Abstract
Background:
Monocarboxylate transporter 8 (MCT8) deficiency is a rare X-linked disease where patients exhibit peripheral hyperthyroidism and cerebral hypothyroidism, which results in severe neurological impairments. These brain defects arise from a lack of thyroid hormones (TH) during critical stages of human brain development. Treatment options for MCT8-deficient patients are limited and none have been able to prevent or ameliorate effectively the neurological impairments. This study explored the effects of the TH agonist sobetirome and its CNS-selective amide prodrug, Sob-AM2, in the treatment of pregnant dams carrying fetuses lacking Mct8 and deiodinase type 2 (Mct8/Dio2 KO), as a murine model for MCT8 deficiency.
Methods:
Pregnant dams carrying Mct8/Dio2 KO fetuses were treated with 1 mg of sobetirome/kg body weight/day, or 0.3 mg of Sob-AM2/kg body weight/day for 7 days, starting at embryonic day 12.5 (E12.5). As controls, pregnant dams carrying wild-type and pregnant dams carrying Mct8/Dio2 KO fetuses were treated with daily subcutaneous injections of vehicle. Dams TH levels were measured by enzyme-linked immunosorbent assay (ELISA). Samples were extracted at E18.5 and the effect of treatments on the expression of triiodothyronine (T3)-dependent genes was measured in the placenta, fetal liver, and fetal cerebral cortex by real-time polymerase chain reaction.
Results:
Maternal sobetirome treatment led to spontaneous abortions. Sob-AM2 treatment, however, was able to cross the placental as well as the brain barriers and exert thyromimetic effects in Mct8/Dio2 KO fetal tissues. Sob-AM2 treatment did not affect the expression of the T3-target genes analyzed in the placenta, but it mediated thyromimetic effects in the fetal liver by increasing the expression of Dio1 and Dio3 genes. Interestingly, Sob-AM2 treatment increased the expression of several T3-dependent genes in the brain such as Hr, Shh, Dio3, Kcnj10, Klf9, and Faah in Mct8/Dio2 KO fetuses.
Conclusions:
Maternal administration of Sob-AM2 can cross the placental barrier and access the fetal tissues, including the brain, in the absence of MCT8, to exert thyromimetic actions by modulating the expression of T3-dependent genes. Therefore, Sob-AM2 has the potential to address the cerebral hypothyroidism characteristic of MCT8 deficiency from fetal stages and to prevent neurodevelopmental alterations in the MCT8-deficient fetal brain.
Introduction
Inactivating mutations in the SLC16A2 gene, which encodes for the monocarboxylate transporter 8 (MCT8), lead to an X-linked rare disease, the so-called MCT8 deficiency or Allan–Herndon–Dudley syndrome (AHDS). 1 –3 MCT8 is the only transmembrane transporter specific for thyroid hormones (TH), transporting thyroxine (T4) and the genomically active form 3,5,3′-triiodothyronine (T3). 4 AHDS patients exhibit a spectrum of symptoms that has been widened recently, which include profound neurodevelopmental delay, cerebral hypothyroidism that leads to motor and intellectual disability, and peripheral thyrotoxicosis leading to a plethora of clinical sequelae. 5,6
Currently, MCT8-deficient patients have limited treatment options and, because of the clinical complexity of the syndrome, with both cerebral hypothyroidism and peripheral hyperthyroidism, the designing of therapeutic strategies is challenging. 7 Among those strategies that have been clinically tested, both TH replacement strategies, such as the combination of PTU and levothyroxine, and TH analog strategies, including diiodothyropropionic acid and triiodothyroacetic acid (TRIAC), have been able to ameliorate key features related to peripheral hyperthyroidism. However, none of them have been able to alleviate neurological outcomes up to date, even though there is still an ongoing clinical trial (NCT02396459) assessing if TRIAC could be able to ameliorate these symptoms in the youngest patients. 8 –12
The brain barriers, and in particular the blood–brain barrier, have been identified as the main restrictions for TH entry to neural cells in the absence of MCT8. 13 –15 For this reason, novel therapeutic strategies have been developed to overcome this limitation. 16 –20 Among these strategies, we previously obtained encouraging results using sobetirome and its pro-drug Sob-AM2. Sobetirome is a TH analog that binds both TH receptors with selectivity for the TH receptor beta over TH receptor alpha (TRA) 21,22 and does not depend on MCT8 for its distribution in the brain. Sob-AM2 is a sobetirome amide prodrug that is converted into the active form sobetirome specifically in the brain by the action of the enzyme fatty acid amide hydrolase (Faah). 23
For these previous preclinical studies, sobetirome and Sob-AM2 were tested in a validated murine model of the disease: the double knockout for MCT8 and deiodinase type 2 (DIO2, Mct8/Dio2 KO). 24 Mct8KO animals are only a partial model of the AHDS, as they replicate the endocrine but not the neurological alterations of patients. 25 The lack of neurological alterations is due to a compensatory mechanism involving a larger presence of the T4 transporter organic anion-transporting polypeptide 1c1 (OATP1C1) at the mouse brain barriers in comparison with humans, and increased activity of DIO2, which converts T4 into T3.
Indeed, Mct8/Dio2 KO and Mct8/Oatp1c1 KO mice replicate both the endocrine and neurological alterations present in patients, thus have been extensively used as a model of AHDS. 24,26 Systemic administration of sobetirome and Sob-AM2 to Mct8/Dio2 KO mice was able to access the brain in the absence of MCT8 and modulate the expression of TH-target genes in juvenile mice. 18 Because the neurological alterations in AHDS patients are already present from fetal stages, 27 patients would benefit from prenatal treatment to prevent these defects. Indeed, some strategies have already evaluated maternal–fetal treatment for MCT8 deficiency 28,29 both in humans and in murine models, with some positive therapeutic outcomes.
This study aimed to evaluate the ability of sobetirome and Sob-AM2 to cross the placental barrier and the fetal brain barriers to exert thyromimetic actions in the brain of MCT8-deficient fetuses as a potential therapeutic strategy to prevent neurodevelopmental alterations. To this aim, pregnant dams carrying Mct8/Dio2 KO mice were treated systemically with sobetirome or Sob-AM2 and their fetuses compared with untreated Mct8/Dio2 KO and wild-type (WT) fetuses. The results indicate that Sob-AM2 can cross the placental barrier and the fetal brain barriers, be converted in the brain into the active form sobetirome and exert thyromimetic effects in the brain of Mct8/Dio2 KO mice in the absence of MCT8 at prenatal stages. Altogether, these data make Sob-AM2 a promising candidate to treat MCT8 deficiency from its most early stages.
Materials and Methods
Reagents
Drugs were prepared to be injected subcutaneously at 5 μL/g body weight concentration. Drug stocks from sobetirome (molecular weight [MV] = 328 g/mol; Sigma; SML1900) and Sob-AM2 (MW = 341 g/mol) 21,23,30 were prepared by dissolving sobetirome and Sob-AM2 at 1 mg/mL in a previously prepared vehicle, composed of distilled water, Kolliphor® (Sigma; C5135), and 1-Methyl-2-pyrrolidinone (Sigma; 328634) in 8:1:1 proportion. Saline was used to dilute the 1 mg/mL drug stocks to the final concentrations of 0.2 mg/mL sobetirome and 0.06 mg/mL Sob-AM2 (corresponding to 1.0 and 0.3 mg/kg dose, respectively). Since treatment with Sob-AM2 results in approximately twofold more sobetirome content in the brain than treatment with its precursor drug sobetirome, 18 the chosen doses were 1.0 and 0.3 mg/kg of sobetirome and Sob-AM2, respectively, to achieve similar content of sobetirome in the brain with both treatments.
Animal models and experimental design
All mice were housed at the Instituto de Investigaciones Biomédicas “Alberto Sols” with ad libitum access to food and water, at 22°C and on a 12:12 diurnal cycle. Mct8/Dio2 KO mice colony was previously established as described in Bárez-López et al. 24 and mice were genotyped for Mct8 and Dio2 genes as described in Ceballos et al. 14 and for fetal sex determination as described in Tunster. 31 Mct8/Dio2 KO breeding was done by mating Mct8+/y Dio2−/− males and Mct8−/+ Dio2−/− females. The morning after mating was considered gestational day E0.5, then, pregnant WT females and Mct8−/+ Dio2−/− females (n = 5) were treated for 7 days (from E12.5 to E18.5, Fig. 1A) with daily subcutaneous injections at 5 μL/g body weight dosing.
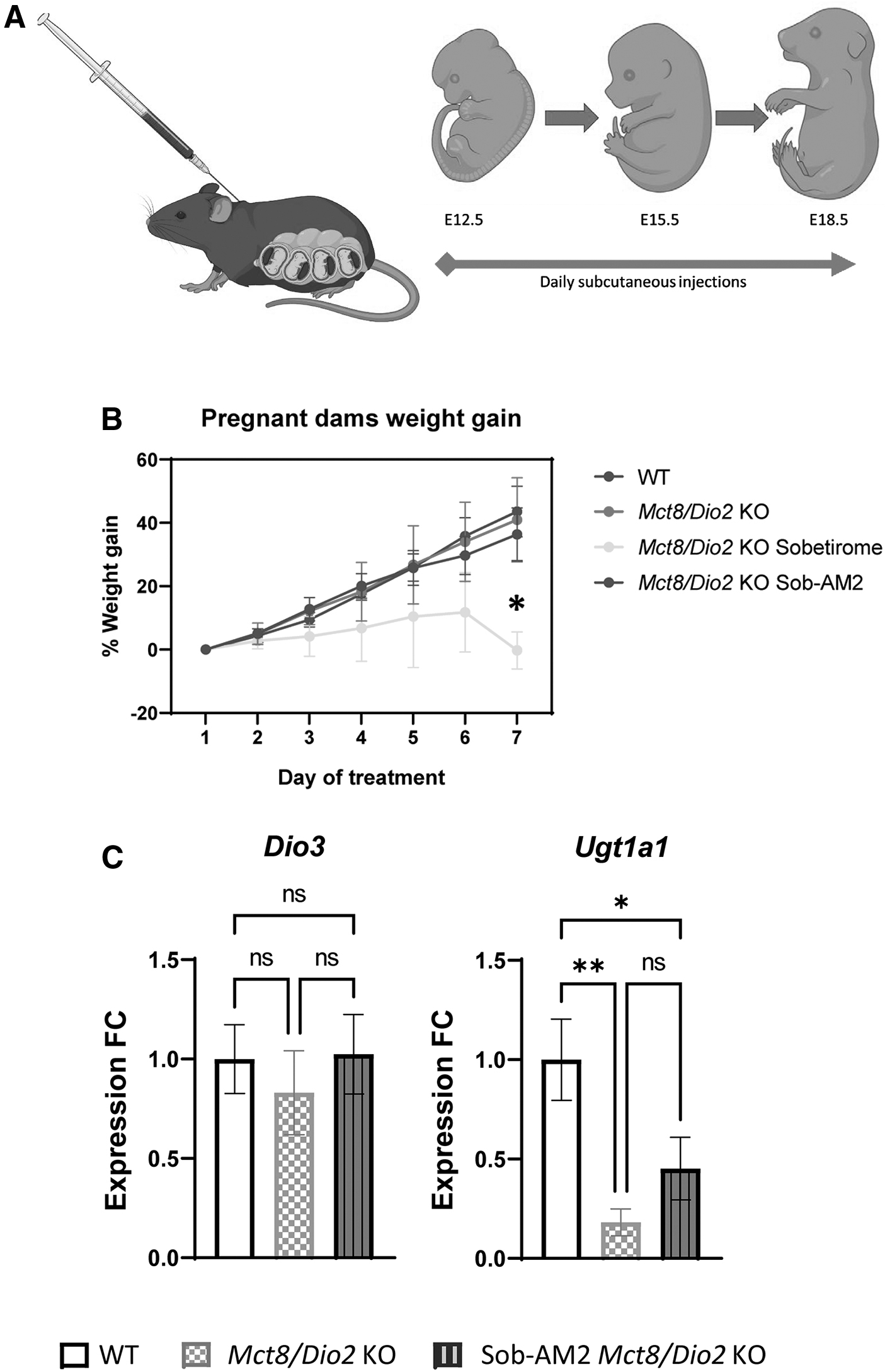
Pregnant dams carrying Mct8/Dio2 KO fetuses were treated with 1 mg of sobetirome/kg body weight/day, or 0.3 mg of Sob-AM2/kg body weight/day. As controls, pregnant dams carrying WT and pregnant dams carrying Mct8/Dio2 KO fetuses were treated with daily subcutaneous injections of vehicle. Dams were euthanized using carbon dioxide asphyxiation at embryonic day E18.5, 4–6 hours after the last injection. Before fetal extraction, blood was extracted by retro-orbital collection and used for the determination of TH levels in maternal plasma. Then, fetuses were extracted into iced saline, and tissues (brain, liver, and placenta) were harvested for further analyses. Only male fetuses were analyzed as MCT8 deficiency is an X-linked pathology only expressed in males.
Animal experimental procedures were performed in strict accordance to the European Union Council guidelines (directive 2010/63/UE) and Spanish regulations (R.D. 53/2013). Approval for these procedures was granted by the ethics committee Comité de Ética y Experimentación Humana y Animal (CEEHA) at CSIC and by the Comunidad Autónoma de Madrid Review Board (Proex 014.1/21) for the use of animals for scientific purposes.
Gene expression assays
RNA was isolated from individual hemi-cortex, liver, and placenta. Total RNA was extracted using TRIZOL reagent (Invitrogen; 15596026) following the manufacturer's instructions with an additional chloroform extraction. Real-time quantitative polymerase chain reaction (qPCR) was performed as described in previous publications. 32
The expression of the following T3-dependent genes was measured using Applied Biosystems TaqMan probes. In the brain: Dio3 (type 3 iodothyronine deiodinase), Faah, Hr (hairless), Kcnj10 (Potassium Inwardly Rectifying Channel Subfamily J Member 10), Klf9 (Kruppel-like factor 9), Mgp (Matrix gla protein), Ncam1 (Neural Cell Adhesion Molecule 1), Nrgn (Neurogranin), and Shh (sonic hedgehog); in the liver: Dio3, Dio1 (type 1 iodothyronine deiodinase), Klf9, Ucp2 (uncoupling protein 2), and Ugt1a1 (UDP glucuronosyltransferase 1 family, polypeptide A1); and in the placenta: Dio3 and Ugt1a1. The mean of the three housekeeping genes 18S ribosomal RNA (18S), Ribosomal Protein Lateral Stalk Subunit P0 (Rplp0), and glyceraldehyde 3-phosphate dehydrogenase (Gapdh) used as internal control to obtain relative mRNA expression. Data were expressed relative to the values obtained in vehicle-treated WT mice tissues (taken as 1.0).
Maternal plasma TH levels determinations
Maternal plasma TH levels were determined using Biovision mouse T4 and T3 ELISA Kits (Biovision, Abcam, K7421, K7422) according to the manufacturer's instructions.
Statistical analysis
Graphs represent mean ± standard error of mean. Normality of the data was assessed by the Shapiro–Wilk test. Differences between means were obtained by one-way analysis of variance (ANOVA) followed by Bonferroni's post hoc test to correct for multiple comparisons. Detection of outliers from a Gaussian distribution was performed by Regression Outlier Removal tests. 33 Outliers were excluded from further analyses (see complete list at Supplemental Methods). All analyses were performed using the GraphPad software (GraphPad Software, Inc., La Jolla, CA).
Results
Sobetirome and Sob-AM2 effects in pregnant dams
Pregnant dams carrying WT or Mct8/Dio2 KO mice were treated either with a vehicle, sobetirome, or Sob-AM2 from E12.5 to E18.5 (Fig. 1A). Sobetirome was observed to exert deleterious effects on sobetirome-treated dams' pregnancy. Weight gain of the pregnant dams was studied every day during the treatment, revealing that sobetirome-treated Mct8+/−/Dio2−/− dams weight gain became stagnant in the period between E15.5 and E18.5 (Fig. 1B), and during this period the treatment seemed to provoke spontaneous abortions.
This secondary effect, probably as a result of a peripheral toxicity effect associated with the dosage, resulted in the impossibility to reach an adequate sample number from sobetirome-treated dams. Sob-AM2-treated dams, however, did not exhibit this complication, as their weight gain was comparable with those of WT and vehicle-treated Mct8+/−/Dio2−/−
dams (Fig. 1B) and they did not suffer deleterious effects and abortions during the treatment. In addition, there were no differences in the size of the litters (Supplementary Fig. S1A
Plasma samples were then analyzed 4–6 hours after the last injection (E18.5). Maternal TH levels were assessed in nontreated and Sob-AM2-treated dams and the analysis revealed that Mct8+/−/Dio2−/− dams presented no differences in T3 and T4 plasma levels compared with WT dams (Supplementary Fig. S1D). Sob-AM2 treatment (once-daily, 7 days) in Mct8+/−/Dio2−/− dams decreased T4 plasma levels to ∼17% and 25% of the WT and Mct8+/−/Dio2−/− vehicle-treated dams, respectively, as previously observed in juvenile animals. 18 T3 levels on Sob-AM2-treated dams, however, could not be assessed due to cross-reactivity between the treatment and the T3 ELISA. Thus, daily treatment with Sob-AM2 for 7 days resulted in depletion of circulating T4.
Effects of Sob-AM2 in the placenta
The expression of two genes related to TH metabolism was analyzed in placental tissue. The expression of Dio3, responsible for metabolizing T4 into T3, 34 was not altered in Mct8/Dio2 KO placentas compared with WT animals (Fig. 1C). Treatment with Sob-AM2 did not change Dio3 expression levels. The expression of the gene encoding the enzyme Ugt1a1, responsible for glucuronidation of both T3 and sobetirome, 35,36 was decreased by 80% in the Mct8/Dio2 KO samples compared with WT animals (Fig. 1C). There were no significant differences in Ugt1a1 expression between basal and Sob-AM2-treated Mct8/Dio2 KO fetuses, indicating minimal effects of Sob-AM2 in placenta.
Effects of Sob-AM2 in the fetal periphery
In the fetal liver, the expression of the T3-dependent genes encoding the TH metabolic enzymes Dio1 and Dio3 responsible for metabolizing T4 into T334 presented no significant differences between Mct8/Dio2 KO and WT fetuses (Fig. 2). Treatment with Sob-AM2 led to a 12-fold and 1.8-fold increase in the expression of Dio1 and Dio3, respectively. The expression of the T3-dependent gene, Klf9, was 20% increased in Mct8/Dio2 KO mice compared with WT fetuses and its expression was reverted to WT values in response to Sob-AM2 treatment. Expression of the T3-dependent gene Ucp2 was not affected by any of the experimental conditions. The expression of Ugt1a1 was also analyzed, showing a near 100-fold decrease in both treated and nontreated Mct8/Dio2 KO fetuses as compared with WT.
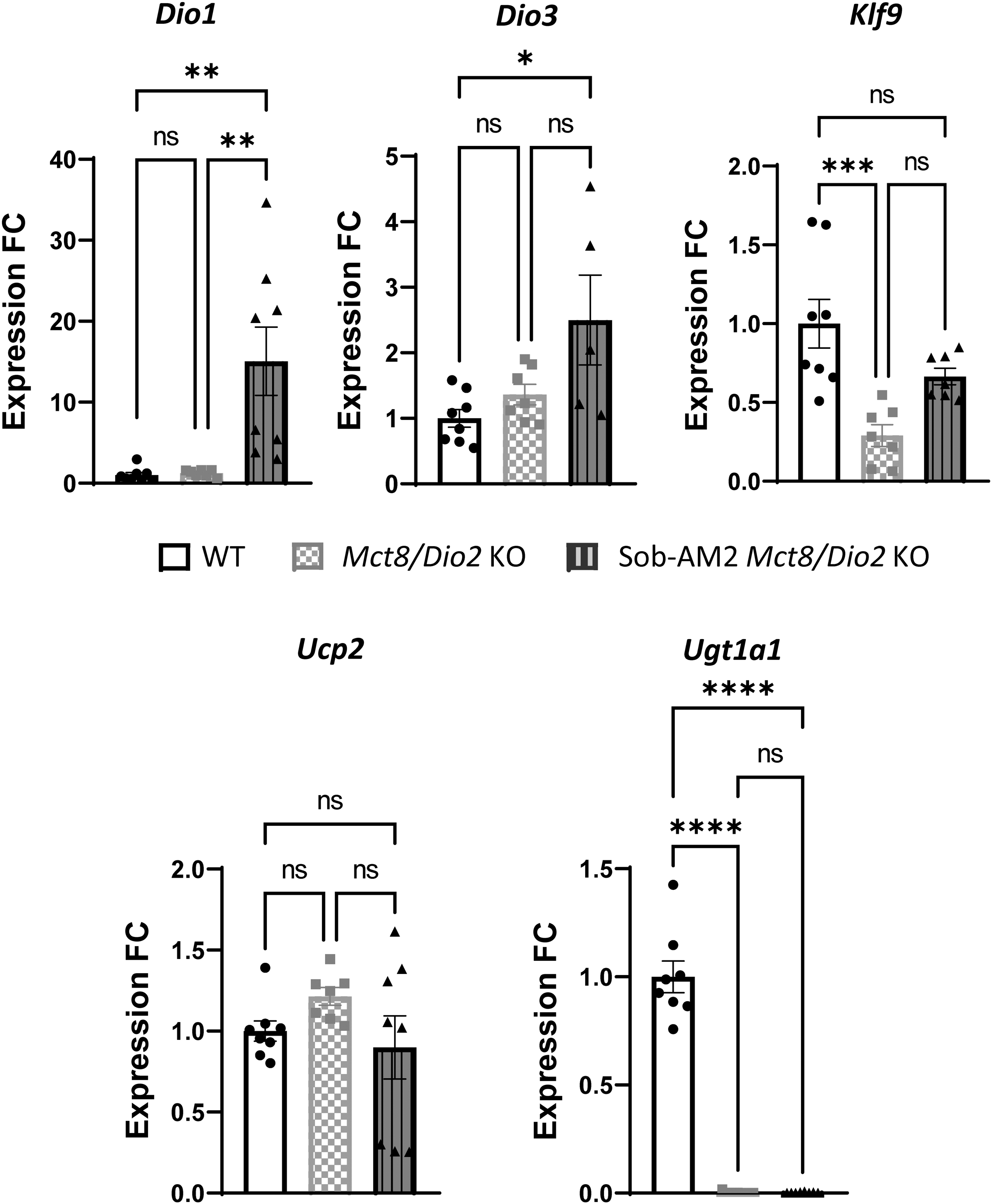
Graphs representing Dio1, Dio3, Klf9, Ucp2, and Ugt1a1 expression in the liver in WT (n = 8) and Mct8/Dio2 KO fetuses untreated (n = 7) and after Sob-AM2 treatment (n = 8). Gene expression levels were obtained by qPCR, and the data are expressed relative to the average of housekeeping genes (18S, Gapdh and Rplp0) as fold changes of WT levels. Outliers identified with ROUT were excluded from the analysis. *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001 were determined by one-way ANOVA and Bonferroni's post hoc test. Dio1, type 1 iodothyronine deiodinase; Klf9, Kruppel-like factor 9; Ucp2, uncoupling protein 2.
Effects of Sob-AM2 on the fetal CNS
Next, we evaluated the ability of Sob-AM2 pregnant dams' treatment to cross the placental barrier, reach the fetuses, and exert actions at the genomic level in the MCT8-deficient brain. T3-dependent genes were selected based on previous findings 18,37,38 and included Dio3, enriched in neurons; Hr, enriched in oligodendrocyte progenitor cells (OPCs) and newly formed oligodendrocytes; Kcnj10, enriched in OPCs; Klf9, enriched in neurons, OPCs, and endothelial cells; Mgp, enriched in endothelial cells; Ncam1, enriched in neurons; Nrgn, enriched in neurons and OPCs; Shh, enriched in neurons and oligodendrocytes; as well as Faah, the enzyme responsible for converting Sob-AM2 into sobetirome, which is enriched in neurons and OPCs. 39 Genes enriched in astrocytes and microglia were not included in the analysis, as these neural populations develop mainly at postnatal stages in the mouse. 40
Untreated Mct8/Dio2 KO animals presented no significant differences in the expression of Dio3, Faah, Hr, Kcnj10, Klf9, Mgp, Ncam1, and Shh, and a 30% decrease in the expression of Nrgn compared with WT (Fig. 3). After treatment, the expression of all the studied genes in Mct8/Dio2 KO was increased. Dio3 expression exhibited a fourfold increase as compared with untreated Mct8/Dio2 KO mice, Hr increased by 50%, Kcnj10 by 60%, Klf9 by 90%, and Shh by 50%.
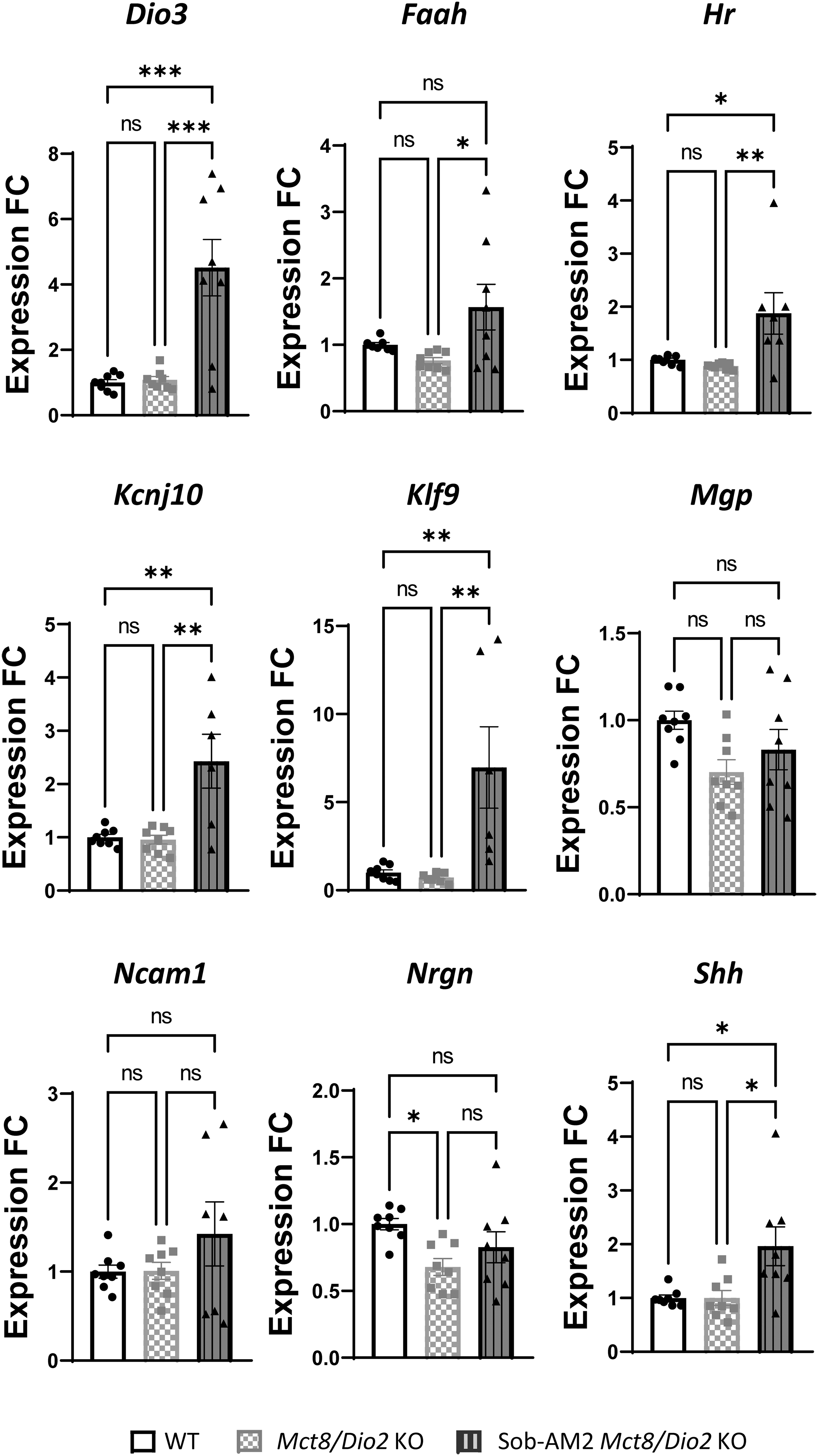
Graphs representing Dio3, Faah, Hr, Kcnj10, Klf9, Mgp, Ncam1, Nrgn, and Shh expression in the brain in WT and Mct8/Dio2 KO fetuses untreated and after Sob-AM2 treatment (n = 8). Gene expression levels were obtained by qPCR, and the data are expressed relative to the average of housekeeping genes (18S, Gapdh and Rplp0) as fold changes of WT levels. Outliers identified with ROUT were excluded from the analysis. *p < 0.05, **p < 0.01, and ***p < 0.001 were determined by one-way ANOVA and Bonferroni's post hoc test. Faah, fatty acid amide hydrolase; Hr, hairless; Kcnj10, potassium inwardly rectifying channel subfamily J member 10; Mgp, matrix gla protein; Ncam1, neural cell adhesion molecule, Nrgn, neurogranin; Shh, sonic hedgehog.
Moreover, Nrgn expression levels were reverted in Mct8/Dio2 KO mice after treatment, with no significant differences from WT values (Fig. 3). Sob-AM2 treatment also appeared to increase Ncam1 and Mgp expression, although this effect was not statistically significant. In addition, Faah expression was analyzed, as this is the enzyme responsible for the conversion of Sob-AM2 to sobetirome in the brain. Faah expression was not altered between WT and untreated Mct8/Dio2 KO mice but was significantly increased by 50% between nontreated and Sob-AM2-treated Mct8/Dio2 KO fetuses, as a response to the access of Sob-AM2 into the brain.
Discussion
This study explored the effects of the TH agonist sobetirome and its CNS-selective amide prodrug, Sob-AM2, in the treatment of pregnant dams carrying Mct8/Dio2 KO mice fetuses. The main focus was to assess their ability to cross the placental barrier, and the fetal brain barriers, and reach the brain to exert a thyromimetic action in the absence of MCT8. In addition, it was important to analyze the treatment effects in peripheral tissues due to the complexity of the thyroidal status of this disease: peripheral hyperthyroidism versus cerebral hypothyroidism.
Our study revealed that Sob-AM2 (0.3 mg/kg) can exert thyromimetic actions on Mct8/Dio2 KO fetuses when administered once daily for 7 days to pregnant dams. While this Sob-AM2 dosing schedule has already been shown to achieve therapeutic effects at postnatal stages, 18,41 transplacental passage of this analog had not been demonstrated until now.
Sobetirome treatment resulted in failures during pregnancy and eventually spontaneous abortions, possibly due to hyperthyroid-like effects. Given that Sob-AM2 did not produce this deleterious effect and that Sob-AM2 can reach the brain almost twofold more than sobetirome, while being threefold less concentrated in the periphery, 18,42 only treatment with Sob-AM2 was further evaluated.
Maternal Sob-AM2 treatment did not alter Dio3 expression values in the placenta, suggesting a lack of Sob-AM2-derived sobetirome effects in this organ. However, mild upregulation of Ugt1a1 could be indicative of sobetirome inactivation in the placenta. Sob-AM2 exerted thyromimetic effects in Mct8/Dio2 KO mice fetuses. In the periphery, Sob-AM2 treatment increased the expression of both genes encoding the Dio1 and Dio3 enzymes in the liver, although it did not further increase the expression of the T3-target gene Ucp2, indicating a thyromimetic effect exerted by maternal Sob-AM2 treatment in fetal peripheral tissues.
Surprisingly, the expression of Ugt1a1 levels in the liver of both nontreated and Sob-AM2-treated Mct8/Dio2 KO fetuses was remarkably reduced in comparison with WT values, possibly by the lack of MCT8 and/or DIO2 interfering with other signaling pathways that regulate Ugt1a1 expression, such as glucocorticoids. 43 In the fetal central nervous system, increased expression of Faah following Sob-AM2 treatment strongly suggests that Sob-AM2 can access neurons and OPCs, neural cell types that express Faah, 39 and enhance the activity of this enzyme to activate the conversion of Sob-AM2 into sobetirome. Moreover, the modulation of TH-target gene expression, which was able to normalize the expression of genes altered in nontreated Mct8/Dio2 KO fetuses, demonstrates that maternal Sob-AM2 treatment can cross both the placental and the fetal brain barriers and induce thyromimetic effects in the fetal brain.
In addition, the data suggest that sobetirome derived from Sob-AM2 is able to exert actions in several neural cell types present at E18, as the modulated genes are expressed in neurons (Dio3, Nrgn, and Shh), OPCs (Hr, Shh, and Kcnj10) and endothelial cells (Klf9). However, it is important to mention that the expression of some TH-target genes have increased expression values in Sob-AM2-treated Mct8/Dio2 KO fetuses over WT values, which could be indicative of a thyrotoxic effect at this dose. This is a proof-of-concept study that supports that Sob-AM2 can cross the placental and the fetal brain barriers and that can be converted into sobetirome in the fetal brain. In further studies, it would be of interest to assess the effect of maternal Sob-AM2 treatment on the thyroidal status of the fetus. Follow-up studies will shed more light into the ability of maternal treatment with Sob-AM2 to prevent the neurological alterations of Mct8/Dio2 KO mice and will aid in identifying the optimum dose.
To sum up, our results indicate that a systemic Sob-AM2 administration to pregnant dams can cross the placental barrier and reach precisely the MCT8-deficient fetal brain to be subsequently converted into the active form sobetirome and exert thyromimetic actions in several neural cell types. As early treatment has been found to be crucial in AHDS clinical outcomes, the availability of a suitable TH analog that can target the fetal brain by maternal administration represents a great step forward in the possibility of treatments that prevent neurodevelopmental damage and alleviate the neurological symptoms of the disease.
Footnotes
Acknowledgments
The authors would like to thank Ana Montero-Pedrazuela and Carmen Grijota-Martínez for their technical assistance and helpful comments and Skylar J. Ferrara for his help in designing drug administration. We thank Susana Alemany and María Gutierrez-Hernández for their help with hormone measurements. We thank the genomics core facility at the Instituto de Investigaciones Biomédicas for her technical help and Cristina Crespo and Coral Pedrero for animal care. ![]() scheme was created with BioRender.com.
scheme was created with BioRender.com.
Authors' Contributions
V.V.-H., S.B.-L., and A.G.-F. conceived and planned the experiments, and analyzed the data. V.V.-H. and M.G.-Y. carried out the experiments. T.S.S. provided the reagents. A.G.-F. supervised and administered the project. V.V.-H. wrote the article with support from T.S.S., S.B.-L., and A.G.-F. All authors discussed the results and commented on the article.
Author Disclosure Statement
T.S.S. is an inventor of a licensed patent on prodrugs of sobetirome. The remaining authors have nothing to disclose.
Funding Information
This study was supported by the Spanish Ministry of Science and Innovation (MCIN)/AEI/10.13039/501100011033 (grant no. SAF2017-86342-R to A.G.-F.); MCIN/AEI/10.13039/501100011033/FEDER “Una manera de hacer Europa” (grant no. PID2020-113139RB-I00 to A.G.-F.); Consejo Superior de Investigaciones Científicas (grant no. 2020AEP044 to A.G.-F.); the Sherman Foundation (grant no. OTR02211 to S.B.-L. and A.G.-F.); Asociación Corriendo con el Corazón por Hugo (grant no. OTR06190 to A.G.-F.), and MCIN/EU (grant no. IJC2020-043543-I) to S.B.-L. V.V.-H. is recipient of a contract from MCIN “El FSE invierte en tu futuro” (grant no. PRE2018-086185) and M.G.-Y. from the MCIN, Programa de Formación de Profesorado Universitario (FPU, FPU19/02006).
Supplementary Material
Supplementary Data
Supplementary Figure S1
