Abstract
Background:
An unmet need for more effective and affordable kinase inhibitors remains in patients with progressive radioactive iodine-refractory differentiated thyroid cancer (RAIR-DTC) in China, where only sorafenib is approved for this indication. This study evaluated the 24-week objective response rate (ORR) to donafenib—a new, domestic multikinase inhibitor—in the treatment of locally advanced or metastatic RAIR-DTC in patients with measurable lesions. Two dose regimens (300 mg twice daily vs. 200 mg twice daily) were used to determine its optimal dosage and safety for further phase III studies.
Methods:
This study was a randomized, open-label, multicenter phase II trial. Thirty-five adult RAIR-DTC patients with at least one measurable targeted lesion according to RECIST 1.1 were enrolled from 12 centers in China and randomized to receive either 200 mg (17 patients) or 300 mg (18 patients) of donafenib orally twice daily for 24 weeks. The primary endpoint was ORR, and the secondary endpoints included progression-free survival (PFS) among others. Additionally, biochemical (serum thyroglobulin) and structural (total tumor diameter [TTD]) responses were assessed, change (ΔTTD) rates were calculated, and safety was evaluated.
Results:
The ORRs for the 200- and 300-mg arms were 12.5% and 13.33% (p = 1.000), respectively. The 300-mg arm had a nonsignificant, longer median PFS than the 200-mg arm (14.98 months vs. 9.44 months) (p = 0.351). There was a trend toward more tumor shrinkage in the 300-mg arm compared with the 200-mg arm (average ΔTTD rate −0.52 ± 0.71 vs. −0.04 ± 1.55 mm/month, p = 0.103). Most treatment-related adverse events (AEs) in both arms were grades 1–2. The most common grade 3 treatment-related AEs in both arms were palmar–plantar erythrodysesthesia and hypertension; the sum occurrence rates of these two AEs in the 200-mg and 300-mg arms were 11.43% and 22.86%, respectively.
Conclusions:
Donafenib was generally well tolerated. Both donafenib regimens demonstrated similar efficacy in terms of the ORR in locally advanced or metastatic RAIR-DTC. The results warrant further studies on donafenib as a new, feasible treatment option for RAIR-DTC patients. Clinical
Introduction
Thyroid cancer is the most common endocrine cancer with a global rise in incidence. The incidence of thyroid cancer in China accounts for approximately one-third of the new global cases, and the five-year survival rate of patients with thyroid cancer in China is lower than in the United States (84.3% vs. 98.3%) (1 –3). Considering the relatively less life-threatening stage distribution in the United States (distant 4% and regional lymph node metastases 28%) and the high proportion of deaths in China, these discrepancies between the two countries may indicate insufficient early intervention or ineffective medication in management of thyroid cancer and might account for the relatively high proportion of late-stage patients and their poor outcomes in China. As a result, there might be a high prevalence of patients with advanced thyroid cancers who warrant more effective treatment options in China.
Over 90% of thyroid cancers are differentiated thyroid cancers (DTCs) (4). Most intermediate/high-risk DTC patients achieve a favorable response after standard treatment, which includes surgery followed by radioactive iodine (RAI) treatment and serum thyrotropin (TSH) suppression with thyroid hormone. However, up to 23% of DTC patients develop distant metastases (5). Approximately one-third of high-risk DTC patients with locally advanced disease or distant metastasis eventually develop resistance to RAI therapy, which results in poor overall survival (less than 10% for 10 years) and a life expectancy of 3 to 5 years, patients who are RAI-refractory (RAIR) (4,6).
Several signaling pathways and activating mutations in genes driving thyroid tumorigenesis have been identified, include the MAPK pathway and proangiogenic factors such as VEGF and VEGFR (7). This finding spurred the development of multikinase inhibitors (MKIs), such as sorafenib and lenvatinib, which target several different tumorigenic drivers and both of which are already approved by the U.S. Food and Drug Administration (FDA) and recommended for progressive RAIR-DTC in the 2015 American Thyroid Association (ATA) thyroid cancer guidelines (4). It was only in 2017 that sorafenib was approved by the Chinese equivalent of the FDA, named the National Medical Products Administration (NMPA), and became the only MKI approved in China for RAIR-DTC. However, sorafenib is not affordable for most Chinese families despite being partially reimbursable. Additionally, progression in virtually all patients was ultimately noticed despite the initial good response to sorafenib or even lenvatinib, indicating the need for an alternative MKI as a rescue treatment option for such patients (8). Considering both financial constraints and possibilities of treatment resistance, more treatment options for RAIR-DTC patients are needed, especially in China.
Donafenib is a novel oral MKI, a compound in which a trideuteriomethyl group is substituted for a methyl on the sorafenib molecule, which exerts similar therapeutic effects as sorafenib by inhibiting phosphorylation of serine/threonine kinases (such as Raf kinase) and by blocking receptor tyrosine kinase signaling (such as VEGFR and PDGFR) (9). Donafenib has proven to be effective and safe at both 200 and 300 mg twice daily in advanced hepatocellular carcinoma (10) and is now in phase III trials for late-stage cancers such as liver [NCT02645981] and colorectal [NCT02870582] cancers. Considering the demonstrated efficacy and safety in the patients mentioned above, a phase II study was initiated in 2017 to determine the 24-week objective response rate (ORR) of donafenib in MKI-naïve Chinese patients with measurable, progressive, locally advanced/metastatic RAIR-DTC lesions. Two dosage regimens (200 or 300 mg twice daily orally) were used to determine its optimal dosage and safety for further phase III studies. Herein, we present the results on the efficacy and safety of donafenib.
Patients and Methods
Patients
The patients included were similar to those enrolled in the phase III sorafenib DECISION trial (11), with the exception that hepatitis B virus carriers (viral load <1000 copies/mL) were also allowed to be enrolled in this study. Patients ≥18 years old with locally advanced/metastatic RAIR-DTC were eligible if they met the following criteria: had at least one measurable lesion according to RECIST 1.1; disease progression as per RECIST 1.1 within the past 14 months; RAIR defined as (i) at least one measurable lesion with no iodine uptake after RAI treatment, (ii) the presence of a measurable lesion that progressed within 14 months after RAI treatment (dose of 3.7–7.4 GBq or 100–200 mCi of 131I) despite iodine uptake, or (iii) had a cumulative 131I dose of ≥22 GBq or 600 mCi without RAI treatment within 6 months before screening; disease not amenable to surgery or radiation therapy; tolerance to TSH suppression therapy and TSH <0.1 mIU/L; Eastern Cooperative Oncology Group performance status (ECOG PS) of 0–2; and adequate bone marrow, liver, and renal function. Patients who received previous tyrosine kinase inhibitor (TKI) therapy, targeted therapy, chemotherapy, or thalidomide were excluded; however, low-dose chemotherapy for radiosensitization was allowed. This study was approved by the Ethics Committee of each participating study center. Written informed consent was obtained from all patients before study entry.
Study design and treatment
This was a randomized, open-label, multicenter phase II study of oral donafenib in patients with RAIR-DTC. A randomized design was utilized for analysis by SAS 9.4 software, and patients from each center were competitively randomized at a ratio of 1:1 to either 300 or 200 mg of donafenib orally twice daily for 24 weeks. Dose modifications were required for adverse events (AEs) of grade 3 or above as defined in the Common Terminology Criteria for Adverse Events (CTCAE), V4.0, with treatment: (i) interrupted for grade 3 or above AEs until the AE had resolved to grade 1 or baseline within two weeks and resumed at a lower dose of 200 or 300 mg daily in each regimen, respectively; (ii) interrupted if grade 3 or above AEs occur for the second time and resumed at a lower dose of 200 or 300 mg once every other day in each regimen, respectively, until the AE had resolved to grade 1 or baseline within two weeks; and (iii) discontinued for AEs that did not resolve within two weeks or grade 3 or above AEs occurring for the third time. Upon completion of the 24-week treatment, eligible patients entered the extension period: the dose of donafenib for patients on the 200-mg regimen was modified to 300 mg twice daily for a possibly better response, while the dose for those on the 300-mg regimen remained. Patients entered a long-term follow-up period after discontinuation of treatment due to disease progression, unexpected medical events, or intolerable toxicity.
Assessments
Efficacy assessments included the ORR as the primary efficacy endpoint, defined as the proportion of patients with the best overall response (partial response and complete response) according to imaging assessments as per RECIST 1.1. The secondary endpoints measured included progression-free survival (PFS), overall response, disease control rate (DCR), Tg/TgAb levels, pharmacokinetics, safety, and the Functional Assessment of Cancer Therapy–General (FACT-G) quality of life scale. Safety assessments included AEs (according to CTCAE, v4.0), vital signs, physical examination results, ECOG PS scores, laboratory examination results (routine blood, routine urine, blood biochemistry, coagulation function, and thyroid function), blood pregnancy test results, and 12-lead electrocardiogram recordings. Structural and biochemical responses were assessed in terms of changes in serum thyroglobulin (Tg) and changes in target lesions measured by changes in the total tumor diameter (ΔTTD), respectively. According to RECIST 1.1, the TTD was defined as the sum of diameters of target lesions (mm), as determined by imaging assessments. Serum Tg levels were measured with an electrochemiluminescence immunoassay according to each center's procedures.
Physical examination results, vital signs, concomitant treatments, AEs, and compliance were assessed at each visit. Laboratory tests (hematology, chemistry, and urinalysis), ECOG PS scoring, thyroid function-related tests, including TSH and Tg/TgAb tests, and electrocardiograms were performed at all visits (except for visit 3, which was for PK sampling only). Tumor imaging assessments by computed tomography (CT)/magnetic resonance imaging (MRI) scans were performed within four weeks before treatment and every eight weeks after treatment. Brain imaging was performed every eight weeks after the start of treatment in patients with brain metastases at baseline or who were suspected of having newly developed brain metastases. All the above-mentioned procedures were performed every 12 weeks in the extension period. CT/MRI scans and TSH and Tg/TgAb tests were performed every 3 months for patients who discontinued treatment until progressive disease, withdrew informed consent, or started another antitumor therapy; the survival status was also updated at these visits.
Statistical analyses
With the assumption of a two-sided alpha value of 0.1, a power of 70%, a threshold response rate of 12% as per the result of the DECISION trial (10), and a 2% ORR of routine TSH suppressive therapy by LT4 (levothyroxine) as a control, a total of 48 subjects were initially planned for enrollment according to Simon's single-stage phase II design. The 95% confidence interval [CI] was used. The main population for the efficacy analysis included all randomized patients who received donafenib for at least two weeks per protocol without serious violations of the protocol. All patients who signed the informed consent form and received at least one dose of donafenib were assessed for safety. The ORRs between the two arms were compared using a Cochran–Mantel–Haenszel (CMH) test, and the CI of the difference between the two arms was calculated. A log-rank test was used to compare PFS between the two arms. Kaplan–Meier methods were used to estimate survival, and the Cox proportional hazards model was used to calculate hazard ratios and CIs between the two arms. DCRs and CIs of the two arms were calculated using the central effect-oriented CMH method.
Results
Patients
Given that the expected primary endpoint as an ORR of 12% was observed earlier in both arms, members of the Steering Committee thought it was convincing enough to initiate a further phase III trial based upon the following reasons: (i) the relatively low incidence of RAIR-DTC, (ii) the lack of an effective TKI in China for RAIR-DTC patients, (iii) the trend of a more promising overall response in the 300-mg regimen that indicated the optimal initial dose of donafenib in its phase III study, and (iv) the overall good tolerance of donafenib among the enrolled RAIR-DTC patients thus far. The decision was unanimously agreed upon by the Steering Committee, which consists of principal investigators, clinicians, and sponsors, and was validated and approved by the Ethics Committee. Hence, enrollment for this phase II trial ended earlier than expected, although 48 patients were planned. In total, 35 patients were ultimately enrolled in this study between March 27, 2017, and March 14, 2018, from 12 centers in China. Approximately 10% (4/35) of the enrolled patients were HBV carriers. The patients' baseline characteristics are listed in Table 1. The average age for the 200- and 300-mg arms was 56.47 and 54.28 years, respectively. The ratios of females to males were 1.43:1 and 2:1 for the 200- and 300-mg arms, respectively. Two of the 17 patients in the 200-mg arm had an ECOG PS score of 2, while all 18 patients in the 300-mg arm had an ECOG PS score of 0–1. The DTC in most patients of both arms was classified as papillary thyroid cancer by histopathology (82.35% and 83.33%, respectively). There were no significant differences in the tumor (p = 0.359), node (p = 0.055), or metastasis (p = 1.000) stage between the two arms. All patients had RAIR-DTC with measurable disease. All patients in both arms presented with distant metastasis, except for one patient in the 300-mg arm who had locally advanced thyroid cancer. The most common sites for metastases were lymph nodes and lungs. All patients were treated with total thyroidectomy and subsequent RAI therapy before enrolling in the study. One patient (5.88%) in the 200-mg arm and two patients (11.11%) in the 300-mg arm received external radiation therapy before enrollment.
Baseline Patient Characteristics (Full Analysis Set)
BID, bis in die; ECOG, Eastern Cooperative Oncology Group; RAI, radioactive iodine.
Efficacy
The ORRs were 11.76% and 11.11% (n = 35) at 200 and 300 mg, respectively. We excluded four patients from the full analysis set. One of the 17 patients from the 200-mg arm was excluded because his baseline bone lesions were not assessed again during the treatment. Three patients from the 300-mg arm were excluded: two withdrew their informed consent (one intolerable grade 3 AE and another grade 2 AE [cholecystitis, which was not likely related to donafenib]) and 1 patient's target lesions could not be reliably assessed due to a tracheotomy performed for dyspnea. Ultimately, 31 patients were included and efficacy analysis was performed.
Both arms achieved a 100% DCR after 24 weeks of treatment (Table 2). A partial response in both arms (2/16 in the 200-mg arm and 2/15 in the 300-mg arm) occurred in the papillary subtype with metastatic target lesions in the lungs. The median PFS time of the 300-mg arm was longer than that of the 200-mg arm (14.98 months vs. 9.44 months), but was not significantly different (hazard ratio [CI]: 0.678 [0.298–1.547]; p = 0.351) (Fig. 1A and Table 2). Four patients from the 200- and 300-mg arms were censored (confirmation of the last time where no disease progression or death was observed). The best overall changes in the sum of diameters of target lesions from the baseline are shown in Figure 1B and C for the 200- and 300-mg arms, respectively. The time taken for patients to show a partial response for the first time after starting treatment ranged from 1.77 to 5.6 months (Supplementary Table 1). The duration for which stable disease was sustained was a mean of 14.15 and 15.22 months in the 200- and 300-mg arms, respectively (Supplementary Table 2).
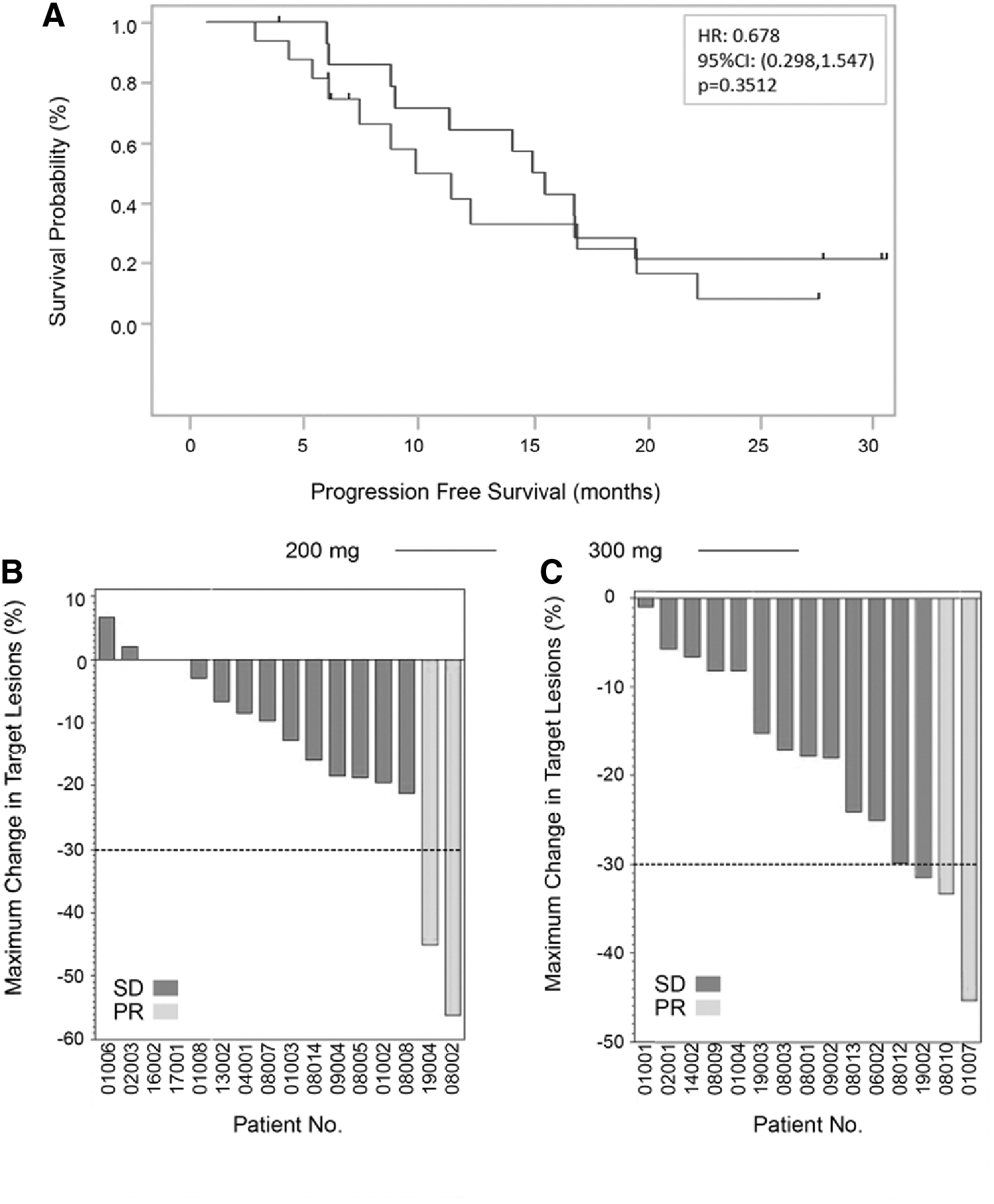
Objective response. (
Tumor Response According to RECIST 1.1
After 24 weeks of treatment.
As of January 31, 2020.
CI, confidence interval; CR, control rate; DCR, disease control rate; HR, hazard ratio; NA, not applicable; ORR, objective response rate; PD, progressive disease; PFS, progression-free survival; PR, partial response; RECIST, Response Evaluation Criteria for Solid Tumors; SD, stable disease.
Target lesion changes according to RECIST 1.1
Individual changes in the TTD (ΔTTD) of target lesions per month for 16 patients in the 200-mg arm and 15 patients in the 300-mg arm after the 24-week treatment period are shown in Figure 2A. Tumor shrinkage tended to occur in more patients in the 300-mg arm (73.3% [11/15]) than in the 200-mg arm (43.8% [7/16]). The average ΔTTD rate per month for the 200-mg arm was lower than that for the 300-mg arm (−0.04 ± 1.55 mm/month vs. −0.52 ± 0.71 mm/month), indicating an ∼10 times higher tumor shrinkage rate in the 300-mg arm (Supplementary Table 3). Consistent with these results, the average tumor shrinkage rate measured as the % change in TTD at the end of the 24-week treatment period from baseline was higher in the 300-mg arm than in the 200-mg arm (300 mg: −13.78% ± 17.01%; 200 mg: −3.90% ± 22.07%) (Supplementary Table 4).
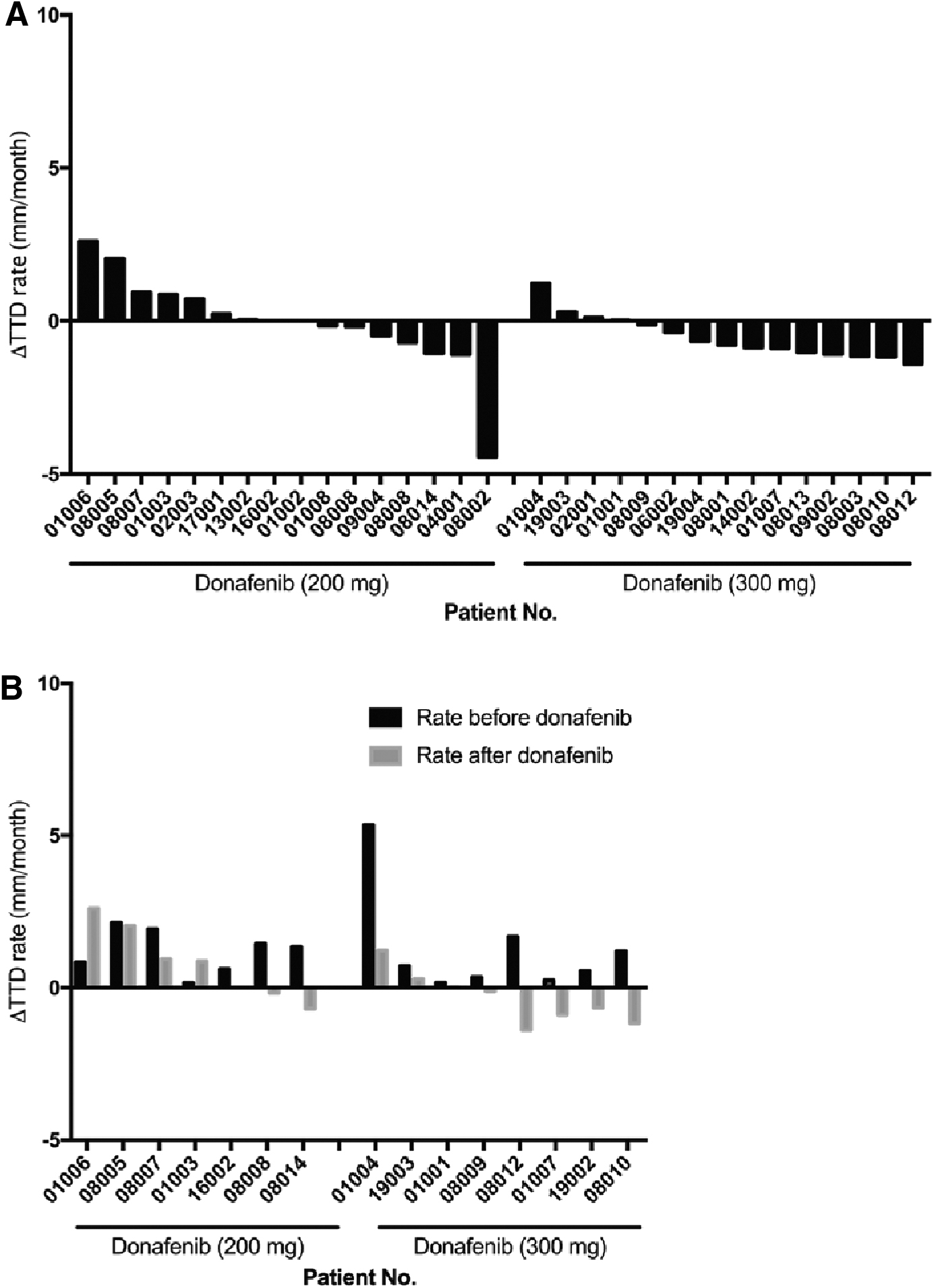
Donafenib's effect on the TTD. (
Treatment-Related Adverse Events (Incidence >20%)
Any AE associated with the investigational drug was defined as definitely related, possibly related, or indeterminable.
AE, adverse event.
A subset of patients (seven patients in the 200-mg arm and eight patients in the 300-mg arm) had positive TTD changes pretreatment, which suggested tumor growth before donafenib administration (Fig. 2B). Treatment with 200 mg of donafenib resulted in tumor shrinkage (negative TTD changes) in 28.6% (2/7) and reduced TTD changes in 42.9% (3/7) of patients, while there was no effect in 28.6% of patients. Meanwhile, treatment with 300 mg of donafenib resulted in tumor shrinkage in 62.5% (5/8) and reduced TTD changes in 37.5% (3/8) of patients. These results suggest that the 300-mg regimen had more superior antitumor activity than the 200-mg regimen.
Thyroglobulin levels in response to donafenib treatment
Of the 31 patients, 22 had thyroglobulin levels that were evaluable (9 from the 200-mg arm and 13 from the 300-mg arm); the rest had values outside of the detection limit or had elevated TgAb that interfered with Tg detection. The biochemical response, evidenced by a decrease in thyroglobulin levels, occurred as early as four weeks after treatment, with up to an 86% and 97% reduction in the 200- and 300-mg arms, respectively (Fig. 3). After 24 weeks of treatment, the thyroglobulin level decreased to below the baseline in all patients on the 300-mg regimen (13/13), while this occurred in less than half (4/9) of the patients on the 200-mg regimen (Fig. 3A, B). Thyroglobulin responders, who were defined as those obtaining ≥25% reduction in thyroglobulin from the baseline when measured on two separate tests eight weeks apart (12), were observed more in the 300-mg arm than in the 200-mg arm (76.92% [10/13] vs. 44% [4/9], p = 0.187).
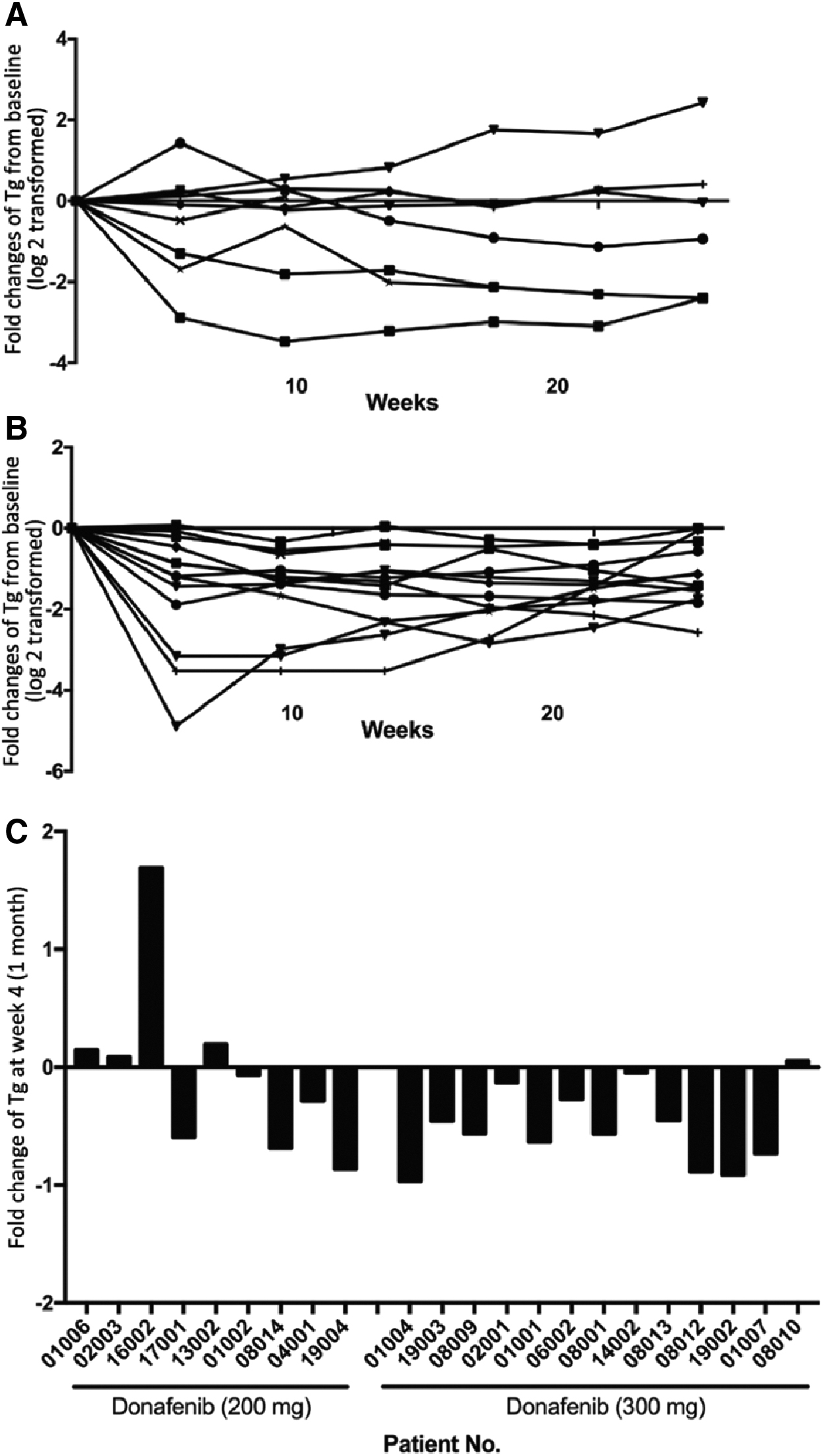
Effect of donafenib on Tg levels after treatment. A subset of patients (9 from the 200-mg arm and 13 from the 300-mg arm) had evaluable Tg measurements. For these patients, the Tg-level fold changes from baseline for each patient were log 2 transformed in (
Safety
All 35 patients were included in the safety analysis: 17 from the 200-mg arm and 18 from the 300-mg arm. Donafenib was generally well tolerated. The most common treatment-related AEs (>20%) are listed in Table 3, most of which were grades 1–2. Common treatment-related AEs (with an occurrence of >20%) in both arms were palmar–plantar erythrodysesthesia (PPE) syndrome (82.86% [29/35]), alopecia (71.43% [25/35]), and increased blood pressure/hypertension (45.71% [16/35]). Nine patients in each arm experienced grade 3 treatment-related AEs. The most common grade 3 treatment-related AEs were PPE and hypertension in both arms, with an occurrence rate of 11.43% (4/35) and 22.86% (8/35), respectively. One patient in each arm presented with serious treatment-related AEs (drug eruptions); both recovered after treatment discontinuation. Overall, 21 treatment-related AEs (8 from the 200-mg arm and 13 from the 300-mg arm) led to dose reductions or permanent discontinuation, and these were mainly PPE syndrome, hypertension, rash, and diarrhea (data not shown). No grade 4 or 5 AE occurred by the end of the 24-week treatment period. No unexpected AEs were reported and those reported were similar to other MKIs in the same category.
Discussion
The aberrant activity of signaling pathways, including MAPK, VEGFR, and PI3K-AKT/mTOR, is well established in thyroid cancer (13,14). These pathways are fundamental in regulating proliferation, angiogenesis, and iodide metabolism. Several MKIs have been developed for RAIR-DTC treatment in recent years, including axitinib, sorafenib, cabozantinib, sunitinib, lenvatinib, and apatinib (10,15 –20).
The global phase III DECISION trial for sorafenib (at 400 mg twice daily) demonstrated improved PFS compared with placebo in RAIR-DTC patients, which led to its approval by the FDA and the European Medicines Agency and subsequently by the China NMPA in 2017. Currently, sorafenib is the only drug available for RAIR-DTC patients in China; however, its use is limited mostly due to its high cost, AEs, or resistance shortly after administration.
Donafenib is a novel oral MKI in which the methyl group of sorafenib is substituted with a trideuteromethyl group. Deuteration enhances metabolic stability and systemic exposure while reducing toxic and reactive metabolites. In the present study, donafenib was well tolerated and demonstrated efficacy in locally advanced or metastatic RAIR-DTC patients. There was no significant difference between the 200- and 300-mg treatment regimens, while the 300-mg regimen appeared to be more promising in efficacy.
Sorafenib was yet to be approved when this study was initiated (in 2017); therefore, a head-to-head comparison could not be conducted. However, of note, almost a quarter of the patients in the DECISION trial were Asian (sorafenib: 22.7% [47/207]); placebo: 24.8% [52/210]) (11). Therefore, China, as one of the participating countries, enrolled 43 Chinese patients, which represented nearly 10% of the patients enrolled, warranting its final approval by the China NMPA in 2017 and thus allowing us to compare this study with the DECISION trial. Upon enrollment of 35 patients, which equals over 80% (35/43) of Chinese subjects recruited in the DECISION trial, both donafenib regimens (200 and 300 mg) reached their primary endpoints with ORRs of 12.5% and 13.33%, respectively, which were similar to the 12.2% for 400 mg of sorafenib from the DECISION trial at merely half to three-quarter dose of sorafenib (11). Considering the promising results at a dose of 300 mg, the rarity of RAIR-DTC worldwide, and the need for alternative TKIs in Chinese patients, a phase III study was initiated with the aim of providing access to donafenib for such patients in China. The median PFS time of patients who received the 200-mg donafenib regimen was 9.44 months, which is comparable with that of patients who received the 400-mg sorafenib, twice daily, regimen (9.44 months vs. 10.8 months); meanwhile, the PFS for patients who received the 300-mg donafenib regimen was longer than that of patients who received sorafenib (14.98 months vs. 10.8 months). The higher efficacy of donafenib at a lower dose can be attributed to increased metabolic stability by the deuterated compound, which prolongs the drug's half-life, but a study comparing the efficacy of the two drugs is necessary to determine if their efficacy is similar or different (21).
Both arms achieved a DCR of 100%. We further examined clinical benefits in terms of the ΔTTD per month, which is indicative of the average tumor growth or shrinkage rate after treatment. The average ΔTTD rate was −0.04 ± 1.55 mm/month for the 200-mg arm and −0.52 ± 0.71 mm/month for the 300-mg arm; this suggests that tumor growth is somewhat prevented by the 200-mg regimen, while the 300-mg regimen led to tumor shrinkage. The higher efficacy of the 300-mg regimen could be attributed to the exposure to higher dose levels. Notably, the 300-mg regimen tended to be associated with longer PFS than the 200-mg regimen (14.98 months vs. 9.44 months), although not statistically significant, suggesting that the dose-dependent efficacy of donafenib remains to be determined with a larger sample size.
Thyroglobulin would be a helpful prognostic marker for RAIR-DTC patients since biochemical changes are more sensitive, more convenient, and more easily monitored than structural changes, especially for DTC after total thyroidectomy and radioiodine therapy. However, its value as a prognostic marker for MKI-treated RAIR-DTC patients remains to be established. In this study, the thyroglobulin levels of all patients who received 300 mg of donafenib fell below baseline levels, while the thyroglobulin levels of patients who received 200 mg of donafenib varied. More thyroglobulin responders were also observed in the 300-mg arm.
There was no significant difference in treatment-related AEs observed between the 200- and 300-mg arms. Donafenib was generally well tolerated, with most of the common treatment-related AEs (>20%) being grades 1–2. A lower proportion of patients withdrew from donafenib than from sorafenib (5.71% vs. 18.8%) treatment due to AEs. Dose modifications to control toxicities were similar between this study and the DECISION trial (occurring in ∼60% of patients); the most common AE for dose adjustments was PPE in both studies. The most common (>20%) AEs observed in the DECISION trial had a higher occurrence rate compared with donafenib treatment (Supplementary Table 5). The donafenib regimens appeared to be less toxic than sorafenib (400 mg twice daily) and had fewer treatment-related AEs overall, even in HBV carriers. The reduced toxicity may be due to reduced reactive metabolites by the trideuteromethyl group.
HBV is prevalent in the Chinese population (∼10% are carriers) (22,23). Of note, HBV carriers were excluded from the DECISION trial and other clinical studies on patients with RAIR-DTC. In this study, ∼10% (4/35) of HBV carriers were enrolled, consistent with HBV prevalence, and donafenib was shown to be tolerated by all patients, including HBV carriers with low viral loads.
In conclusion, donafenib demonstrated promising efficacy as well as favorable safety in patients with RAIR-DTC, including HBV carriers, with the 300-mg regimen appearing to be more clinically beneficial than the 200-mg regimen. Considering its safety, comparable efficacies observed between the two regimens, and local accessibility, donafenib may be a plausible option for RAIR-DTC patients in China. A multicenter, randomized, double-blind, placebo-controlled phase III study at 300 mg in a larger sample is currently ongoing to further explore the efficacy and safety of donafenib in patients with RAIR-DTC (NCT03602495).
Footnotes
Acknowledgments
The authors thank the patients, their caregivers, and the investigators who participated in this study. We thank Dr. D. Sun for her assistance during the submission and revision process.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was sponsored by the Suzhou Zelgen Biopharmaceuticals Co., Ltd.
Supplementary Material
Supplementary Table 1
Supplementary Table 2
Supplementary Table 3
Supplementary Table 4
Supplementary Table 5
