Abstract
Resuscitation with large volumes of crystalloids during traumatic hemorrhagic shock might increase the mortality by inducing rebleeding. However, few studies have addressed this problem during hypothermic conditions. Sixty-eight Sprague-Dawley rats were exposed to a standardized femoral artery injury and resuscitated with low (LRe), medium (MRe), or high (HRe) intensity using lactated Ringer's solution after being cooled to 30°C. An additional MRe group was also given desmopressin since this drug might reverse hypothermic-induced impairment of the primary hemostasis. The rats were rewarmed after 90 minutes and observed for 3 hours. The incidence, on-set time, duration, and volume of bleedings and hemodynamic changes were recorded. Rebleedings occurred in 60% of all animals and were more voluminous in the HRe group than in the LRe group (p=0.01). The total rebleeding volume per animal increased with the rate of fluid administration (r=0.50, p=0.01) and the duration of each rebleeding episode was longer in the HRe group than in the LRe group (p<0.001). However, the mortality tended to be higher in the LRe group (LRe=6/15, MRe=1/15, HRe=2/15, p=0.07). Desmopressin did not change the bled volume or the mortality. Overall, the mortality increased if rebleeding occurred (10/35 rebleeders died vs. 1/25 nonrebleeders, p=0.015). Liberal fluid administration increased the rebleeding volume while a trend toward higher mortality was seen with the restrictive fluid program. Desmopressin had no effect on the studied parameters.
Introduction
Among the drugs tested as adjuncts to decrease the bleeding, recombinant-activated Factor VIIa (Ryan et al., 2006; Dutton and Conti, 2009) and tranexamic acid (Roberts et al., 2011) shows promising results. Studies in vitro indicate that desmopressin reverse the HT-induced impairment of the primary hemostasis (Ying et al., 2008). In the current study we wanted to investigate whether these effects can be reproduced in an in vivo model.
Resuscitation studies in animals show that large volumes of crystalloid fluid increase the hemorrhage and the mortality (Bickell et al., 1991; Riddez et al., 1998). Only a few animal studies address the problem during HT (Kim et al., 1997; Hachimi-Idrissi et al., 2004) and, therefore, no guidelines exist for patients with both HS and HT that may coincide if rewarming is not possible to perform.
In a new uncontrolled bleeding model in the rat (Heinius et al., 2011) we have shown that HT increases rebleeding. In the present study, the same model was used to compare the additional effect of three different intravenous (i.v.) infusion regimes of lactated Ringer's solution (LR) on rebleeding. The effect of desmopressin was tested in a group of rats receiving the intermediate amount of fluid.
The mimicked clinical situation is accidental HT outside hospital in a cold climate that develops after a hemorrhagic insult, such as a car accident in wintertime. The hypothesis was that animals subjected to HT will rebleed even when a small volume of fluid is infused.
Methods
The study protocol was approved by Umeå djurförsöksetiska nämnd (Umeå Animal Care and Use Committee 040130 A7-04, 061129 A125-06) (Fig. 1). Sixty-eight male albino Sprague-Dawley rats weighing of 309±35 g (mean
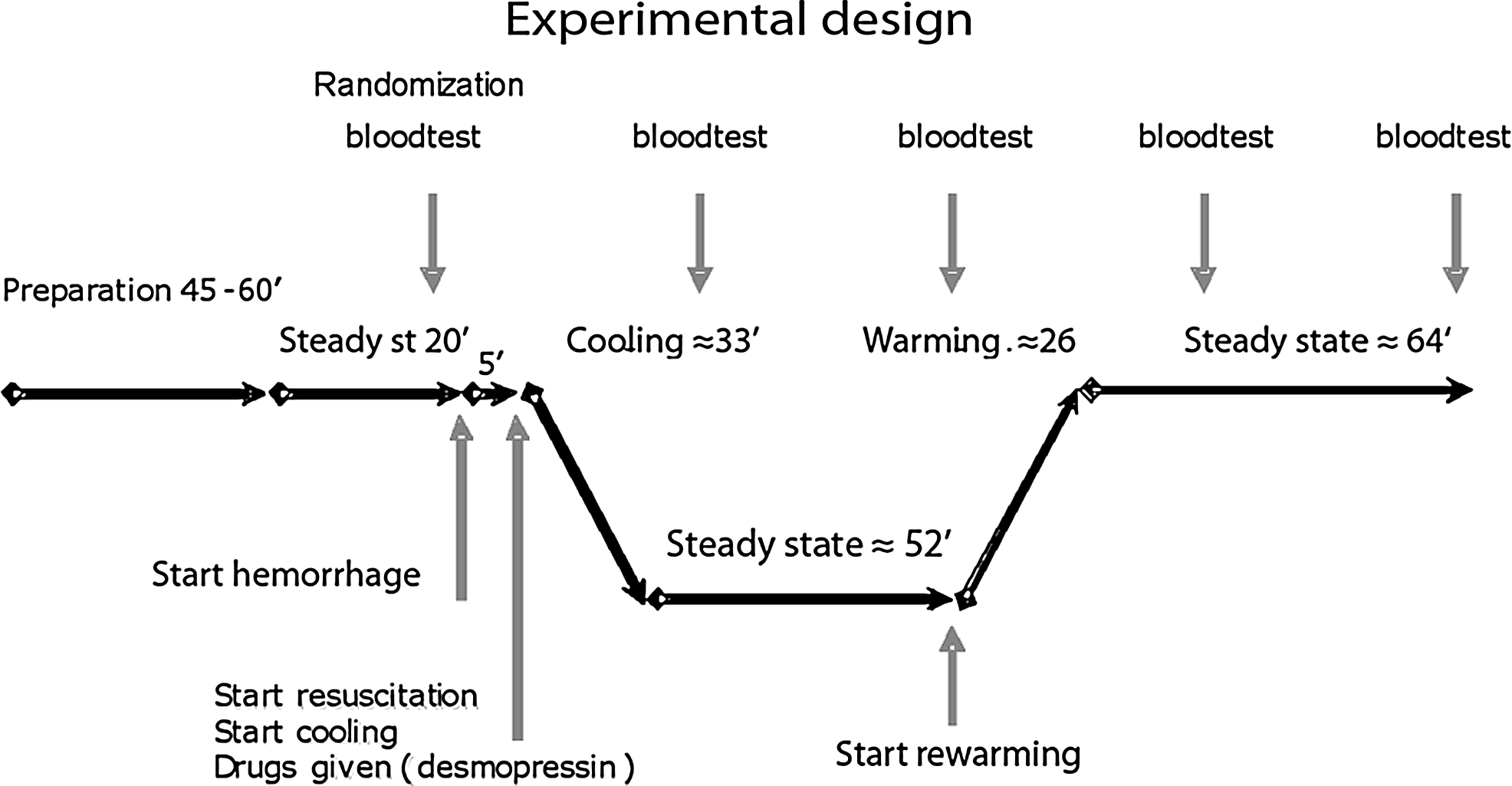
The experimental design.
Surgical preparation
Rats were given free access to food and water before the experiment. Anesthesia was induced by an intraperitoneal injection of 75 mg/kg of ketamine (Ketalar®; Pfizer, Strängnäs, Sweden) followed by 0.1 mg/kg glykopyrron (Robinul®; Meda AB, Solna, Sweden) and 5 mg/kg midazolam (Dormicum® Roche, Basel, Switzerland).
Animals were placed in the supine position and allowed to breathe spontaneously. Oxygen (<0.2 L/min) was administered through an open catheter placed 2 cm from the animal's nose. The oxygen flow was titrated so that no rise on blood pressure occurred (Bitterman et al., 1991). A heating pad, connected to a temperature controller with a rectal temperature (Tr) probe (CMA 150; Carnegie Medicin AB, Stockholm, Sweden), was used to maintain the body temperature at 37.5°C±0.5°C.
Via an incision on the neck, an MRE 040 catheter (Agn Tho's AB, Stockholm, Sweden) was placed ∼1 cm into the left carotid artery to continuously record arterial blood pressure and heart rate (HR). Second, a polyethylene catheter (PE 50) was inserted into the left jugular vein. The catheter was connected to a pump that infused ketamine at a rate of 50 mg/kg/h, and rate could be titrated to prevent movement on paw pinch. Third, a PE 50 catheter was advanced into the inferior vena cava via the left femoral vein and connected to an infusion pump that administered a maintenance dose of 1 mL/h of LR. Both infusions started immediately after the catheters had been inserted. Finally, the femoral artery in the inguinal region was carefully exposed within a length of 1 cm in a standardized fashion. The incision site on the neck was closed with continuous suture, whereas the incision site in the groin was left open. Before each incision the skin and subcutis had been anesthetized by 0.3–0.5 mL of bupivacaine (Marcain®; AstraZeneca, Södertälje, Sweden).
After the surgical preparation, an additional subcutaneous dose of midazolam (2.5 mg/kg) and glykopyrron (0.1 mg/kg) was given, whereafter all animals were cleaned from mucus in the lower hypopharynx, using a thin suction catheter. A period of 20 minutes was allowed for the animals to reach hemodynamic steady state. Baseline data on mean arterial pressure (MAP), HR, Tr, respiratory rate, and blood chemistry variables were then collected. The glycopyrron injection (0.1 mg/kg) and the cleaning of hypopharynx were repeated after 105 minutes.
Randomization
Five minutes after the baseline measurements had been performed the rats were randomized into five groups. Four of them comprised 15 animals each while a sham group contained 8 animals. The fluid resuscitation was designed to set a minimum infusion rate of LR that was to be increased if a proportionality factor multiplied by the continuously measured blood loss indicated that a higher infusion rate should be used. When the rebleedings had subsided all animals were given LR at a rate of 1 mL/kg for the remaining observation period.
1. Low intensity resuscitation (LRe); 1 mL/h of LR i.v. was given continuously during the entire observation period. 2. Medium intensity resuscitation (MRe); the minimum infusion rate of LR was 8 mL/h and the maximum rate corresponded to twice the blood loss. 3. High intensity resuscitation (HRe); the minimum infusion rate was 15 mL/h and the maximum rate corresponded to 4 times the blood loss. 4. Medium intensity resuscitation with desmopressin (MRe + D). This group received the MRe fluid program, but also desmopressin 3 μg/kg i.v. (4 μg/mL, Octostim®; Ferring AB, Malmö, Sweden) as a single dose 5 minutes after exanguination. The animals in groups 1–3 were given 0.5 mL of LR i.v. as placebo at the corresponding point in time. 5. The sham group was treated in the same way as the LRe group, but without exsanguination.
HS, HT, and resuscitation
HS was induced directly after the randomization (time zero) by puncturing the left femoral artery with a 0.5 mm Æ surgical needle (Fig. 1). A gauze sponge with known weight was gently placed over the artery and replaced every 15th second during the first minute, and then every 30th second until the bleeding stopped. The sponges were immediately weighed on an electronic scale (Mettler Toledo PM6100; AEA Investors, Inc., New York, NY).
The resuscitation started at 5 minutes according to the randomization. At the same time all the animals were exposed to surface cooling by removing the heating pad, placing a gauze bandage over their bodies, and moistening them with water at regular intervals. An electric fan blew room-tempered air over the rats as a means of reducing their body temperature by causing evaporative heat loss. Cooling was stopped when Tr reached 30.3°C and was then maintained at 30.0°C±0.5°C.
At 90 minutes, rewarming was initiated by returning the heating pad and by using a heat lamp to raise the air temperature to 41°C±2°C close to the rat. When Tr reached 37.5°C±0.5°C, the heat lamp was turned off, and Tr was maintained at the same level throughout the remainder of the experiment. All animals were followed for 3 hours after which survivors were then sacrificed.
Rebleeding events and blood chemistry
Rebleeding episodes were treated in the same way as the initial bleeding. These events were documented with regard to start/stop time, start/stop MAP, volume of blood lost, and Tr at the onset of the rebleeding.
Arterial blood samples (0.3 mL each) were drawn from the carotid artery to monitor Hgb (Hb analyzer; HemoCue, Ängelholm, Sweden), pH, pCO2, pO2, SaO2, base excess, hematocrit, and the serum concentrations of potassium and lactate (GEM premier 3000; Instrumentation Laboratory, Lexinton, KY) at baseline and at 45, 90, 135, and 180 minutes. These values were determined at 37°C without correction for Tr.
NaCl 0.9% was given as a 1:1 replacement fluid for sampled blood and for intermittent flushing of catheters. HR, MAP, and Tr were recorded at 5 minutes and thereafter every 10 minutes during the observation period.
Calculations and statistics
Blood density was estimated to 1.05 mg/mL and, hence, the volume of all blood losses was calculated using the formula 1 g=0.952 mL of blood. The blood volume of the animals at baseline was estimated to be 6.8 mL/100 g body weight (Probst et al., 2006).
Data are expressed as the mean and SD, or as median and 25th–75th interquartile range when appropriate. Categorical data were analyzed using the χ2 test. Life table analysis was used to compare the survival rate. When comparing three or more groups, Kruskal–Wallis test was used to analyze differences between variables having a skewed distribution. One-way ANOVA followed by the Scheffe's test was used for data with normal distribution. When comparing two groups (such as between rebleeders and nonrebleeders) Mann–Whitney U test was used for skewed data analyzes and independent sample t-test for normal distributed data. The linear correlation between parameters was studied by simple regression (where r=correlation coefficient). In all tests, p<0.05 were considered statistically significant. All analyses were performed in SPSS 17.0 software.
Results
There were no significant differences in baseline data between the four study groups (Table 1). All animals in the sham group survived the observation period; they did not differ in baseline data from the study groups (data not shown).
Values expressed as mean±SD or as median and interquartile range when appropriate.
Significant difference between LRe and HRe.
Significant difference between MRe and HRe.
Significant difference between LRe and MRe.
LRe, low intensity resuscitation; MRe, medium intensity resuscitation; HRe, high intensity resuscitation; NS, nonsignificant; BE, base excess.
The initial bleeding, caused by puncturing the femoral artery, was quite similar between groups with regard to bled volume (24%±4% of estimated blood volume), duration of bleeding (2:28±0:29 minutes), and MAP at the beginning (101±9 mmHg) and the end (35±8 mmHg) of the hemorrhage.
Rebleeding
Approximately 60% of the animals had one or more rebleeding event (Fig. 2). The MRe group had fewer rebleeders than the other groups, but this difference did not reach statistical significance; the data were LRe=10/15 (67%), MRe=6/15 (40%), HRe=11/15 (73%; p=0.14).
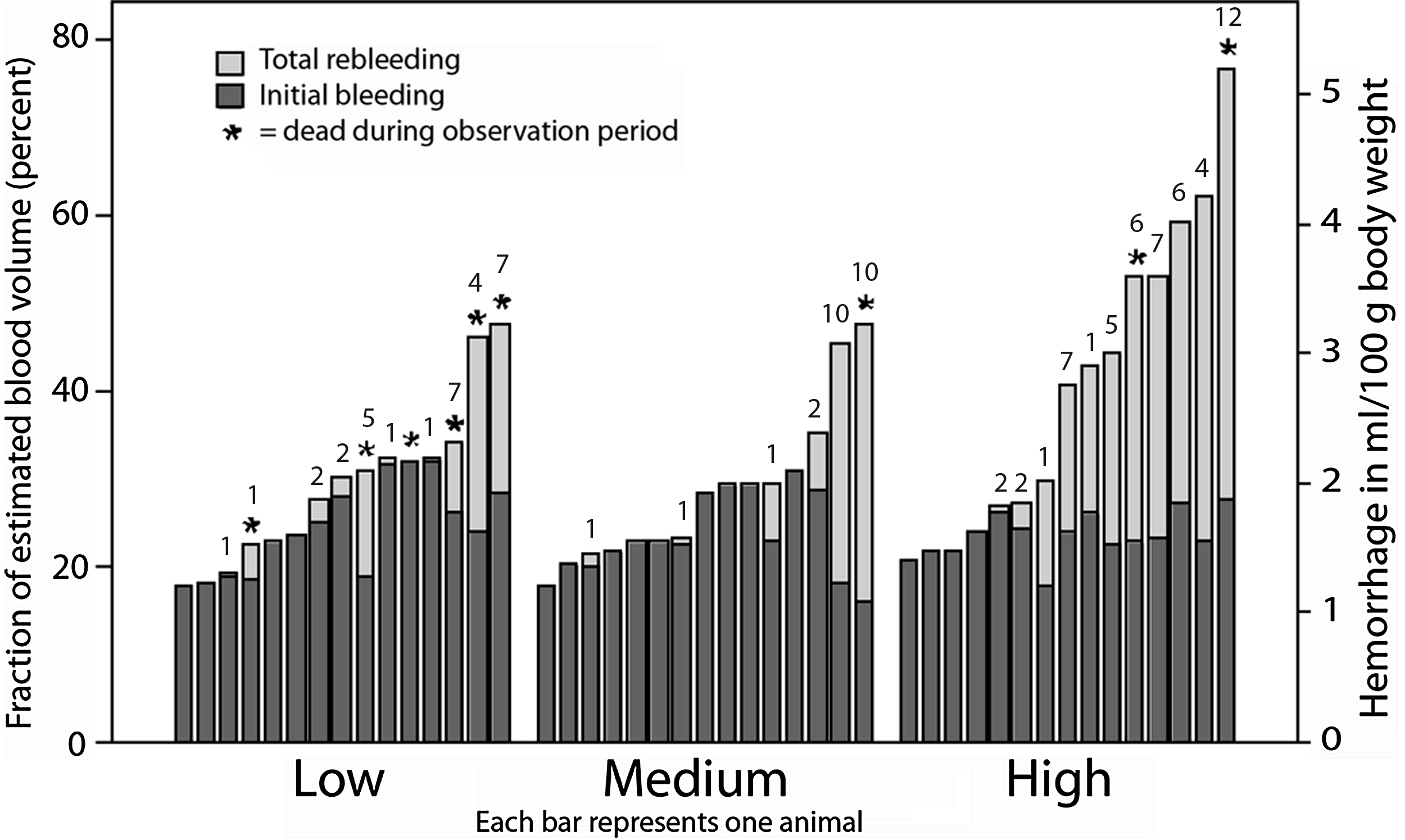
Total bleeding volume depending on the intensity of the fluid resuscitation. Black bars represents the initial bleeding and grey bars the total rebleeding volume. The number of rebleeding events is given on top of each bar.
The total rebleeding volume was larger in the HRe group (median 17% of the blood volume) than in the MRe (0%) and LRe (1%) groups (p=0.046).
Analyzing the rebleeders separately, the total rebleeding volume was larger in the HRe group than in the LRe group (p=0.01) but the difference between the HRe and MRe groups was not statistically significant (p=0.15; Table 2).
Values expressed as median and interquartile range.
Significant difference between LRe and HRe.
MAP, mean arterial pressure.
There was a statistically significant linear correlation between the total rebleeding volume per animal and the rate of the fluid administration (r=0.50, p=0.01).
The duration of the bleeding for each rebleeding episode was longer in the HRe than in the LRe group (p<0.001; Table 2).
In contrast, the number of rebleedings per animal and the bleeding rate did not differ between groups (p=0.47 and 0.45, respectively).
Hemodynamics
The initial bleeding always caused a sudden drop in MAP.
For the animals without rebleeding, MAP rose to above baseline at 30 minutes when the HT had been achieved (Fig. 3, top). During steady state HT (40–90 minutes) the HRe group had a significantly higher MAP than both the MRe and LRe group. These differences remained during post-HT (130–180 minutes).
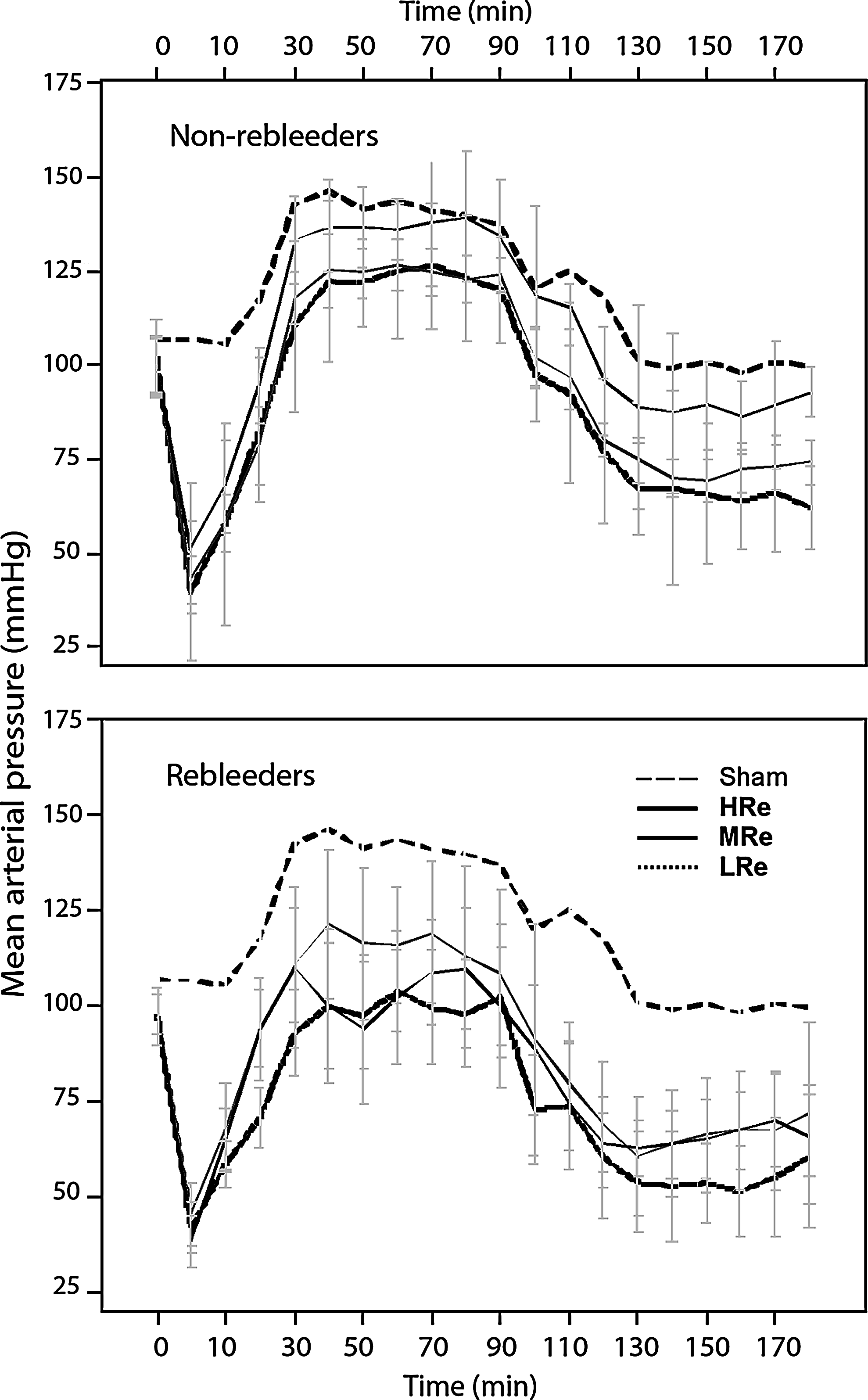
Mean arterial pressure (MAP) among nonrebleeding (top) and rebleeding animals (bottom). Error bars represent 95% confidence interval (CI).
In the animals with rebleeding, the LRe group had a significantly lower MAP than both the MRe and HRe groups during post-HT (Fig. 3, bottom).
HR did not differ between study groups during the study period.
Blood chemistry
The Hgb decreased more in the HRe group compared with the MRe and LRe groups at 45 minutes (end of cooling), 90 minutes (start of rewarming), 135 minutes (post-HT), and 180 minutes (end of study period). The decrease at 180 minutes amounted to 2.0, 3.1, and 5.1 g/dL in the LRe, MRe, and HRe groups, respectively (p=0.001; Table 1).
From 135 minutes and onward, the serum lactate and potassium concentrations were significantly higher and the base excess was significantly lower in the LRe group compared with the MRe and HRe groups (Table 1).
Survival
The highest survival rate (93%) was observed in the MRe group and the lowest survival rate (60%) in the LRe group. When comparing all three groups simultaneously the difference did not quite reach statistical significance (p=0.07; Fig. 4, top).
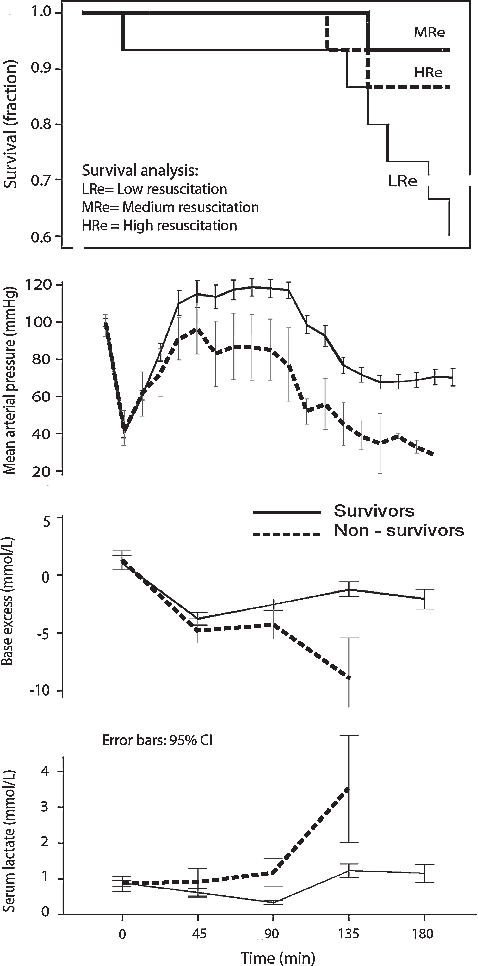
Survival plots depending on the intensity of the fluid resuscitation (top) and the MAP, base excess, and the serum lactate concentration (bottom panels) for survivors and nonsurvivors.
Desmopressin
When comparing the MRe with MRe + D, ∼50% of the animals in each group had one or more rebleedings. The number of rebleeders did not differ significantly between groups (Table 3), neither did the total rebleeding volume expressed as percent of estimated blood volume (p=0.32).
Data are expressed as median values and interquartile range.
D, desmopressin.
When analyzing rebleeders separately, no difference was found in the total rebleeding volume between groups, nor in any of the other parameters analyzed, i.e., duration of each rebleeding, bleeding rate, or bleeding volume per occasion. However, the MAP at onset of rebleeding was higher in the MRe group (Table 3).
At the end of observation period (180 minutes) Hgb was higher in the MRe group (9.3±1.1 g/dL) compared with the MRe + D group (8.4±0.9 g/dL; p=0.04) but these two groups did not differ significantly with respect to base excess, serum lactate, or to survival (MRe 93% vs. MRe + D 87%, p=0.50).
Survivors versus nonsurvivors
The 49 survivors and 11 nonsurvivors had a similar initial hemorrhage, being 24 (22%–28%) and 26 (11%–28%) of the blood volume, respectively (median). However, the mortality increased if the animal had one or more rebleedings (10/35 rebleeders died vs. 1/25 nonrebleeders, p=0.015) that increased the difference in hemorrhage between survivors and nonsurvivors; the total bleeding in these groups amounted to 28 (23%–32%) and 48 (32%–64%) of the blood volume, respectively (p=0.001). The survivors and nonsurvivors also differed with regard to MAP, base excess, and serum lactate, and this difference became more pronounced with time (Fig. 4, lower panels).
Discussion
This study used a new model for uncontrolled hemorrhage in rats that allows detailed studies of rebleeding events (Heinius et al., 2011). The topic was how different i.v. regimes of LR affect rebleeding during HT and also if desmopressin, an inexpensive and widely marketed hemostatic drug, decreases rebleeding under these conditions.
The results confirm that rebleeding promotes mortality by increasing the total blood loss (Riddez et al., 2002; Drobin et al., 2005). The rebleeding volume increased with the intensity of the fluid resuscitation due to prolongation of the rebleeding episodes, which might be due to increased blood flow rates (Riddez et al., 2002), dilution coagulopathy (Maegele et al., 2007), or higher MAP (Stern et al., 1993). Moreover, there was no evidence that desmopressin reduced the number or the size of rebleedings during HT.
The LRe regimen did not prevent rebleeding, but those that occurred became less voluminous and had shorter duration. However, rats with rebleeding in this group seemed to have a particularly high risk of dying (Fig. 2). Hence, there was a clear trend toward higher mortality in LRe group, where laboratory data indicated poor tissue perfusion. Base excess and serum lactate, which are markers of anaerobic metabolism that predict poor outcome of HS (Rutherford et al., 1992; Manikis et al., 1995; Reynolds et al., 2008), indicated already at 135 minutes that the LRe group did more poorly than the others.
When surviving animals were compared with nonsurvivors, base excess and serum lactate levels began to differ already at the end of the HT period (Fig. 4). The number of rebleedings appeared to be the most important factor explaining the larger bleeding volume in the nonsurvivors. Similar findings were observed by us in an aortic tear model in pigs during NT conditions (Drobin et al., 2005) and supports the view that repeated rebleeding is an important cause of death in uncontrolled hemorrhage.
Since the work by Shires and others (Shires et al., 1964; Dillon et al., 1966), the standard care for trauma victims in HS has been to resuscitate with large volumes of crystalloids, even before surgical hemostasis has been achieved. This view is challenged by more recent studies of uncontrolled hemorrhage (Bickell et al., 1991; Riddez et al., 1998; Varela et al., 2003). Very few randomized studies on humans have been performed, but the European expert group still recommended a target systolic blood pressure of 80–100 mmHg until major bleeding has been stopped (Spahn et al., 2007). This corresponds to MAP of ∼65–75 mmHg.
The impact of different fluid regimes on rebleeding during HT is even more sparsely investigated. Kim et al. (1997), using the “rat tail cut” model, showed improved survival with the combination of a low volume of LR (target MAP 40 mmHg) combined with HT at 30°C. Hachimi-Idrissi et al. (2004) used the same tail cut model to compare NT (38°C) and HT (34°C) groups resuscitated to MAP 80 or MAP 40 mmHg. Better survival was seen in the HT group resuscitated to MAP 40 mmHg. However, the animals in these studies were anesthetized with N2O/halothane which, like many other anesthetic agents, partly blocks the sympathetic drive (Longnecker and Sturgill, 1976). As a possible result of this, MAP was unaffected by HT. In healthy conscious humans, MAP increases by 20% and cardiac output by up to 50% on exposure to cold water or cold air (Budd and Warhaft, 1966; Muza et al., 1988). Also in the present study, where the anesthesia was chosen to spare the sympathetic drive, animals without rebleeding quickly reached a MAP above baseline. The hypertensive effect of the HT is particularly apparent in the sham group where no exsanguination was undertaken.
The slightly elevated MAP could make the present results difficult to generalize, as “permissive hypotension” is the standard of care in trauma where uncontrolled hemorrhage might be present (Spahn et al., 2007). On the other hand, “permissive hypotension” could be difficult to practice in HT when resuscitating an initial hemorrhage amounting to only 25% of the blood volume, at least without having blunted the HT-induced vasoconstriction with drugs. From other studies we know that survival is optimal when MAP is maintained at 50–60 mmHg in barbital-anesthetized normothermic rats subjected to uncontrolled hemorrhage (Li et al., 2011).
Other limitations include that the study intends to mimic accidental HT that occurs outside hospital in a cold climate. Trauma victims are usually not as hypothermic, and therapeutic HT in these patients aims to reach a body temperature of 32°C–34°C (Finkelstein and Alam, 2010). Another issue is that no coagulation parameters were measured. Finally, present study was not designed to titrate the fluid administration to reach a target MAP but to compare the effect of different fluid programs on rebleeding. The rationale behind this choice is that rebleeding, besides arterial pressure, is promoted by increases in the blood flow rate that are dependent on the intensity of the fluid resuscitation (Riddez et al., 1998, 2002).
The strength with the current new model is the high reproducibility and the ease by which rebleeding events can be detected and quantified. Thus, the model has the possibility to disclose details and bleeding patterns that can be important for understanding how rebleeding occurs. An asset is that the bleeding intensity is monitored continuously, which makes it possible to match the fluid resuscitation with the amount of lost blood, as was done in the present study.
The choice of desmopressin as a possible drug to prevent rebleeding during HT is based on a previous finding showing that rebleeding volumes are larger among HT rats at temperatures between 35°C and 37.5°C (Heinius et al., 2011). The coagulation enzymes are then not affected (Watts et al., 1998) but there is rather a negative effect on platelet function (Wolberg et al., 2004) that desmopressin prevents in vitro (Ying et al., 2008). However, an exploratory analysis of this temperature interval showed that, just as for the entire study period, desmopressin did not decrease the number of rebleedings or the size or duration of each rebleeding episode (data not shown). Desmopressin is an inexpensive drug with immediate onset and few and mild adverse effects (Lethagen, 1997). A high dose (10 times the standard in man) was used to compensate for the higher drug clearance in the rat and the possible loss of drug of rebleedings (Ryan et al., 2006).
In conclusion, the results from our “one vessel injury model,” using three different i.v. regimes of LR during cooling and HT, show that rebleeding events increased the amount of blood lost and the mortality. The intensity of the fluid resuscitation increased the rebleeding volume and the duration of each rebleeding episode. The medium infusion rate appeared to be most suitable regime, as many animals receiving fluid resuscitation of low intensity apparently died of shock. Desmopressin does not prevent rebleeding episodes or decrease bleeding volume during these circumstances.
Footnotes
Acknowledgment
Financial support was received from the Swedish Defence and Research Agency (FOI).
Disclosure Statement
R.G. Hahn has received speaker's honorarium from Baxter Medical AB. There are no other conflicts of interest.
