Abstract
Alveolar cleft repair is a key step in multiple disciplinary treatment for patients with cleft lip/and palate. Although autologous bone grafting has been used worldwide over the past half century, alternative advanced techniques, such as the use of bone substitutes and guided tissue regeneration, have shown their great potentials and have been recommended by a growing number of physicians and surgeons. The employment of new therapeutic approaches and devices in clinical routine requires tremendous experimental efforts and appropriate animal models with similar sizes and sites of deformity to that of human both anatomically and physiologically. The aim of this study is to develop a juvenile porcine model with surgically created alveolar clefts imitating congenital alveolar cleft in the cleft lip and palate. Alveolar defects between second incisor and canine were surgically created in two miniature pigs (unilateral cleft in P1 and P2); bilateral alveolar defects were surgically created between first and third incisor in one miniature pig (P3) using piezo surgery. Pigs were sacrificed (P1 at 1 month after the surgery and P2 at 3 months postoperatively) and the evaluation of defects were performed by assessing result from the computed tomography (CT) scan and histopathological examination. Postoperative CT scan results showed that the size of the defect remained the same, whereas the edge of the defect became irregular 3 months after the surgery. In all pig subjects, histopathological examination found no sign of osteogenesis in the area of defect, indicating that our surgical procedure was successful in establishing porcine models for alveolar cleft in congenital cleft lip and palate. In conclusion, we developed alveolar cleft in porcine models to mimic the size, site, and environment of congenital alveolar cleft in cleft lip and palate. The novel animal model can be employed in pilot studies for the purpose of optimizing the current surgical treatment techniques as well as developing new treatment procedures and test the bone substitute materials. The bilateral model can be applied in further control studies.
Impact statement
Cancellous iliac bone graft was the most popular surgical technique as well as the gold standard to reconstruct alveolar cleft. Nevertheless, several disadvantages exist regarding the additional surgical field of donor side and delayed age of alveolar bone grafting. Bone tissue-engineered strategy offers a promising alternative to address the gap in the current limitation of autologous bone to treat the growing craniofacial skeleton. Among different species of laboratory animals, porcine is suitable for oral and maxillofacial bone and implant-related research, where alveolar defect can be surgically developed simulating the size and site of alveolar cleft occurring together with cleft lip and palate. In this proposal, a reproducible porcine model of alveolar bone defect imitating congenital alveolar cleft during craniofacial growing stage is successfully constructed that will show great potential application in the field of tissue engineering and regenerative medicine. The model for bilateral alveolar cleft can be potentially applied in a controlled study in future.
Introduction
Cleft lip and/or palate is a one of the most common congenital malformations in the oral and maxillofacial region. Alveolar cleft is the covert phenotype in cleft lip and palate, which may cause several different types of secondary deformities in patients, including but not limited to: the collapse of nasal floor on the affected side, the discontinuity of the maxilla, misplacement of teeth, and the communication between oral and nasal cavity by oral vestibule fistula. Alveolar defect repair is a crucial middle step in the process of multidisciplinary treatment of cleft lip/and palate.
Although cancellous iliac bone grafting has been commonly used worldwide over the past 50 years and has been considered the gold standard for alveolar cleft repair, the procedure has received criticisms in that it requires a second surgical site (the donor site) and may introduce doner-site complications. Importantly, the procedure cannot be performed in young patients because of the insufficient cancellous bone within the iliac crest in very young children, which may lead to delayed treatment. As children become aware of their appearance at around age 5, this delay of treatment may potentially introduce further psychosocial impact.
There is a growing demand for advanced alternative techniques, including the use of bone substitutes and guided tissue regeneration. The potential contributions that tissue engineering proffers for children afflicted with congenital craniofacial malformations are significant. 1 Bone tissue-engineered strategies offer a gap-filling alternative to remedy the limitation of autologous bone grafting for the surgical treatment of the growing craniofacial skeleton. The operation can be performed without a secondary donor-site injury, and it is no longer limited by the age of children or the size of defect.
To evaluate the usability of bone institutes as alternatives for the current gold standard method in alveolar cleft surgeries, their safety and efficacy must be carefully tested and validated. The selection of suitable animal species, as well as the establishment of appropriate surgical models, are quite complex processes since the clinical demands vary and the models must be built based on the particularities of different circumstances. In other words, human and the selected animals should share anatomical and physiological similarities; the laboratory animal models must present the “right” anatomic, histopathologic, and clinicopathologic features that meet the specific research demands.
Pig has been widely used as animal models in maxillofacial surgeries since they show similar physical and biological properties to those of humans.2,3 A systematic review by Mardas et al. provided critical appraisal of the literature regarding porcine models covering the evaluation of craniomaxillofacial osseous defect healing to regeneration. 4
Despite the rich data documentation, research on animal models for alveolar cleft is rarely reported. Alveolar cleft is one of the congenital malformations that appears in cleft lip and palate, which differs from the osteoid lesion caused by trauma, dissection of tumor, or other acquired defects as reported in previous literature. The aim of this study is to develop juvenile porcine models with surgically created alveolar clefts imitating congenital alveolar cleft in cleft lip and palate patients to simulate the defect location, dentition situation, the defect environment, and the stage of craniofacial growing in human.
Overview of the Procedure
A schematic diagram demonstrating the experimental design was provided in Figure 1. Bama miniature pigs with an average age of 5 months (±1 month) were selected as our animal subjects. Two animal models were created with their alveolar cleft surgically developed on the left side; a third model was generated wherein the alveolar cleft was surgically created for both sides. The two unilateral models were computed tomography (CT) scanned at 1 month (P1) and 3 months (P2) after the surgery, respectively. Histological examination was performed following the execution right after the CT scan. The bilateral alveolar cleft model (P3) underwent CT examination immediately after the surgery. A second scan was also performed 1 month after the surgery under general anesthesia, to observe the morphology change of the bone defect.
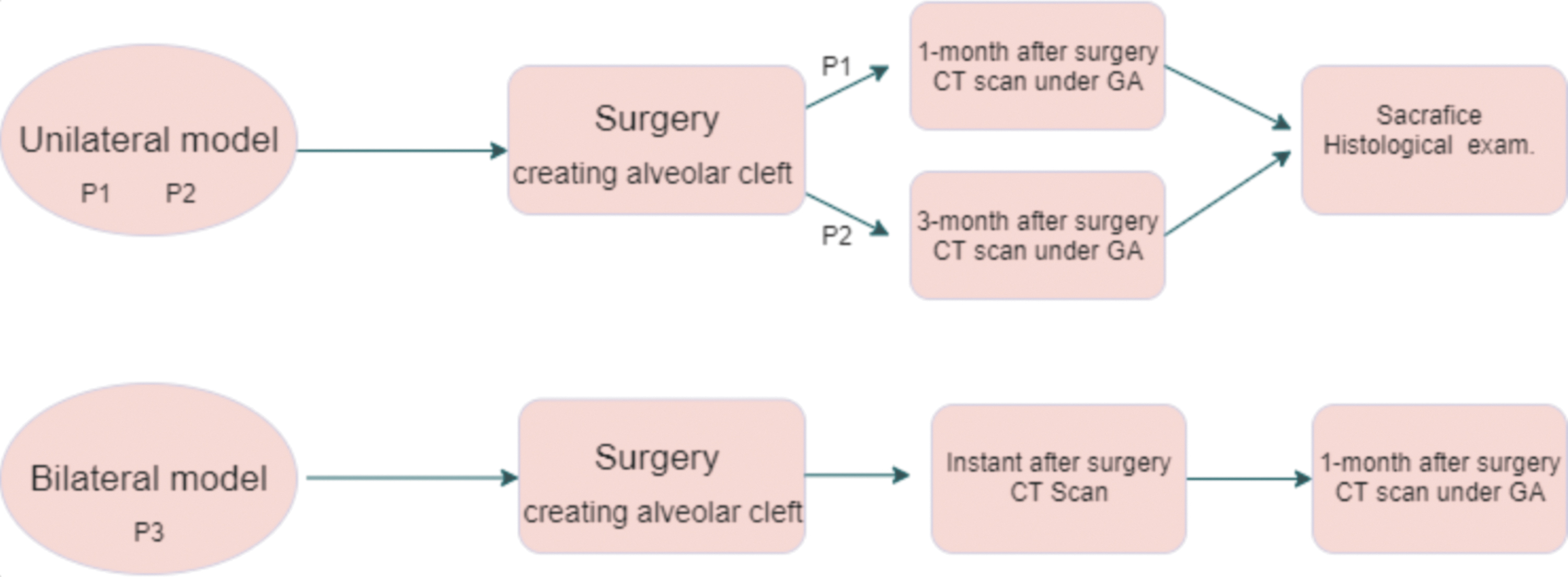
Overview of the procedure. CT, computed tomography. Color images are available online.
Experimental Design
Selection of the animal
The validation of new therapeutic approaches and medical devices requires great amount of animal testing. It is essential that researchers choose the appropriate animal model that have anatomical and physiological similarity to human in size, site, and their nature of deformity. Selection of the appropriate animal model is therefore considered the essential preliminary step in the field of tissue engineering, as well as the most fundamental step in developing new surgical techniques for oral and facial reconstructive surgeries.
Ovine is one of the commonly used animals for establishing the surgical models. Research conducted by Watson et al. reported a reproducible ovine model of in vivo bioreactor technology toward customized bone generation, but only mandibular defect creation was mentioned. 5 However, ovine is not a proper choice imitating the congenital alveolar cleft because of their upper dentition structures: (i) there is no incisors and canine in the anterior part of the palate, only palate plate instead. (ii) The upper deciduous and permanent incisors in ovine are absent and replaced by a dental pad providing an occlusion surface for mandibular incisors. (iii) The upper canines are absent during evolution. 6 In this study, we chose pig instead of ovine as our animal subject. The difference of premaxilla between ovine and pig is shown in Figure 2.7,8
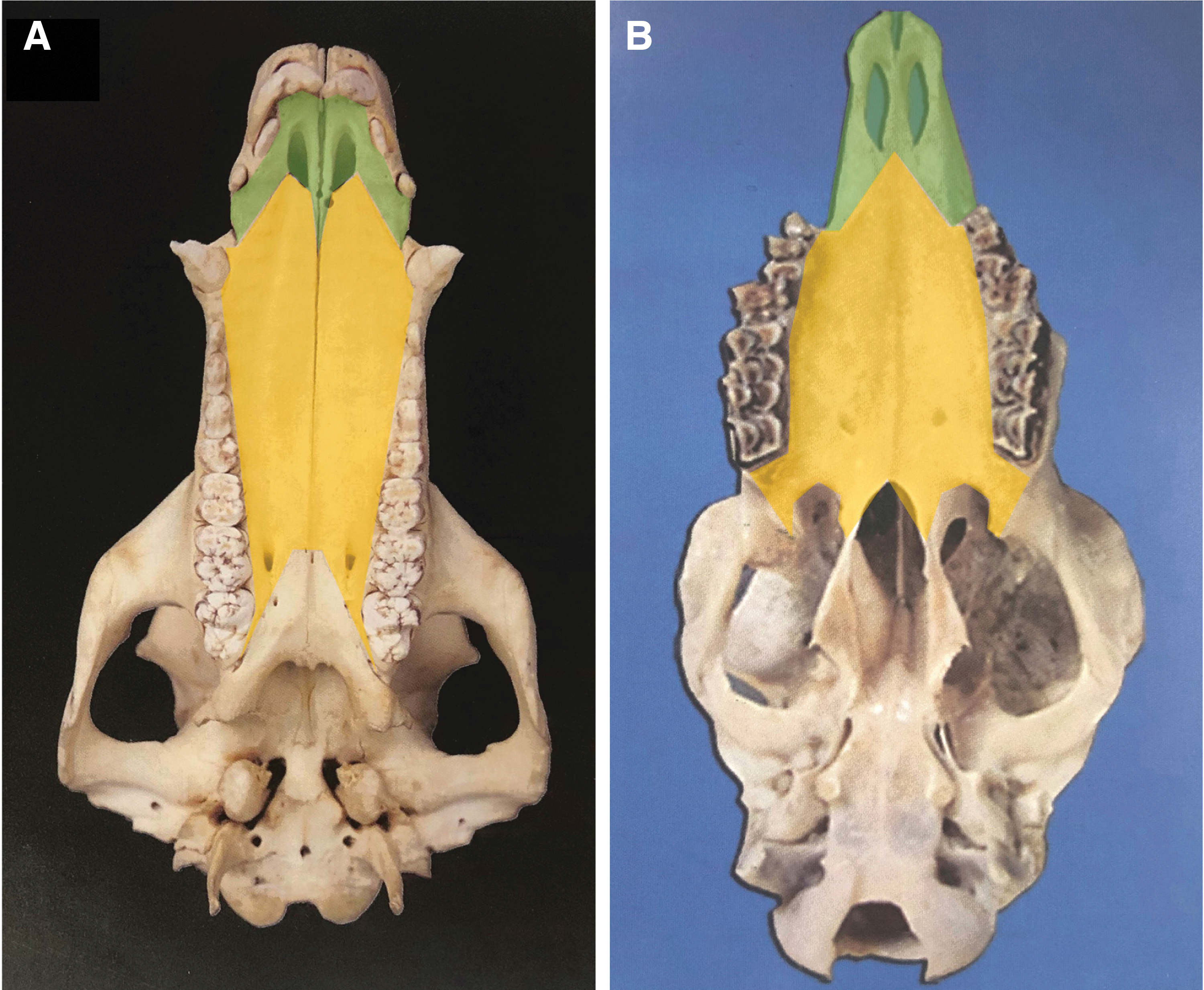
Palatal view of craniomaxillary skull in adult pig
Because of the similarities between human and pig skeleton, swine has been considered one of the major animal species used in various research fields, as documented by Martiniakova's group. 4 In our study, three pigs were chosen as the experimental animals, with an average age of 5 to 6 months. Importantly, these animals were selected with the appropriate teeth condition to meet the condition of dentition as well as the age of human patients with cleft lip/and palate at the time of surgical operation (i.e., alveolar bone grafting).
Based on the nature of eruption sequence of deciduous and permanent teeth replacement in human, the relationship between the age of humans and that of the experimental pigs can be estimated; an age of 1 month for pig is equivalent to 1 year for human (Tables 1 and 2).9,10 Figure 3 presents the palatal view of an adult pig skull showing the dentition situation and named teeth. 11
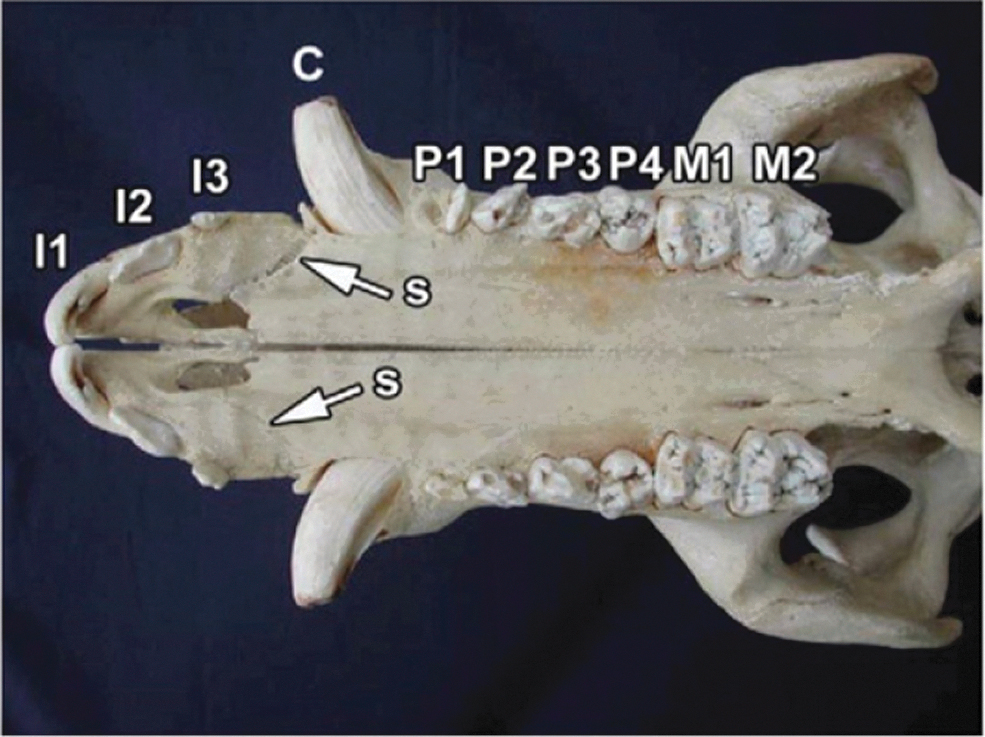
Showing permanent dentition in upper jaw of adult pig skull. S. suture between premaxillary bone and maxillary bone. I: Incisor; P: premolar; M: Molar.
The Eruption Time of Deciduous and Permanent Teeth of Upper Jaw in the Miniature Pig
D: Deciduous; I: incisor; C: canine; P: premolar; M: molar.
The Eruption Time of Deciduous and Permanent Teeth of Upper Jaw in Human
D: Deciduous; I: incisor; C: canine; P: premolar; M: molar.
Surgical creation of alveolar bone defect
Bone defect in congenital alveolar cleft is different from the bone defect caused by trauma or tumor dissection. Presently, in most studies, the bone defect in animal models were developed surgically and reconstructed with bone substitutes or bioreactors simultaneously,12,13 regardless of how small or large the animal is. In this study, we proposed porcine models with unilateral and bilateral alveolar clefts. To bring the environment of surgically created defect closer to that of congenital bone defects, we continue to feed the laboratory pigs for 1–3 months after the surgical operation.
The location of the created defect was similar to the site of congenital alveolar cleft in human. In all porcine models, the three incisors were localized on the premaxilla bone, while canine, premolars, and molars were on the maxillary bone. 11
Unilateral model
The length of surgically created defects ranged from 1.7 to 2.0 cm. The cleavage site was located between the second incisor and canine; the third incisor was removed; the alveolar crest bone was completely removed, and the intact nasal mucosa remained medial and apical.
Bilateral model
The fissure of bilateral alveolar crest deformity was modeled based on the features of incomplete alveolar cleft, which was located between the first and third incisors. We removed the block alveolar bone (maxilla), retained the continuity of the bone on the medial side and floor of the nose, and stabilized the premaxilla.
Material Required
Animal species
Three Bama miniature pigs (labeled as P1, P2, and P3) collected from Bainong Experimental Animal Breeding Technology Co., Ltd. were used for the establishment of unilateral and bilateral alveolar defect models. P1 was male (5–6 months) with a weight of 23.3 kg, P2 was female (5–6 months) with a weight of 25.7 kg, and P3 was male (5–6 months) with a weight of 25.3 kg at the time of alveolar cleft creation.
The surgical team
We collaborated with Beijing TongHe Litai Biotechnology Co., Ltd., and the procedure was performed at the operation room at the company building.
The surgical team involved five qualified medical experts, including two senior oral and maxillofacial surgeons, who were experienced with the surgical procedure for alveolar cleft treatment and alveolar bone grafting, an assistance to assist with the setup of surgical field, a scrubbing nurse, and a coordinator in charge of taking intraoperative photographs and assisting the scrub nurse.
Equipment and instrument
Large animal anesthesia equipment (Tianjin Sendi Hengsheng Technology Development Co., Ltd. Type of machine: SD-M2000).
Ultrasonic osteotome (Beijing Yingjiehua Technology Co., Ltd. Type of machine: GE-350).
Electrocautery system.
Suction equipment.
Sterile gown, gloves, mask, and head cover.
Surgical instruments: two retractors, periosteal elevator, tooth forceps, mosquito forceps, needle holder, tissue scissors, thread scissors, tooth extraction forceps, tooth elevator, and 3–0 coated VICRYL (absorbable thread).
CT scanner and image processing software for three-dimensional (3D) reconstruction (Shanghai United Imaging Health care Co., Ltd.; model: uCT760).
Protocol
Preoperative preparation
Pigs were selected by a cooperative company based on the research goals and demands of the experiments. An acclimation period of 1 week was scheduled allowing the animals to stabilize in the new environment before surgical manipulation. Surgical instruments were sterilized using autoclave and then were cooled at room temperature. Work equipment and devices were examined in advance to ensure normal functioning.
Surgical induction and animal preparation
Food and water were removed from pig 12 h before surgery.
The animal was anesthetized by intramuscular injection of 0.4 ml/kg Beite sumianxin (Changsha Beita Biotechnology Research Institute Co., Ltd.), 125 mg Shutai 50 (Tiletamine hydrochloride), and 125 mg Zolazepam hydrochloride (Vik trading Shanghai Co., Ltd.). The sedated animal was then weighed.
Endotracheal intubation was performed, and the tube was held by ropes around the neck to avoid fall off and was linked with anesthesia machine by an extension tube.
Anesthesia was maintained throughout the operation with isoflurane inhaled (Jiangsu Hengfengqiang Biotechnology Co., Ltd.).
The pig was placed in left lateral decubitus position on the operation table. The pig was secured to the table, and the limbs were tied with ropes.
The face and oral cavity were sterilized with betadine.
Alveolar defect creation (unilateral)
The surgical field was exposed to air, and the incision line was marked (Fig. 4A).
Five microliters of local anesthetics containing 1% Lidocaine (Shanghai Chaohui Pharmaceutical Co., Ltd.) with 1:200,000 epinephrine (Ningbo Second Hormone Factory) was injected before the defect creation procedure, to decrease pain and bleeding during the operation. 6 An incision was made along the marked line and deep down to the surface of bone.
Two buccal and palatal periosteum mucosa membrane were elevated exposing the alveolar in premaxilla region, from second incisor to canine (Fig. 4B).
A line was marked on the bone surface with ultrasonic osteotome to determine the area of osteotomy (Fig. 4C).
Osteotomy was performed using ultrasonic osteotome from the second incisor to canine (about 1.8 cm in length). A trapezoid-shaped bone block was removed while the nasal mucosa and palatal bone plate were kept intact (Fig. 4D, E).
The region of surgically created defect was washed using saline. Bleeding was fully stopped using electrocoagulator.
The buccal periosteum mucosa membrane was released.
The wound was closed with 3–0 VICRYL suture interruptedly (Fig. 4F).
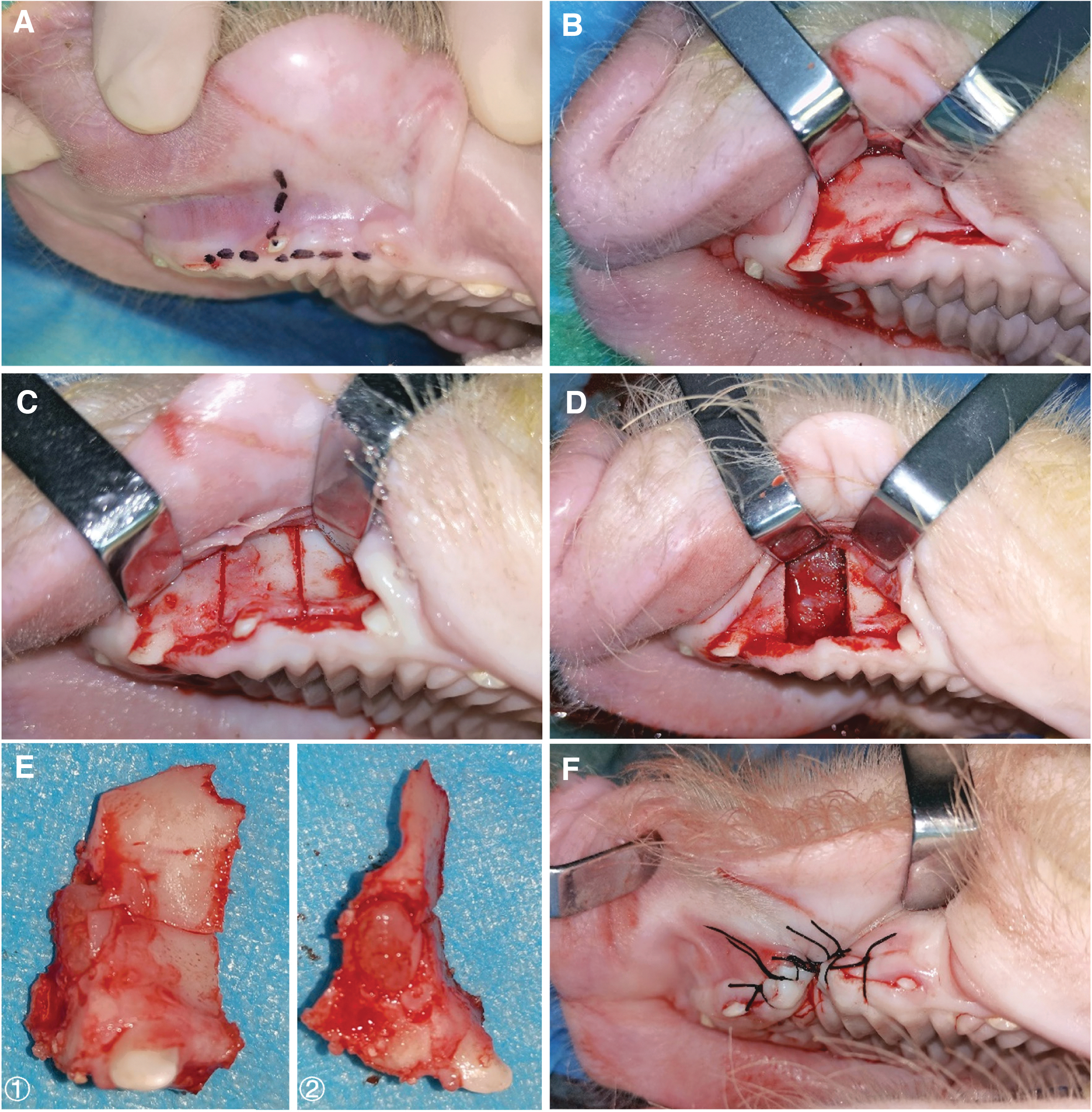
Image of created alveolar defect procedure.
Note: The surgical procedure for bilateral model (P3) creation was the same as the unilateral models (P1 and P2). During the operation, the position of the pig was altered when creating the defect on the contralateral side. The location of the defect in the bilateral model lay between the first and the third incisors and the bone continuity was retained on the medial side and floor of the nose, when the block alveolar bone (maxilla) was removed. Second incisor was extracted.
It is important to note that, the 1.7–2 cm width of the defect created on both sides of the alveolar resulted in loose bone support on the floor of the nose and therefore led to instability of the central anterior maxillary segment. For this reason, the bilateral bone defect model confronted with difficulties regarding postoperative feeding, which is likely to cause death of the experimental animals. Evidence has shown that a baby pig with congenital complete bilateral alveolar cleft exhibits difficulties to survive. 11
Postoperative care
We closely observed the changes of the condition of the pig until the pig resumed normalcy in behavior after awakening from the surgery.
Administer Penicillin G (dose: 800,000 unite) intramuscularly once per day for 5 days postoperatively (CHINO pharmaceutical Group Shijiazhuang Pharmaceutical Co., Ltd., Batch number: 0161909404).
During the first week after the operation, feed pellets were softened by adding water. After this, the animal was fed on a regular hard pellet diet.
Caution: For bilateral alveolar cleft model, special postoperative care should be carried out, including a longer period of soft diet feeding, placing the stomach tube in site if necessary, and paying more attention to nutrition.
Sacrificing and specimen retrieval
Food and water were removed from pig 12 h before sacrifice.
The pig was anesthetized by intramuscular injection of 0.4 mL/kg Beite sumianxin (Changsha Beita Biotechnology Research Institute Co., Ltd.) and Shutai 50 containing 125 mg Tiletamine hydrochloride and 125 mg Zolazepam hydrochloride (Vik trading Shanghai Co., Ltd.) for a CT scan.
The pig was sacrificed through air embolism (100 mL/IV.) under sedation.
Note: the criteria we used to define the death of the animal were the following: (i) cardiac arrest, (ii) pupil dilation, and (iii) systemic cyanosis.
The anterior 2/3 maxilla of the pig, including the region of the created alveolar defect were isolated by an electric saw. The specimen was stored in 10% formalin solution.
Note: P1 and P2 were sacrificed 1 and 3 months after the alveolar cleft was created, respectively. P3 underwent CT scan at 1 month after the operation under sedation since the pig was implanted with orthodontic device for another experiment.
Data analysis
3D reconstruction of the CT scan
Postoperative CT was taken under general anesthesia at 1 month (P1) and 3 months (P2) after the surgery following the execution. For the bilateral cleft model (P3), CT examination was taken immediately after the surgery, and then at 1 month after the surgery to assess the morphological changes of the fissure (if any) and its relationship with the nasal cavity.
Histological examinations
Specimens were decalcified, eosinophilia were stained, hypoploidy observation of tissue composition and cell type at the fissure region and normal tissue junction, and high magnification observation of meaningful areas were done to further determine the type of cell.
Anticipated Results
3D reconstruction of CT scan
Figure 5A gives P1's 3D reconstruction results of its CT. The CT images were taken 1 month after the surgical creation of the unilateral cleft (left side). Compared with the control side (the right side), the 3D reconstructed CT showed that the third incisor on the cleft was missing, and no bone existed between the created defect and nasal cavity. In Figure 5B we presented the 3D reconstruction result for P2. The postoperative CT was taken 3 months after the surgery. Compared with control (the right side), it can be observed from the figure that the third incisor was missing. The anterior and posterior margins of the defect became rough and irregular.
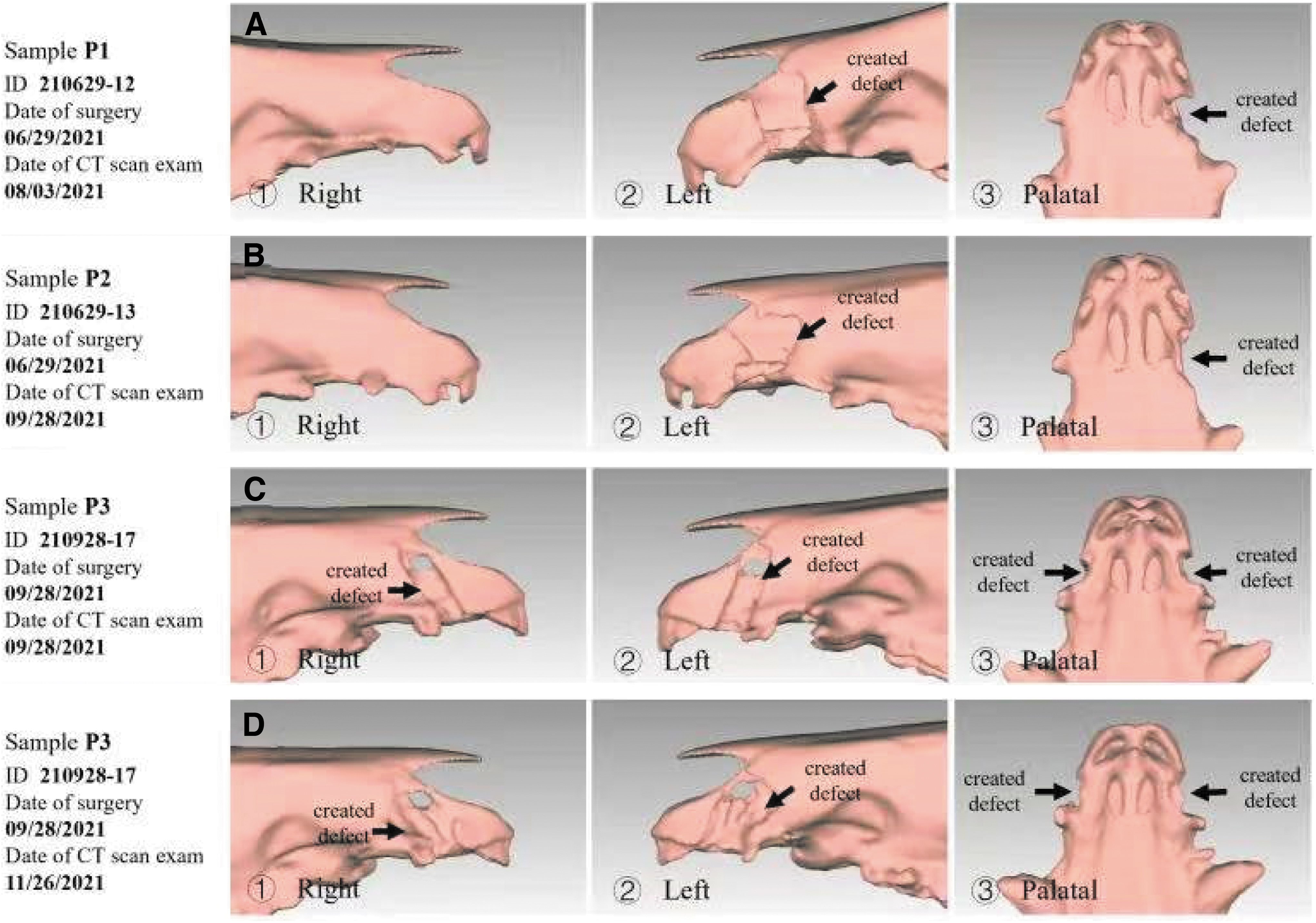
CT 3D image of P1, P2, and P3.
In Figure 5, we provided the 3D reconstructed CT image for the bilateral alveolar cleft model (P3). In this model, the CT scan was taken twice. The first scan was taken immediately after the surgery (Fig. 5C), and the results showed regular margin of the bone defects. We also observed that the second incision teeth were missing for both sides. When seen from the palatal view, bone remained between the surgically developed defect and the nasal cavity. A second CT examination was conducted at 1 month after the surgery for P3 (Fig. 5D). The CT images showed irregular margin of the walls of the bone defect. Overall, the height, width, and thickness of the defect did not present significant change 1 month after the surgery.
Histology and histomorphometry results
Figure 6A showed the histological manifestations in the defect side 1 month after the surgery (P1). Osteoclasts were observed on the surface of the root of the tooth, as well as the surface of the alveolar bone and the bone traps; aggregation of inflammatory cell was found on the surface of the tooth root, osteoclasts were closed to the absorbed bone and dentin. Epithelial cell hyperplasia (crawling along the fistulation channel, changed after being stimulated by inflammation) could also be seen. Figure 6B showed the histological manifestations on the defect side 3 months after the surgery (P2). As it was shown in the figure, we observed surface ulcers, small foci of bacteria, and infiltration of inflammatory cells. Compared with the histological signs 1 month after surgery (Fig. 6A), suspected osteogenesis was seen locally and no osteoclasts were found.
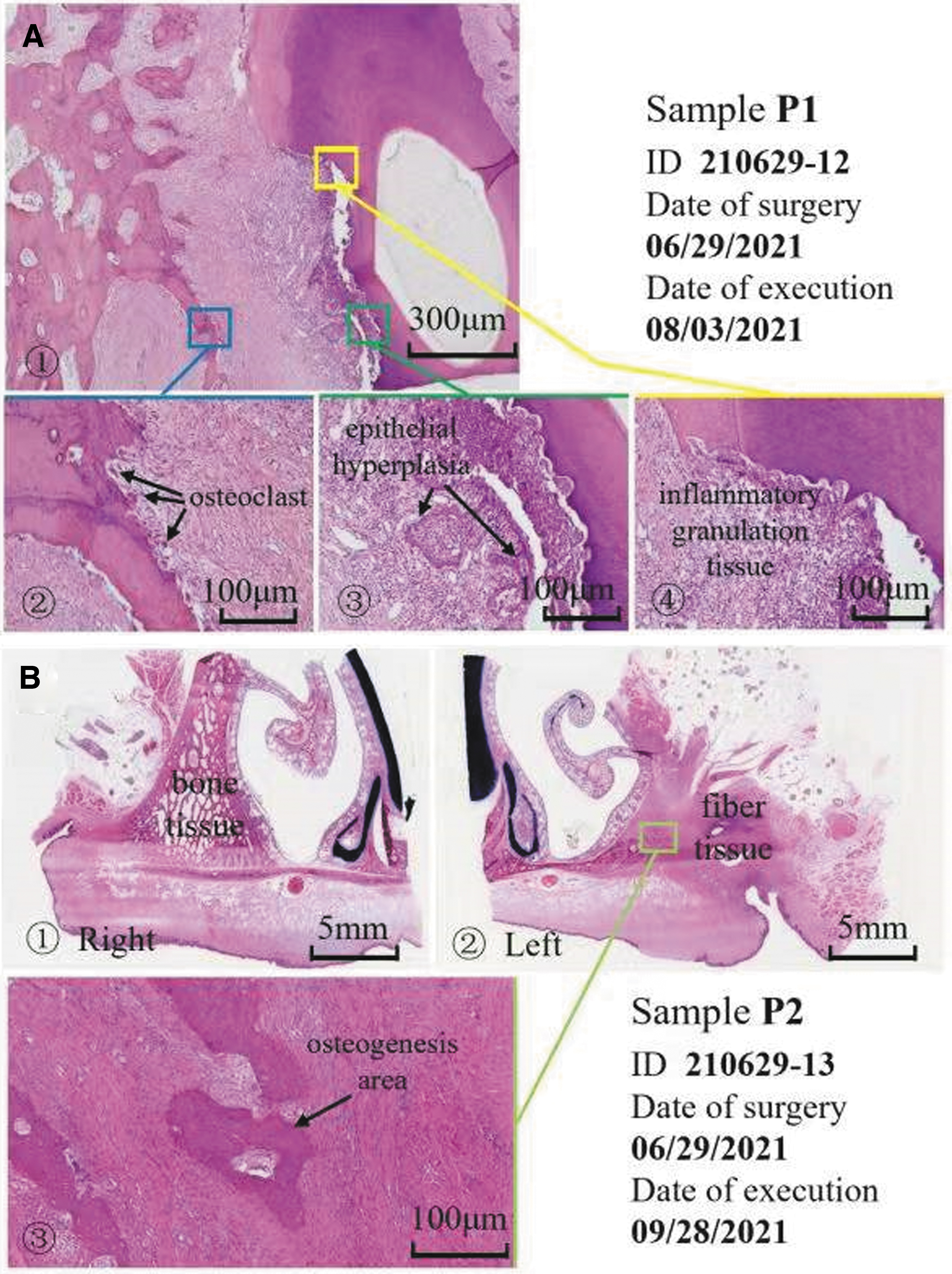
Histological characteristics in the surgically created alveolar cleft defect 1 and 3 months after operation (sample P1, P2).
Conclusion
We developed a porcine model imitating congenital alveolar cleft associated with cleft lip and palate, and demonstrated the procedure in a step-by-step fashion. The size of surgically created defects remained the same 1–3 months postoperatively and there was no sign of bone self-healing in the defect area. Results suggested that our proposed novel animal model can be employed in pilot studies for the purpose of optimizing the current treatment techniques as well as developing new treatment procedures and test the bone substitute materials. The bilateral model can be applied in further control studies.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr. Jianyun Zhang for his contribution of performing the histological analysis and Dr. Zhihui Zhang for the help with the animal experiment.
Disclosure Statement
No competing financial interests exist.
Funding Information
This work was funded by the National Key R&D Program of China (No. 2020YFC1107603).
