Abstract
In native articular cartilage, chondrocytes (Chy) are completely capsulated by a pericellular matrix (PCM), together called the chondron (Chn). Due to its unique properties (w.r.t. territorial matrix) and importance in mechanotransduction, the PCM and Chn may be important in regenerative strategies. The current gold standard for the isolation of Chns from cartilage dates from 1997. Although previous research already showed the low cell yield and the heterogeneity of the isolated populations, their compositions and properties have never been thoroughly characterized. This study aimed to compare enzymatic isolation methods for Chy and Chns and characterizes the isolation efficiency and quality of the PCM. Bovine articular cartilage was digested according to the 5-h (5H) gold standard Chn isolation method (0.3% dispase +0.2% collagenase II), an overnight (ON) Chn isolation (0.15% dispase +0.1% collagenase II), and an ON Chy isolation (0.15% collagenase II +0.01% hyaluronidase). Type VI collagen staining, fluorescence-activated cell sorting (FACS) analysis, specific cell sorting, and immunohistochemistry were performed using a type VI collagen staining, to study their isolation efficiency and quality of the PCM. These analyses showed a heterogeneous mixture of Chy and Chns for all three methods. Although the 5H Chn isolation resulted in the highest percentage of Chns, the cell yield was significantly lower compared to the other isolation methods. FACS, based on the type VI collagen staining, successfully sorted the three identified cell populations. To maximize Chn yield and homogeneity, the ON Chn enzymatic digestion method should be combined with type VI collagen staining and specific cell sorting.
Impact statement
Since chondrocytes are highly dependent on their microenvironment for maintaining phenotypic stability, it is hypothesized that using chondrons results in superior outcomes in cartilage tissue engineering. This study reveals the constitution of cell populations obtained after enzymatic digestion of articular cartilage tissue and presents an alternative method to obtain a homogeneous population of chondrons. These data can improve the impact of studies investigating the effect of the pericellular matrix on neocartilage formation.
Introduction
Articular cartilage regeneration is a large and fast-growing research field within orthopedics. During the past 10 years alone, over 2200 research articles have been published on cartilage regeneration and repair, with the number of articles increasing each year. Considering the clinical problems and socioeconomic burdens associated with damage to articular cartilage, the growing interest in finding solutions for cartilage regeneration is well supported.
Regenerative strategies aim to recreate the cartilage extracellular matrix, mimicking the biomechanical, biochemical, and structural properties of the native tissue. Most of these are cell-based approaches, 1 and the cell type is very important. The use of stem cells like mesenchymal stem cells (MSCs) and adipose-derived stem cells has been widely explored since these are accessible cell sources.2,3 However, depending on their original differentiation line, stem cells often do not differentiate well and have tendencies toward the bone lineage. 4 Chondrocytes (Chy), the native cell type of articular cartilage, are therefore often preferred. Within cartilage, Chy are responsible for maintaining the ECM, which mainly consists of type II collagen and proteoglycans.
However, current clinical procedures using Chy, for example, autologous chondrocyte implantation (ACI) and matrix-induced autologous chondrocyte implantation, often result in fibrocartilage or hypertrophy.5,6 Less than half of the procedures leads to hyaline-like cartilage or hyaline-like cartilage with fibrocartilage, and in 9% of the cases, reoperation is needed. 7 This is caused, in part, by the inability of Chy to overcome dedifferentiation during in vitro monolayer expansion.7,8
In native cartilage, Chy are completely encapsulated by a pericellular matrix (PCM), together called the chondron (Chn). The main collagen in the PCM is type VI collagen, which plays an important role in the Chn's integrity and cell signaling between the Chy and the extracellular matrix.9–11
Using these Chns, considered as the functional unit of articular cartilage, may be advantageous for cartilage regeneration. Using an enzymatic isolation method developed by Lee et al., 12 it has been shown that the PCM of the Chy can be preserved. This resulted in a significant increase of matrix production and led to a mechanically functional neocartilage construct in vitro.13–15 During ACI in osteoarthritic (OA) patients, human OA Chns, obtained using a 5-h (5H) isolation method with 0.18% Dispase II and 0.2% collagenase, outperformed OA Chy in terms of cell survival and biosynthesis, 16 which can be explained by the protecting and mechanosensing role of the PCM.10,17,18 Autologous Chns, obtained with a rapid isolation method for 45 min, mixed with MSCs showed to have similar or even superior outcomes compared to ACI, suggesting a stimulatory effect of MSCs on Chns in regenerating cartilage.19,20
Not only a significant increase in GAG content and GAG/DNA was found when Chns were cocultured with MSCs compared to Chy cocultured with MSCs in vitro but also the loss of type VI collagen of the PCM of the Chns when cocultured with MSCs was reduced.21,22 Similarly, coculturing Chns and Chy in a 1:1 ratio led to increased levels of aggrecan and type II collagen gene expression and GAG production in vitro and improved outcomes after implantation. 23 Using Chns over Chy in cartilage regenerative strategies is therefore hypothesized to provide superior outcomes.
As previously described, Chns are mostly isolated using enzymatic digestion of cartilage tissue. The current gold standard for isolation of Chns from articular was described by Lee et al. in 1997, proposing a mild 5H enzymatic isolation protocol using a mix of dispase and collagenase type II. 12 However, both Chn and Chy isolation methods result in a heterogeneous mix of Chns and Chy, respectively; 62% and 29% of cells were Col VI positive. 17 Although this heterogeneity can influence their use, the properties and compositions of these cell populations have never been thoroughly characterized. The aim of this study is to compare enzymatic isolation methods for Chns and Chy and characterize the results based on isolation efficiency and structural quality of the PCM.
Materials and Methods
Experimental design
Cell populations resulting from different enzymatic isolation methods were analyzed using type VI collagen immunostaining to discriminate Chns (with PCM) from Chy (without PCM). Flow cytometry was used to collect quantitative data of the population composition and to sort the different cell populations. The stained cells were visualized using microscopy to assess the structure of the PCM. In addition, the sorted cell populations were imaged to validate the composition of the cell populations identified with flow cytometry.
Cell isolation
Articular Chy and Chns were isolated from fresh bovine articular cartilage from multiple donors (n = 4, age 8–12 months from slaughterhouse material). The usage of this slaughterhouse material for research purposes was not subjected to the Medical Research Involving Human Subjects Act or subject to IRB approval. The protocols were consistent with the EC regulations 1069/2009 regarding slaughterhouse animal material for diagnosis and research as supervised by the Dutch Government (Dutch Ministry of Agriculture, Nature and Food Quality) and were approved by the associated legal authorities of animal welfare (Food and Consumer Product Safety Authority). Articular cartilage was harvested from the metacarpophalangeal joint, weighted, minced (1–3 mm), and divided into three groups. Chy were isolated by overnight (ON) enzymatic digestion at 37°C in 0.15% collagenase type II (LS0004174; Worthington Biochemical Corporation, Lakewood, NJ) and 0.01% hyaluronidase (CAS 37326-33-3; Sigma-Aldrich, Zwijndrecht, the Netherlands) in Dulbecco's modified Eagle's medium (DMEM, 41966-029, Gibco™; Thermo Fisher Scientific, Landsmeer, the Netherlands), supplemented with 10% fetal bovine serum (FBS, BCBV7611; Sigma-Aldrich) and 1% penicillin/streptomycin (P/S, 15070063; Thermo Fisher Scientific). Chns were isolated using an ON (Chn ON) or a 5H (Chn 5H) protocol, 12 using 0.15% dispase II (17105-041, Gibco) and 0.1% collagenase type II in DMEM, supplemented with 1% P/S or 0.3% dispase and 0.2% collagenase II in DMEM, supplemented with 1% P/S, respectively.
Undigested tissue remnants were filtered out using a 70 or 100 μm cell strainer (542070/542000; Greiner Bio-One, Alphen aan den Rijn, the Netherlands) for the Chy and Chn isolations, respectively. All cells were washed twice with phosphate-buffered saline (PBS, P4417; Sigma-Aldrich), and cell counts were measured using a NucleoCounter® NC-100™ (ChemoMetec, Allerod, Denmark), according to the standard manufacturer protocol, to determine the total number of cells obtained from each isolation. The cell yield was calculated by dividing the total number of cells by the weight of cartilage of each sample. The collected cells were centrifuged at 150 g for 5 min and kept ON in a loose pellet culture in a 50 mL Falcon tube with DMEM, supplemented with 10% FBS and 1% P/S, at 4°C.
Type VI collagen staining
To analyze the cell populations, all cells were labeled with a conjugated type VI collagen antibody. Cells were resuspended after loose pellet culture and washed with 3% bovine serum albumin (BSA, 10735086001; Sigma-Aldrich) in Ca++ and Mg++ free PBS (DPBS, 14190250, Gibco). Cells (106) were labeled with fluorescein isothiocyanate (FITC)-conjugated rabbit anti-type VI collagen polyclonal antibody (50-199-23-71; Life Technologies Europe, Bleiswijk, the Netherlands) at a dilution of 1:100 for 30 min at 4°C. Cells were washed twice using the BSA/DPBS solution and were finally resuspended in 500 μL of 0.5% BSA and 2 mM ethylenediaminetetraacetic acid in DPBS, resulting in a final concentration of 2 × 106 cells/mL.
A positive control for the type VI collagen staining was obtained using bovine cartilage tissue cryosections of 5 μm thickness.
Flow cytometry
To analyze and sort the full cell population, flow cytometry was used. Before sample analysis, 1 μL of propidium iodide (PI, P4864; Sigma-Aldrich) was added to selected samples to discriminate between viable and dead cells upon analysis. Flow cytometry was performed using a fluorescence-activated cell sorting (FACS) Aria III (BD Biosciences, Franklin Lakes, NJ). Of each sample a total of 10,000 events were analyzed.
To validate the PCM structure of the present cell populations, cell sorting was performed with the FACS AriaIII based on the intensity of the type VI collagen staining. Using a type VI collagen-FITC-A, three distinct cell populations could be defined. The cells were collected into Eppendorf tubes containing 500 μL DMEM and were kept at 4°C until analysis. After sorting, the resulting cell populations were characterized with flow cytometry.
Flow cytometry data were analyzed with FlowJo (BD Biosciences v10.6.1). First, cells were separated from debris using a forward scatter (FSC)/side scatter dot plot (Fig. 1A). Dead cells marked by the PI staining were excluded from analysis (Fig. 1B). Based on the intensity of the type VI collagen staining, the cells were characterized as Chy or Chns (Fig. 1D). Within the type VI collagen positive cell population, two separate populations of Chns with low- (partial) and high-intensity (intact) type VI collagen staining were found (Fig. 1E). The relationship between FSC-A and FSC-H was used to define single-cell and multicellular events (Fig. 1C). To obtain the cell yield of all analyzed samples, the results of the flow cytometry analysis were normalized to the calculated cell yield.
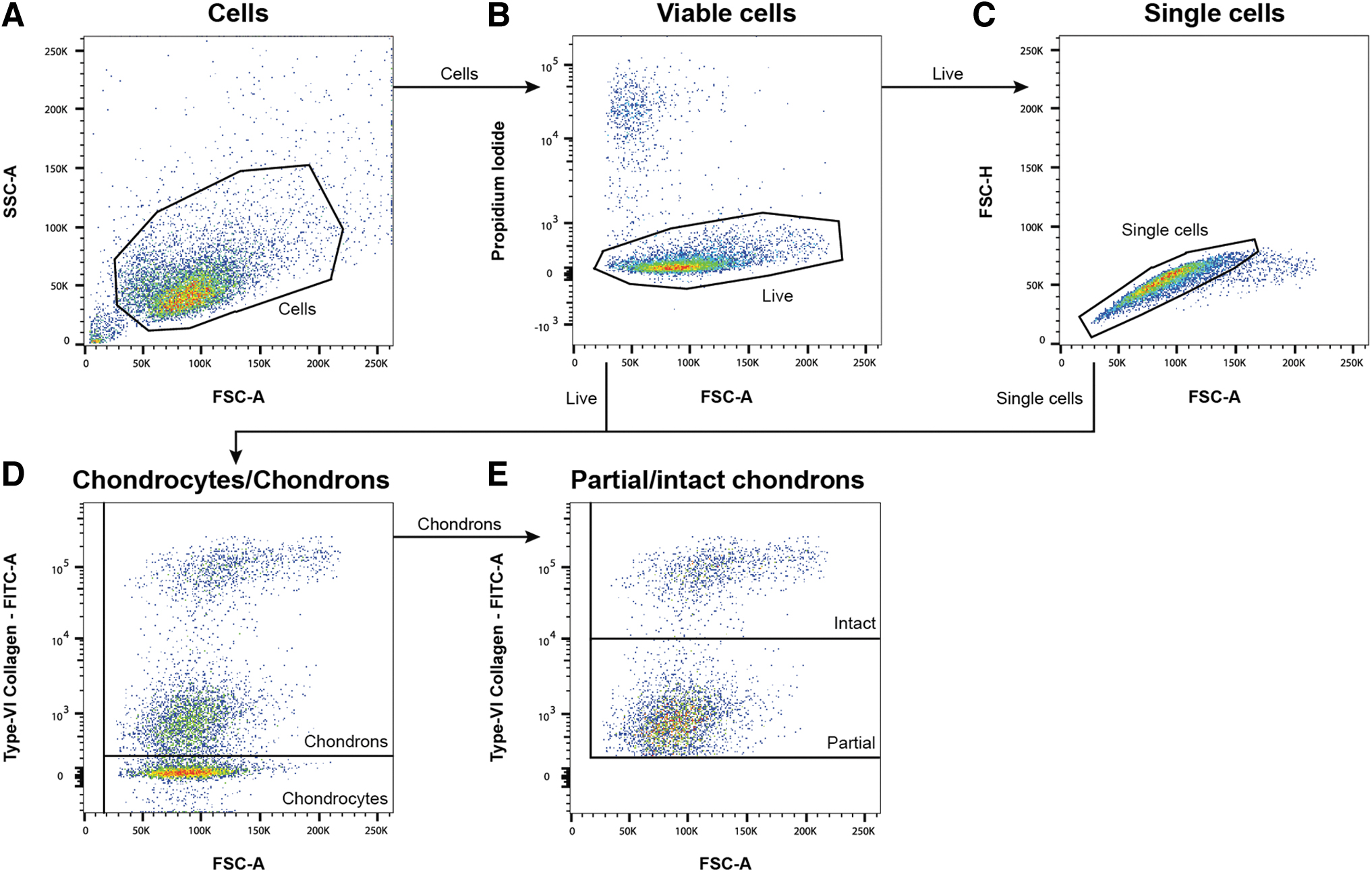
Gating strategy of flow cytometry data analysis. First, cells were separated from debris
Microscopy
To analyze the structural integrity of the PCM, the type VI collagen stained cells were visualized using microscopy. Following the staining, cells were fixed in suspension using 1 mL of 4% paraformaldehyde at room temperature for 15 min. Cells were washed with DPBS and permeabilized with 0.5% Triton X-100 in PBS for 10 min. Cells were washed with DPBS, cell nuclei were stained with 4′,6-diamidino-2-phenylindole at a 1:500 dilution, and cells were resuspended at a concentration of 107 cells/mL. Before visualization, a droplet of cell suspension was put on a microscopy slide and covered with a cover glass. A general overview of PCM structure was obtained using widefield fluorescent microscopy (20 × , 0.4 NA, Axio Observer 7; Zeiss, Oberkochen, Germany), and detailed images were obtained with confocal laser scanning microscopy (40 × , 1.1 NA, SP5X; Leica Microsystems, Wetzlar, Germany).
Statistical analyses
Data are presented as mean (±standard deviation). To test for normality, a Shapiro–Wilk test was done for all datasets. For comparisons between digestion methods when analyzing cell yield, viability, and different cell populations, a two-way analysis of variance (ANOVA) with Tukey post hoc test was performed with the significance level set to p < 0.05. All statistical analyses were performed with R (v4.1.1) and GraphPad Prism (v 8.0.2; San Diego, CA).
Experiment
Five-hour Chn isolation had a much lower cell yield compared to other isolation methods
Articular Chy and Chns were successfully isolated from bovine articular cartilage. Cell yield of each isolation method, determined using the weight of the cartilage before digestion and the resulting cell count, was significantly different. The total cell yield for the Chy method was 14.2 × 106 cells/g cartilage (±4.1 × 106; n = 4), Chn ON was 20.1 × 106 cells/g cartilage (±2.1 × 106; n = 4), and Chn 5H was 2.8 × 106 cells/g cartilage (±1.8 × 106; n = 4) (Fig. 2). The cell yield of Chn 5H was an order of magnitude lower compared to both Chy (p < 0.001) and Chn ON (p < 0.0001). Cell viability, based on PI staining (Fig. 3), was highest for the Chy method (91.2 ± 3.27%; n = 4), which was significantly higher compared to Chn 5H (77.2 ± 8.42%; n = 4; p < 0.05). There were no significant differences between the viability of Chn ON (78.8 ± 15.2%; n = 4) and the other two isolation methods.
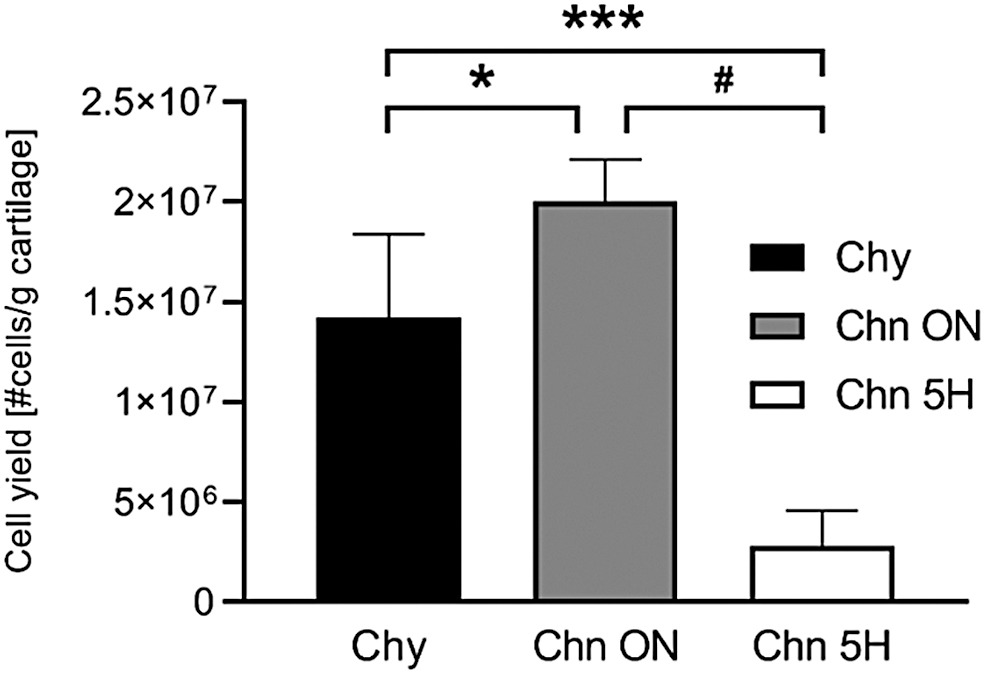
Cell yield after Chy isolation using 0.15% collagenase type II and 0.01% hyaluronidase in DMEM, supplemented with 10% FBS and 1% P/S ON and Chn isolation using 0.15% dispase II and 0.1% collagenase type II in DMEM, supplemented with 1% P/S ON or 0.3% dispase and 0.2% collagenase II in DMEM, supplemented with 1% P/S for 5H. *p < 0.05; **p < 0.01; ***p < 0.001; #p < 0.0001. 5H, 5 h; Chn, chondron; DMEM, Dulbecco's modified Eagle's medium; FBS, fetal bovine serum; ON, overnight; P/S, penicillin/streptomycin.
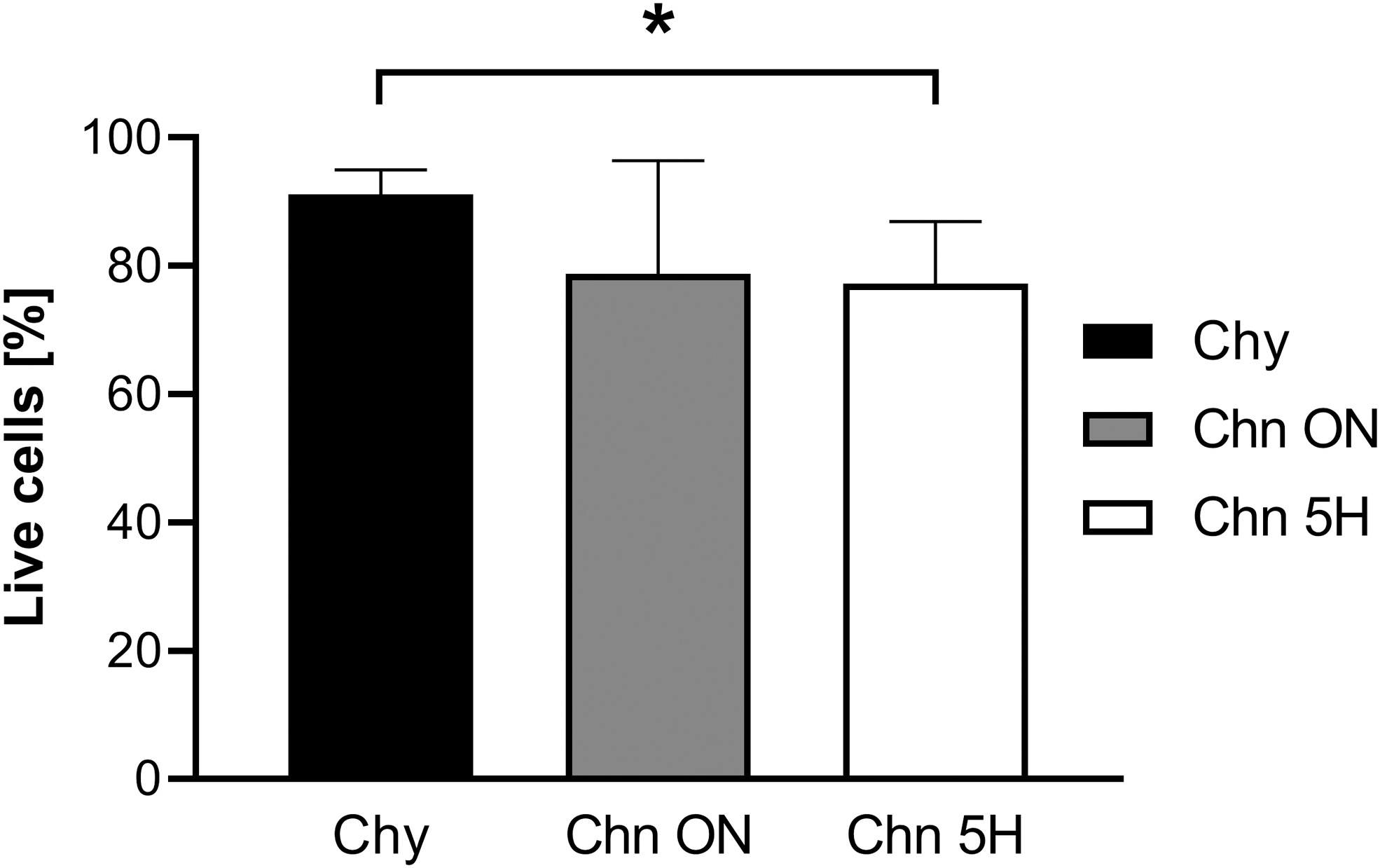
Flow cytometry analysis of PI staining shows lower cell viability when using the Chn 5H isolation method. *p < 0.05; **p < 0.01; ***p < 0.001; #p < 0.0001. PI, propidium iodide.
Enzymatic digestion of articular cartilage results in a heterogeneous mixture of Chns (partial and intact) and Chy; efficiency depends on the isolation method
All isolation methods resulted in a heterogeneous mixture of Chy and Chns. Three cell populations could be defined with flow cytometry, based on the intensity of the type VI collagen staining (Fig. 4A–C). The group containing the lowest intensity of type VI collagen was identified as Chy, and classified as the “type VI collagen negative (Col-VI neg) population.” The low-intensity and high-intensity type VI collagen positive cells were classified as partial and intact Chns, respectively.
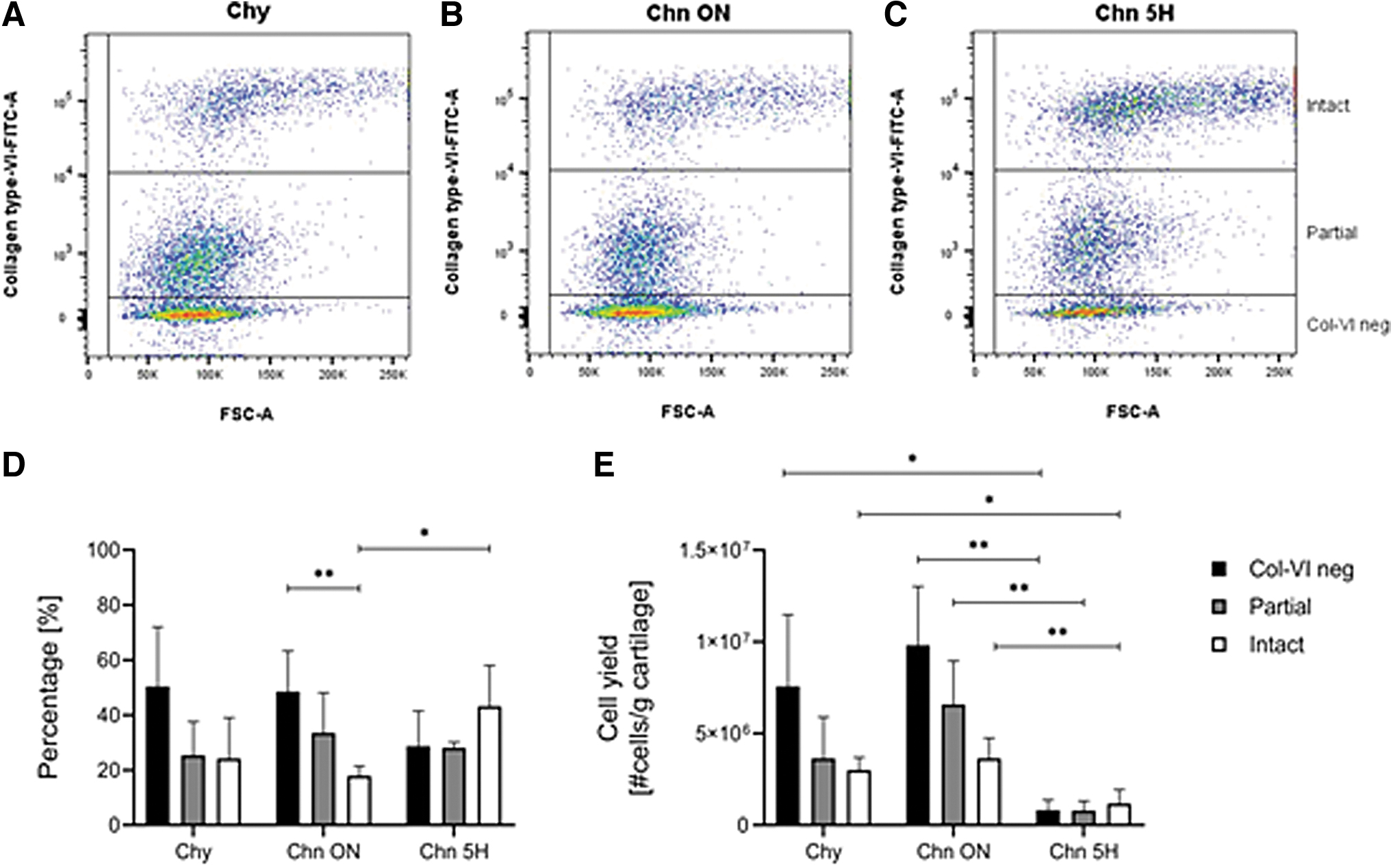
Enzymatic digestion of articular cartilage results in a heterogeneous mixture of Chns (partial and intact) and Chy, efficiency depends on the isolation method. Flow cytometry data of isolation efficiency of the enzymatic isolation methods Chy
Analysis of the flow cytometry data resulted in the percentages of Col-VI neg, partial Chns, and intact Chns after the three isolation methods (Fig. 4D, Table 1). The Chn 5H isolation method resulted in the highest percentage of intact Chns, which was significantly higher compared to the Chn ON isolation method (p < 0.05). Chn ON resulted in significantly more Col-VI negative cells than intact Chns (p < 0.01). No other significant differences were found between the isolation methods (p > 0.05).
Summary of the Percentage of Col-VI Negative, Partial Chondrons, and Intact Chondrons After All Three Isolation Methods
Data are shown as mean (±SD).
5H, 5 h; Chn, chondron; Chy, chondrocytes; ON, overnight; SD, standard deviation.
Normalization of these data to the yield of each sample resulted in the yield of each population after the three isolation methods (Fig. 4E, Table 2). When considering the yield of the isolation methods, the relative distribution over the three isolation methods changed (Fig. 4E). All three cell populations were significantly lower after Chn 5H isolation compared to Chn ON (p < 0.01). When comparing the Chy and Chn 5H isolations, the Col-VI neg and intact Chn population was significantly lower after the Chn 5H isolation (p < 0.05).
Summary of the Total Number of Cells and the Number of Col-VI Negative Cells, Partial Chondrons, and Intact Chondrons After All Three Isolation Methods, per Gram of Cartilage Tissue Used for Isolation
Data are shown as mean (±SD).
To validate our assumption on the existence of three different populations (intact and partial Chns, and Chy), immunocytochemistry was performed after the different isolation methods. This indeed showed a heterogeneous mixture of Chns and Chy. Overall, Chy (Fig. 5A, D) and Chn ON (Fig. 5B, E) showed more Chy without a PCM compared to the Chn 5H method (Fig. 5C, F). Besides intact Chns, partially digested Chns were observed, with PCMs not fully encapsulating the cell. This was observed after all isolation protocols in all conditions.
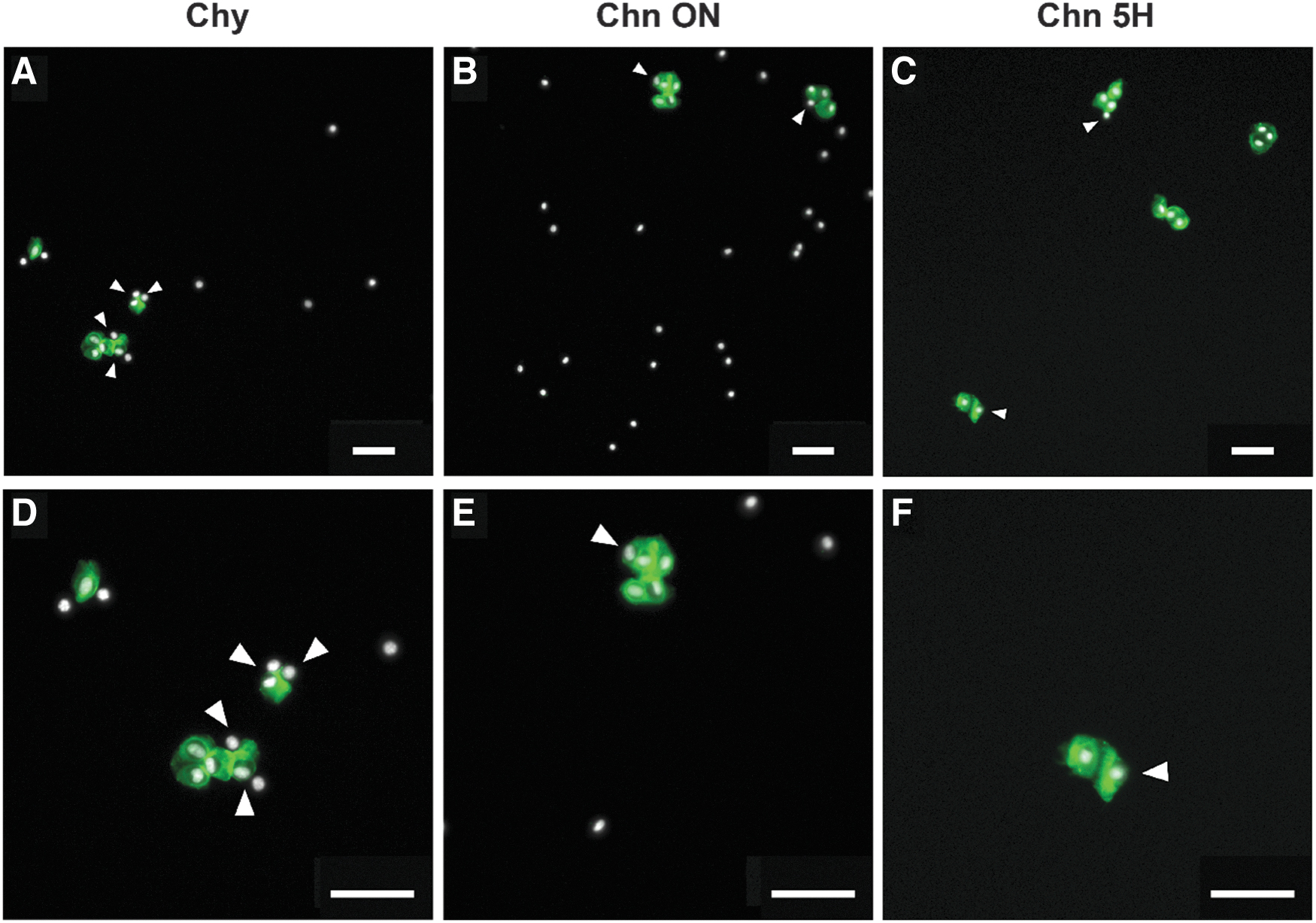
Immunocytochemistry of cell populations obtained after different methods of enzymatic isolation shows a heterogeneous mixture of Chns and Chy.
Intact Chns mainly consist of multicellular Chy clusters
Microscopy data revealed the existence of single cells and clustered cells, which could indicate the regional origin of the Chn. Using the relationship between the FSC-A and FSC-H of the flow cytometry data, the events with a single cell and multiple cells were analyzed. All isolation methods resulted in a similar distribution of events and mixture of single-cell and multicellular events (Fig. 6A–C). Previously, the flow cytometry data showed that there was a heterogeneous mixture of type VI collagen negative, partial Chns, and intact Chns (Figs. 2 and 4, Tables 1 and 2). When combining the values of FSC-H, FSC-A, and intensity of the type VI collagen staining (FITC-A), the distribution of the three groups (Chy, partial and intact Chns) was very different when comparing the single-cell and multicellular events (Fig. 6D, E, Tables 3 and 4).
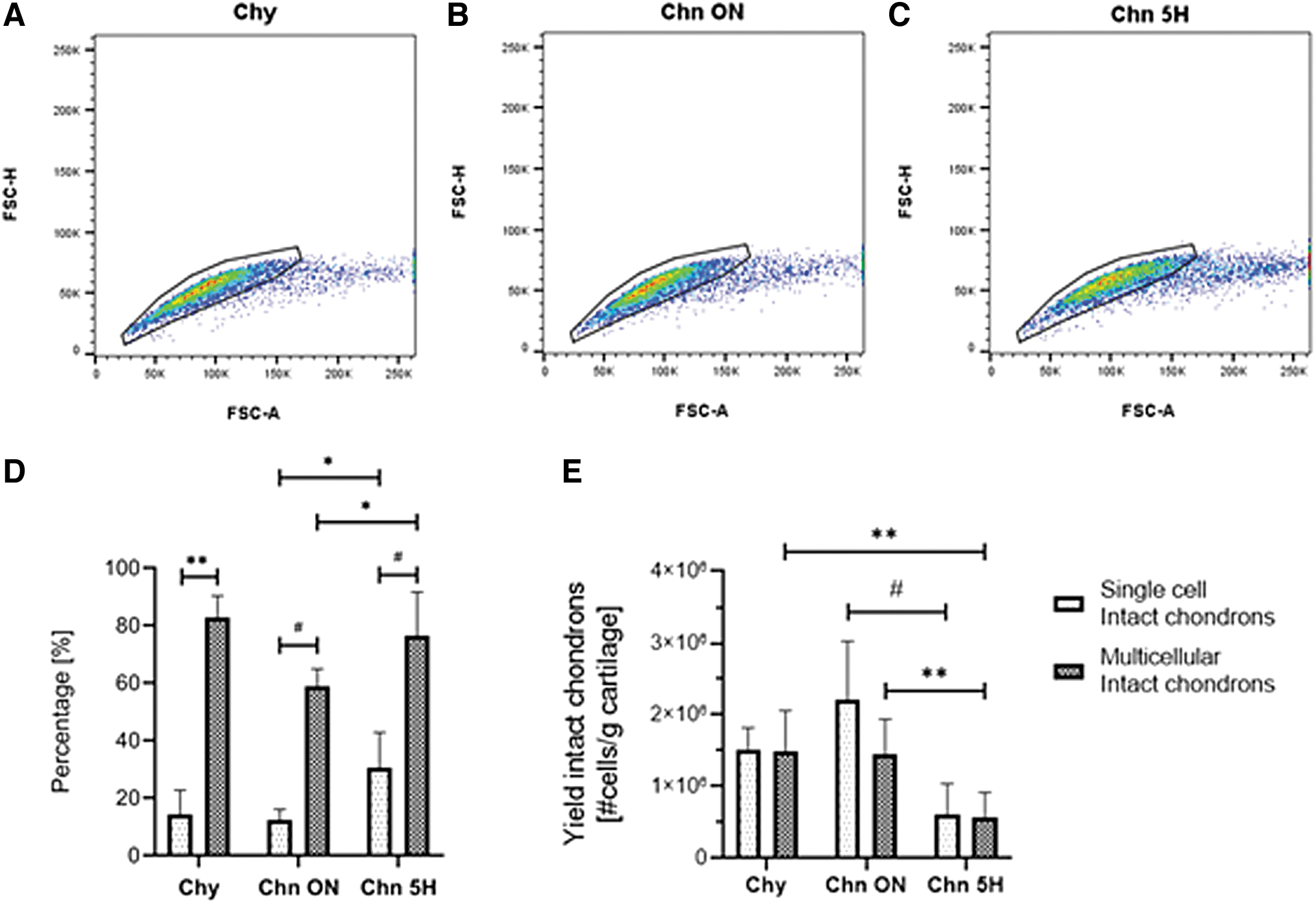
Intact Chns mainly consist of multicellular Chns. Selection of single-cell and multicellular events in the flow cytometry data using the relationship between FSC-A and FSC-H after the Chy
Summary of the Percentage of Col-VI Negative, Partial Chondrons, and Intact Chondrons in the Single-Cell and Multicell Populations After All Three Isolation Methods
Data are shown as mean (±SD).
Summary of the Number of Col-VI Negative, Partial Chondrons, and Intact Chondrons in the Single-Cell and Multicell Populations After All Three Isolation Methods
Data are shown as mean (±SD).
When comparing the percentages of intact Chns in these populations, there was a significantly higher percentage of intact Chns in the multicellular population compared to the single-cell population for all isolation methods (Fig. 6D, Table 3). This difference was smaller for the Chn 5H isolation (p < 0.01) than for the Chy (p < 0.0001) and Chn ON (p < 0.0001) isolations. When comparing the isolation methods, there were significantly more intact Chns in both the single-cell (p < 0.05) and multicellular (p < 0.05) populations after the Chn 5H isolation compared to the Chn ON isolation.
To get insight into the yield of the isolation methods, the flow cytometry data were normalized to the number of cells yielded per gram of cartilage (Fig. 6E, Table 4). This resulted in a shift of the distributions among the populations and isolation methods due to differences in yields between donors. There were no significant differences between the amount of intact Chns in the single-cell and multicellular populations after all isolation methods (Fig. 6E). Comparison of isolation methods showed that the Chn ON isolation yields significantly more single-cell intact Chns compared to the Chn 5H isolation (p < 0.0001). In addition, significantly more multicellular intact Chns were yielded by both the Chy (p < 0.01) and Chn ON (p < 0.01) isolations than by the Chn 5H isolation.
Fluorescence-activated cell sorting shows the different cell populations present after enzymatic digestion of articular cartilage
Finally, a cell sorting procedure was adopted to confirm the validity of our classification system, that is, the presence of three distinctly different groups (Chy, and Chns with a partial or intact PCM). Therefore, the three cell populations were sorted based on the intensity of the type VI collagen staining (to note, only the Chy isolation procedure was used for this validation step, as a proof of concept). The settings used for sorting resulted in a purity between 98% and 99%. FITC histograms, based on type VI collagen intensity, reveal that there was some overlap to the other groups after sorting (Fig. 7A, D, G, J).
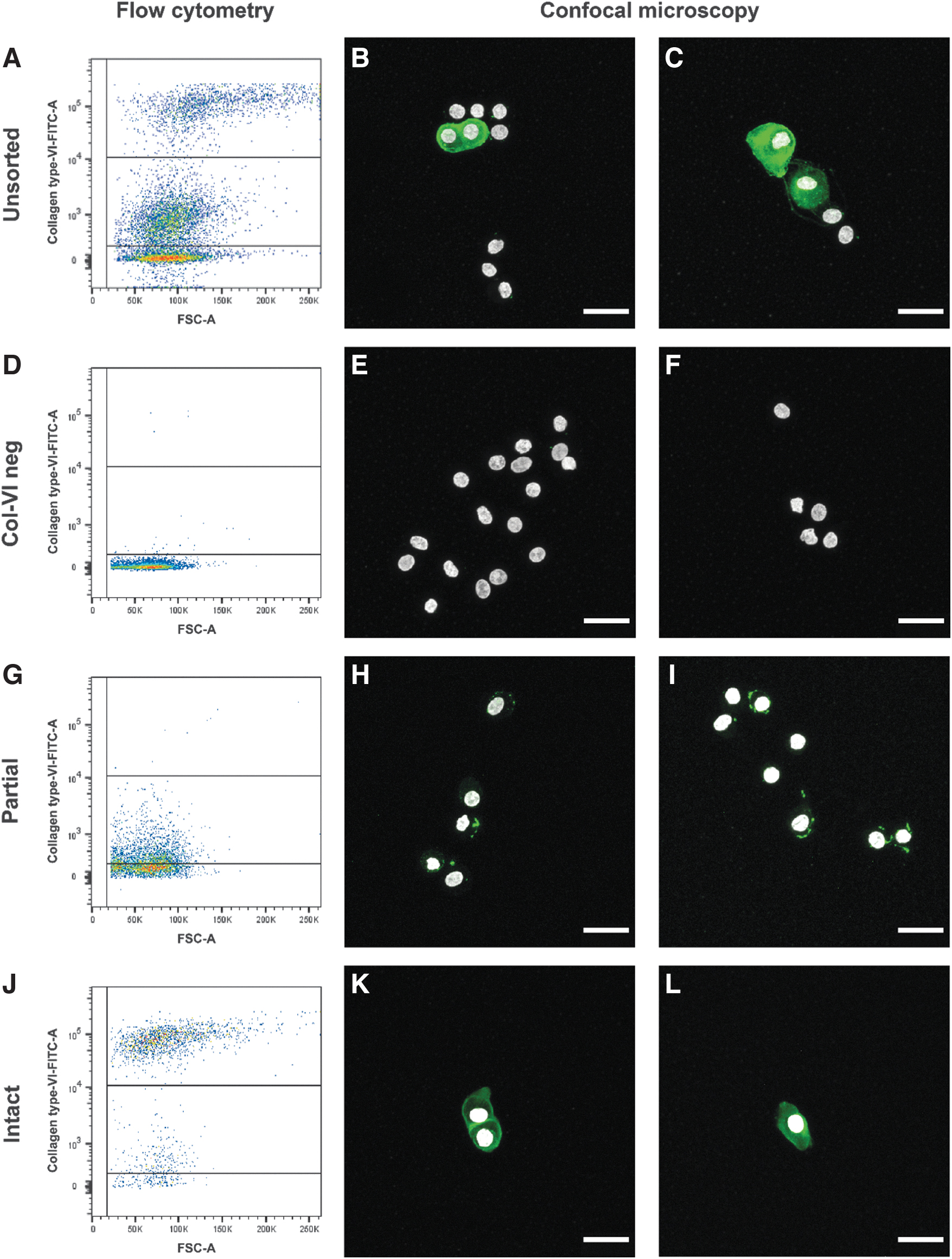
FACS can separate three distinctly different cell populations based on type VI collagen staining, which can be confirmed with confocal microscopy. Flow cytometry analysis before
Immunocytochemistry confirmed that FACS could separate the cell populations. A heterogeneous population for the unsorted cell population was observed (Fig. 7B, C), as shown before (Fig. 5). Confocal microscopy showed that characteristics of all three cell populations were present before FACS. After sorting, the type VI collagen negative cells were devoid of type VI collagen staining, although some sporadic remnants of type VI collagen could be observed (Fig. 7E, F). The partial Chns showed small type VI collagen rich PCM but this did not envelop the cells (Fig. 7G–I). The population of intact Chns showed a thin PCM enveloping the cells and mainly consisted of clustered Chns (Fig. 7K) with also some single Chns (Fig. 7L).
Discussion
This study quantified the presence of Chy and Chns within cell populations obtained after different enzymatic isolation protocols to digest articular cartilage, which were designed to preferentially isolate either Chy or Chns. It was shown that all isolation methods result in a heterogeneous mixture of Chns and Chy, using a combination of flow cytometry and microscopy. A new insight was that two populations of type VI collagen positive cells exist, which were considered to be partial and intact Chns. Using FACS-sorting, a homogenous population of intact Chns was isolated from bovine articular cartilage for all three different enzymatic isolation methods using a type VI collagen staining, however leading to a very low cell yield.
Peters et al., already revealed the heterogeneity of the isolated Chn/Chy population using the Lee protocol. 17 They however did not show the population of partial Chns as shown in this research. When adding up the partial and intact Chns, the percentage of Chns from the current study are similar to that of Peters et al., 17 meaning that this partial population was likely present in the study of Peters. Moreover, the current study showed the presence of a Chn population containing multiple Chy within the PCM. Within this group of multicellular Chns, most type VI collagen positive cells were grouped as intact Chns (Fig. 6D, Tables 3 and 4).
Although these methods have existed since decades, this outcome is not surprising. Enzymatic isolation is a variable process and is highly dependent on the number of active units, size of the tissue to be digested, the exact composition of the ECM, and digestion time. The data of this study suggest that certain regions of the cartilage tissue are either under- or overrepresented in the Chn population, since not all parts of the tissue will digest at an equal rate. Considering that Chy from different cartilage zones express particular phenotypes, 24 this could potentially affect study outcomes. Uncovering the native zonal location of Chns obtained using enzymatic isolation could improve the interpretation of future studies.
From the current study, it is clear that without proper sorting of enzymatically isolated cells, a mixture of Chns and Chy always exists, the exact ratio depending on the used isolation protocol. This heterogeneous mixture is believed to influence the interpretation of the results of previous studies. When a pure population of Chns is compared to Chy, the beneficial results of Chns are expected to become more pronounced. However, the specific application is key, as earlier studies have shown that the ratio between cell types when coculturing Chns with Chy or MSCs affects the outcomes both in vitro and in vivo.19,21–23 This stresses the importance of the homogeneity of the initial cell population and thus the isolation method, as the underlying mechanisms remain elusive. Moreover, using Chns to study cell-PCM interactions on cellular level can also benefit from the suggested isolation method presented in this current study.
For the application in cartilage tissue engineering, high yields are required to reach the necessary high cell seeding densities.25–27 From the current study, it is suggested to use the ON Chn isolation method. With the addition of FACS sorting, based on type VI collagen staining, a cell yield of ∼3–4 × 106 cells/g cartilage tissue (Fig. 4E, Table 2) could be attained. Since this is a pure population of Chns, it would be suitable for in vitro studies focusing on Chns and ECM production. Moreover, the use of FACS is also highly recommended for single cell studies and more fundamental research.
However, with clinical applications like ACI, only a small cartilage piece is available, and unfortunately, the yield of the combination of this isolation method and purification steps is too low for these purposes. 28 This low yield has been an issue from the introduction of the enzymatic procedure of isolating Chns by Lee et al., back in 1997. 12 Nonetheless, the use of Chns in cartilage tissue engineering do result in superior outcomes compared to only Chy or MSCs.19,21–23 Different methods to obtain Chns which increase purity and yield should therefore be explored. Since the available digestive enzymes are not specific enough to digest only the cartilage ECM components, a tissue engineering approach could be a favorable alternative to the current methods. Chy are known to form a PCM during in vitro culture when embedded in agarose and hyaluronic acid hydrogels.29,30 When this process is optimized, tissue engineered Chns could be a more homogeneous cell source compared to enzymatically isolated Chns.
In this study, partial Chns were defined as cells with a low-intensity type VI collagen staining. As can be seen after cell sorting, this population of cells has small remnants of PCM attached to the cell. It is however questionable whether these cells should be classified as Chns or Chy. In their study, Hing et al. 31 observed cells with similar punctuates of type VI collagen, which were indicated as Chy. It remains unknown what the effect is of partial PCM digestion on biophysical stimuli in neocartilage formation. Full disruptions of the PCM could have significant effects on Chy when presented with osmotic challenges during joint loading. 31 Since the PCM has a stress-shielding effect on Chy, 32 deformation of Chy in partial Chns could increase during compression. However, since PCM components are still present in a partial Chn, its growth factor modulating role and cell–matrix interactions might still be functional.33–37
In conclusion, this study demonstrated the heterogeneity of cell populations obtained after enzymatic digestion of articular cartilage. It is suggested that an ON Chn isolation protocol results in a superior yield of intact Chns, compared to the gold standard 5H protocol. Cell sorting methods like FACS are suitable for obtaining a purified population of intact Chns. This gives the most homogeneous Chy populations. However, because partial Chns and Chy are filtered out, it also comes with a lower yield. Therefore, this method is less suitable for applications which demand large numbers of cells and have a limited tissue supply, like in vivo studies and clinical applications. For these purposes alternative isolation methods with higher yields may be more applicable. However, this comes at the expense of less homogeneity in the cell population, with larger numbers of partial Chns and Chy. Thus, care must be taken with interpretations of potential beneficial effects of Chns. Increasing the homogeneity of Chn populations could have a positive impact on future in vitro studies investigating cell-PCM interactions and comparisons between Chy and Chns.
Footnotes
Acknowledgment
The authors thank Bart Tiemeijer for providing support during cell sorting.
Authors' Contributions
M.v.M.: conceptualization; formal analysis; investigation; methodology; validation; visualization; writing—original draft; writing—review and editing. G.H.S.: conceptualization; formal analysis; investigation; methodology; validation; visualization; writing—original draft; writing—review and editing. L.P.V.V.: resources; methodology; writing—review and editing. L.A.V.: methodology; writing—review and editing. C.C.v.D.: conceptualization; supervision; writing—review and editing. K.I.: conceptualization; funding acquisition; supervision; writing—review and editing. J.F.: conceptualization; funding acquisition; supervision; writing—review and editing.
Disclosure Statement
No competing financial interests exist.
Funding Information
This research was financially supported by the Gravitation Program “Materials Driven Regeneration,” funded by the Netherlands Organization for Scientific Research (024.003.013), the framework of Chemelot InSciTe (Project MimiCart), supported by the partners of Regenerative Medicine Crossing Borders and powered by Health∼Holland, Top Sector Life Sciences & Health.
