Abstract
Patients presenting with ST elevation myocardial infarction (STEMI) are routinely treated with percutaneous coronary intervention to restore blood flow in the occluded artery to reduce infarct size (IS). However, there is evidence to suggest that the restoration of blood flow can cause further damage to the myocardium through reperfusion injury (RI). Recent research in this area has focused on minimizing damage to the myocardium caused by RI. Therapeutic hypothermia (TH) has been shown to be beneficial in animal models of coronary artery occlusion in reducing IS caused by RI if instituted early in an ischemic myocardium. Data in humans are less convincing to date, although exploratory analyses suggest that there is significant clinical benefit in reducing IS if TH can be administered at the earliest recognition of ischemia in anterior myocardial infarction. The Essex Cardiothoracic Centre is the first UK center to have participated in administering TH in conscious patients presenting with STEMI as part of the COOL-AMI case series study. In this article, we outline our experience of efficiently integrating conscious TH into our primary percutaneous intervention program to achieve 18 minutes of cooling duration before reperfusion, with no significant increase in door-to-balloon times, in the setting of the clinical trial.
Introduction
T
Therapeutic hypothermia (TH) has been shown to have beneficial effects on left ventricular myocardial salvage in animal models of coronary artery occlusion and reperfusion (Dae et al., 2002; Gotberg et al., 2008; Erlinge, 2011). The exact mechanism of this benefit remains unclear, although it has been suggested that at lower core body temperatures, there is significant reduction in reactive hyperemia (Olivecrona et al., 2007) and also complex molecular regulation, including suppression of destructive enzymes, free radicals, protection of the cellular phospholipid bilayer membrane, and reduction of intracellular acidosis, resulting in reduced RI (Dempsey et al., 1987; Chopp et al., 1989). There seems to be a strong relationship between the timing of hypothermic temperature achievement and reduction in IS, with 100% reduction in IS demonstrated if TH is achieved before onset of ischemia, although of course this is not clinically feasible (Dixon et al., 2002; Gotberg et al., 2008; Erlinge et al., 2013). There is no reduction in IS if TH is initiated after reperfusion. However, some report that up to 80% reduction in IS can be achieved if TH can be initiated at the earliest recognition of onset of ischemia and TH achieved before reperfusion (Dixon et al., 2002; Gotberg et al., 2008, Erlinge et al., 2013). Therefore, it can be postulated that an efficient transfer of patient suffering STEMI and initiating TH simultaneously early in the catheter laboratory before reperfusion are likely to offer the most potential therapeutic benefit of this treatment.
Studies of TH in humans presenting with STEMI have so far not demonstrated significant clinical benefit (Dixon et al., 2002; Erlinge et al., 2013; Erlinge et al., 2014). In the latest multicenter, randomized controlled trial (Erlinge et al., 2014), it was shown that TH administered simultaneously at the time of primary percutaneous intervention (PPCI) did not reduce the IS significantly. However, exploratory analysis demonstrated that there was a significant 33% reduction in IS if TH was administered within 4 hours of symptom onset in patients with anterior STEMI, and also a significant reduction in incidence of heart failure was noted in this group. Future studies on TH will focus on early presenting anterior STEMI, using more powerful TH devices enabling a more rapid cooling profile, and to achieve a lower temperature before reperfusion specifically in these larger infarcts. Some investigators are also looking at commencement of TH at an earlier stage, including during ambulance transfer (Testori, et al., 2013). It has been learnt that an important aspect of future potential success in this area is efficiency of the clinical team in administering TH simultaneously with PPCI. The time of coronary intervention following symptom onset is vital in dictating prognosis and therefore any delay following hospital admission should be minimized. In this article, we focus our discussion on our experience of running a TH interventional research trial in the setting of patients presenting with STEMI.
Materials and Methods
The Essex Cardiothoracic Centre (CTC) is a tertiary heart attack center (HAC) serving a population of 1.7 million in the county. Every year the center carries out ∼750 PPCIs. It is the first UK center that received ethical approval to conduct a feasibility study of TH in patients presenting with STEMI, which was sponsored by ZOLL Circulation.
The purpose of the study is as follows:
(1) Investigate the feasibility of administering TH simultaneously with PPCI and before reperfusion. (2) Organize an efficient team to minimize time delays to TH without compromising patient care. (3) Controlling shivering in conscious patients who are actively cooled with an intravenous cooling device.
Recruitment model
Preparation
All 24/7 HACs are well trained and efficient in the assessment and treatment of the STEMI patient. Teamwork in this potentially stressful clinical arena is vital, and currently the roles and responsibilities of all PPCI team members are well described and understood. Implementing a TH service in the setting of the STEMI clinical trial adds further complexity. To ensure efficient enrollment and cooling, it is paramount that these extra tasks are defined and delegated.
The day is started by briefing all members of the cardiac catheter laboratory and research team that if a suitable patient with STEMI is admitted, he/she will be approached to participate in the study. Members of the team are given clear instructions about their roles during various stages of the recruitment. All medications, including buspirone, pethidine, and cold intravenous saline, are checked to ensure adequate supply. The Essex CTC is notified in advance by the ambulance crew when a patient suffering STEMI is identified and boarded for transfer with an expected arrival time. The ZOLL Thermogard (TG) cooling machine is precooled to ensure immediate availability at the optimum temperature. A typical patient flow diagram is illustrated in Figure 1.
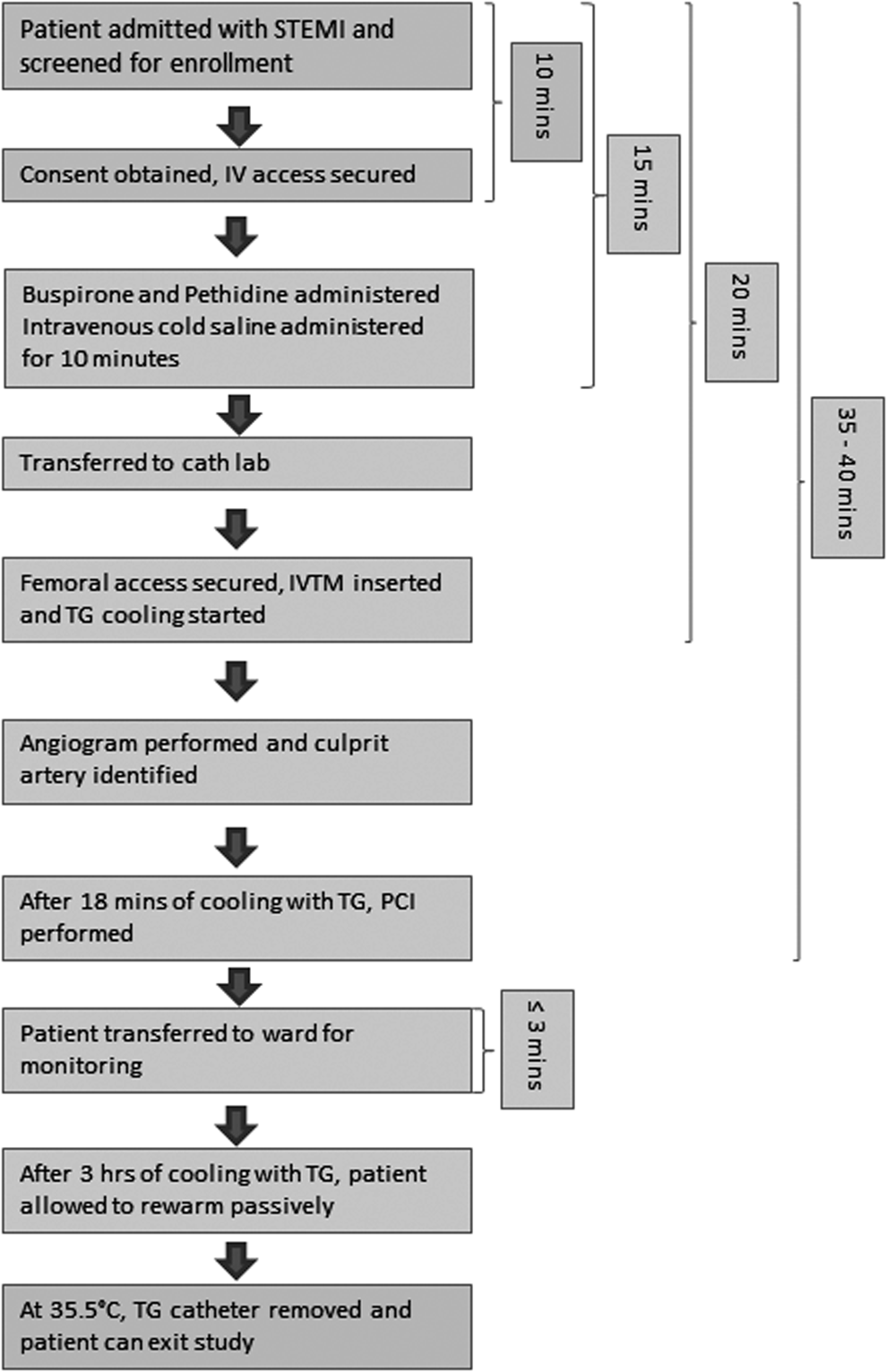
Flow diagram of COOL-AMI EU patient flow and time aims for completing each part of the study protocol. IVTM, ZOLL's Intravascular Temperature Management; PCI, percutaneous intervention; STEMI, ST elevation myocardial infarction; TG, The ZOLL Thermogard.
Stage 1: holding bay/consent
On arrival, the patient is taken to the holding bay outside the catheter laboratory for initial assessment by the study team. If hemodynamically stable, the patient is screened against the inclusion/exclusion criteria of the ZOLL COOL-AMI EU study. An informed consent is obtained if the patient decides to take part in the study and baseline data are collected. Two intravenous access sites are secured, and the patient is then transferred to the catheter laboratory for urgent TH and coronary intervention. We aim for the consenting and IV access steps to be completed within 10 minutes where possible.
Stage 2: catheter laboratory
Once included into the trial, the focus is on delivering the maximum dose of cooling before early reperfusion therapy. A Bair Hugger is already placed on the catheter laboratory table in anticipation for the patient's arrival and it is set at 43°C. Using warming blankets on the skin, while cooling within the vena cava with a cooling catheter, is a recognized strategy for preventing shivering. For successful and rapid reduction in patient core temperature, shivering must be prevented. Once shivering occurs, core temperature will reduce no further and may in fact increase until the shivering is suppressed. The antishiver regimen includes 60 mg Buspirone orally, with the esophageal temperature probe inserted simultaneously as it is easier to pass through the tube when the patient is swallowing, which minimizes the gag reflex. A bolus of pethidine is then administered at a dose of 1 mg/kg, followed by intravenous cold saline to initiate TH. The patient is then wrapped up in blankets to provide surface warming. We aim to have the patient on the catheter laboratory table, consented, antishiver regimen delivered, intravenous iced saline commenced, and patient ready for cooling catheter insertion within 15 minutes from arrival. The layout of the cardiac catheter laboratory during PPCI and simultaneous application of endovascular TH is shown in Figure 2.
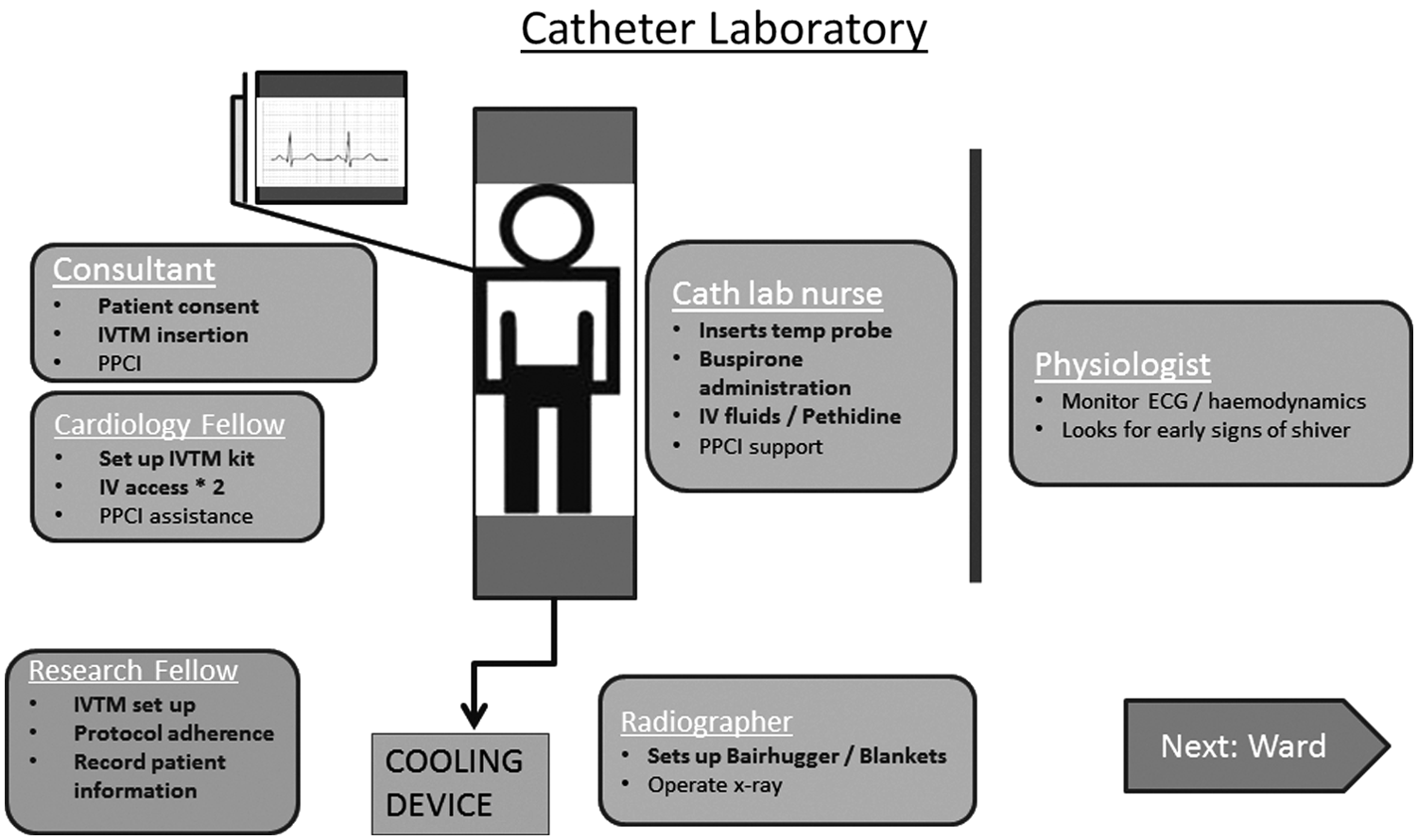
An illustration of the catheter laboratory layout during primary percutaneous intervention and simultaneous administration of endovascular therapeutic hypothermia. PPCI, primary percutaneous intervention.
The volume of cold saline administered is dependent on the suspected area of myocardial infarction. In inferior myocardial infarction, up to 2 L of cold saline is administered compared with 1 L in anterior myocardial infarction. The rationale for this is related to the territory of the myocardium supplied by the coronary arteries. The right coronary artery (inferior STEMI) supplies predominantly the right ventricle and consequently the left ventricle is more susceptible to volume depletion due to reduced right ventricular contractility (Cohen et al., 1995), whereas the left anterior descending artery supplies the bulk of the left ventricle and it is more susceptible to volume overload following a myocardial infarction (Opie et al., 2006).
Following routine sterilization procedures, the consultant cardiologist secures right femoral vein access to pass the cooling catheter (ZOLL Quattro catheter). The optimal catheter position in the inferior vena cava is confirmed under X-ray guidance. The catheter is then connected to the TG cooling device to initiate cooling with a set point temperature of 32°C. The esophageal temperature probe is connected to the TG cooling device, which then displays core body temperature changes. Peripheral tympanic temperatures are also obtained at regular intervals according to the study protocol and this allows checking for any discrepancy in temperature measurement. We aim for the cooling catheter to be inserted and cooling commenced within 20 minutes of hospital arrival.
Arterial access is then obtained preferably through the right radial artery as there is a lower bleeding risk for the procedure and it is easier to manage the radial sheath on the ward and monitor arterial blood gases if required. Baseline blood is taken and sent off to the laboratory for analysis. Coronary angiogram images are obtained and the occluded artery is identified. Following 18 minutes of cooling through the TG cooling device, the consultant cardiologist proceeds to appropriate coronary intervention. We aim for a door-to-balloon (DTB) time in this trial of between 35 and 40 minutes, with 18 minutes of cooling before reperfusion.
According to the study shiver management protocol, a pethidine infusion is set up that can be titrated between 24 and 32 mg/h depending on the level of shivering and respiratory drive and administered 15 minutes after the initial loading dose. The patient is closely monitored during this active cooling phase to ensure hemodynamic stability and adequate shiver management, and end tidal CO2 is monitored through a sensor within the oxygen mask to ensure the patient has adequate ventilation.
Stage 3: transfer from catheter laboratory to the ward
At the end of the coronary intervention, the patient is prepared for transfer to the ward. This phase of the cooling maintenance is vital. The TG cooling device currently needs to be turned off for transfer, and as a result, patients can increase in temperature by about 0.5°C within 5 minutes. We have developed an ice-cold saline protocol where, during transfer, the patient is given a bolus of 250 mL of ice-cold saline to cover the transfer time. We have learnt from our experience that this intervention minimizes any rebound temperature gain during the transfer from the catheter laboratory to the ward. The Essex CTC is well designed for a quick and efficient transfer between the laboratory and the coronary care unit, and the average journey time is 3 minutes or less.
Stage 4: ward
On arrival at the ward, the TG cooling device and the Bair Hugger machine are both switched on immediately to continue the active cooling therapy to a target temperature of 32°C±1°C. The pethidine infusion rate is tailored to the patient's clinical condition. The radial sheath is kept in situ to monitor arterial blood pressures and blood gas on the ward. The right femoral vein site is inspected on regular intervals for any vascular complications. After 3 hours of active cooling, the patient is rewarmed passively. The TG cooling is turned off, but the esophageal temperature probe is still kept in situ until core temperature warms to above 35.5°C, defined as normothermia. A cooling curve of a typical patient enrolled in the study is shown in Figure 3. The femoral catheter is then removed in a sterile manner, and manual pressure is applied for 10–15 minutes. At this point, usual post-PCI care is resumed by the ward team. All steps of research participation are documented in the medical notes and formally handed over to the clinical team for continuation of care.
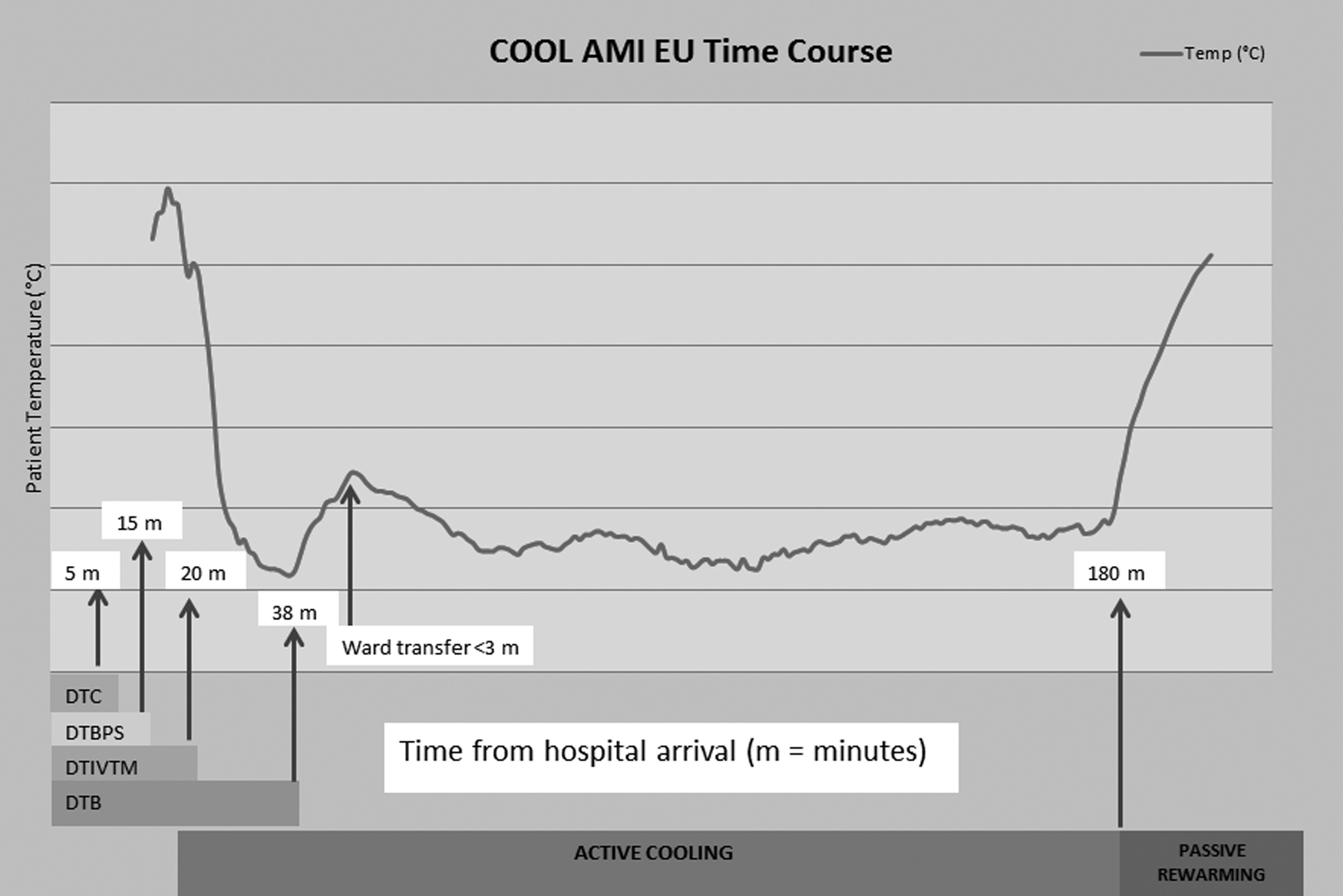
Cooling curve of a typical patient recruited to the COOL-AMI trial; mild temperature rise during ward transfer. DTC, door to consent; DTBPS, door to buspirone, pethidine, ice-cold saline; DTIVTM, door to IVTM; DTB, door-to-balloon.
Discussion
It is feasible to deliver efficient TH within the setting of a clinical trial to patients presenting with STEMI in a large HAC. The average DTB time for the first 11 patients recruited into the trial was 38 minutes with an average of 16.5 minutes of intravenous catheter cooling before reperfusion. This compares with a DTB mean of 37 minutes for all patients presenting with STEMI without administration of TH at the Essex CTC during 2014. This observed 1-minute delay is favorable to the mean 9-minute delay observed in the CHILL MI trial, although their sample size was much larger across multiple sites (Erlinge et al., 2014). More importantly, this minor delay is well within the 90-minute DTB time target that PPCI centers are expected to meet (Canadian Cardiovascular Society et al., 2008).
Evidence from external monitors suggests that our performance improved significantly over time in terms of achieving minimal delay in DTB time and shiver management. This can be attributed to a combination of factors, including strict adherence to protocol, co-ordination of the team, and clearly defined roles. Following the enrollment of each patient, we conduct a debrief session where we discuss different aspects of the patient treatment and areas for improvement. We, as a team, got better in speed and efficiency by practicing our roles meticulously over the duration of the study. There has been no compromise to the safety of the patients and there have been no trial-related serious adverse events.
The prevention of shivering is the key to successfully administering TH in these patients. The antishivering regimen provided by the sponsor is a useful guide to ensuring that safe doses of antishivering medications are administered and in the correct order. Early signs of shivering can be identified from the ECG monitors, where the isoelectric line may demonstrate signs of microshivering. This then allows the clinical team to administer bolus doses of pethidine if clinically indicated. However, the patient's clinical condition and safety take priority over shiver control. Therefore, it is essential to monitor patient's clinical condition before any pethidine bolus is administered. The application of multiple layers of blanket ensures maximum surface warming as the skin is the largest organ for heat exchange, and if the skin perceives warmth, then it appears that shivering is minimized.
The efficient transfer of the patient from the catheter laboratory to the ward is another key element to success as this is a time when active cooling needs to be turned off and therefore allows the potential for rebound hyperthermia. We introduced the administration of 250 mL of cold saline into our transfer regime to minimize temperature rebound. In addition, the CCU is prenotified before transfer to ensure all equipment, including the Bair Hugger and TG cooling device, can be restarted instantly on patient's arrival. The continuous presence of a clinician throughout the course of the patient monitoring during active cooling and passive rewarming adds to the safety of this trial; however, this may be a challenge in a center with limited resources.
With a well-trained team within this feasibility trial, it is possible to deliver 18 minutes of TH to STEMI patients before PPCI reperfusion without a significant prolongation of DTB times. This work paves the way for a larger randomized controlled study where important clinical outcomes, including left ventricular IS and function, patient safety, and mortality, can be measured against standard PPCI care.
Conclusion
In summary, RI is associated with an increase in morbidity and mortality. TH has shown promise particularly in patients presenting early with anterior STEMI. The delivery of TH in the conscious patient is challenging, and previous trials have been criticized by delaying reperfusion to allow for delivery of TH, the benefits of which may be canceled out by the adverse effects of lengthening the symptom-to-reperfusion time. This review outlines our experience at a busy HAC in delivery of TH in conscious STEMI without delaying reperfusion, albeit in the setting of a clinical trial. We hope that this review will help other centers deliver similar results, thus giving TH the best chance of succeeding in reducing the adverse effects of RI in STEMI.
Footnotes
Acknowledgments
The study team would like to acknowledge all members of the cardiac catheter laboratory at the Essex CTC, who kindly accommodated the TH intervention in this busy tertiary unit. Special gratitude is conveyed to the medical team on the ward, who went beyond their usual routine to help look after these patients and ensured safety and smooth running of the trial procedures; the research and development office that provided logistic support, including ethics application support, stationery, and facilitated data monitoring at regular intervals; and last but not least, the sponsor, ZOLL Circulation, for providing the study guidelines, equipment, and data monitoring.
Author Disclosure Statement
No competing financial interests exist.
