Abstract
Background:
The Hippo pathway has a fundamental role in tissue homeostasis, but little is known about how this signaling cascade is controlled in the thyroid. PAX8 is an essential driver of thyroid differentiation and is involved in the control of genes crucial for thyroid hormone biosynthesis, including the sodium/iodide symporter (NIS; SLC5A5). A role for the Hippo mediator transcriptional coactivator with PDZ-binding motif (TAZ) as a coactivator of PAX8 to promote thyroglobulin expression has been previously described. Here, we studied the role of TAZ on thyroid differentiation focusing on PAX8-mediated Slc5a5 transcription.
Methods:
Gene silencing and overexpression assays were performed in rat PCCl3 thyroid follicular cells (TFCs) to determine the role of TAZ in the regulation of Slc5a5. Transcriptional activity of the Hippo mediators was investigated by chromatin immunoprecipitation and promoter-reporter gene activity. Hippo component levels and location were analyzed in PCCl3 cells and in mouse thyroid under different treatment conditions.
Results:
By suppressing the expression of PAX8 and its binding to the Slc5a5 upstream enhancer, TAZ inhibits Slc5a5 expression, impairing NIS membrane location and activity. Other Hippo effectors such as YAP1 and TEAD1 were not required for the repressor effect of TAZ. We also found an interplay between the Hippo, thyrotropin (TSH), and transforming growth factor β1 (TGFβ) pathways in TFCs. TSH via cyclic adenosine monophosphate activated Hippo signaling pathway and, consequently, TAZ was excluded from the nucleus. We confirmed this in hypothyroid mice, characterized by elevated TSH serum levels, which showed downregulated activation of Hippo signaling in thyroid. Conversely, TAZ nuclear retention was promoted by TGFβ, a potent NIS repressor, and TAZ silencing markedly relieved the TGFβ-induced inhibition of the symporter.
Conclusions:
We demonstrate that the effects of TAZ are promoter specific, as it functions as a corepressor of PAX8 to modulate Slc5a5 expression in TFCs. Overall, our data place TAZ as an integrator of the different signaling pathways that control NIS expression, pointing to a role for TAZ in thyroid differentiation and identifying the Hippo pathway as a relevant target to recover NIS levels in thyroid cancer cells.
Introduction
Hippo signaling is an evolutionary conserved signaling pathway that controls organ and tissue growth by regulating cell proliferation, apoptosis, and differentiation (1,2). In mammals, the Hippo core machinery consists of a kinase cascade, including STE20-like protein kinase 1/2 (MST1/2), adaptor salvador 1 (SAV1/WW45), large tumor suppressor 1/2 (LATS1/2), and Mps one binder (MOB1). These kinases modulate the phosphorylation (activation) of the major pathway effectors, the transcriptional coactivators YAP1 (Yes-associated protein) and TAZ (transcriptional coactivator with PDZ-binding motif).
When the Hippo pathway is off, nonphosphorylated YAP and TAZ enter the nucleus, and stimulate target gene transcription to drive proliferation. Activation of Hippo signaling triggers YAP/TAZ phosphorylation by LATS1/2 kinases, resulting in their 14-3-3-mediated cytoplasmic retention and/or proteasome degradation (3,4). YAP and TAZ lack a DNA-binding domain and function by interacting with multiple transcription factors, primarily members of the TEAD family of transcription factors (5), although many other transcription factors have been linked to the activity of YAP/TAZ (6 –9). YAP/TAZ are widely described as coactivators, but they can also act as transcriptional corepressors in some contexts (10,11).
Hippo regulation in the thyroid is poorly understood, and the few available reports have mainly focused on the role of YAP/TAZ in thyroid development and carcinogenesis (12 –14). A better understanding of the function and regulation of the Hippo pathway in normal thyroid physiology should help in discerning its role in thyroid pathology.
PAX8, NKX2.1, and FOXE1 are lineage transcription factors crucial for the differentiation of thyroid follicular cells (TFCs) by modulating the expression of thyroglobulin, the sodium/iodide symporter (NIS), and thyroid peroxidase (15 –17). All three proteins are essential for thyroid hormone (TH) biosynthesis and are stimulated by thyrotropin (TSH), which is the main positive regulator of TFC proliferation and differentiation. By interacting with its G protein-coupled receptor (GPCR), TSH activates mainly Gαs-mediated cyclic adenosine monophosphate (cAMP) production (18).
Contrastingly, transforming growth factor β1 (TGFβ) is a negative regulator of TFC growth (19) and differentiation by inhibiting TSH-induced FOXE1 expression (20) and by repressing NIS through multiple molecular mechanisms (21). Iodide uptake by NIS is crucial not only for TH synthesis but also for radioactive iodine therapy of differentiated thyroid carcinomas (22).
TAZ is expressed in the adult thyroid and is reported to act as a coactivator of PAX8 and NKX2.1 (23,24). Given that PAX8 is the major regulator of SLC5A5, we investigated the potential role of TAZ in PAX8-mediated NIS regulation and explored the signaling pathways controlling TAZ expression in TFCs.
Materials and Methods
Cell culture, transfection, and infection
PCCl3 and HeLa cells were used as a stable (rat) follicular thyroid cell line (25) and as a heterologous system, respectively. Details of culture methods, gene silencing, and transfection are described in Supplementary Data.
Plasmids, site-directed mutagenesis, and luciferase assays
Promoter activity was evaluated by luciferase assays using the constructs described in Supplementary Data.
Protein extraction, Western blotting, immunofluorescence, and immunohistochemistry
Protein detection was performed following experimental techniques described in Supplementary Data. Antibodies are listed in Supplementary Table S1.
Quantitative real time-polymerase chain reaction
RNA extraction and quantification were performed as described in Supplementary Data. Primers are listed in Supplementary Table S2.
Immunoprecipitation assays, radioiodide transport assay, and in vivo mice studies
Immunoprecipitation, iodide uptake, and mouse studies were conducted as described in Supplementary Data.
Statistical analyses
GraphPad Prism (La Jolla, CA) was used for all statistical analyses. Results are represented as mean (standard error of the mean or standard deviation) of at least three independent experiments. Differences between measurements were analyzed by Student's two-tailed t-test. Significant differences were considered at p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), and p < 0.0001 (****).
Results
TAZ acts as a corepressor of PAX8-mediated Slc5a5 transcription
TAZ coactivates PAX8 in the transcriptional regulation of TG (23). As PAX8 is key for thyroid differentiation, we questioned if TAZ regulates other PAX8-target genes. We focused on Slc5a5, which is directly regulated by PAX8 by its binding to the NIS upstream enhancer (NUE) (26). Transient transfection assays in HeLa cells showed that PAX8 augmented Slc5a5 promoter activity, whereas TAZ had no affect (Fig. 1A). Notably, PAX8-induced Slc5a5 activation was significantly lower when cells were cotransfected with TAZ (Fig. 1A), suggesting that TAZ corepresses PAX8 in the regulation of Slc5a5 transcription, contrary to its canonical role as a coactivator.
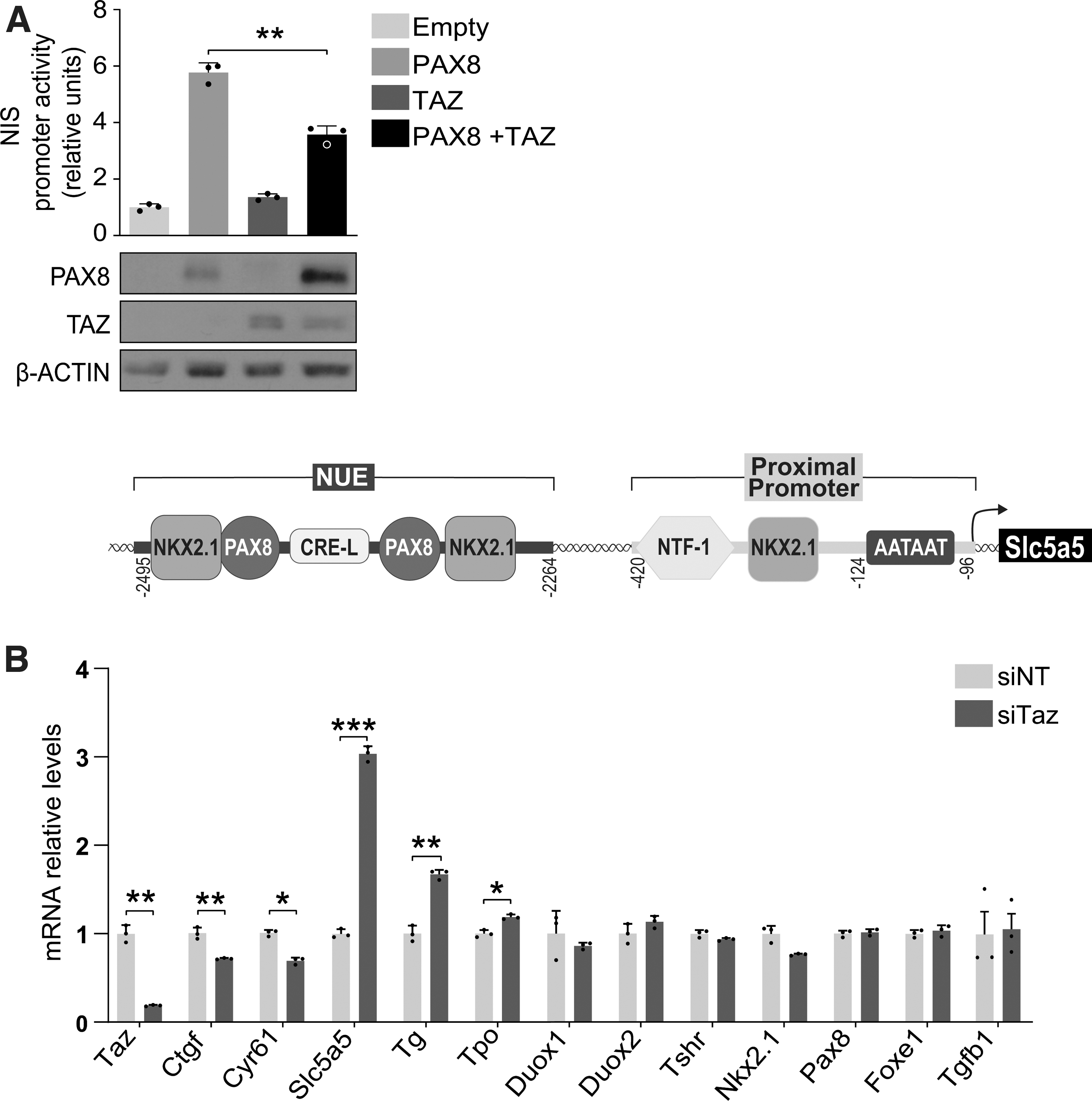
TAZ acts as a corepressor of PAX8-induced Slc5a5 transcription. (
We assessed whether this activity of TAZ is exclusive for Slc5a5 or whether it represses other specific/nonspecific thyroid genes by surveying the expression of a range of genes in rat thyroid PCCl3 cells transiently transfected with an siRNA against Taz. The gene expression of Taz was significantly lower in Taz-silenced PCCl3 cells (Fig. 1B), as was the expression of the TAZ-target genes Ctgf and Cyr61 (27). Examination of thyroid-related genes revealed that Slc5a5 expression was significantly higher (three-fold) in Taz-silenced cells, and Tg and Tpo were also significantly higher but to a lesser extent (Fig. 1B).
Other genes controlling thyroid function, including Duox1, Duox2, Tshr, and Tgfb1, and the transcription factors Nkx2.1, Pax8, and Foxe1, were unaffected by Taz silencing. Comparable results for thyroid differentiation gene expression were found when Taz was stably silenced with a small hairpin construct (Supplementary Fig. S1). These data suggest a promoter-specific corepressor action for TAZ, acting principally on Slc5a5.
Knockdown of Taz increases NIS expression in the cell membrane, improving iodide uptake and relieving the repression of NIS by TGFβ
TGFβ strongly downregulates Slc5a5 expression (21) and we show that TAZ is also involved in its transcriptional regulation. The functional crosstalk between YAP/TAZ and the TGFβ pathway in multiple cellular systems (6,28) prompted us to test TAZ as a mediator of NIS repression by TGFβ.
Confirming the RNA analysis (Fig. 1B), NIS protein levels were higher in Taz-silenced PCCl3 cells than in control cells (Fig. 2A). We also detected a greater abundance of TAZ in control cells treated with TGFβ (Fig. 2A), particularly in the nucleus (Supplementary Fig. S2A, B), suggesting that TAZ participates in the repression of Slc5a5 by TGFβ. Indeed, the suppression of NIS by TGFβ in control cells was relieved in Taz-silenced cells (Fig. 2A). Nonetheless, combined Taz knockdown and TGFβ pathway blockade with the TβRI inhibitor SB431542 additively increased NIS levels (Fig. 2A), suggesting that rather than mediating TGFβ-induced NIS inhibition, TAZ independently represses the symporter.
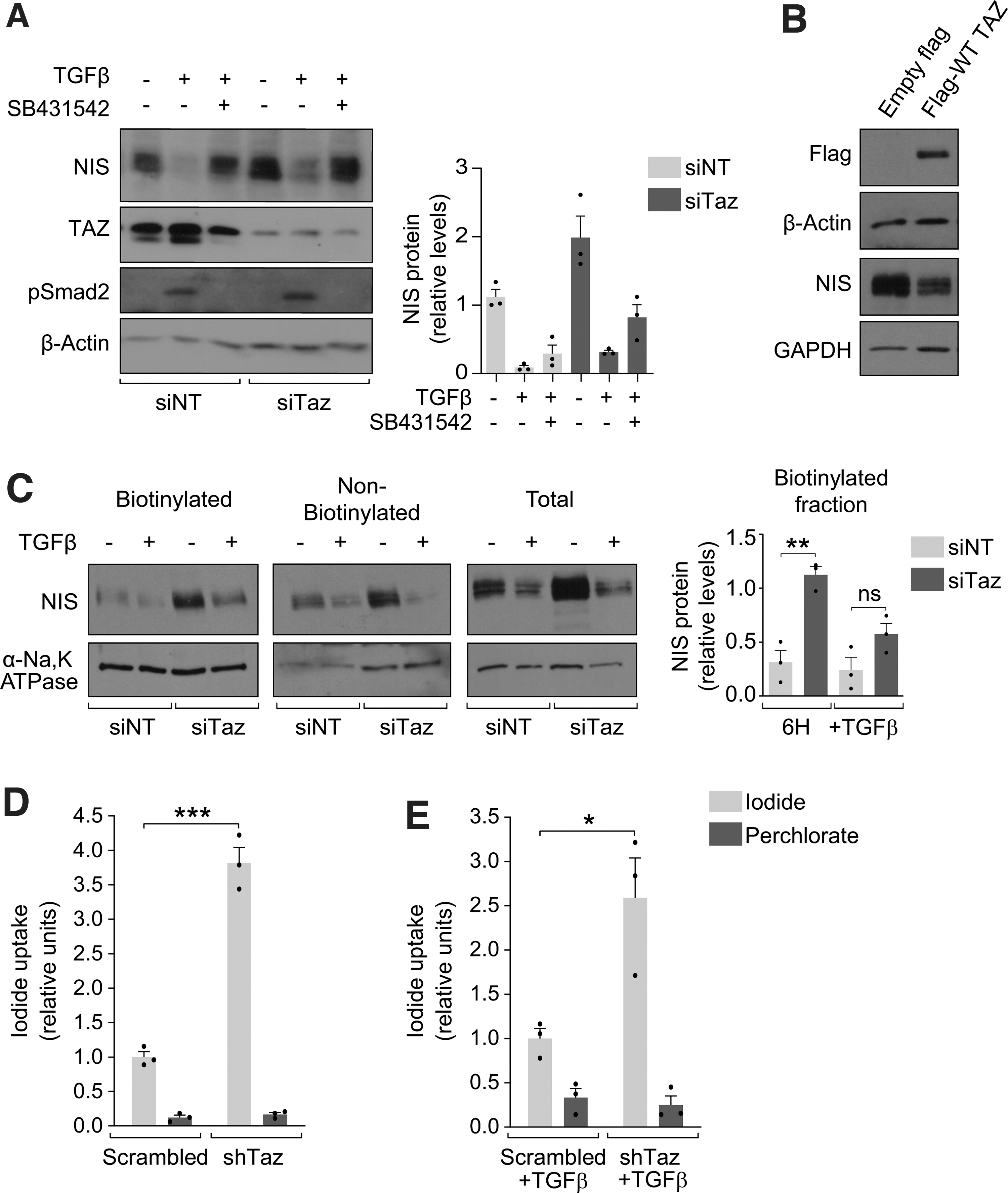
TAZ knockdown increases NIS expression and function, relieving TGFβ-induced inhibition. (
To further confirm that TAZ regulates NIS expression, we overexpressed wild-type (WT) human TAZ in PCCl3 cells, finding that NIS protein expression was diminished in the presence of high TAZ levels (Fig. 2B). As TAZ is negatively regulated by its upstream kinase LATS1, we silenced Lats1 expression as a strategy to increase endogenous TAZ protein levels, finding a moderate decrease in NIS expression (Supplementary Fig. S3). Overall, these results corroborate that TAZ mediates NIS repression.
To function as an iodide symporter, NIS must be located at the basolateral membrane of the TFC. To assess whether NIS membrane localization was dependent on TAZ expression, we performed cell surface biotinylation assays in PCCl3 cells. NIS protein levels were higher in the biotinylated fraction of Taz-silenced cells, even in the presence of TGFβ (Fig. 2C). To question whether this translated into enhanced NIS function, we generated stable short hairpin Taz-silenced PCCl3 cell lines to analyze iodide uptake, finding a significant 3.5-fold increase upon Taz silencing (Fig. 2D). Also, loss of Taz improved NIS active transport in cells concurrently treated with TGFβ (Fig. 2E). Overall, these data support the idea that TAZ is a potent regulator of NIS expression, location, and function. As these findings point to TAZ as a key target to mitigate TGFβ-induced NIS inhibition, we studied the underlying mechanism.
TAZ mediates NIS repression by downregulating PAX8 and blocking its binding to the Slc5a5 promoter
We show that TAZ hinders PAX8-induced Slc5a5 transcription; however, Pax8 mRNA expression is unaffected by Taz silencing. We questioned whether the TAZ-mediated inhibition of NIS expression induced by PAX8 could be explained by its regulation of PAX8 localization. Western blotting showed that the levels of PAX8 in nuclear fractions were increased in short hairpin Taz-silenced PCCl3 cells, even after TGFβ treatment (Fig. 3A), which is similar to our observations on NIS expression (Fig. 2A). We performed chromatin immunoprecipitation assays to examine whether PAX8 upregulation was accompanied by enhanced binding to the NUE.
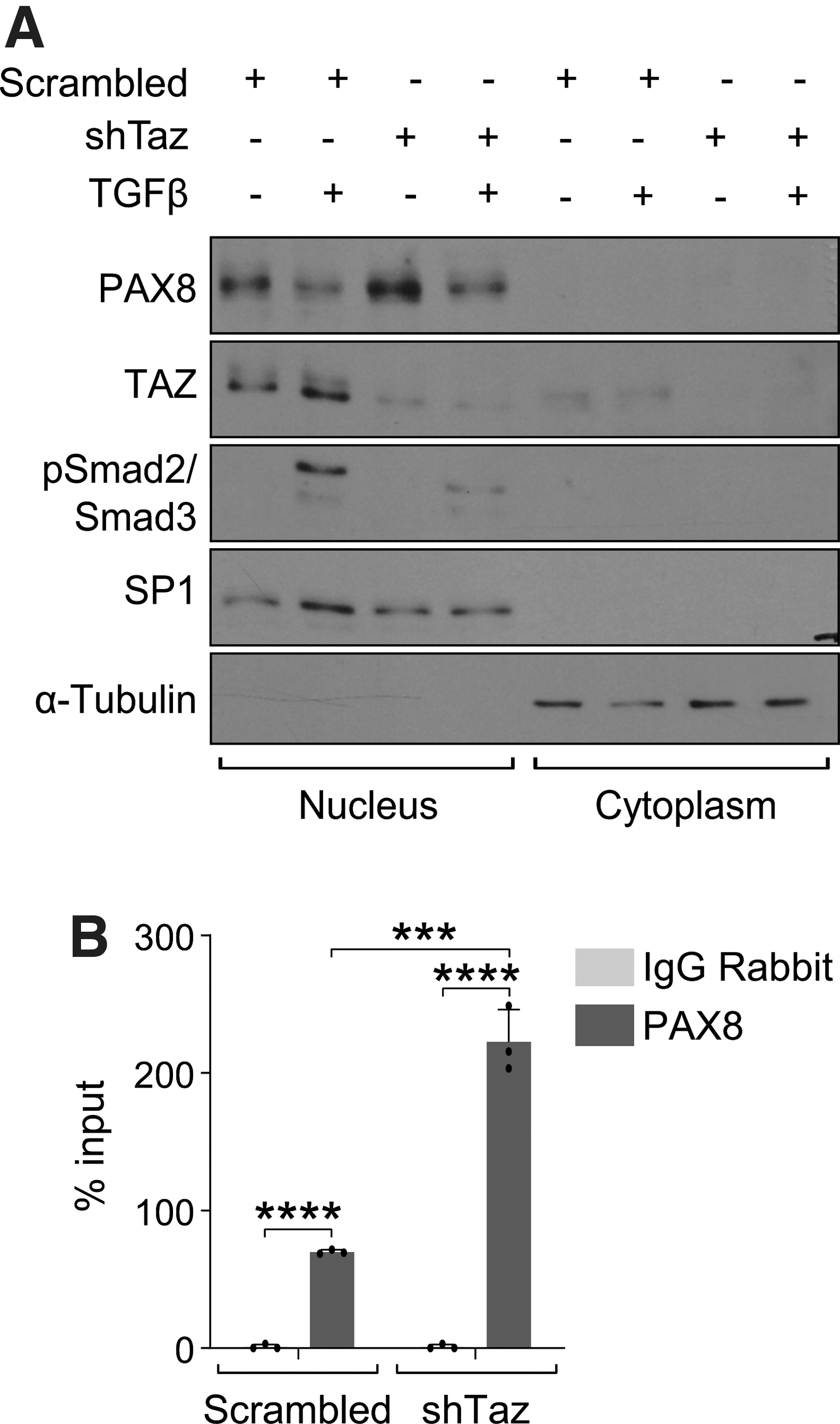
TAZ mediates NIS inhibition by impairing PAX8 binding to the Slc5a5 promoter. (
Results showed that the binding of PAX8 to the NUE was significantly greater in short hairpin Taz-silenced PCCl3 cells (Fig. 3B). These data indicate that TAZ tempers PAX8 expression and binding to the NUE, impairing Slc5a5 transcription.
TEAD-binding residues are critical for TAZ-mediated downregulation of NIS expression, but is independent of TEAD1 and YAP1
As TAZ does not directly bind DNA, we analyzed the role played by its main interactor partner, the TEAD family of transcription factors. We measured Slc5a5 promoter activity in transfection assays using two human TAZ expression vectors carrying either the nonphosphorylatable S89A mutation, which traps TAZ in the nucleus (Supplementary Fig. S4A) (6), or the F52/53A mutation, which impairs TEAD binding (Supplementary Fig. S4B) (29). Slc5a5 promoter activity was significantly inhibited both by WT TAZ and the S89A mutant (Fig. 4A).
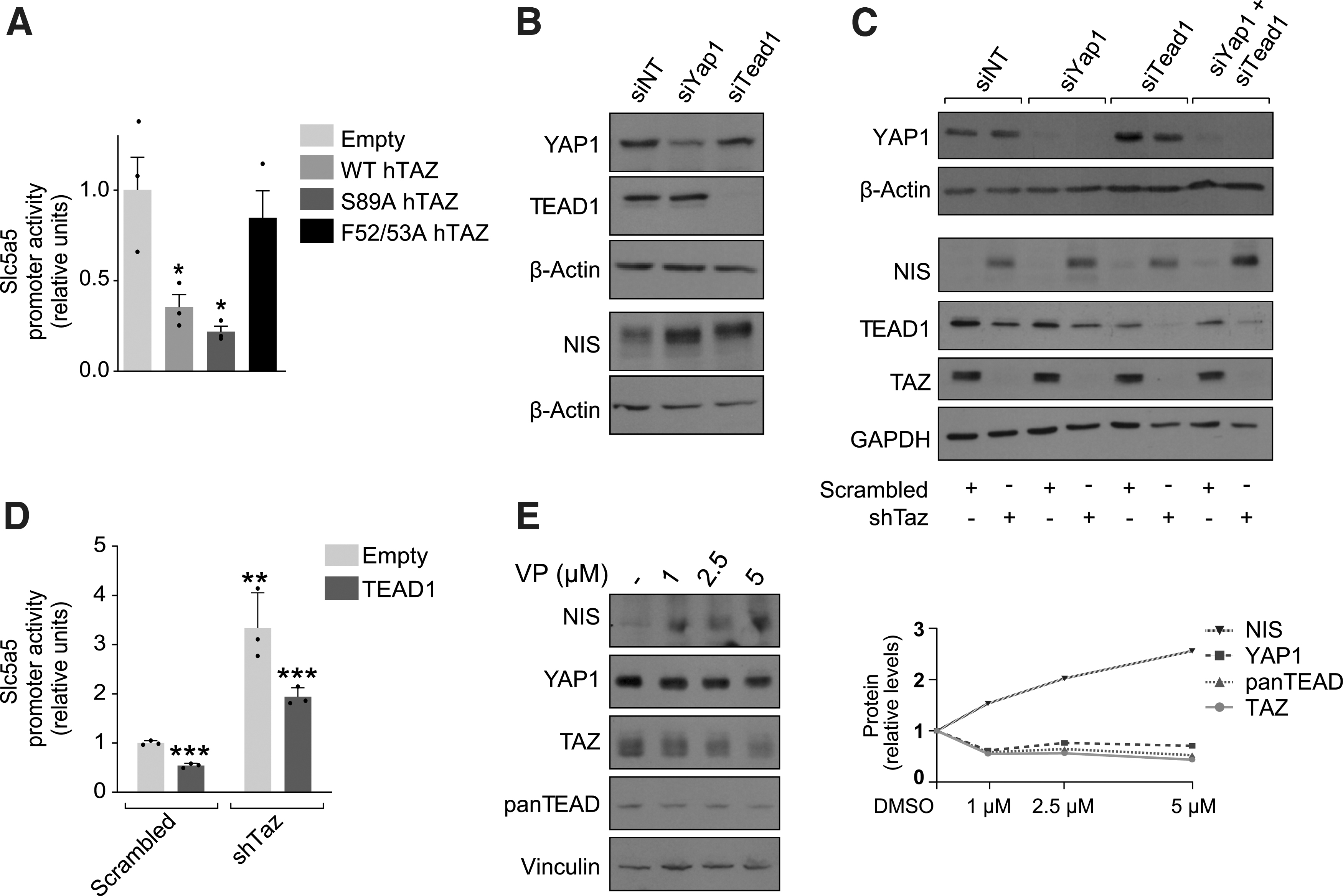
TAZ is sufficient for NIS inhibition and is dependent on its F52/53 residues. (
By contrast, the repressive effects of TAZ on Slc5a5 promoter activity were lost in cells cotransfected with the F52/53A mutant (Fig. 4A), indicating that TAZ relies on TEAD-binding residues to inhibit Slc5a5 expression.
As the TAZ mutant study highlighted the importance of a complex between TAZ and TEAD transcription factors to negatively affect Slc5a5 transcription, we silenced Tead1 in PCCl3 cells. We focused on TEAD1 because it shows the greatest expression of the four TEAD paralogs in endocrine tissues (
Results showed that NIS expression was moderately enhanced when Tead1 or Yap1 was individually silenced, (Fig. 4B, C), but was greater when both were silenced simultaneously with Taz (Fig. 4C). Overexpression of human TEAD1 in short hairpin Taz-silenced PCCl3 cells failed to completely suppress Slc5a5 promoter activation (Fig. 4D), and we failed to detect an interaction of TEAD1 with the NUE (Supplementary Fig. S4C). These data suggest that TAZ is sufficient for Slc5a5 inhibition. Finally, we confirmed the effect of the Hippo pathway on NIS expression using the YAP/TAZ inhibitor verteporfin (30), which enhanced NIS protein levels even at the lowest concentration tested (Fig. 4E).
TSH promotes Hippo pathway activation, triggering TAZ nuclear exit and degradation
The mechanisms underlying the activation of Hippo signaling are not fully established, but it has been reported that metabolites and hormones can regulate YAP/TAZ via GPCR signaling (31). We questioned whether TSH, via its GPCR, affected Hippo pathway activation. Western blotting and immunocytochemistry analysis revealed an increase in phosphorylated and nonphosphorylated MST1/2, pLATS1/LATS1, and MOB1 expression in PCCl3 cells after TSH treatment, which was mimicked by the cAMP inductor forskolin (Fig. 5A, B).
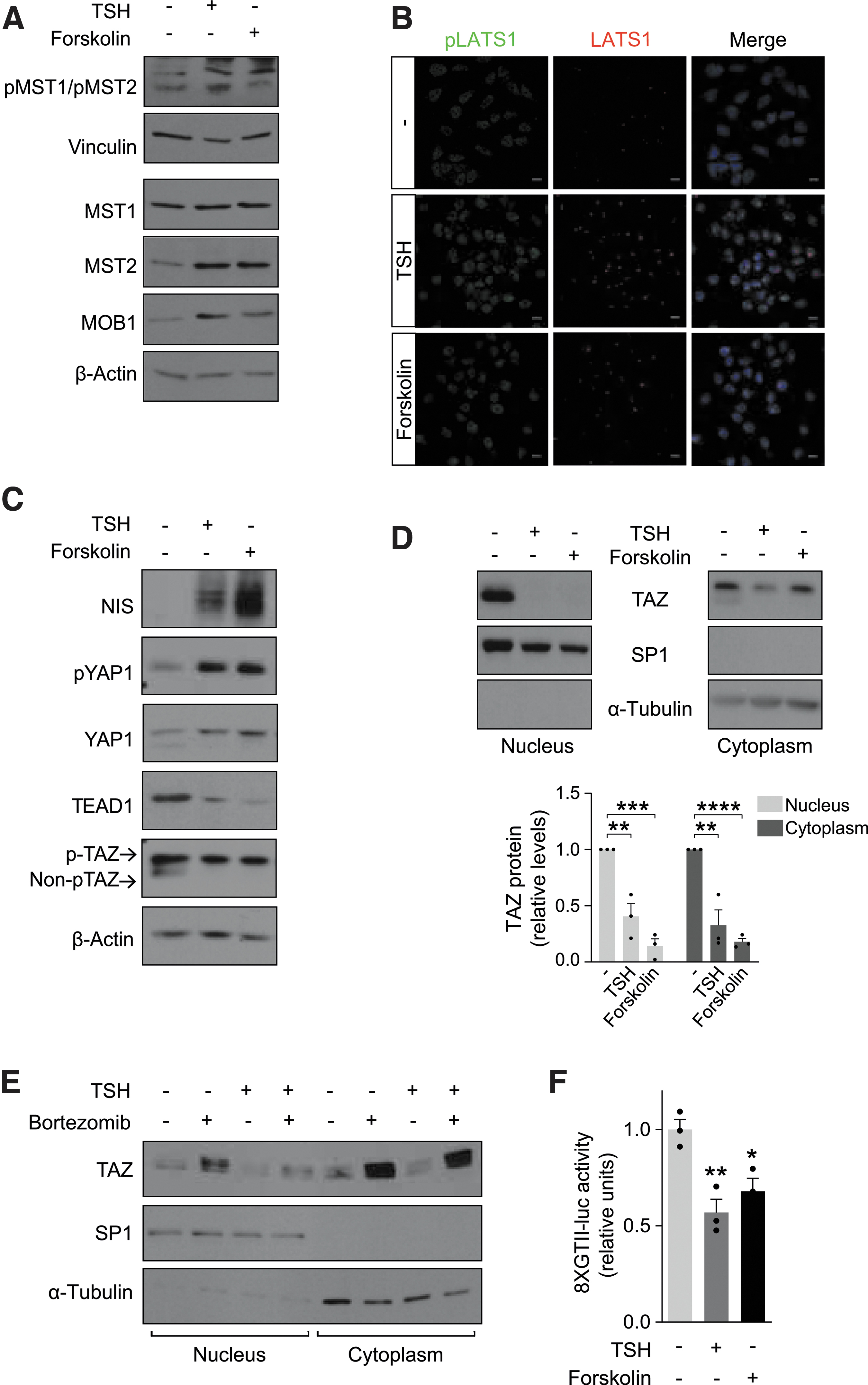
Thyrotropin/cAMP pathway activates Hippo signaling and decreases TAZ nuclear levels. (
Both TSH and forskolin also modified the downstream effectors of the Hippo cascade, as revealed by a strong increase in YAP1 phosphorylation, and by lower TEAD1 levels (Fig. 5C). Moreover, the nonphosphorylated form of TAZ was undetectable after either treatment, which was accompanied by the expected increase in NIS expression (Fig. 5C). In line with this was the finding of TAZ nuclear exclusion in TSH- and forskolin-treated cells (Fig. 5D).
We tested how proteasomal inhibition affects TAZ degradation, finding that both MG132 and bortezomib treatments led to a recovery of total TAZ levels (Supplementary Fig. S5), even after TSH stimulation and TAZ nuclear accumulation (Fig. 5E). This indicates that TSH decreases TAZ stability, promoting its proteasomal degradation.
We questioned whether the transcriptional activity of the two Hippo mediators was impaired by TSH using transactivation assays in PCCl3 cells with a synthetic YAP/TAZ-responsive promoter construct (32). Both TSH and forskolin significantly decreased promoter activity (Fig. 5F). Taken together, these data demonstrate that TSH, through cAMP, promotes TAZ phosphorylation and nuclear exclusion, leading to proteasomal degradation and lower activation of target promoters.
TSH regulation of Hippo effectors occurs in vivo
To confirm the in vitro effects of TSH on Hippo mediators, we induced hypothyroidism in mice using a low-iodine diet (LID). Histological staining of thyroid sections revealed large and flat follicles in age-matched control animals, and smaller thyroid follicles with a columnar-like epithelium in LID mice, demonstrating hypothyroidism (Fig. 6A). This was confirmed by high TSH, low thyroxine (T4) and maintained triiodothyronine (T3) serum levels (Fig. 6B–D) (33). In addition, NIS expression was notably increased (Fig. 6E).
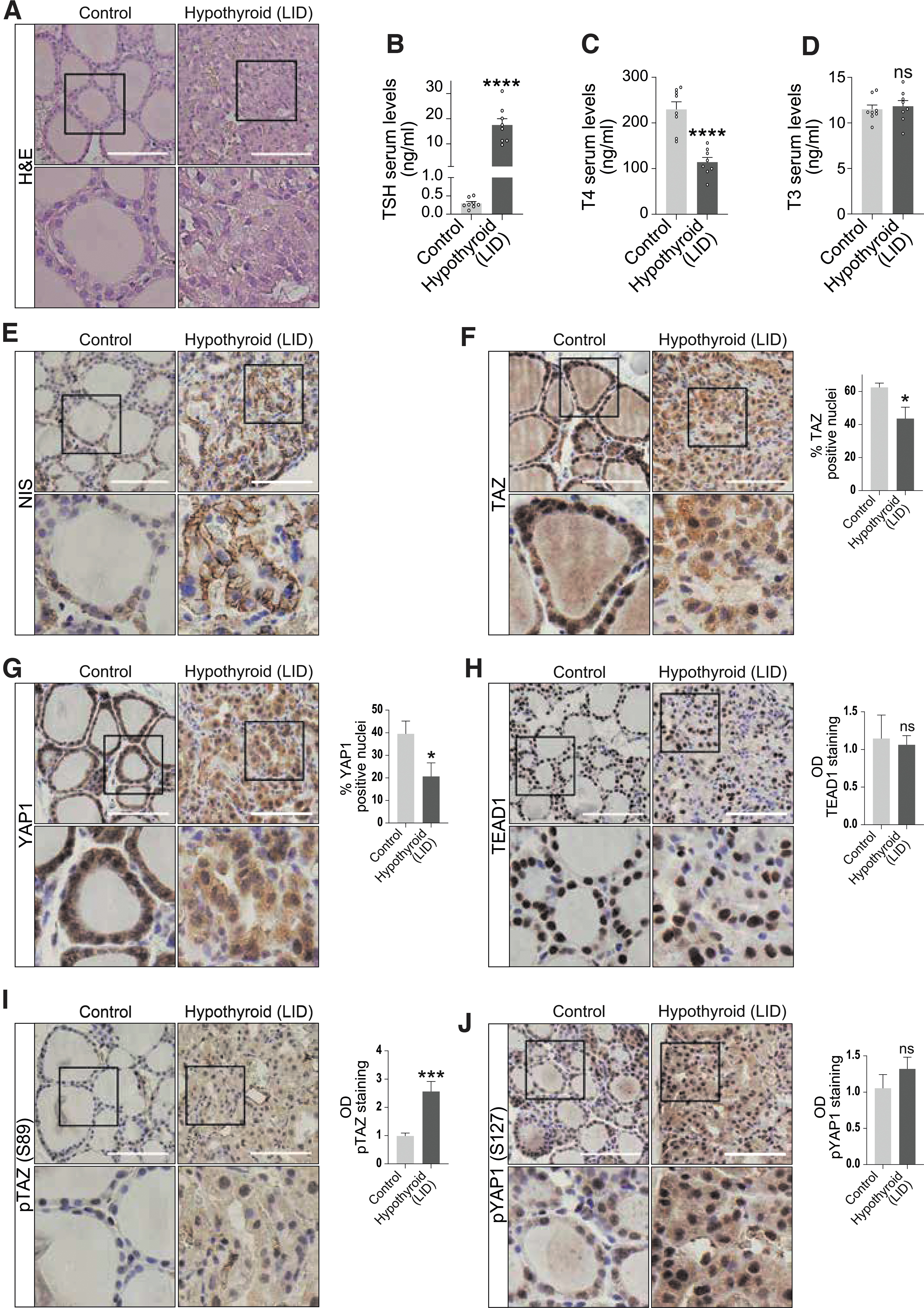
Hypothyroid mice show YAP/TAZ nuclear exclusion and pTAZ accumulation. Hypothyroidism was induced in experimental mice by LID. Thyroid histological sections from control and hypothyroid mice were analyzed for differences in Hippo signaling components. (
TAZ and YAP1 nuclear staining in thyroid sections was significantly weaker in hypothyroid mice than in control mice (Fig. 6F, G), concomitant with a small decrease in the expression of the YAP/TAZ-targets CTFG and CYR61 (Supplementary Fig. S6A, B). No significant changes were observed for the expression of TEAD1 (Fig. 6H), providing further evidence of its likely minor effect on NIS regulation. Moreover, pTAZ staining was significantly greater (two-fold) in hypothyroid mice (Fig. 6I) and a trend for an increase was observed for pYap1 (Fig. 6J), which might indicate that TAZ expression in the thyroid is more sensitive than YAP1 to changes in TSH levels.
Similar results (Supplementary Fig. S7) were obtained in hypothyroid mice generated by MMI/perchlorate treatment (34). Overall, these findings agree with our in vitro data and strongly suggest that TSH decreases the nuclear distribution of Hippo cofactors and triggers TAZ inactivation in the thyroid.
Discussion
The function of NIS in iodide transport requires both its expression and its correct location on the basolateral membrane (35). We show that loss of Taz stimulates NIS function, as reflected by its correct membrane location and iodide transport. While it is not known how TAZ regulates NIS trafficking to the membrane, we hazard that it is an indirect process. What is clear, however, is that TAZ suppresses Slc5a5 transcription by limiting PAX8 binding to its promoter through a mechanism involving PAX8 repression in the nucleus. Depletion of Taz in PCCl3 cells also upregulates Tg and Tpo expression, although to a lesser extent, but not Duox1/2 or other thyroid transcription factors.
These findings differ from those of previous studies where TAZ acts as a coactivator (23,36). Although TAZ interacts with PAX8 in PCCl3 cells, its role as a coactivator of PAX8 on TG and DUOX1/2 transcription was established in HeLa and HEK293T cells, respectively (23,36). TFCs are the only cellular system that simultaneously expresses PAX8, NKX2.1, and FOXE1 transcription factors, and thyroid-specific genes (37). Heterologous cell systems may lack a protein interaction that critically modulates TAZ function and is specific to thyroid cells. Our data are in accordance with those described in zebrafish where taz abolition does not affect nkx2.1 or pax8 levels, but increases tg, tpo, and slc5a5 expression (14).
Our findings establish TAZ as a new player in thyroid differentiation, and it will be interesting to define its interactions with other regulators. TAZ was also observed to positively modify NKX2.1 regulation of the TG promoter (23), although in HeLa cells, and the presence of PAX8 in the thyroid might interfere with the TAZ-NKX2.1-mediated interplay. SOX9, a transcription factor also involved in thyroid development and differentiation (38), is a downstream target of YAP/TAZ-TEAD, and the transcriptional regulation of SOX9 by these factors—either activating or repressing its promoter—depends on the cell type (39,40). These data support the notion that the function of TAZ as a cofactor should be studied in a promoter- and cell-specific context.
We found that the other main mediators of the Hippo pathway, YAP1 and TEAD1, are not necessary for the inhibitory action of TAZ on NIS. While the lack of an inhibitory effect of the TAZ F52/53A mutant reveals dependency for TEAD-binding residues for TAZ-induced NIS repression, this is not related to TAZ-TEAD1 interactions, and the role of other TEAD family members should be addressed in future studies.
Whereas a wide range of GPCR ligands are known to regulate the Hippo pathway (31), regulation by TSH has not previously been addressed to our knowledge. We show that TSH/cAMP triggers activation of the Hippo cascade, reducing TAZ nuclear levels and its transcriptional activity. Accordingly, TSH treatment blocks TAZ-mediated NIS inhibition. TSH stimulates NIS expression by increasing PAX8 binding to the Slc5a5 promoter (26), and inhibiting TAZ nuclear expression is an additional mechanism by which TSH promotes this binding (Fig. 7, right).
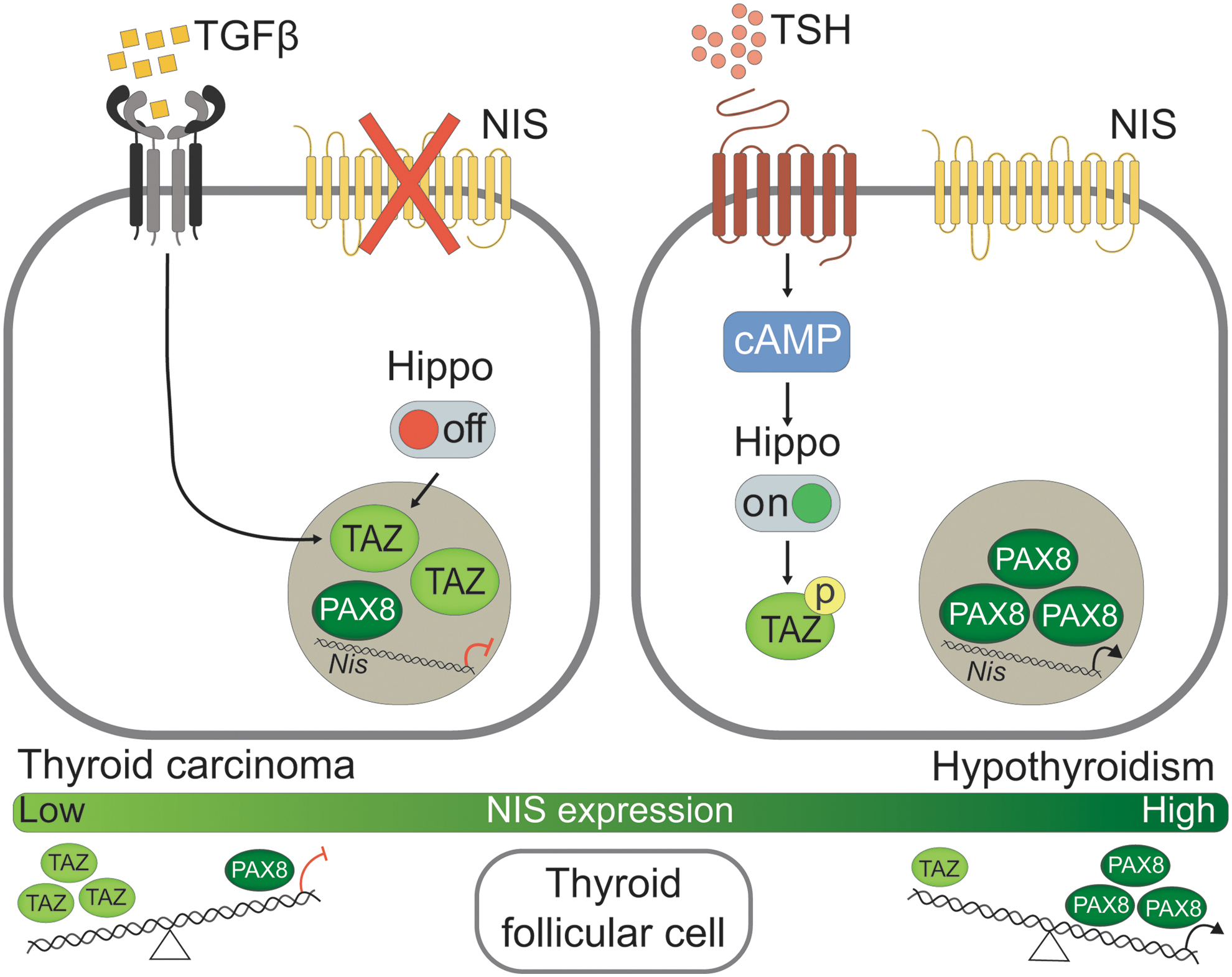
Model of the role of TAZ in the adult thyroid differentiated follicular cell. Left panel: Following TGFβ treatment of TFCs and in the absence of Hippo signaling, TAZ protein is elevated and accumulates in the nucleus, from where it decreases PAX8 protein levels and impairs PAX8 binding to the NUE, thus limiting the presence of NIS on the basolateral membrane. Right panel: Upon TSH stimulation, cAMP signaling is triggered in the cytoplasm of TFCs, leading to Hippo activation and TAZ phosphorylation, allowing PAX8 to enter the nucleus and promote NIS expression. Lower panel: A balance between PAX8 and TAZ expression is critical to maintain physiological levels of NIS. When TSH levels are high, as in hypothyroidism, PAX8 overexpression and TAZ downregulation lead to NIS upregulation. By contrast, in a pathological scenario with increased TGFβ, as in thyroid carcinoma, TAZ nuclear expression is upregulated, which compromises PAX8 and hence NIS levels. TFC, thyroid follicular cells.
The distribution of Hippo cofactors follows a similar pattern in hypothyroid mice, characterized by elevated serum TSH: YAP/TAZ are downregulated in the nucleus, and pTAZ levels are increased. YAP/TAZ are known to respond to cell stretch in confluent epithelial monolayers by accumulating in the nucleus (41), as was observed in euthyroid follicles.
TAZ participates in zebrafish thyroid specification, as morpholino-based knockdown of taz results in smaller thyroid follicle size and number (14). Although no thyroid defects were described in Taz-null mice (2,42), the small size of these mice together with their reduced physical activity suggests an impairment of the pituitary/thyroid axis. In agreement with this, TAZ has gained importance in thyroid development as its activation in human embryonic stem cells by ethacridine leads to TFC specification by upregulating NKX2.1 and PAX8. These effects are triggered by an increase in chromatin accessibility of their promoters through an unknown mechanism (43,44).
Hence, while TAZ appears to have a positive role in promoting early differentiation during development (43,44), it appears to have a negative role in differentiation maintenance in adulthood, as we demonstrate here (Fig. 7). In normal thyroid tissue, not all of the follicles express NIS concurrently, and this heterogeneity reflects the tight regulation by multiple feedbacks to control TH production (45).
Defining TAZ regulation in TFCs is crucial to understand how changes in signaling pathways in tumorigenesis could lead to the increased expression of this cofactor, which is associated with thyroid cancer (12). TGFβ is an inhibitor of thyroid growth and function by counteracting TSH actions (19,21,46), but it also plays a key role in thyroid carcinogenesis by decreasing NIS expression (47,48). We show that TGFβ promotes TAZ nuclear retention in PCCl3 cells (Fig. 7, left), in accordance with findings in other cell types (6,49). In fact, TAZ forms a complex with the SMADs proteins and modulates their nucleocytoplasmic shuttling upon TGFβ stimulation (6).
One possibility might be that TAZ interacts with SMADs in the nucleus of TFCs, enhancing their transcriptional activity, thereby reinforcing their NIS inhibitory effects and also preventing their cytoplasmic translocation. Our work shows that loss of TAZ in PCCl3 cells relieves TGFβ-induced NIS inhibition, and this might represent a new therapeutic option to improve radioiodine uptake in advanced thyroid carcinomas.
In conclusion, we provide evidence of crosstalk between the Hippo pathway, TGFβ, and TSH in TFCs, which ultimately controls NIS expression by maintaining a balance between TAZ and PAX8. In a pathological context, such as hypothyroidism or thyroid cancer where TSH and TGFβ are, respectively, upregulated, TAZ expression might be altered, which might influence the differentiated state of the cell (Fig. 7, lower).
Footnotes
Acknowledgments
We are grateful to members of the Instituto de Investigaciones Biomédicas, Drs. Miguel A. Zaballos and Adrián Acuña-Ruiz, for ideas and critical discussion with this work; Andrea Martinez-Cano and Raquel Arocha Riesco for technical assistance in the laboratory and with mouse experiments, respectively; and Mónica Martín-Belinchón, and Lucía Guerrero López for confocal images acquired in the Servicio de Microscopia Optica y Confocal. We greatly appreciate Dr. José C. Moreno (Instituto de Genética Médica y Molecular (INGEM), IdiPAZ, Madrid, Spain) for providing the Low Iodine Diet and for his advice with the in vivo experiments. We thank Dr. Vicente Barrios (Endocrine Service, Hospital Infantil Niño Jesus, Madrid, Spain) for helping with TSH, T4, and T3 assays and Dr. Kenneth McCreath for constructive comments on the article.
Authors' Contributions
C.F.-M. conducted the experiments, and acquired and analyzed the data. C.F.-M. and P.S. designed the research studies and wrote the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by grants SAF2016-75531-R, Fondo Europeo de Desarrollo Regional, and PID2019-105303RB-I00/AEI/10.13039/501100011033 from the Ministerio de Ciencia e Innovación (MICIN), Spain; and by grant S2017/BMD-3724 from Comunidad de Madrid. C.F.-M. holds an FPI fellowship from Universidad Autónoma de Madrid (Spain).
Supplementary Material
Supplementary Data
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Table S1
Supplementary Table S2
