 A randomized phase II study of sunitinib using a 4/2 schedule versus a 2/1 schedule in metastatic renal cell carcinoma revealed that sunitinib administered with a 2/1 schedule is associated with less toxicity and higher failure-free survival at 6 months than a 4/2 schedule, without any compromise in efficacy in terms of ORR and time to progression. 16
Recently, a pharmacodynamic study on patients receiving sunitinib in a 4/2 schedule reported achieving maximum antiangiogenic activity after 14 days on sunitinib, with no further reduction thereafter until 28 days. Of note, however, most patients developed rebound tumor blood flow after a 2-week treatment break. 17 This could have been avoided in part with a 2/1 schedule. In a phase I study of sunitinib, while sunitinib blood concentrations reached a steady state within 14 days of treatment, sunitinib and its active metabolites were detectable after a 7-day break, likely indicating prolonged drug exposure. This result would appear to contrast with complete elimination following the 2-week off drug period under the 4/2 schedule, 18,19 and in turn to suggest that the 2/1 schedule is optimal for sunitinib.
A similar approach would be a reasonable way to find an optimal schedule of lenvatinib. A phase I dose escalation study of the lenvatinib 2/1 schedule showed that all patients had a measurable plasma lenvatinib concentration (>0.08 ng/mL) for as long as 7 days after administration of a single dose, and for as long as 14 days after twice-daily administration. This finding indicates that a 7-day break may allow a considerable extension of exposure to the drug. Although there is no pharmacodynamic study of the 4/2 schedule of lenvatinib, complete elimination after the 2 weeks would be expected based on the results of the pharmacodynamic study on patients receiving sunitinib in a 4/2 schedule.
Based on these results, a 2/1 schedule would be a reasonable treatment schedule for lenvatinib.
Further investigation of the 2/1 schedule of lenvatinib is warranted.
In conclusion, we found that planned drug holidays for lenvatinib were associated with significantly better clinical outcomes than daily oral administration, including OS, TTF, TFS, and PFS. Further investigation of the optimal treatment schedule of lenvatinib is warranted.
Footnotes
Acknowledgments
The authors thank the participating investigators and patients. A full list of investigators and patients recruited in this study is provided in the Supplementary Data S1. The authors thank Guy Harris, DO, of Dmed (![]() ) for editing a draft of the article, Miho Akita of the Public Health Research Foundation for study support, and Katsuyuki Haino for statistical data analysis. Additionally, the authors convey their heartfelt condolences on the passing of Prof. Yasuo Ohashi, who served as the chief of statistical data analysis in the COLLECT study.
) for editing a draft of the article, Miho Akita of the Public Health Research Foundation for study support, and Katsuyuki Haino for statistical data analysis. Additionally, the authors convey their heartfelt condolences on the passing of Prof. Yasuo Ohashi, who served as the chief of statistical data analysis in the COLLECT study.
Authors’ Contribution
M.T. handled conceptualization, data curation, investigation, project administration, resources, supervision, visualization, writing-original draft. H.T. oversaw project administration and supervision. Y.I., T.O., I.S., K.S., S.T., H.T., H.T., H.H., A.M., and H.Y. involved in resources, and investigation. Y.O. conducted formal analysis, project administration and supervision. T.I. managed project administration, investigation and supervision.
Author Disclosure Statement
M.T. reports grants and personal fees from Eisai during the conduct of the study; grants and personal fees from Ono Pharmaceutical, BMS, Bayer, MSD, Eli Lilly, GSK, Pfizer, AstraZeneca, Rakuten Medical, and Merck Biopharma; grants from Novartis; and personal fees from Boehringer Ingelheim and Genmab outside the submitted work. H.T. reports personal fees from Eisai during the conduct of the study. Y.I. reports personal fees from Eisai, during the conduct of the study; personal fees from Eli Lilly, outside the submitted work. Takahiro Okamoto reports scholarship donations from Eisai during the conduct of the study. I.S. reports personal fees from Eisai during the conduct of the study; grants and personal fees from Bayer; grants from Kyowa-Kirin and Pfizer; and personal fees from Lilly, Medtronic, Takeda Pharmaceutical, Asuka Pharmaceutical, Ono Pharmaceutical, Johnson & Johnson, and Covidien outside the submitted work. K.S. reports personal fees from Eisai during the conduct of the study; and personal fees from Bayer and Eli Lilly outside the submitted work. S.T. reports grants and personal fees from Bayer during the conduct of the study; grants and personal fees from Daiichi-Sankyo, Eisai, Novartis, MSD, Chugai, AstraZeneca, Bristol Myers Squib, Ono Pharmaceutical and TAIHO outside the submitted work. H.T. reports grants and personal fees from Eisai during the conduct of the study; and personal fees from Bayer; personal fees from Lilly, Medtronic and Ethicon outside the submitted work. T.I. reports personal fees from Eisai during the conduct of the study; and personal fees from Bayer and Eli Lilly outside the submitted work. Others declare no conflict of interest. M.T. is a current editorial board member of Cancer Science.
Funding Information
This study was sponsored by the Public Health Research Foundation with funding support from Eisai Co., Ltd.
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
Supplementary Table S2
Supplementary Data S1
