Abstract
Localized brain hypothermia (HYPO) can be achieved by infusing cold saline into the carotid artery of animals and patients. Studies suggest that HYPO improves behavioral and histological outcomes in focal ischemia models. Given that ischemic stroke and intracerebral hemorrhage (ICH) share pathophysiological overlap, we tested whether cold saline infusion is safe and neuroprotective when given during collagenase-induced ICH. Eighty-five adult male Sprague-Dawley rats were used. Experiment 1 investigated brain and body temperature changes associated with a cold saline infusion paradigm that was scaled from patients according to brain weight and blood volume (3 mL/20-minute infusion). Experiment 2 determined whether HYPO aggravated bleeding volume. Experiment 3 investigated if cerebral edema or elemental concentrations were altered by HYPO. We also collected core body temperature and activity data through telemetry. Experiment 4 investigated whether behavioral outcomes (e.g., skilled reaching) and tissue loss were influenced by HYPO. Our HYPO protocol decreased the ipsilateral striatal temperature by ∼0.20°C (p < 0.001), with no other effects. HYPO did not affect hematoma volume (p = 0.64), cerebral edema (p = 0.34), or elemental concentrations (p = 0.49) at 24 hours post-ICH. Although ICH caused persistent behavioral impairments, HYPO did not improve behavioral outcomes (measured by a neurological deficit scale, cylinder, and the staircase test; p > 0.05 for all). Brain tissue loss was not different between groups on day 28 post-ICH (p = 0.90). Although cold saline infusion appears to be safe in the acute post-ICH period, there was no evidence that this therapy improved outcome. However, our treatment protocol was relatively mild and additional interventions might help improve efficacy. Finally, our findings may also speak to the safety of this cooling approach in focal ischemia where hemorrhagic transformation is a risk; future studies on this issue are needed.
Introduction
Stroke is the second leading cause of death worldwide (Feigin et al., 2017). Intracerebral hemorrhage (ICH) accounts for ∼15% of all strokes, and has a 30-day mortality of 35–50% (An et al., 2017). Only 12–40% of ICH survivors become functionally independent. ICH has no clinically proven neuroprotective interventions, and treatment is limited to medical management (Hemphill et al., 2015). Without the development of tissue-saving interventions, ICH will continue to incur great individual and societal costs.
Therapeutic hypothermia (TH) may be a useful neuroprotective intervention for ICH. TH can be induced by cooling the surface of the body, or more selectively (e.g., using cooling helmets; Song and Lyden, 2012). Selective techniques can rapidly achieve target temperatures, while avoiding complications with systemic cooling methods like shivering, and pneumonia (Jain et al., 2018). However, there are feasibility concerns for some selective approaches, including difficulty achieving and maintaining target temperatures, especially for deep brain structures and during prolonged treatments (van der Worp et al., 2019).
Intra-arterial cooling can rapidly and selectively induce cooling following ischemic stroke (Dang et al., 2011). In animals, this method induces HYPO by introducing a catheter into the external carotid artery and infusing saline at the carotid bifurcation or beyond. In patients, the catheter begins in the femoral vein and is advanced into the internal carotid artery before infusion. Importantly, because of the length and limited insulation of the catheter, the infusate warms up due to contact with surrounding blood before it exits the catheter tip (Choi et al., 2010). Models have suggested that 5°C saline can warm to ∼25°C at the catheter tip. Unless the flow rate is increased, or catheter insulation is improved, exit temperature is an important limiting parameter for modeling this HYPO method. So far, most research has focused on using cold saline infusion in ischemic stroke models as a supplemental therapy to mechanical thrombectomy and/or thrombolytic therapy (Chen et al., 2016).
TH provides numerous protective effects (Andresen et al., 2015). For example, studies in ICH suggest that TH reduces behavioral deficits, perihemorrhagic edema, and blood–brain barrier injury (John and Colbourne, 2016; Melmed and Lyden, 2017). Furthermore, there is a large body of evidence for neuroinflammatory injury in ICH (Mracsko and Veltkamp, 2014), and previous studies have suggested that saline infusion may promote neuroprotection by flushing vasculature of inflammatory mediators (Ding et al., 2002).
Although TH can be beneficial, harmful effects can occur (e.g., bleeding complications, in addition to those listed above). TH can affect hemostasis by inhibiting clotting factors and altering platelet function (Alam et al., 2012). We showed that prolonged hypothermia (HYPO) aggravates bleeding in the collagenase ICH model (John et al., 2015). Therefore, depth, duration, and onset of TH must be considered when TH is used in populations with a high risk of hemorrhage. Some researchers have suggested that brief cooling may be advantageous, perhaps especially in the context of hemorrhagic complications (Lyden et al., 2019).
In this study, our aim was to investigate the safety and efficacy of cold saline administration early after bleeding started. We used an established intracarotid saline infusion method for rats (Chen et al., 2015). We hypothesized that while focal cooling may mitigate injury and edema, cooling and use of heparinized saline may induce coagulopathy, thereby worsening outcome. The collagenase model creates a bleed that begins within minutes and evolves over hours. Thus, it is desirable for investigating whether a treatment approach aggravates bleeding.
Methods
Ethics statement
All procedures were approved by and comply with the University of Alberta's Animal Care and Use Committee and Canadian Council on Animal Care guidelines. Experiments were designed with the 3R's of animal ethics in mind and are reported using the RIGOR and ARRIVE guidelines (Balls et al., 1995; Kilkenny et al., 2010; Lapchak et al., 2013).
Group sizes were calculated a priori using G-Power (version 3.1) for each primary brain tissue endpoint to ensure that we had 80% power to detect a 20% increase in hematoma volume, a 35% change in lesion volume, and a 1% decrease in edema at an alpha level of 0.05. Group size calculation parameters (expected mean, SD) were based on previous data from the surgeon (L.L.) using the same collagenase doses and volumes (Liddle et al., 2019). We used randomization and blinding throughout our experimentation. For randomization, group allocations were drawn from an opaque box immediately following insertion of the catheter into the carotid artery (to ensure no bias leading up to that point). For blinding, all endpoints were assessed by an experimenter who was not involved in the surgical preparation and thus, was unaware of treatment group allocation.
Subjects
Eighty-five male Sprague-Dawley rats were used in four experiments and were obtained from Charles River Laboratories (Saint-Constant). Animals were housed individually during Experiments 1 and 2 (for intensive monitoring) and during Experiment 3 (telemetry experiment) due to limitations with radio frequency receivers. In Experiment 4, animals were housed 4 per cage. The vivarium was light (12-hour light/12-hour dark cycle), temperature, and humidity controlled.
Experiment 1 examined the impact of cold saline infusion on brain and body temperature in naive anesthetized rats. A 3 mL/20 minute infusion protocol was based on clinical studies (Chen et al., 2016), with the assumption that no more than 1 L of saline can safely be infused into the patient's middle cerebral artery at a rate of 30 mL/minute, within a reasonable period of time. We scaled our dose to rats based on brain weight (1 L = ∼70% of human brain weight), total blood volume (∼20% blood volume), and body mass (∼1.25% body mass). We selected a 3 mL dose, which is between a ∼1.4 mL (scaled to brain weight) and a 4.4 mL infusion (scaled to body mass). We used nine animals in Experiment 1 (weight: 550 ± 46 g; age: 14 weeks).
Experiment 2 examined whether our HYPO protocol influenced bleeding at 24 hours post-ICH. We used 10 animals per group (weight: 381 ± 56 g; age: 9 weeks).
Experiment 3 examined whether cerebral edema or elemental concentrations were altered by the HYPO protocol at 24 hours post-ICH. In addition, we collected temperature and activity data through telemetry to investigate whether systemic processes were affected by cold saline infusion. We used 16 animals per group (weight: 300 ± 26 g; age: 8 weeks).
Experiment 4 investigated whether cold saline infusion improved functional and/or histological outcomes following a long-term (28 days post-ICH) survival. We used 12 animals per group (weight: 287 ± 24 g; age: 8 weeks).
Surgical procedures
Surgical procedures were conducted aseptically under isoflurane anesthesia (4% induction and 2% maintenance; 60% N2O, balance O2). All surgical regions of interest were shaved, coated with Betadine, and draped before incision. Body temperature was monitored using a rectal probe and maintained near 37°C. Analgesia was achieved using Bupivacaine (0.5 mg S.C.; SteriMax, Inc.). Following core probe implantation, Meloxicam (0.2 mg S.C.; Boehringer Ingelheim Ltd.) provided additional postoperative analgesia.
Brain temperature monitoring
Three days before brain temperature monitoring, 23 G guide cannulae were embedded 0.5 mm anterior and 3.5 mm bilateral to Bregma. We implanted the cannulae before the cold saline infusion to avoid body temperature decreases related to prolonged anesthesia. Brain temperature data were collected using 30 G hypodermic temperature probes (HYP-1; Omega). The tip of the brain temperature probe was ∼6–7 mm below the skull's surface (Fig. 1).
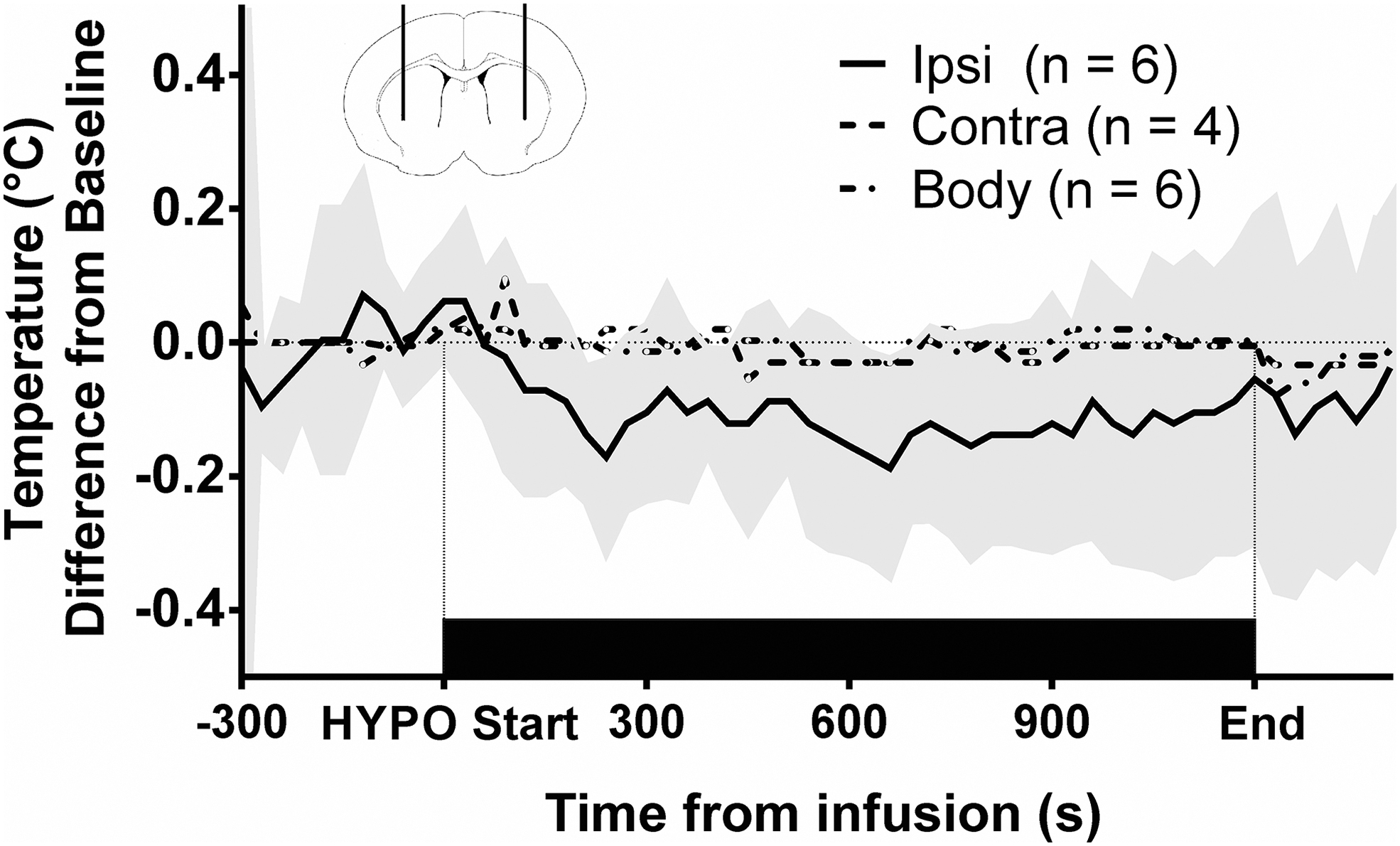
Effect of our cold saline infusion protocol on brain and body temperature in naive rats. A 3 mL/20-minute infusion was selected based on protocols that would be clinically feasible, and then scaled based on relative brain weight and body mass in rats. During the infusion, the greatest brain temperature decrease was ∼0.20°C below baseline. Shaded area indicates the 95% confidence interval for ipsilateral brain temperature. Top in-set panel: schematic diagram of brain temperature probe placement in rats according to the stereotaxic rat atlas (Paxinos and Watson, 2006). Probes were placed 0.5 mm anterior and 3.5 mm bilateral to Bregma, at a depth of ∼6 mm relative to the surface of the skull. (n = 6 in ipsilateral BG and n = 4 in contralateral basal ganglia after exclusions). BG, basal ganglia; BL, baseline.
ICH induction
ICH was induced through striatal collagenase infusion (Type IV-S; Sigma) as described previously (Liddle et al., 2019). A 30 G Hamilton syringe delivered 1 μL of collagenase (0.6 U/mg in Experiment 2 and 0.2 U/mg in Experiment 3 and 4) into a burr hole 0.5 mm anterior and 3.5 mm lateral to Bregma, and 6.5 mm beneath the skull's surface. In Experiments 2 and 3, the left striatum was the site of ICH. In Experiment 4, ICH was induced in the striatum contralateral to the rat's preferred forepaw (assessed during skilled reaching baseline). Collagenase was infused over 5 minutes, and the needle remained in place for an additional 5 minutes before retraction to prevent backflow up the needle tract. Following the removal of the syringe, a #0 screw sealed the burr hole, and wound clips closed the scalp.
Cold saline infusion (HYPO) procedure
A midline incision was made into the neck and the ipsilesional common carotid, internal carotid, and external carotid arteries were isolated (Chen et al., 2015). Following isolation, the common carotid and internal carotid artery were temporarily occluded using S&T vascular clamps (Fine Science Tools), and the external carotid artery was electrocauterized (ME 102; KLS Martin). Following electrocoagulation, a PE-10 catheter was advanced into the external carotid artery and placed near the carotid bifurcation. Vascular clips were removed before the infusion of 3 mL, 21°C heparinized saline (1 U/mL; Sandoz) over 20 minutes. Our saline temperature was selected because many studies infuse 0°C saline directly into the carotid artery, which is not feasible clinically, given current technology and methodology. Control animals received similar treatment, but the infusion was not initiated and the catheter was left in place for 20 minutes. The saline infusion or control procedure began ∼50 ± 10 minutes post-ICH. Following the infusion, the internal and common carotid arteries were briefly occluded to remove the catheter and seal the external carotid artery using sterile suture and electrocoagulation. Sterile suture closed the incision.
Spectrophotometric hemoglobin assay
Hematoma volumes were assessed using a modified spectrophotometric hemoglobin assay (Choudhri et al., 1997; Liddle et al., 2019). Individual differences in vasculature were considered by subtracting the blood volume in the contralesional hemisphere from the ICH hemisphere.
Core probe implant
Core temperature telemetry probes (Model TA10TA-F40, Data Sciences International) were implanted into the abdomen (Wilkinson et al., 2019). Probes were implanted at least 3 days pre-ICH and baseline data were collected between implantation and ICH. Data were collected every 30 seconds and averaged into one-hour intervals.
Tissue wet-dry measurements and inductively coupled plasma mass spectrometry
Twenty-four hours post-ICH, rats were anesthetized and Magnevist® (gadopentetate dimeglumine; 2.5 mL/kg; Bayer) was infused into the femoral vein, and allowed to circulate for 10 minutes before euthanasia and brain extraction. Magnevist® will not pass through a healthy blood–brain barrier (Wardlaw, 2010). Once extracted, brains were placed into a brain matrix and separated into 6 mm blocks (2.5 mm anterior and 3.5 mm posterior to Bregma). The ipsilesional and contralesional basal ganglia and cortex were weighed before and after baking for 24 hours at 100°C. The cerebellum was control tissue. Following wet-dry weight measurement, the dehydrated ipsilesional and contralesional basal ganglia were dissolved in high-purity nitric acid before analysis of cerebral elemental concentrations (e.g., Gd, Fe, Na, and K) using inductively coupled plasma mass spectrometry (ICPMS; Thermo Scientific ICAP-Q quadrupole ICPMS, University of Alberta) (Nadeau et al., 2019).
Staircase task
The staircase test was our a priori primary behavioral endpoint (Montoya et al., 1991). All rats in Experiment 4 were food deprived to 90% of their free-feeding weight to encourage reaching for fortified food pellets during training and testing sessions (21 pellets maximum per forelimb; 45 mg; Bioserv). Rats can be food deprived to a 90% level within 24 hours. Rats were trained for 15 days before ICH (2 trials per day separated by 4 hours). Average performance on the last 3 training days was used as baseline data. Following skilled reaching training, rats were maintained on ad libitum feeding for 2 days before ICH. Rats typically return to free-feeding weights within 1 day of ad libitum feeding following 90% food deprivation. On day 6 and 20 post-ICH, 90% food deprivation was temporarily reestablished to enable skilled reaching testing. Post-ICH testing sessions occurred similar to baseline on days 7, 8, 9, 21, 22, and 23. Post-ICH testing was analyzed as week 1 and 3 post-ICH skilled reaching averages. Skilled reaching performance was gauged by pellets eaten using the following formula: eaten pellets = 21– remaining pellets.
Neurological deficit scale
A neurological deficit scale (NDS) gauged impairment on days 1, 3, and 10 post-ICH. The NDS score was based on hindlimb retraction, forelimb flexion, forepaw grasping, locomotor ability, and circling behavior (MacLellan et al., 2006). Zero indicates no deficits with a maximum impairment score of 14. The circling component of the NDS score was videotaped for analysis of forelimb placement (described in the Cylinder task section).
Cylinder task
This was used to assess spontaneous forelimb exploratory behaviors on days 1, 3, and 10 post-ICH. Animals were placed in a cylinder for 5 minutes and videotaped. Paw placements were categorized as left, right, or both, and a bias score was computed based on baseline paw preference on the staircase task. The bias score was calculated as follows: Bias Score = [(# of touches with dominant limb) ÷ (# of total touches)] * 100%. A bias score of 100% indicates exclusive use of the pre-ICH preferred forelimb, whereas a bias score of 0% represents exclusive unaffected forelimb use (MacLellan et al., 2006).
Histological analysis
On day 28 post-ICH, rats were overdosed with pentobarbital (80 mg/kg; I.P; Bimeda-MTC) and perfused with 0.9% saline and 10% neutral buffered formalin. Tissue was cryosectioned at 40 μm, stained with cresyl violet, and analyzed using ImageJ (version 1.51; NIH). Lesion volume was determined according to our previously published methods (MacLellan et al., 2006) and determined as follows: Tissue loss = Volume of tissue in unaffected hemisphere–volume of tissue in affected hemisphere. Tissue volume in the unaffected hemisphere was calculated as (total hemisphere volume–ventricular volume). Tissue volume in the affected hemisphere was calculated as (total hemisphere volume– ventricular volume–lesion volume).
Statistical analysis
Analyses were performed using R (version 3.5.1; Vienna, Austria). When assumptions of ANOVA were met, data were analyzed using parametric techniques. Otherwise, nonparametric and ordinal data were analyzed using nonparametric techniques. Data are expressed either as mean ± 95% confidence interval or as medians and interquartile ranges with all values shown. A p-value less than 0.05 was considered significant.
Results
Experiment 1
Our saline infusion protocol decreased the temperature of the ipsilateral basal ganglia by a maximum of 0.20°C (p < 0.001; Fig. 1). No changes were observed in the contralateral basal ganglia (p = 0.59). Core body temperature (p = 0.079) was not affected by the infusion. All data from three animals were excluded due to inadequate intraoperative body temperature control, and contralateral striatum temperature data could not be collected in two animals due to cannula blockage.
Experiment 2
Animals that received the saline infusion had a comparable hematoma volume at 24 hours post-ICH (p = 0.64; Fig. 2A).
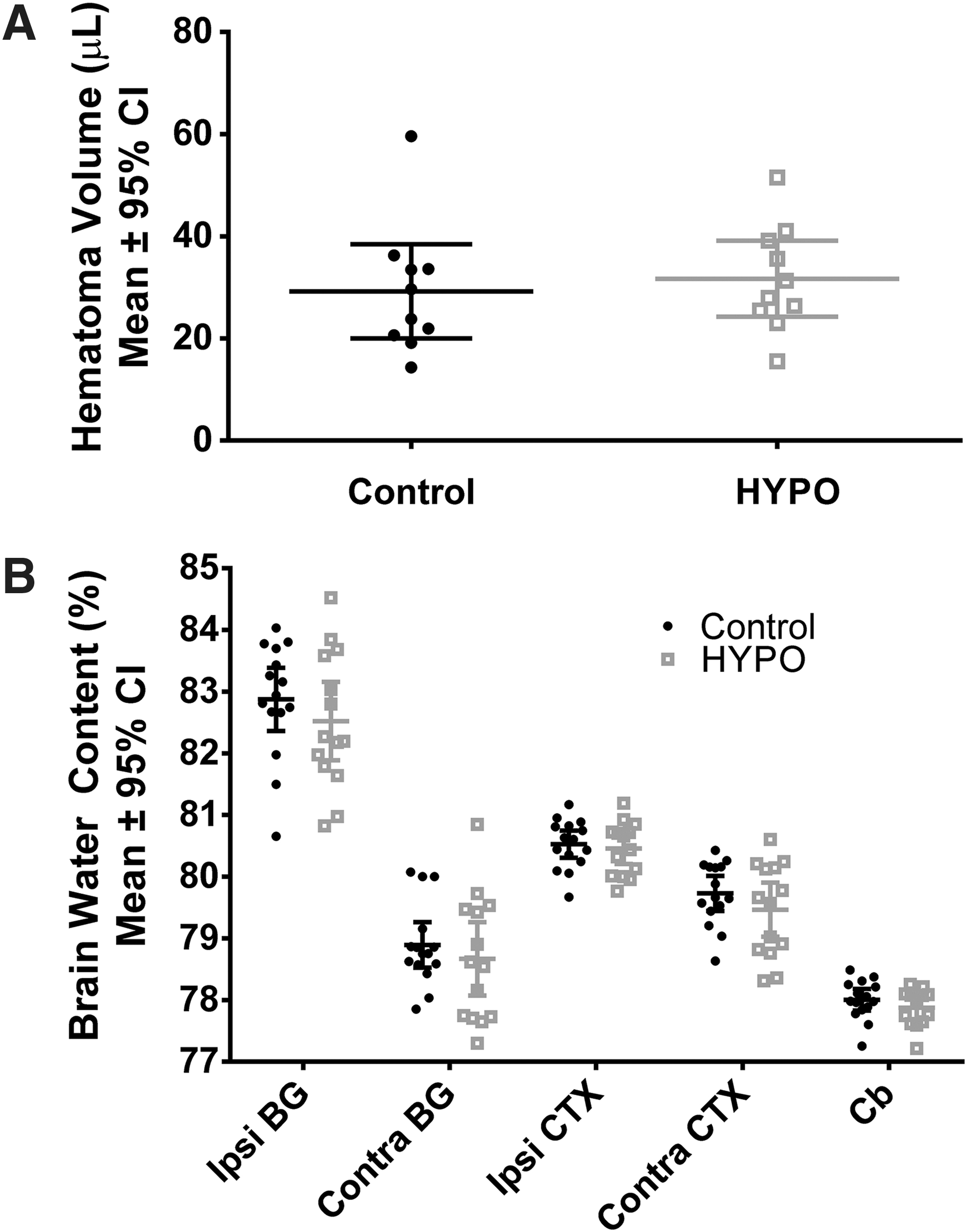
Key tissue endpoints in Experiment 2 and 3.
Experiment 3
Twenty-four hours post-ICH, there was an effect of region of interest, as expected (e.g., greater edema in ipsilesional than contralateral basal ganglia; p < 0.0001). In the ipsilesional basal ganglia, brain water content was not different between groups (p = 0.34; Fig. 2B). The interaction was not significant (p = 0.08).
ICPMS analysis of the ipsilesional basal ganglia showed that elements existed in different concentrations, as expected (p < 0.0001). There was no effect of group (p = 0.49; Fig. 3). There was no interaction effect (p = 0.85).
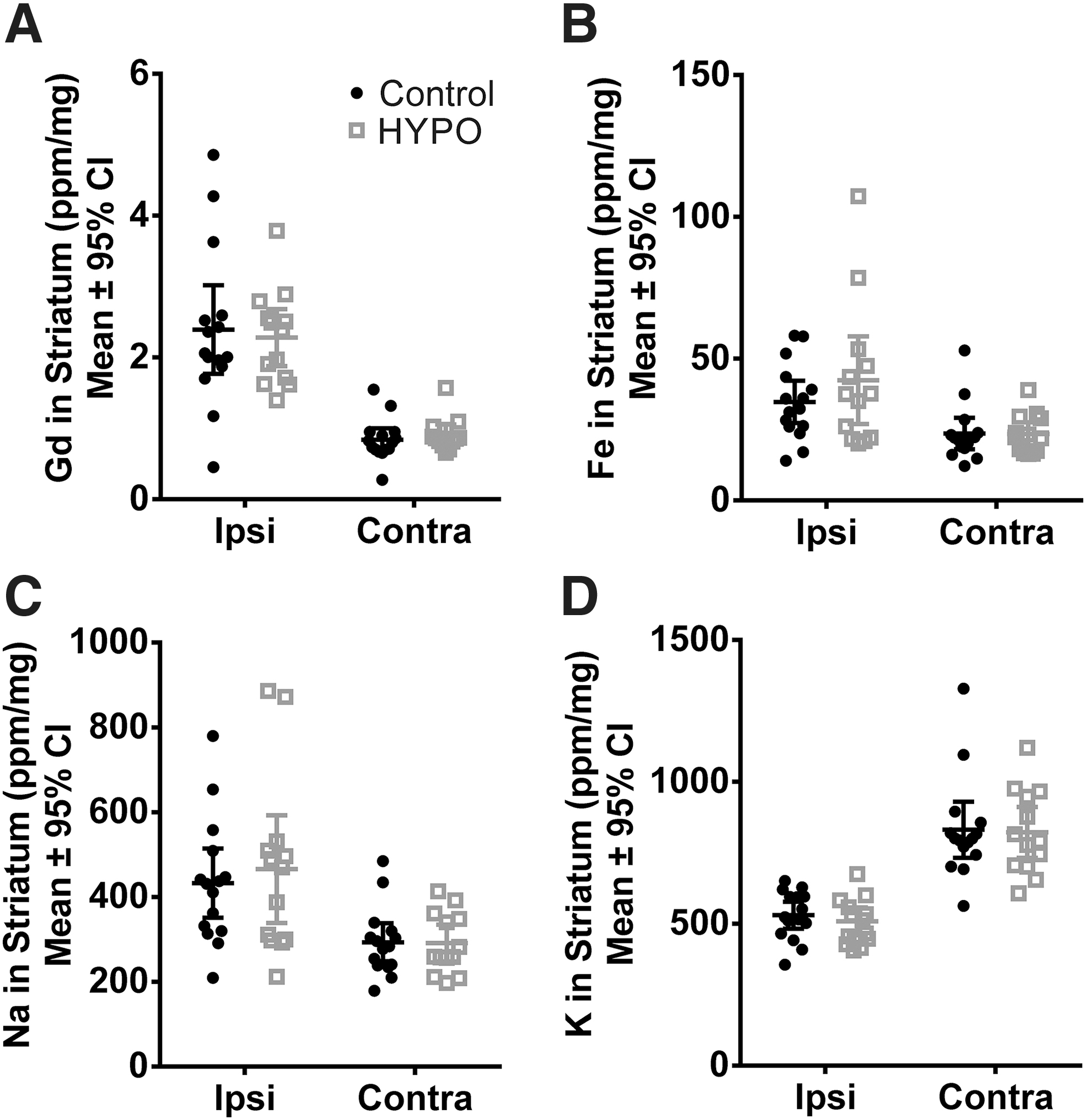
Cerebral elemental concentrations measured 24 hours post-ICH in Experiment 3 using ICPMS to determine
In terms of post-ICH body temperature measured using telemetry, there was a significant effect of time (p < 0.0001; Fig. 4A), but not group (p = 0.32) or the interaction (p = 0.58).
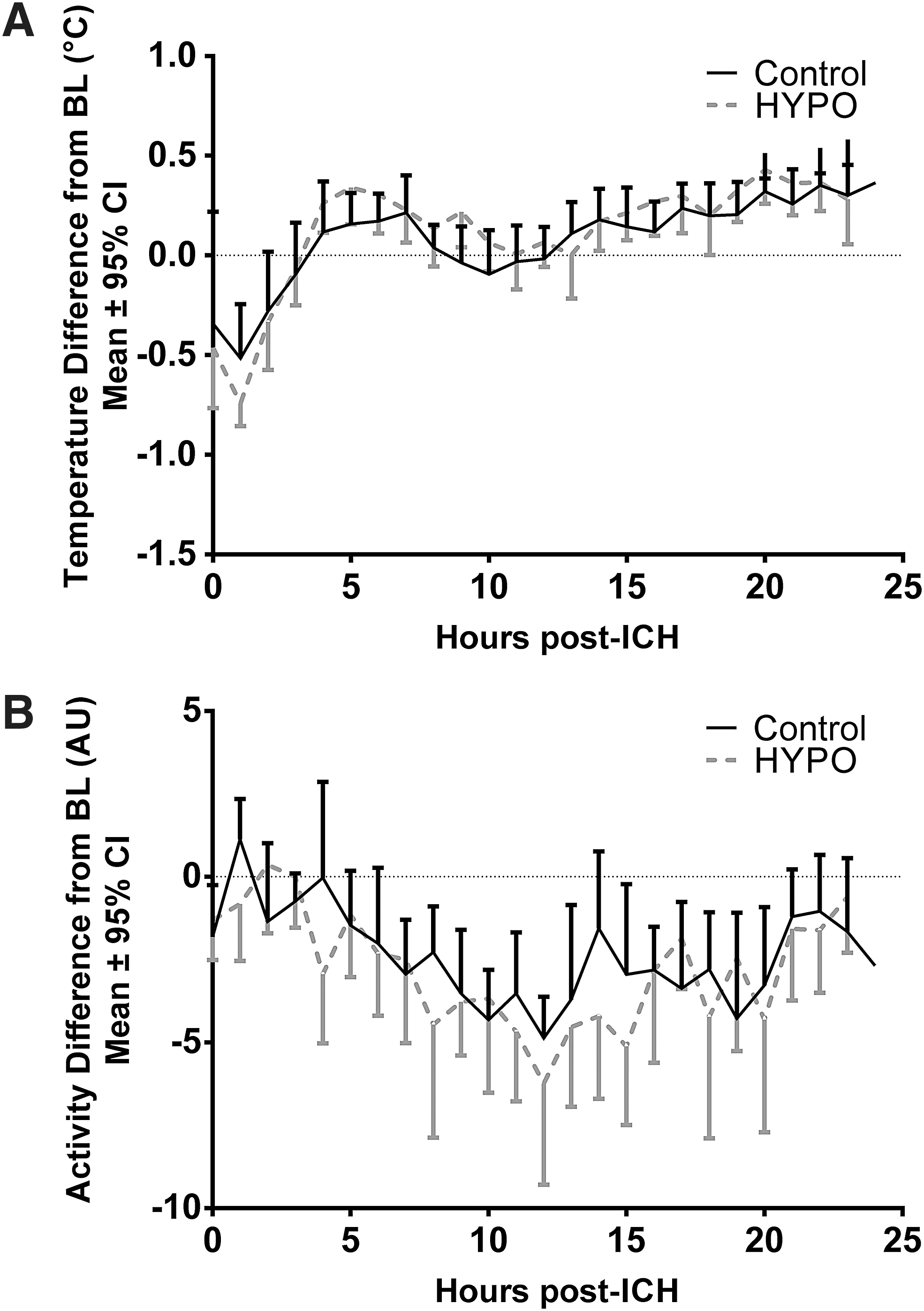
Temperature and activity data collected simultaneously from core telemetry probes (n = 14 per group). (
Similar to the temperature data, our analysis of activity data revealed that there was a significant effect of time (p < 0.0001; Fig. 4B), but group (p = 0.38) and interaction terms (p = 0.47) were not significant.
Two animals died from surgical error before randomization and catheter insertion into the carotid artery (one anesthetic-related death and one failed neck preparation). One control animal died presumably from a large parenchymal hematoma with ventricular extension following the saline infusion, leaving 14 animals in the HYPO group and 15 animals in the control group for brain water content measurements. In addition, one HYPO animal was excluded from ICPMS analysis due to sample contamination during tissue processing (elemental concentrations were 5–15 × higher than average, and >3 SD away from the mean), leaving 15 animals in the control group and 13 animals in the HYPO group for ICPMS. In addition to the deceased animal, telemetry data could not be collected in 1 animal due to probe failure. These exclusions left 14 animals per group.
Experiment 4
One animal in the HYPO group was excluded due to surgical error, and was euthanized 2 days post-ICH. On the staircase task, animals were excluded if they could not eat at least seven pellets with either forelimb at baseline. One animal did not meet this criterion. We excluded any animal that did not touch the cylinder walls at least six times, because limb use estimates become unreliable. Three animals failed to reach this criterion on day 3 post-ICH. There were no other behavioral exclusions.
During our analysis of skilled reaching on the staircase task, we did not find an effect of group (p = 0.64; Fig. 5A). There was an effect of time (p < 0.001), with post hoc tests showing about 55% and 40% worse performance at week 1 and 3, respectively, compared to baseline (p < 0.001 for both). There were no differences between groups at baseline (p = 0.78). The interaction term was not significant (p = 0.33). Our analysis of NDS revealed neurological impairments, (time effect p < 0.0001; Fig. 5B), but the deficits did not differ between groups, (p = 0.20), and the interaction term was not significant (p = 0.44). Our analysis of the cylinder task revealed no effect of group (p = 0.43; Fig. 5C). There was a significant time effect, indicating that post-ICH forelimb use was more asymmetric after ICH (p < 0.001). Post hoc tests showed that animals more frequently relied on their unaffected limb on day 3 and 10 post-ICH compared to baseline (p < 0.001 for both). The interaction term was not significant (p = 0.68) and groups were not different at baseline (p = 1.00).
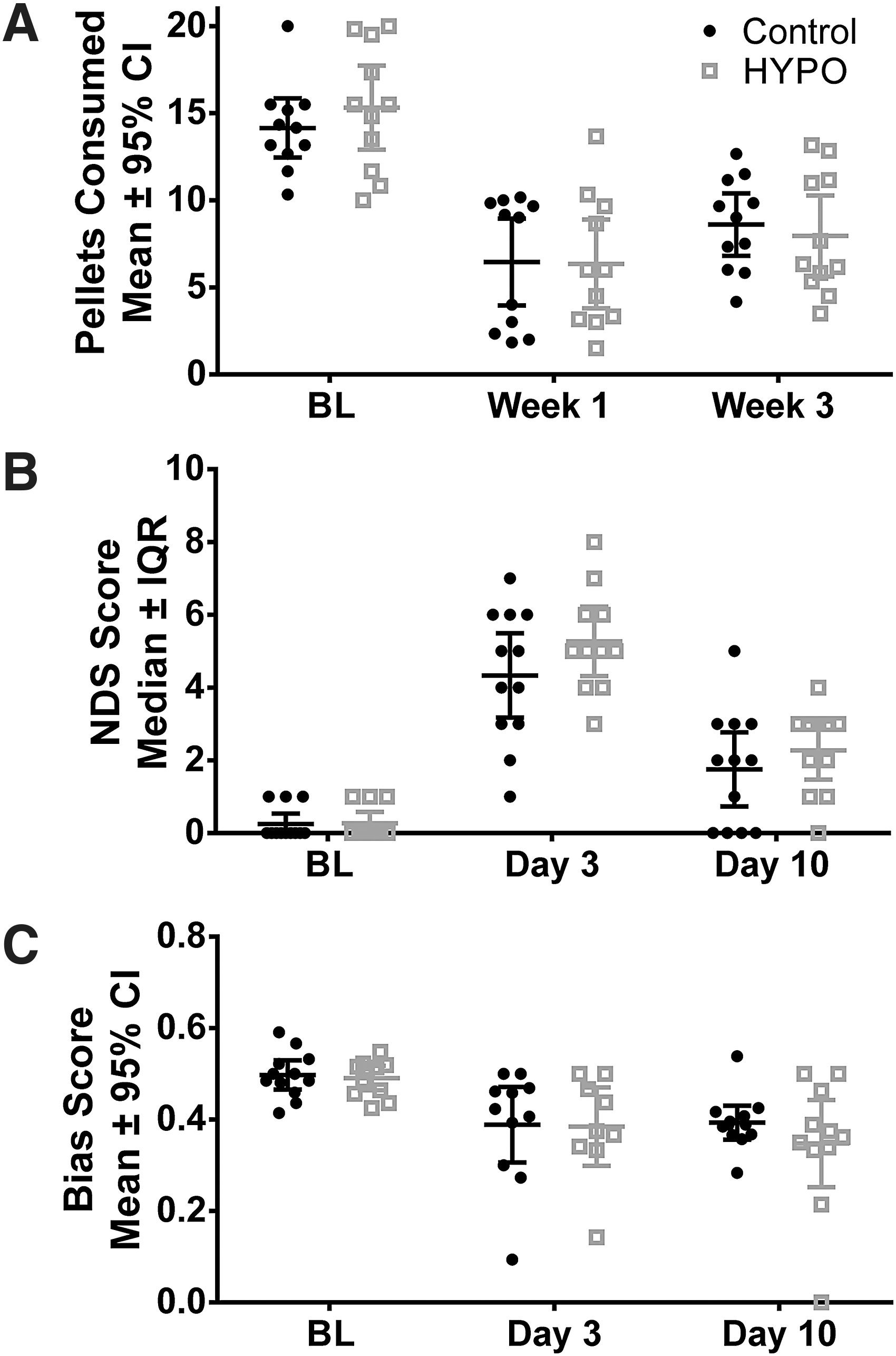
Behavioral results from Experiment 4. Results from all three behavioral tasks showed significant and persistent deficits in both groups, with similar recovery trajectories.
Our lesion volume analysis revealed that tissue loss did not differ between groups (p = 0.90; Fig. 6). Damage was mostly confined to the basal ganglia, and ventriculomegaly occurred in all animals (Fig. 6, top insert).
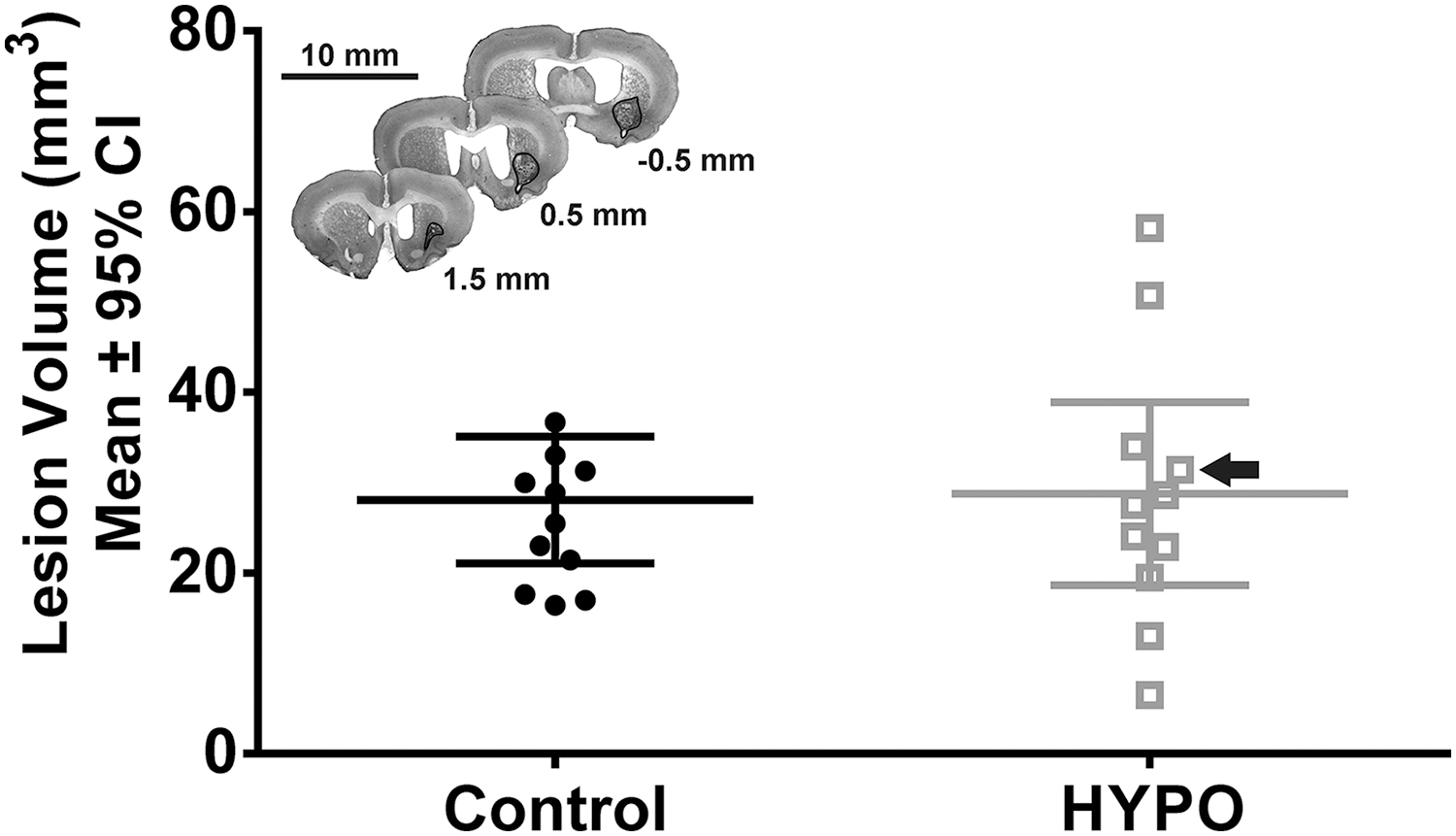
Results of quantification of tissue loss between groups in Experiment 4 (n = 12 in control and n = 11 in HYPO group). Black arrow points to animal used in representative histological image. Top insert: Representative histological photograph of injury at 28 days post-ICH. This animal had a lesion volume of 31.44 mm3. Hemorrhagic injury was largely confined to the striatum; a cavity surrounding the lesion is apparent (outlined with black trace). Ventriculomegaly can also be observed in all sections. Scale bar = 10 mm.
Discussion
Our data suggest that cold saline delivered in the acute period of collagenase-induced ICH appears to be safe, but does not mitigate injury. We found similar hematoma volumes, suggesting that hemorrhaging is not worsened by infusion of cold heparinized saline during the acute post-ICH period. We did not find improved blood–brain barrier integrity measured by brain water content and ICPMS measurements. We also found similar behavioral trajectories and ICH-induced brain damage during our histological analysis. Our failure to find benefit from cold saline infusion in the context of ICH is in stark contrast to the efficacy of saline infusion in focal ischemic stroke models, where studies often show behavioral and histological benefit (Ding et al., 2004; Dang et al., 2011; Duan et al., 2019).
We believe there are two major reasons why we did not find neuroprotection in ICH compared to focal ischemia studies. First, our inability to observe neuroprotection is most likely due to the mild nature of the HYPO treatment. Indeed, most studies employing TH in ICH use paradigms that induce HYPO for hours, with lower target temperatures (e.g., 33–35°C; Melmed and Lyden, 2017). Second, despite many similarities, there are key pathophysiological differences between ICH and ischemia. For example, the infusion of saline may open cerebral collateral vasculature and flush affected vasculature of inflammatory mediators (Ding et al., 2002). Thus, this treatment may be sufficient to promote neuroprotection in ischemic stroke models, but not in ICH. Indeed, the evidence for cerebral ischemia in ICH is controversial and therefore, collateral engagement may not promote neuroprotection (Prabhakaran and Naidech, 2012). Moreover, although inflammation is considered to be a major post-ICH injury process, our treatment is probably insufficient to prevent neuroinflammatory injury owing to its mild nature. Inflammation begins early after ictus, and peaks 2–3 days post-ICH (Wang and Doré, 2007). Therefore, saline infusion may be useful for initial vascular flushing, but probably does not have sufficient hypothermic depth or duration to promote neuroprotection on its own in ICH.
Our findings suggest that there may be difficulty decreasing parenchymal temperature in patients, given current constraints related to infusion rate and inadequate catheter insulation. Perhaps supplemental TH methods may increase the depth and duration of temperature reduction achieved by cold saline infusion. Previous studies have reported temperature drops of 2–5°C using similar methods, but the infusion volumes and rates were relatively large. Moreover, temperature was measured in ischemic tissue, which would be more prone to HYPO because of ischemia and impaired metabolism. Altogether, our findings underscore the challenges in achieving significant poststroke cooling, and point to critical differences in the pathophysiology of ICH and ischemia.
Experiment 1 revealed that our HYPO paradigm (based on clinical realities) does not greatly reduce temperature in our targeted tissue. Our data showed a temperature decrease of only about 0.20°C for the duration of the saline infusion. Without technological improvements or supplemental HYPO methods, the temperature reduction achieved by cold saline infusion is likely inconsequential in terms of providing neuroprotection in ICH.
Our hemoglobin assay data in Experiment 2 and ICPMS Fe data in Experiment 3 suggested that our HYPO protocol does not appear to influence bleeding. This is despite administering the infusion during a period of active hemorrhaging, which begins within minutes in the collagenase model of ICH (MacLellan et al., 2008). This experiment was a necessary step in understanding the safety profile of this method and its use in ICH because hematoma volume is a robust predictor of patient death and disability (Hemphill et al., 2015). Moreover, hematoma expansion predicts poorer functional outcomes and increases mortality risk in ICH patients.
In the context of ischemic stroke, these data are encouraging for those interested in the use of cold saline infusion when hemorrhagic complications are possible, but must be explored further because hemorrhagic transformation predicts greater risk of death and disability for ischemic stroke patients (Jickling et al., 2014). Our study is advantageous in providing a worst-case scenario (active hemorrhage) to clarify whether cold saline infusion may produce worse outcomes. Moreover, the onset, duration, and total hemorrhage volume are well characterized in the collagenase model, whereas the onset, duration, and volume of secondary hemorrhaging in other models may be less clear (Sun et al., 2011; Fagan et al., 2013; Zhang et al., 2014). Finally, although it is a model of primary hemorrhage, the collagenase model shares several overlapping mechanisms with secondary hemorrhage in ischemic stroke [e.g., increased matrix metalloproteinase levels, blood–brain barrier breakdown, extensive edema, and inflammation; (Sun et al., 2011; Zhang et al., 2014)]. Thus, a treatment that impacts bleeding in the collagenase model may have a comparable impact in secondary hemorrhage models, but additional study is needed. In sum, although cold saline infusion appears to be safe in the context of primary hemorrhage, its safety profile is important to establish not only in hemorrhagic stroke (and its models) but also in ischemic stroke (and its models).
In Experiment 3, we showed that edema was not affected by cold saline infusion. We also showed that ion concentrations were not affected. We focused our analysis on Gd to gauge blood–brain barrier integrity and Fe as a secondary (crude) endpoint to gauge hematoma size, and measured Na and K levels to investigate whether key contributors to the membrane potential were improved by the procedure. Unfortunately, chloride concentrations cannot be measured with our ICPMS method. Gd, Na, and K concentrations were similar between groups, suggesting that blood–brain barrier permeability was similar in HYPO animals compared to controls. Fe levels were also similar, suggesting that our results were not confounded by bleeding differences. Our findings suggest that although TH can improve blood–brain barrier integrity in ICH (Melmed and Lyden, 2017), cold saline infusion alone may not provide a high enough “dose” of TH to protect the blood–brain barrier in ICH.
Finally, Experiment 4 assessed whether behavioral and histological outcomes were influenced by cold saline infusion. Across each of our behavioral tasks, we showed that animals in each group had similar baseline functioning, and that there was significant (and persistent) post-ICH behavioral deficits, but no differences between groups. We used a number of tests in both the short and long term to ensure that we had sufficient temporal resolution to detect possible neuroprotective effects. Importantly, we selected the staircase task as our primary behavioral endpoint because it is persistently sensitive to striatal ICH (MacLellan et al., 2006).
After behavioral testing, we assessed lesion volume at a long-term survival period, as recommended in published guidelines for translational neuroprotection research (Lapchak et al., 2013). Long-term tissue loss analysis is a critical final piece to understanding the efficacy of any neuroprotectant because injury occurs for weeks following ICH (MacLellan et al., 2008). The lesion volume analysis showed that our HYPO and control groups had similar brain injury, which may partially explain the similar behavioral deficits observed in this study.
Limitations and future directions
Our study has multiple limitations. First, because we were interested in post-ICH bleeding complications, we used the collagenase model of ICH. This treatment method may have a differential impact on outcomes in the blood infusion model of ICH. Moreover, although saline infusion may engage collateral vasculature and flush out inflammatory mediators, we did not measure these phenomena. However, there were no differences in any of our study endpoints. Therefore, although measuring these phenomena would be helpful, the efficacy of cold saline infusion in ICH is limited in terms of long-term outcome measures.
Also, the treatment was induced relatively early (∼1 hour) following ICH. We performed the saline infusion during the same preparation as ICH to reduce confounds associated with reanesthetizing the animal. Future studies may vary the timing of saline administration following ICH, bearing in mind a clinically realistic paradigm results in a mild dose of TH, regardless of onset timing. Finally, although our results suggest that saline infusion may be safe in the context of primary hemorrhage, our findings may not apply to secondary hemorrhage that is observed in ischemic stroke, and future studies should investigate this further.
Conclusion
Our experiments show that cold saline infusion seems safe when administered during the acute post-ICH period, but more work is needed to establish whether there are circumstances under which it is contraindicated.
Footnotes
Acknowledgment
The authors thank Cassandra Wilkinson for providing consultation regarding ICPMS measurement and analysis, and their useful feedback and support throughout the project.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the Canadian Institutes of Health Research (Grant number 166087). F.C. is supported by a Canada Research Chair award. L.J.L. is supported by a Canadian Institutes of Health Research (CGS-M) award.
