Abstract
Mouse embryonic fibroblasts (MEFs) have been widely used as feeder cells in embryonic stem cell cultures because they can mimic the embryonic microenvironment. Milk fat globule-epidermal growth factor 8 (MFGE8) is expressed during mouse gonadal development, 10.5–13.5 embryonic, and is also found in MEF-conditioned medium (MEF-CM). Feeder-less culture of human-induced pluripotent stem cells (iPSCs) with MEF-CM significantly decreased the number of adherent cells when an inhibitory antibody against MFGE8 was used. The concentration of mouse MFGE8 in MEF-CM, as measured by an ELISA (Enzyme-Linked Immunosorbent Assay), was 0.16-1.24 μg/mL. Mouse MFGE8 and human MFGE8 have partially different molecular structures. Both the recombinant mouse MFGE8 and human MFGE8 significantly promoted cell adhesion of human iPSCs at medium-added concentrations of 2 μg/mL. This cell adhesion was also strongly inhibited by Arginylglycylaspartic acid (RGD) inhibitors, suggesting that it is dependent on the RGD sequence. The integrin αVβ5 expressed in iPSCs was thought to be involved in binding to the RGD sequence. MEF-CMs have long been an essential bio-derived material for the feeder culture method of iPSC culture. This study demonstrates that MFGE8 in MEF-CM is a functional factor in the promoting of cell adhesion of human iPSCs. Furthermore, the use of MFGE8-containing media demonstrates that iPSCs can be established and cultured while maintaining pluripotency and inducing three germ layer differentiation. The results of this study suggest the possibility of using MFGE8 as a scaffold material suitable for inducing differentiation when reproducing in vivo maturation in vitro.
Impact Statement
For the practical application of regenerative medicine using human-induced pluripotent stem cells (iPSCs), it is important to establish a method that can induce the differentiation of cells and tissues that function in vivo. This article demonstrates new functions of biomaterials expressed in the embryo. Milk fat globule-epidermal growth factor 8 (MFGE8) is expressed during mouse gonadal development, 10.5-13.5 embryonic, and is also found in MEF-conditioned medium (MEF-CM). This study demonstrates that MFGE8 in MEF-CM is a functional factor in the promoting of cell adhesion of human iPSCs.
Introduction
Mouse embryonic fibroblasts (MEFs) have been used as scaffolds for cell culture, from inner cell mass cells in blastocysts to pluripotent embryonic stem (ES) cells. 1 MEFs are not thought to affect the pluripotency of undifferentiated cells. Therefore, the classical method of ES cell2,3 and induced pluripotent stem cell (iPSC)4,5 culture has been the on-feeder culture method using MEFs.6,7 However, instead of using MEFs as feeder cells and coculturing them, a method was investigated that allowed ES cells and iPSCs to be cultured in a culture dish on their own. MEFs were found to secrete factors that contribute to the maintenance of hPSCs during overnight incubation, and MEF conditioned medium (MEF-CM) 8 was developed. It was also found that MEF-CM can be used to culture ES cells and iPSCs alone when FGF2 (fibroblast growth factor 2) is added to the medium before use to achieve Xeno-free culture. 9 Integrin function has been reported to strongly influence the pluripotency of iPSCs 10 and more undifferentiated naive iPSCs. 11 It has been reported that feeder-less cultures of MEF-CM with Matrigel® 12 or laminin 13 result in a similar pattern of integrin expression on the plasma membrane surface as feeder MEF cultures. 14 Feeder MEF cultures use gelatin-coated plates as a scaffold material. A comparison of Matrigel® and gelatin scaffolds showed no difference in the pluripotency of mouse ES cells. 15 However, iPSCs adhere to the gelatin in the presence of MEF-CM, but not in the absence of MEF-CM. We have also reported that iPSCs have reduced OCT3/4 (Octamer-binding transcription factor 3/4) mRNA expression levels in the cell mass state without cell adhesion. 16 Therefore, it has been speculated that MEF-CM contains factors that promote adhesion of iPSCs to gelatin. Therefore, in this study, we analyzed the components of MEF-CM to search for components that promote cell adhesion of iPSCs in MEF-CM that have not been previously reported.
In this study, a total of 1487 different proteins were identified by a shotgun proteome analysis of the protein components in MEF-CM. Among the 1487 proteins, an attempt was made to identify factors that influence iPSC proliferation or cell adhesion. Milk fat globule-epidermal growth factor 8 (MFGE8) is a cell-secreted protein derived from mouse mammary epithelium, and its human homologue has been isolated. MFGE8 consists of two contiguous epidermal growth factor (EGF) domains and two discoidin domains. The second EGF domain contains RGD sequences that bind to the plasma membrane proteins integrin αvβ3 and integrin αvβ5. The discoidin domain is highly homologous to the C domain of coagulation factors V and VIII and is known to bind phosphatidylserine (PS), a plasma membrane phospholipid exposed on the cell surface. MFGE8 uses these two cell adhesion domains for cell-cell adhesion. Expression of MFGE8 has been found in mammary epithelia, as well as in various organs and cells, but its function remains unclear. From an embryological point of view, a transient local development was observed during the gonadal development of the mouse, from about 10.5 to 15.5 days of embryonic life. The presence of MFGE8 was thought to induce cell adhesion in cells expressing integrins or PS. Apoptosis is also known to be induced during development. 17 In addition, iPSCs express the integrin αvβ5, 10 a target of RGD sequences. In this context, MFGE8 may act as a cell adhesion-promoting factor in MEFs derived from 13.5-day-old mouse embryos and the components in MEF-CMs. MFGE8 may be a factor that promotes cell adhesion of iPSCs in the MEF-CM. In this study, we report the identification of MFGE-8 as a functional factor for pluripotent stem cells as a component of MEF-CM.
Methods
Reagents
StemFit AK03N was purchased from Ajinomoto Healthy Supply Co., Inc. (Tokyo, Japan). iMatrix-511 was purchased from Matrixome Inc. (Osaka, Japan). A 10-mmol/L Y-27632 solution, D-PBS (phosphate-buffered saline) (-), and 0.5 mol/L-EDTA solution (pH 8.0) were purchased from Nacalai Tesque (Kyoto, Japan). TrypLTM Select Enzyme (1×), Essential 8™ Flex Medium Kit, and CTSTM Essential 8TM Medium were purchased from Thermo Fisher Scientific K.K. (Waltham, MA, USA). Human GloLIVE TRA-1-60 (R) NorthernLightsTM NL557-conjugated antibody, recombinant human MFGE8, and mouse MFGE8 were purchased from R&D Systems (Minneapolis, MN, USA). GLS250 gelatin solution (1.0 mg/g) was purchased from Nitta Gelatin Inc. (Osaka, Japan). Kyoto probe 1 (KP-1) was purchased from Goryo Chemical, Inc. (Sapporo, Japan). Cellstain®-Hoechst 33342 solution was purchased from Dojindo Laboratories (Kumamoto, Japan). RGD peptides (H-Gly-Arg-Gly-Asp-Asn-Pro-OH) were purchased from PEPTIDE INSTITUTE INC., (Osaka, Japan). MEFs were purchased from REPROCELL Inc. (Kanagawa, Japan).
Preparation of cell culture plates
The following reagents were added to each well of a 6-well plate to be coated with 9.6 µL iMatrix-511 (0.5 µg/µL) + 1.5 mL PBS. Gelatin and MFGE8 supplemented medium was prepared by adding 0-10% GLS250 gelatin solution (1.0 mg/mL) and 0-4 μg/mL of recombinant human or mouse MFGE8 to the medium.
Maintenance culture of human mononuclear cells
These experimental techniques have been previously reported by us.16,18 Normal human PBMCs (Peripheral Blood Mononuclear Cells) from Japanese donor, purified and characterized (10 M cells/vial), were obtained from FUJIFILM Wako Pure Chemical Corporation. The medium for human mononuclear cell medium consisted of stem cell factor/c-kit ligand (final concentration 50 ng/mL), thrombopoietin (final concentration 10 ng/mL), Flt3L (final concentration 20 ng/mL), IL-6 (final concentration 50 ng/mL), IL-3 (20 ng/mL), and G-CSF (Granulocyte colony-stimulating factor) (10 ng/mL) in a mixture of liquids A and B of StemFit AK03N (Ajinomoto Healthy Supply Co., INC., Tokyo, Japan).
The following protocol is a method for culturing PBMCs and is a brief description of the procedure normally followed at our institution. Thaw a frozen vial of normal human PBMCs in a 37°C water bath for 1 min. Suspend the PBMCs in 5 mL of a mixture of liquid A and liquid B of StemFit AK03N and then centrifuge the sample (440 × g for 5 min at 22°C). After removing the supernatant, 1 mL of human mononuclear cell culture medium is added, mixed, and the cells are counted. In our case, a total of 3 × 106 cells/mL human mononuclear cell culture medium was used. Cells are seeded in 24-well plates at 1 mL/well and incubated at 37°C in 20% O2 and 5% CO2 for 5 days.
Establishment of hiPSCs
These experimental techniques have been previously reported by us.16,18 Human-induced PSCs were established using CytoTune®-iPS 2.0 Vector, according to the manufacturer’s instructions (ID Pharma Co., Ltd., Tokyo, Japan). Briefly, 1 × 105 cells were added to microtubes and centrifuged (300 × g for 5 min). According to the data sheet provided with the kit, 7.14 μL of KOS (Klf4, Oct4 and Sox2) tube, 6.66 μL of KLF4 (KLF Transcription Factor 4) tube, and 10.00 μL of C-MYC (MYC proto-oncogene, bHLH transcription factor) tube, the vector provided with the kit, were added to 2 mL of human mononuclear cell medium. A total of 1 × 105 cells were then seeded into a 6-well plate. The cells were cultured in human mononuclear cell medium supplemented with different types of vectors [MOI (Multiplicity of Infection) = 5] at 37°C, 20% O2, and 5% CO2 for 2 h. Next, 2/3 volume of StemFit AK03N medium was added on days 1, 3, 5, and 7 of culture at 37°C for 2 h at 20% O2 and 5% CO2. The medium was replaced with StemFit AK03N medium on days 9, 11, and 13 of culture. Cell passaging and colony picking were performed on day 15 of culture.
Maintenance culture of hiPSCs
These experimental techniques have been previously reported by us.16,19 PBMC-derived iPSCs and hiPSC (line 201B7) were obtained from the CiRA Foundation (Kyoto, Japan). A publicly available method (CiRA_Ff-iPSC_protocol_Eng_v140310) was used to culture iPSCs (https://www.cira.kyoto-u.ac.jp/j/research/img/protocol/Ff-iPSC-culture_protocol_E_v140311.pdf).
Preparation of MEF culture supernatant
MEFs were cultured on a 100-mm culture plate using an Essential 8 Flex Medium Kit (the number of cells was 1.5 × 107 cells/plate). Cells were cultured for 24 h in Essential 8™ Flex Medium Kit (Thermo Fisher Scientific A2858501). After 24 h, the culture supernatant was aspirated with a pipette and centrifuged (1500 g, 30 min, 4°C) to remove the cells. After the centrifugation of the medium, the supernatant was concentrated 20 times using Amicon Ultra-15, PLGC Ultracell-PL membrane, and 10 kDa (UFC901008) (MERCK, Kenilworth, NJ, USA), and a concentrated solution of culture supernatant was obtained.
Shotgun liquid chromatography–tandem mass spectrometry
These experimental techniques have been previously reported by us. 19 The experiment was performed by Apro Science Group/Pharma Foods International Co., Ltd (Tokushima, Japan). For cell lysis, an EzRIPA Lysis Kit (ATTO Corporation, Tokyo, Japan) was used according to the manufacturer’s instructions. A protein solution of 2362 μg/mL was obtained from the concentrated solution of the culture supernatant of MEFs. Finally, 2.0 μg of protein was used for liquid chromatography-tandem mass spectrometry (LC-MS/MS) (EASY-nLC 1200 system; Thermo Fisher Scientific K.K.). The samples were subjected to Nanoflow-LC ESI using a Q Exactive plus (Thermo Fisher Scientific K.K.) at the Pharma Foods International Co., Ltd. (Tokushima, Japan). Based on the results of protein quantification, Tris buffer containing dithiothreitol was added to 2 μg of sample to obtain a final concentration of 5 mM, and the sample was reduced at 35°C for 2 h. Tris buffer containing iodoacetamide to a final concentration of 14 mM was added to the reduced sample for 30 min at room temperature (light shielded), followed by trypsinization. The collected sample solution was solvent exchanged on a cation exchange column (EASY-Spray column, 15 cm × 75 μm ID, 3 μm particles, 100 Å pore size; Thermo Fisher Scientific K.K.), desalted, and concentrated, and a portion was subjected to LC-MS/MS. Solvent A was 0.1% formic acid, and solvent B was 80% acetonitrile/0.1% formic acid. Peptides were eluted in a 229-min gradient from 5% solvent B in solvent A to 40% solvent B in solvent A at 300 nL/min. In-house database searches were performed using the MASCOT server (http://www.matrixscience.com/help_index.html) with product ion measurement data for all MS/MS spectra obtained from LC-MS/MS.
Cell differentiation assays
To functionally verify the ability of a hiPSC to differentiate into three germ layers (ectoderm, mesoderm, and endoderm), the STEMdiff Trilineage Differentiation Kit (Stemcell Technologies Inc., Vancouver, Canada) was used.
qPCR (quantitative PCR) array
These experimental techniques have been previously reported by us.16,19 RNA was prepared using a SuperPrep II Cell Lysis & RT Kit for qPCR (Toyobo Co., Ltd., Osaka, Japan) according to the manufacturer’s instructions. Real-time PCR was performed using a StepOnePlus system (Life Technologies, Carlsbad, CA, USA).
For the mRNA expression analysis, a TaqManTM Array 96-Well FAST Plate (Human Stem Cell Pluripotency; Applied Biosystems) was used with TaqMan Fast Advanced Master Mix (Thermo Fisher Scientific, K.K.) according to the manufacturer’s instructions. The PCR protocol was as follows: (1) denaturation at 95°C for 20 s, further denaturation of double-stranded DNA; (2) annealing of primers at 60°C for 20 s, and repeating steps (1) to (2) 40 times.
For the analysis of real-time PCR data using a TaqManTM Array 96-Well FAST Plate, 18S, GAPDH, HPRT1, and GUSB were used as housekeeping genes. The maximum CT value was set at 40. The ΔCT value for undetected was calculated by subtracting the average of the CT values of the four housekeeping genes from the maximum CT value (40). The ΔCT value of the target was calculated by subtracting the average CT values of the four housekeeping genes from the CT values of the different genes under each culture condition. To calculate the ΔΔCT values of the target, the average ΔCT values of the different genes under control culture conditions were subtracted from the ΔCT values of the different genes under each culture condition. The ΔΔCT values were then calculated using the Excel software program (Microsoft Corporation, Redmond, WA, USA).
Cell proliferation assays
Cell proliferation was measured using a Countess (Thermo Fisher Scientific K.K.). Cell proliferation was measured using a Cell Counting Kit-8 (Dojindo, Kumamoto, Japan) according to the manufacturer’s instructions. Briefly, the cells were seeded into 96-well plates, medium supplemented with gelatin, and MFGE8 was prepared by mixing 5% GLS250 gelatin solution (1.0 mg/mL) and 0-4 μg/mL of recombinant human or mouse MFGE8 into the medium. Each well contained a 200 µL volume of StemFit AK03N medium at a cell concentration of 1.3 × 104 cells/mL. Absorbance was measured using a microplate reader at a wavelength of 450 nm.
Statistical analyses
Statistical analyses were performed using Student’s t-test to compare the means of two samples. Comparisons between multiple groups (more than two groups) were performed by one-way analysis of variance using the StatPlus software program (AnalystSoft, Walnut, CA, USA). Statistical significance was set at *p < 0.05 or **p < 0.01 for all tests.
Results
Identification of MFGE8 protein in MEF-CM
MEF-CM is a conditioned medium containing proteins secreted by MEFs into the culture medium. A total of 1487 different proteins were identified by a shotgun proteome analysis of the protein components in MEF-CM (Supplementary Table S1). Peptide fragments of MFGE8 contained in the MEF-CM were detected by LC-MS/MS (Fig. 1A). The position of the peptide sequence identified by the Scaffold 6 software program (MATRIX SCIENCE, Boston, MA, USA) was visualized. At 12% coverage of MEF-CM samples, peptide chains were detected at positions 66–87, 216–226, 353–364, and 376-386. The peptide chains detected in the MEF-CM sample were the EGF-like domain and the F5/8 type C-terminal domain. The EGF-like domain (a cell adhesion site) has a cell adhesion active sequence (RGD motif) at positions 28-57 and 68-106 of the peptide sequence. The F5/8 type C domain (discoidin-like domains) at positions 115-265 and 273-425, which binds to type I collagen20,21 or PS,22,23 was expressed on the surface of dead cells (Fig. 1B). Both the EGF-like domain and the F5/8 type C domain (discoidin-like domains) were detected in MEF-CM samples. Mouse MFGE8 and human MFGE8 are reported to share 63% amino acid sequence homology. 24
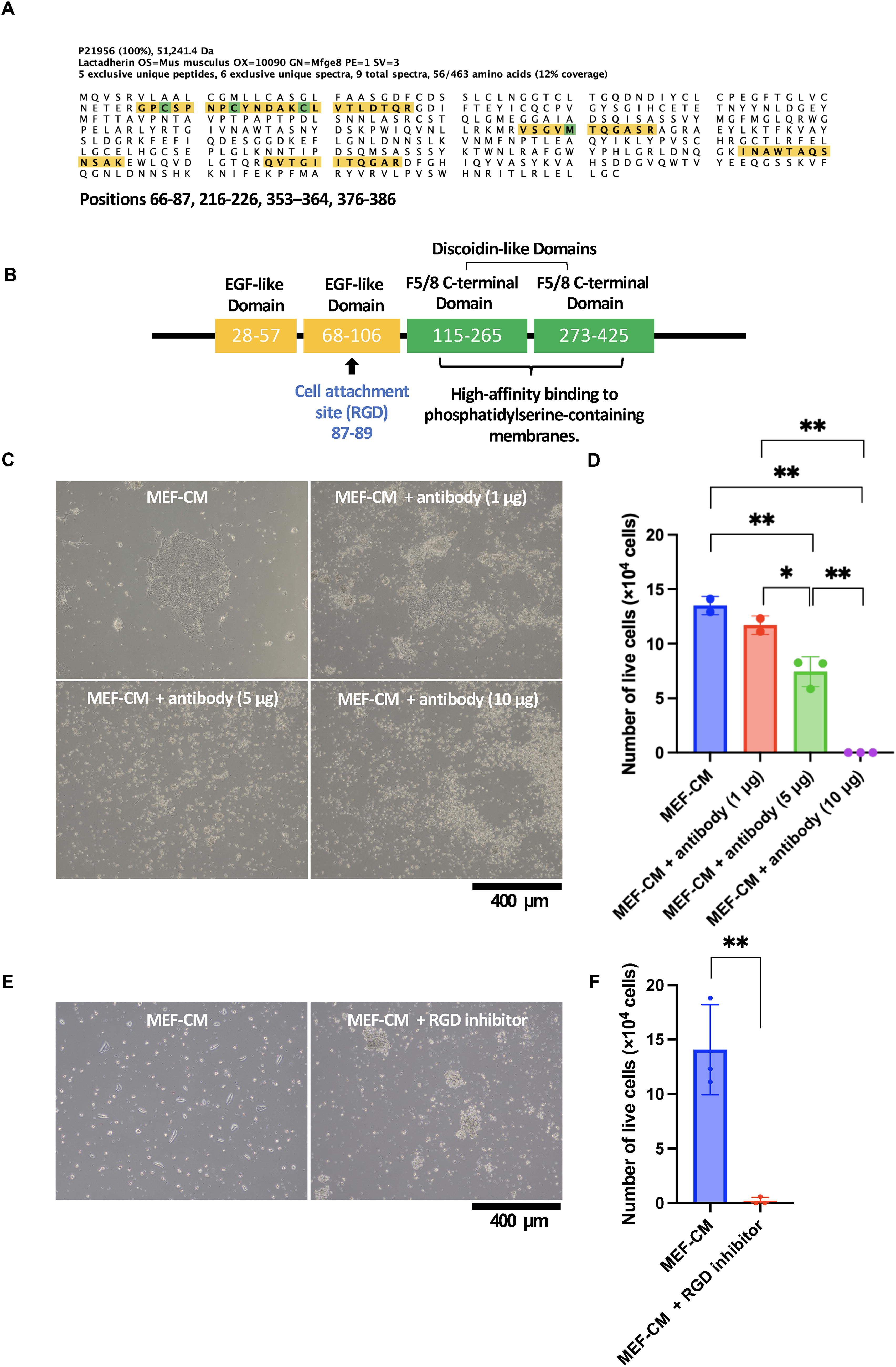
Identification of MFGE8 in MEF-CM.
MEF-CM has the effect of promoting cell adhesion of hiPSCs to uncoated 6-well plates. To investigate the effect of MFGE8 on cell adhesion, an anti-MFGE8 antibody was added to the culture medium to determine whether it inhibited cell adhesion. PBMC-derived iPSCs were cultured in 6-well plates by adding MEF-CM only. On day 7 after cell seeding (5 × 104 cells), PBMC-derived iPSCs formed adherent colonies (Fig. 1C, upper left panel), and the number of viable cells was 1.35 × 105 cells (Fig. 1D). PBMC-derived iPSCs were cultured in 6-well plates with 5 μg of MFGE8 inhibitory antibody and MEF-CM. On day 7 after cell seeding (5 × 104 cells), PBMC-derived iPSCs did not form adherent colonies (Fig. 1C, lower left panel), and the number of viable cells was significantly reduced to 7.45 × 104 cells (Fig. 1D). PBMC-derived iPSCs were cultured in 6-well plates with 10 µg of MFGE8 inhibitory antibody using MEF-CM. No PBMC-derived iPSCs were observed at day 7 after cell seeding (5 × 104 cells) (Fig. 1C, lower right panel), and the number of viable cells was significantly decreased at 0.00 × 104 cells (Fig. 1D). MEFs secrete activin A, collagen I, HSPGs (heparan sulfate proteoglycans), fibronectin, TGF-β (Transforming Growth Factor-β), IGF1 (insulin-like growth factor-1), and IGF2 (insulin-like growth factor-2) using human serum albumin in the culture medium as a nutrient. 10 These results suggest that MFGE8 is essential for maintaining the cell adhesion and survival of iPSCs under MEF culture conditions. iPSCs were cultured in 6-well plates by adding MEF-CM only. On day 3 after cell seeding (5 × 104 cells), iPSCs (201B7) formed adherent colonies (Fig. 1E, left panel), and the number of viable cells was 1.41 × 105 cells (Fig. 1F). iPSCs (201B7) were cultured in 6-well plates with 10 μg of RGD peptide (H-Gly-Arg-Gly-Asp-Asn-Pro-OH) (PEPTIDE INSTITUTE INC., Osaka, Japan) with MEF-CM. On day 3 after cell seeding (5 × 104 cells), iPSCs did not form adherent colonies (Fig. 1E, right panel), and the number of viable cells was significantly reduced to 0.20 × 104 cells (Fig. 1F). These results suggest that MFGE8 is the major factor promoting cell adhesion of iPSCs in MEF-CM and that the RGD sequence in the MFGE8 molecule is its functional site.
Cell adhesion and proliferation of hiPSCs cultured in MFGE8-containing medium
Mouse MFGE8 has been shown to promote the cell adhesion and cell proliferation of human iPSCs. We next tested whether this effect also occurs with human MFGE8. Next, MFGE8-containing medium was prepared to study the function of MFGE8. Gelatin was randomly selected as the medium additive because it is required for MEF on-feeder culture methods. Gelatin was added to the culture medium. Mouse or human MFGE8 (0, 0.1, 0.2, 0.4, 0.8, 1.0, 2.0, and 4.0 μg/mL) was added to the medium (StemFit AK03N) containing 5% gelatin solution, and iPSCs adhered to the wells were observed by optical microscopy on day 5 (Fig. 2A). An MTT assay was also performed (Fig. 2B) to examine cell viability activity. Both mouse and human MFGE8 significantly promoted cell adhesion and cell proliferation of human iPSCs when the medium concentration was 2.0 μg/mL. These results suggest that mouse and human MFGE8 have similar effects on human iPSCs, although their molecular structure homology is only 63%. Therefore, recombinant human MFGE8 was used in the following experiments.
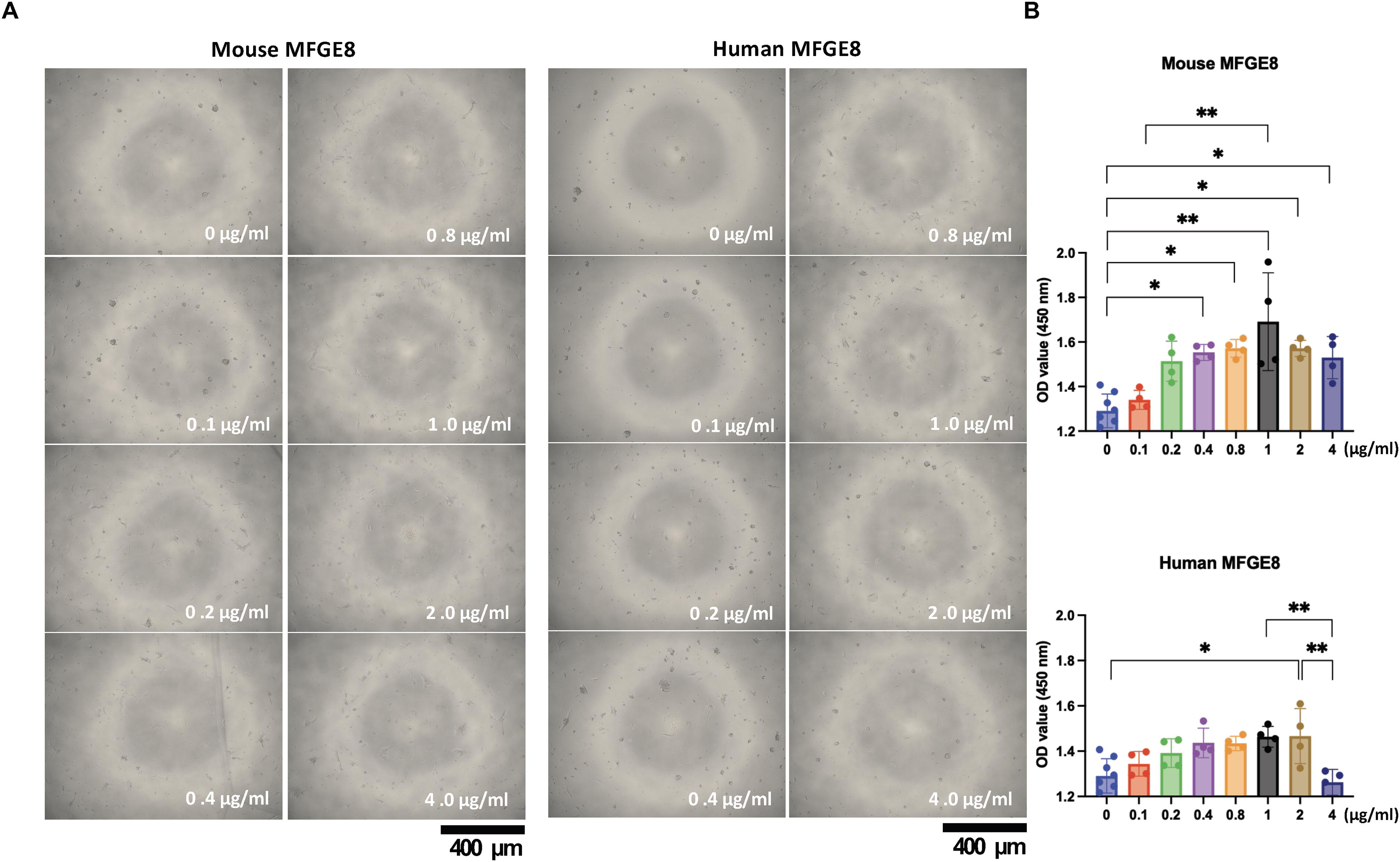
Cell proliferation of hiPSCs cultured with gelatin+MFGE8.
MFGE8-containing media maintain undifferentiated hiPSCs and allow for establishment and passage
We first investigated the optimal concentrations of gelatin to add to the medium. MFGE8 (2.0 μg/mL) was added to the medium. This was the maximum amount that could be added. Human-induced PSCs in medium (StemFit AK03N) containing 10% gelatin solution showed no cell adhesion to uncoated dish surfaces at day 6 after cell seeding (Fig. 3A, upper most left panel). However, in medium (StemFit AK03N) containing 5-10% gelatin solution and MFGE8 protein (2 μg/mL), hiPSCs showed cell adhesion to the uncoated dish surface at day 6 after cell seeding (Fig. 3A, upper middle and upper right panels).
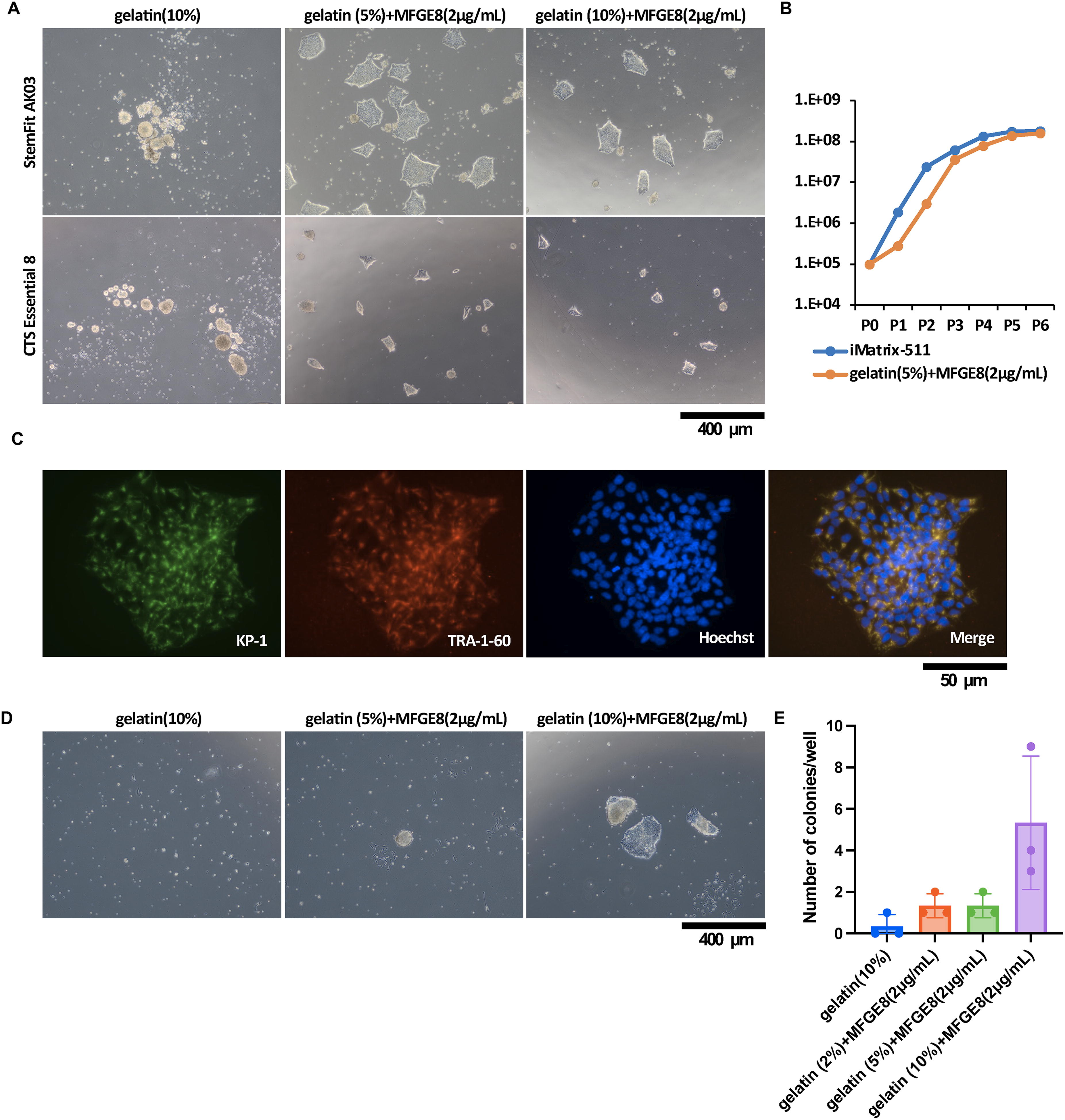
Characterization of hiPSC cultured with gelatin + MFGE8. (
Experiments were then performed with different media. The optimal medium addition showed concentrations of gelatin and MFGE8 protein. Human-induced PSCs in medium (CTS Essential 8) containing 10% gelatin solution showed no cell adhesion to uncoated dish surfaces at day 6 after cell seeding (Fig. 3A, lower most left panel). However, in medium (CTS Essential 8) containing 5-10% gelatin solution and MFGE8 protein (2 μg/mL), hiPSCs showed cell adhesion to the uncoated dish surface at day 6 after cell seeding (Fig. 3A, lower middle and upper right panels).
For the cell proliferation assay, cells were detached from 6-well plates, cell suspensions were prepared, and cell numbers were calculated. Cells were seeded at a concentration of 1 × 105 cells at each passage. Cells from passages 0-5 were counted. PBMC-derived iPSCs cultured on iMatrix-511-coated plates are indicated by blue lines, and PBMC-derived iPSCs cultured in medium containing gelatin (5%) + MFGE8 (2 μg/mL) are indicated by red lines (Fig. 3B). The results showed that when medium containing MFGE8 (2 µg/mL) and gelatin solution (5-10%) was used, hiPSCs adhered to uncoated dishes and were able to grow cells without any difference from those grown on iMatrix-511-coated plates.
CTS Essential 8 medium supplemented with gelatin solution 10% + MFGE8 (2 µg/mL) was used for the culture. 201B7 cells were subjected to KP-1, TRA-1-60, and Hoechst staining on day 6 after seeding at 5.2 × 104 cells/well (12-well plate). Fluorescence microscopy was then performed at 400 × magnification (Fig. 3C). The results showed that KP-1-positive cells (undifferentiated cells) and TRA-1-60-positive cells (a marker for iPSCs) costained in the cytoplasm around the nucleus in the merged photographs. These results indicate that pluripotent iPSCs can be grown in either StemFit AK03N medium or CTS Essential 8 medium with gelatin solution 5-10% + MFGE8 (2 µg/mL) as a medium supplement.
The next step was to determine if it was possible to establish iPSCs using a medium containing 5-10% gelatin solution + MFGE8 (2 µg/mL). iPSCs were established in StemFit AK03N medium containing gelatin solution (10%) + MFGE8 (2 µg/mL) as a medium additive. Gelatin solution (10%) (Fig. 3D, left panel) or gelatin solution (5%) + MFGE8 (2 µg/mL) (Fig. 3D, middle panel) or gelatin solution (10%) + MFGE8 (2 µg/mL) (Fig. 3D, right panel) was used as medium additive. Cells were reprogrammed by adding Sendai virus vector to a total of 1 × 105 human PBMCs in StemFit AK03N medium. Optical microscopy images of the cells were collected 16 days after reprogramming. The number of TRA-1-60-positive colonies/well at 16 days after reprogramming was shown (Fig. 3E). The establishment of iPSCs by cell reprogramming was possible in StemFit AK03N medium containing gelatin solution (5-10%) + MFGE8 (2 µg/mL) as medium additive.
Cultured hiPSCs in MFGE8-containing media were capable of three germ layer differentiation
The pluripotency of 201B7 cells cultured in StemFit AK03N medium containing gelatin solution (10%) + MFGE8 (2 µg/mL) as a medium supplement was investigated. Induction of three-embryo differentiation was initiated 6-8 days after cell seeding of the 201B7 cells at 5 × 104 cells/well (6-well plate) in StemFit AK03N medium containing 10% gelatin solution + MFGE8 (2 µg/mL) as a medium supplement. Photographs were taken of endoderm on day 5 after induction (Fig. 4A, left panel), mesoderm on day 5 after differentiation induction (Fig. 4A, middle panel), and ectoderm on day 7 after differentiation induction (Fig. 4A, right panel). mRNA was extracted from these cells, and cDNA was synthesized, and the results of an mRNA expression analysis are shown. Expression was determined by the ΔΔCt method. Data were normalized by converting the average expression of various mRNAs in 201B7 cells cultured on iMatrix-511 using StemFit AK03N medium to 1. 201B7 cells at 5 × 104 cells/well (6-well plate) in StemFit AK03N medium containing gelatin solution 10% + MFGE8 (2 µg/mL) as a medium additive were used as a control before differentiation induction. The mRNA levels of various endoderm markers (Fig. 4B), mesoderm markers (Fig. 4C), and ectoderm markers (Fig. 4D) were measured using real-time PCR. If the mRNA level was four times (or more) higher or lower, it was considered abnormal and plotted in yellow on the heat map. The difference identified was that the expression level of AFP (alpha-fetoprotein) (endoderm marker), T (a mesoderm marker), and PAX6 (Paired box protein Pax-6) (an ectoderm marker) was increased more than 4-fold in the differentiation induction medium compared with the culture in the StemFit AK03N medium. These results confirm that hiPSCs cultured in MFGE8-containing media have trichoderm differentiation potential.
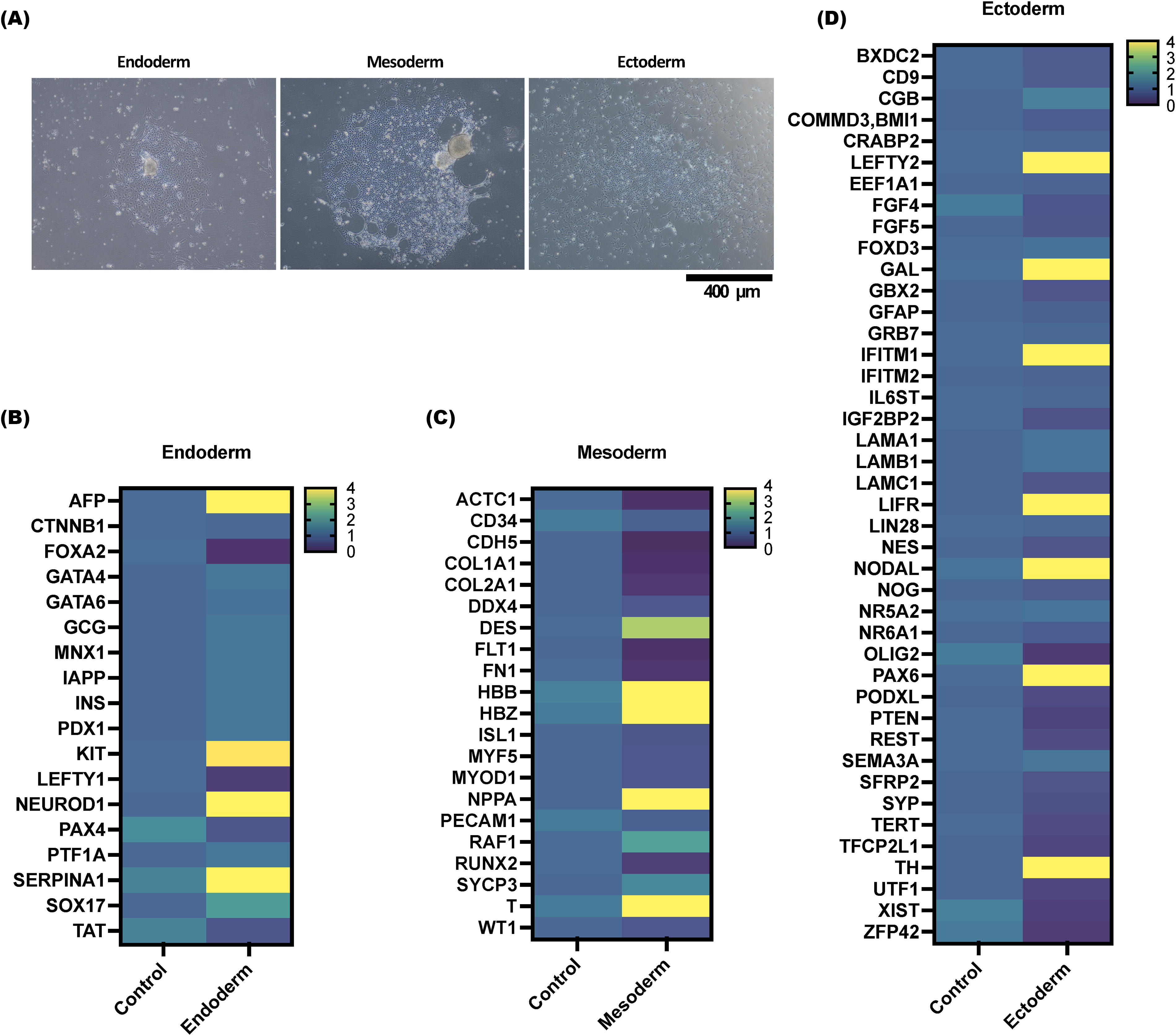
Evaluation of the gene expression and differentiation potential after culture using MFGE8-based iPSC culture methods.
Discussion
In the medium supplemented with MFGE8, the RGD motifs in the EGF-like domain enable the integrin-mediated cell adhesion of the iPSCs. RGD motifs have been reported to bind to the integrins αvβ3, αvβ5, αvβ6, αvβ1, αvβ8, α5β1, αIIbβ3, and α8β1.25–27 In the present study, a multiomics analysis was conducted, revealing the activation of fibronectin ligand integrin α5β1 28 in iPSCs cultivated in medium augmented with gelatin and MFGE8 media additives (data not shown). Previous reports have indicated that the presence of RGD motifs is also present in heat-treated gelatin molecules, and integrins α5β1 and αVβ3 have been observed to bind via magnesium. 29 In addition, integrin α5β1 in iPSCs activates the PI3K/AKT signaling pathway to promote iPSC survival. Furthermore, activation of integrin α5β1 in iPSCs consequently activates laminin-322, laminin-511, laminin-521, and collagen IV secretion. 10 A substantial body of research has indicated that αvβ3 and αvβ5 integrins function as MFGE8 receptors, as evidenced by studies.30–32 Integrin αvβ5 has been demonstrated to be activated by vitronectin, thereby promoting self-renewal. 33 Consequently, it can be deduced that the effects of gelatin and MFGE8 media additives may result in gelatin-mediated activation of integrin α5β1 and MFGE8-mediated activation of integrin αvβ5.
An investigation was conducted into the potential application of the RGD sequences present in MFGE8 as a constituent of the medium. The results of this investigation revealed that the incorporation of MFGE8 (2 µg/mL) and gelatin (1 mg/mL solution at 5-10%: 50-100 µg/mL) into the medium significantly promoted the adhesion of iPSCs (Fig. 1C). This technique does not necessitate the chemical binding of the RGD motif to the plate surface, as the gelatin diffuses in the medium as type I collagen and settles to the bottom of the plate. The protein-protein binding of MFGE8 to the type I collagen coating the plate surface facilitates the formation of the RGD motif on the plate surface. The weak cell adhesion mechanism of iPSCs cultured in medium supplemented with MFGE8 and gelatins suggests that these cells may have a particularly high potential to differentiate into the nervous system compared with iPSCs cultured on iMatrix-511. 19 MFGE8 has been shown to exert a vitronectin-like molecular function in iPSC culture. In addition, gelatin exerts fibronectin-like molecular functions via the fibronectin receptor integrin α5β1. Consequently, MFGE8 emerges as a promising component for promoting cell adhesion in iPSC culture, making it a viable medium supplement.
Footnotes
Authors’ Contributions
Study design, Y.N. and M.T.; study conduct, Y.N.; data collection, Y.N.; data analysis, Y.N.; data interpretation, Y.N. and M.T.; drafting of the article, Y.N. and M.T.; revision of the article for content, Y.N. and M.T.; and approval of the final version of the article, Y.N. and M.T. Y.N. takes responsibility for the integrity of all data analyses.
Data Availability Statement
The unique materials generated in this study are available from the lead contact upon reasonable request after signing of a material transfer agreement.
Disclosure Statement
The authors declare that they have no competing interests related to this study.
Funding Information
This research was supported by the Agency for Medical Research and Development (AMED) under grant numbers JP22bm0104001, JP23bm1323001h0101, and JP24bm1323001h0102.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
