Abstract
Recent biotechnical advances in the in vitro culture of cholangiocytes and generation of bioengineered biliary tissue have a high potential for creating biliary tissue to be used for disease modeling, drug screening, and transplantation. For the past few decades, scientists have searched for a source of cholangiocytes, focused on primary cholangiocytes or cholangiocytes derived from hepatocytes or stem cells. At the same time, the development of scaffolds for biliary tissue engineering for transplantation and modeling of cholangiopathies has been explored. In this review, we provide an overview on the current understanding of cholangiocytes sources, the effect of signaling molecules, and transcription factors on cell differentiation, along with the effects of extracellular matrix molecules and scaffolds on bioengineered biliary tissues, and their application in disease modeling and drug screening.
Impact statement
Over the past few decades, biliary tissue engineering has acquired significant attention, but currently a number of factors hinder this field to eventually generate bioengineered bile ducts that mimic in vivo physiology and are suitable for transplantation. In this review, we present the latest advances with respect to cell source selection, influence of growth factors and scaffolds, and functional characterization, as well as applications in cholangiopathy modeling and drug screening. This review is suited for a broad spectrum of readers, including fundamental liver researchers and clinicians with interest in the current state and application of bile duct engineering and disease modeling.
Introduction
The biliary tract is a highly complex architectural and functional system in the human body, comprised by the liver, bile ducts, and gallbladder. After production by hepatocytes, bile is secreted into the bile canaliculi and then flows through the bile ducts that are lined by biliary epithelial cells, termed cholangiocytes, responsible for the final bile composition. 1 Their function is previously reviewed in detail and includes bile modification 2 and immune tolerance. 3 In addition, the biliary tree harbors putative stem cell niches, crucial for tissue homeostasis. 4 Human cholangiocyte function is regulated by bile salts, cytokines, growth factors, peptide hormones, nucleotides, and neurotransmitters.5,6
Cholangiopathies are a group of diseases originating from cholangiocyte injury, often causing liver failure. The most common cholangiopathies include cystic fibrosis, polycystic liver disease, primary biliary cholangitis, primary sclerosing cholangitis, biliary atresia, and cholangiocarcinoma (CCA). These cholangiopathies are caused by various factors (e.g., genetic, immune-mediated, idiopathic, infectious) resulting in cholestasis (impaired bile formation), followed by inflammation, fibrosis of bile ducts, and liver dysfunction, eventually leading to liver failure or end-stage liver disease.7,8 End-stage liver disease is a leading cause of death and liver transplantation is the only curative strategy. However, liver transplantation is hampered by a lack of suitable healthy donors and life-long immunosuppressive therapy with its adverse effects.9,10 Although precise numbers are lacking, cholangiopathies account for a substantial number requiring a liver transplant. The lack of sufficient donor livers poses a significant health and economic burden.
To study cholangiopathies, testing of new drugs and the (clinical) application of tissue engineering approaches, in vitro long-term maintenance of cholangiocytes and generation of biliary tissue is of utmost importance.11,12 Thereto, the anatomical and functional features of the tissue should be replicated in vitro, which is currently hindered by a lack of adequate in vitro culture systems and suitable scaffolds. 12 The possibility to induce differentiation of cholangiocytes from pluripotent stem cells (PSCs) has resulted in novel opportunities to establish reliable in vitro culture systems of cholangiocytes.13–15 Cholangiocytes and cholangiocyte-based organoids derived from induced pluripotent stem cells (iPSCs) are advancements in the field of liver cell culture and the emerging field of tissue engineering, specifically bile duct engineering, offering a variety of choices regarding cell source and desired differentiated cell type.16–18 However, heterogeneity of morphological features, functional assays, and marker-expression of stem cell-derived cholangiocytes is still observed in vitro. In this review, we focus on strategies of human cholangiocyte differentiation and the heterogeneity of obtained cell types. Subsequently, the use of these cells in various in vitro and in vivo applications studying cholangiopathies such as cholestasis is described.
Cell and Tissue Sources for Bile Duct Engineering
Cholangiocytes are primarily responsible for bile modification and transport. Furthermore, the biliary tree contains stem cell niches harboring the bipotential precursors able to differentiate into cholangiocytes and hepatocytes. During embryogenesis, hepatocytes and cholangiocytes differentiate via distinctive pathways from a shared progenitor cell, referred to as hepatoblasts. 19 This bipotential progenitor cell source is the product of a series of differential steps, starting with specification of cells in the mesendoderm forming the definitive endoderm, which gives rise to the foregut, midgut, and hindgut. Subsequently, a part of the foregut generates progenitors that are capable of differentiating toward pancreatic progenitors and hepatoblasts (Fig. 1). Details on these intermediate cell types and signaling pathways involved in embryogenesis are reviewed elsewhere. 20
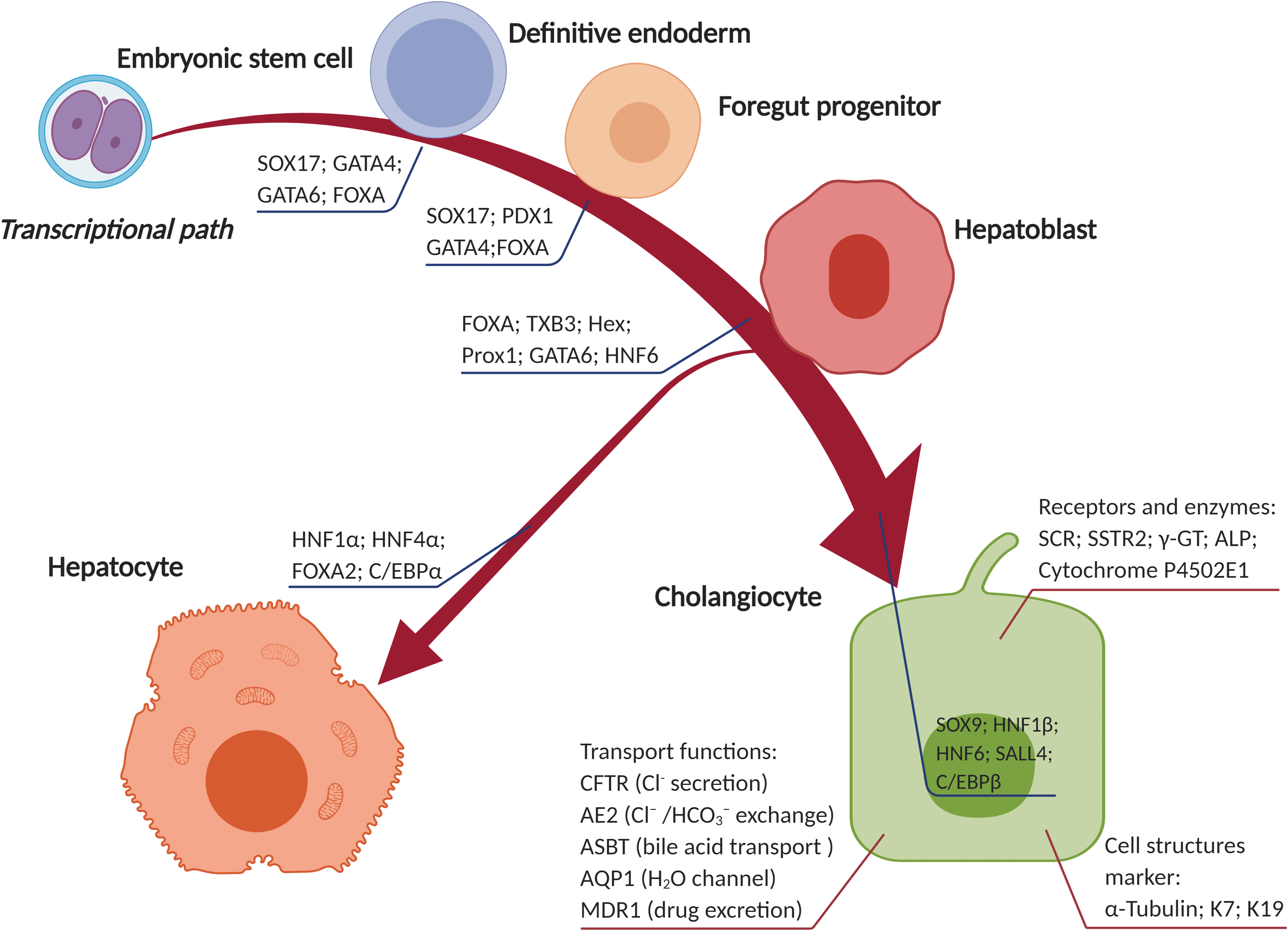
Differentiation of embryonic stem cells (ESCs) into cholangiocytes. The key transcription factors that guide ESCs differentiate via a series of stages toward hepatoblasts, serving as a bipotential progenitor cell source that is able to differentiate into both cholangiocytes and hepatocytes. Several structural features, transport function, receptor- and enzyme-markers are shown that can be used to identify mature cholangiocytes, and to evaluate the differentiation process.
For bile duct tissue engineering, the preferred cell source is primary cholangiocytes closely mimicking native cell physiology.21–26 Shortage of primary cholangiocytes hampered bile duct engineering and encouraged researchers to seek for substitutes. Alternative cell sources include differentiated hepatic cells, hepatic progenitor cells (HPCs), iPSCs, embryonic stem cells (ESCs), and immortalized or tumor-derived cholangiocyte cell lines. Table 1 presents an overview of cell sources used for bile duct tissue engineering, discussed in the next paragraphs.
Cell Source for Bile Duct Tissue Engineering
2D, two-dimensional; 3D, three-dimensional; EGF, epidermal growth factor; ESC, embryonic stem cell; HPC, hepatic progenitor cell; iPSCs, induced pluripotent stem cells.
Adult cell sources
Primary cholangiocytes
Primary cholangiocytes, isolated from biliary tract tissue, have been used widely in biliary tract regenerative medicine.23,24,26 Purified primary cholangiocytes proliferate, but lose their differentiated/functional phenotype in culture. Especially in two-dimensional (2D) culture conditions, lack of proliferation after a few passages is observed and functionality is lost due to absence of the native microenvironment of the cholangiocytes. One well-known nonimmortalized human cholangiocyte line is the NCH-3 cells, which has been used for biliary disease modeling and to uncover that inhibition of the Wnt-ligand production should be considered as a therapy strategy against scar formation. 27 Three-dimensional (3D) culture on collagen gels 28 or fibrous scaffolds 29 delayed dedifferentiation and allowed long-term proliferation compared with conventional cell cultures. In 3D cultures, primary human cholangiocytes formed spheroids with a central lumen phenotype (cholangioids), which were characterized as suitable 3D models of bioengineered biliary tissue. 26 However, utilization of isolated cholangiocytes remains suboptimal, encouraging alternative sources.
Hepatocytes
Hepatocyte-to-cholangiocyte transdifferentiation has been evidenced by an increase of cells expressing the cholangiocyte marker cytokeratin 19 in the spleen after hepatocyte transplantation in rats. 30 Furthermore, transdifferentiation was demonstrated upon severe injury of the biliary epithelium, 31 chronic liver injury, 32 or after activation of Notch signaling. 33 Although transdifferentiation of human HepaRG-derived hepatocyte-like cells into biliary-like cells was observed a decade ago, 34 no additional work has been done to investigate the use of human hepatocytes as a cell source for cholangiocytes. Primary human hepatocytes (PHHs) with high proliferation ability in vivo are hampered in in vitro utilization by their rapid dedifferentiation and loss of specific hepatic functions. 35 Some efforts have been made to the in vitro expansion of PHHs, which retain their hepatic features.36–39 Although the PHHs were dedifferentiated to a biphenotypic state by defined media conditions, the biliary differentiation has not been studied in-depth for this cell source.
Cholangiocyte-derived/like cell lines
For long-term experiments investigating the function of cholangiocytes, immortalized or tumor-derived cell lines were used as cell source, such as the SV-40 immortalized human intrahepatic biliary epithelial cell line H69. 40 H69 cells presented key biliary markers and were used to study anion exchanger (AE) activity 41 and epidermal growth factor (EGF) receptor inhibition. 42 In addition, MMNK-1 cell line, a SV40T, and hTERT transformed human fetal liver cell clone, expressed key biliary markers and demonstrated specific cholangiocyte functions, including responses to lipopolysaccharide, 43 clarification of the transforming growth factor-β1 (TGF-β1) signaling pathway targets, 44 and effects of cholesterol-derivatives 45 and were used in genotoxicity studies. 46 Interestingly, MMNK-1 cells formed tubular structures upon seeding in Matrigel.43,44
The HepaRG cell line was isolated from a liver tumor of a female patient suffering from hepatocarcinoma and hepatitis C virus infection. 47 The HepaRG cell line is commonly used for its ability to differentiate toward hepatocytes 34 and cholangiocytes. 48 In monolayers, HepaRG-derived hepatoblasts were treated with human growth hormones, EGF, interleukin 6, and sodium taurocholate to differentiate them toward the cholangiocyte lineage. These cholangiocyte-like cells (CLCs) showed high levels of cholangiocyte markers and formed primary cilia. They also formed a lumen with apico-basal polarity and transporter functions. 48 Due to its stable phenotype, it is suitable for drug discovery/metabolism, as reviewed elsewhere. 49
CCA is an epithelial biliary malignancy that may arise anywhere along the biliary tree. 50 The majority of research in CCA was reported with two CCA-derived cell lines, EGI-1, and TFK and included drug discovery, such as sorafenib or bosutinib combined with gemcitabine or cisplatin to suppress proliferation. 51 Immortalized or tumor-derived cell lines could be very useful in the modeling cholangiopathies, however, due to safety concerns, they cannot be used for cell therapy purposes. 52
Stem cells
Stem cells, including ESCs, iPSCs, and adult stem cells (ASCs), have been utilized for biliary tissue engineering. Proliferation, a hallmark of stem cells, allows scalable populations of cells to high quantities. In addition, stem cells can be directed to differentiate into cholangiocytes by mimicking embryonic development.
Embryonic stem cells
ESCs are used extensively in recent years for their self-renewal and differentiation into cells of all germ cell layers. 53 Zhao et al. directed the differentiation of human ESCs via a bipotent hepatic progenitor stage into CLCs. These CLCs lacked some cholangiocyte functions, but expressed the cholangiocyte markers KRT7 and KRT19, formed cysts and polarized tubule-like structures under 3D in vitro culture, and demonstrated multidrug resistance gene-1 (MDR1; P-glycoprotein) transport function. 54 Noushin et al. directed ESCs-derived hepatoblasts into functional CLCs with cilia expression and bile acid transport. Under 3D in vitro culture conditions, these CLCs form polarized tubule-like structures. 48 However, ESCs research is restricted by ethical and immunological guidelines, issues that can be circumvented by iPSCs.
Induced pluripotent stem cells
iPSCs are derived from somatic cells upon reprogramming by, for example, Yamanaka factors (OCT3/4, SOX2, KLF4, and c-MYC), 55 can be cultured long-term, and differentiate into various cell types under controlled conditions. To differentiate iPSCs toward CLCs, a stepwise, specific growth factor dependent, protocol mimicking embryonic development was used, via definitive endoderm, to initiate hepatoblast specification.13,14,16,56 In vivo, these hepatoblasts can give rise to both hepatocytes and cholangiocytes during the embryonic development (Fig. 1). Hepatoblasts eventually form cholangiocytic cysts with basolateral polarity and cholangiocytic marker expression.56,57 Sampaziotis et al. used patient-derived iPSCs for drug screening and to model polycystic and cystic fibrosis-associated liver disease. 14
Adult stem cells
After severe or chronic liver injury, ASCs replenish hepatocytes or cholangiocytes and/or support regeneration. 58 Cholangiocytes have been generated from HPCs in vitro.59–61 Unfortunately, the low number of HPCs limits their isolation and purification and hampers research on progenitor cells-derived cholangiocytes. Isolation of HPCs from human tissue is based on the membranous expression of epithelial cell adhesion molecule (EpCAM), 62 a marker for ASCs. 63 EpCAM-positive cells are found in different parts of the human biliary tree, including the canals of Hering and the peribiliary glands. 59 Transplantation of these cells in the liver of immune-deficient mice demonstrated differentiation toward hepatocytes and cholangiocytes with de novo bile duct formation.59,62 The low percentage of human HPCs (0.5–2.5%) in a liver biopsy 62 appeared insufficient for a human application, thus restricting this research to mouse transplantations. 62 Therefore, isolation and expansion protocols require improvements to obtain adequate cell numbers for disease models, drug screening, or regenerative therapies. This is where the organoid culture system comes into focus.60,64
Organoids/cholangioids
Organoids are self-organized 3D tissue cultures that can be developed from various species, organs, and cell types 65 when seeded in Matrigel or other hydrogels. The cholangiocyte-specific in vitro organoid model, termed cholangioids, formed proliferating spheroid-like structures with epithelial polarity. In addition, K7 and K19 remained present on the membrane during culture, and the cholangioids respond to secretin and rhodamine 123 accumulation indicating active transport. Lgr5-positive stem cells isolated from damaged mice liver tissue were able to differentiate and expand in vitro, forming liver organoids and further differentiated into hepatocytes and cholangiocytes. 66 Of great importance, primary human bile duct-derived organoids remained genetically stable in long-term cultures. 64 In another study, primary extrahepatic cholangiocytes in Matrigel formed extrahepatic cholangiocyte-organoids (ECOs) with native biliary features and functions. Furthermore, the ECOs populated on scaffolds created biliary tract tissue suitable for transplantation. Loarca et al., used patient-specific cholangioids to study the pathogenesis of primary sclerosing cholangitis, showing the cholangioids' potential for in vitro disease modeling. 18
Enhancing Directed Differentiation Toward Cholangiocytes
Differentiation protocols for the generation of CLCs from PSCs have been reported.13,14,48,67 A thorough examination of the signaling pathways used in directed differentiation toward cholangiocytes is listed in Tables 2 and 3, summarizing nine protocols by stage-specific differentiation steps, culture conditions, characterization, and application purposes. Early differentiation protocols (Table 2) aimed at the bipotential differentiation capacity of hepatoblasts or HPCs, protocols from 2014 onward (Table 3) primarily focused on the differentiation of PSCs toward CLCs. A consensus view was reached on the intermediate steps in the differentiation protocol.
Differentiation of Stem/Progenitor Cells Toward Hepatocytes and Cholangiocytes
AE2, anion exchanger 2; BMP, bone morphogenetic protein; CFTR, cystic fibrosis transmembrane conductance regulator; CLC, cholangiocyte-like cell; DE, definitive endoderm; FGF, fibroblast growth factor; GGT, gamma glutamyl transpeptidase; HB, hepatoblast; HE, hepatic endoderm; Hep, hepatocyte; HGF, hepatocyte growth factor; PSC, pluripotent stem cell; SCID, severe combined immunodeficiency; SCTR, secretin receptor; Shh, sonic hedgehog; OSM, oncostatin M; VEGF, vascular endothelial growth factor.
It is beyond the scope of this review to discuss all stages of liver development. However, some information regarding signaling pathways and transcriptional regulation of these processes offer valuable suggestions for differentiation of stem cells into cells of the hepatic lineage. First, the effect of different signaling molecules and transcription factors (TFs) will be described, followed by a brief examination of the effects of extracellular matrix (ECM) molecules. Finally, clues for scaffolds induced differentiation are reviewed. Although most of these processes is performed in animal models, the focus in this review is on human cell differentiation and the development of human tissue engineering applications, unless otherwise stated (Fig. 2).
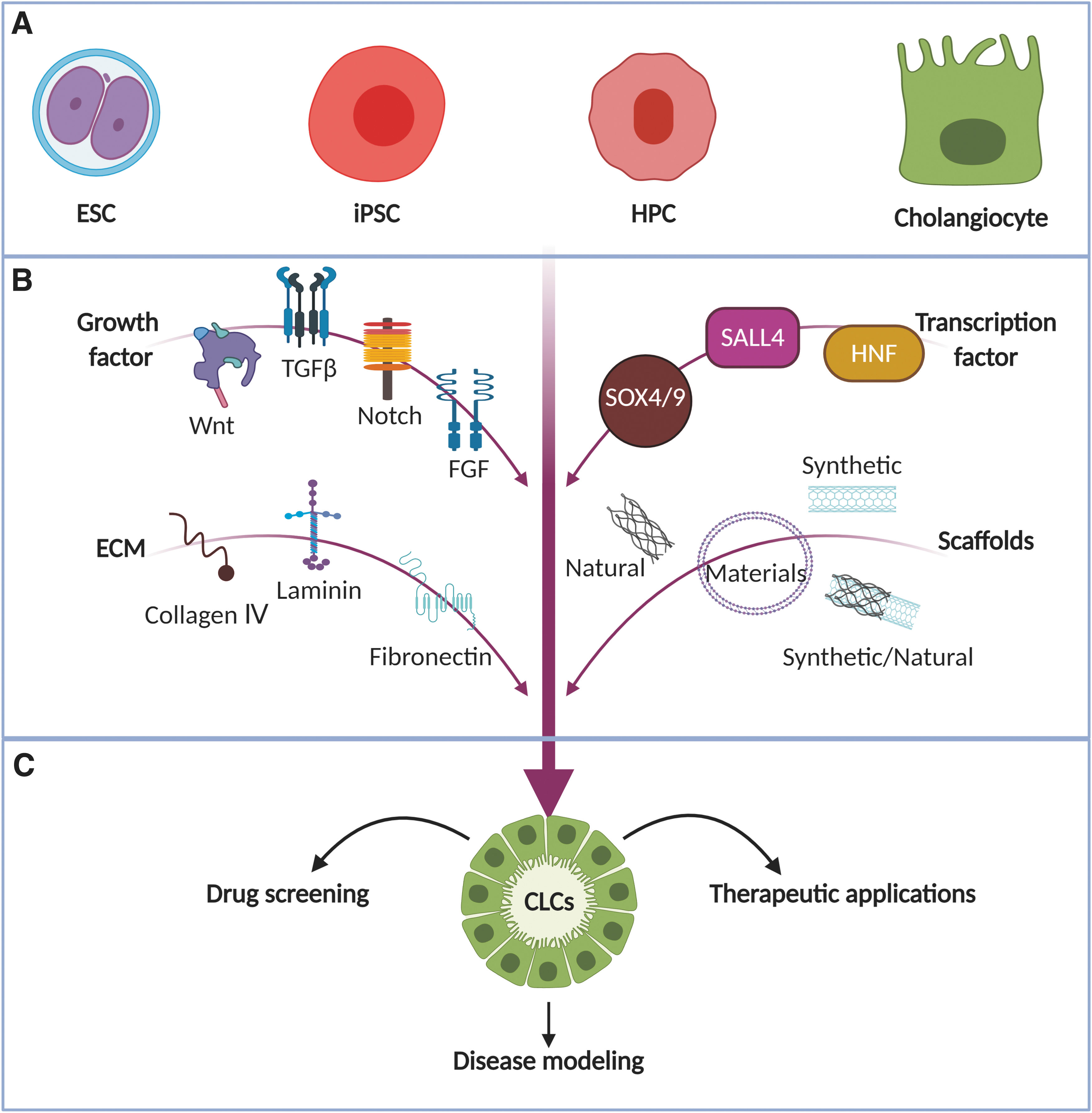
Factors influencing the differentiation of PSCs and adult (stem) cells toward cholangiocytes. Within the approaches to differentiate cholangiocytes,
Signaling pathways
Wnt/β-catenin signaling
Wnt/β-catenin signaling is involved during hepatic development. 68 To date, an excellent overview of stage-specific effects during in vitro ESC differentiation toward hepatoblasts is provided by Touboul et al.. 69 Inhibition of the Wnt/β-catenin signaling during early stages of differentiation (definitive endoderm into hepatic cells) and stimulation of the Wnt/β-catenin signaling during later stages of differentiation (hepatic cells into hepatoblasts) resulted in an optimal proliferative hepatoblast population. Inhibition of Wnt signaling reduced the differentiation of hESC-derived HPCs toward hepatocytes as evidenced by decreased expression of albumin and HNF1A. 70 Conversely, enhanced Wnt signaling during this differentiation process improved hepatocyte differentiation and decreased expression of the cholangiocyte marker KRT7. 71 The importance of Wnt/β-catenin signaling was confirmed in organoids, 64 in which its inhibition induced growth arrest that could be reversed by exogenous addition of Wnt3a. Activation of Wnt/β-catenin signaling during differentiation protocols should be done with careful considerations, since Wnt signaling is also linked to cholangiocarcinogenesis.72,73 Initial manipulation of Wnt/β-catenin signaling is required to induce differentiation of PSCs into definitive endoderm. 74 Together, the use of Wnt/β-catenin signaling is limited to the part until hepatoblast differentiation, and no beneficial effects of Wnt/β-catenin signaling on further cholangiocyte differentiation has been described.
TGF-β signaling
The TGF-β signaling family includes various ligands such as TGF-βs, bone morphogenetic proteins, and activins. The role of TGF-β ligands on hepatic tissue is stage-specific. Being important during liver development and liver regeneration, high levels of TGF-β in healthy hepatic tissue are linked to cell death and involved in the onset of various liver diseases (reviewed in detail by Fabregat et al.. 75 ). Empirical evidence showed the necessity of TGF-β in cholangiocyte differentiation protocols.13,67,76 After differentiation of cholangiocytes, however, the effect of TGF-β shifts toward decreased proliferation, as shown in H69-derived cholangiocytes. 77 Alternatively, inhibition of TGF-β can be used to differentiate hepatoblasts into hepatocytes 69 and TGF-β inhibition extends the culture of human liver organoids. 64 Kazuo et al. showed enhanced expression of cholangiocyte markers upon stimulation of human hepatoblast-like cells with TGF-β ligands, together with reduced expression of hepatocyte markers. 78 TGF-β-induced senescence in hepatocellular carcinoma (HCC) cell lines has been reported, and this factor could be used as a therapeutic target for early HCCs. 79 These studies indicate a counter-acting role of TGF-β signaling in cholangiocyte and hepatocyte differentiation and provide a therapeutic strategy in cholangiopathies.
Notch signaling
Notch signaling is essential in development and regeneration. 80 In mammals, four Notch receptors (Notch1–4) and five membrane-bound Notch ligands (Jagged1–2, Delta-like 1, 3, 4) are described. Particularly Notch2 and Jagged1 seem to play an essential role in biliary fate during embryonic development.81,82 Jagged1 (JAG1), expressed on the nascent portal mesenchyme, stimulates the transdifferentiation of Notch2 expressing hepatoblasts into a biliary phenotype. Upon JAG1/Notch2 interaction, the hairy and enhancer of split-1 (HES1) genes are transcribed subsequently, activating the cholangiocyte-specific TFs HNF1β and SOX9 (sex determining region Y [SRY]-box 9), resulting in development of the ductal plate.81–83 In Alagille syndrome patients, mutations in Notch ligand (JAG1) or receptor (NOTCH2) cause immaturely developed bile ducts despite normal hepatocyte differentiation. 77 Downregulation of Notch is linked to enhanced hepatocyte differentiation in cultured HPCs.71,84
Fibroblast growth factor signaling
Within the fibroblast growth factor (FGF) family, FGF2 (also known as bFGF) is the most important member involved in hepatic specification. FGF2 induced the expression of K19 during in vitro culture of chicken hepatoblasts, 85 while hepatic markers were not detected. A comprehensive evaluation of the working mechanism of FGF2 on human iPSCs was performed by Kirk et al.. 86 Since FGF induced naked cuticle homolog 1 expression to transiently inhibit the canonical Wnt/β-catenin signaling during hepatic specification of the endoderm, FGF2 is used in early steps of the cholangiocyte differentiation protocol for PSCs.13,14,48,67,87
Transcriptional regulation
Among the plethora of TFs for embryonic biliary specification, hepatocyte nuclear factors (HNFs) are the most important TFs, further divided in four subfamilies (Table 4). Because the HNF4 subfamily is exclusively involved in hepatocyte differentiation,88–90 it is not further discussed in this review.
Directed Differentiation of Human Stem/Progenitor Cells Toward Cholangiocytes
AA, ascorbic acid; AChR M3, M3 muscarinic acetylcholine receptors; AFP, alpha-fetoprotein; ALB, albumin; AQP1, aquaporin 1; ASBT, apical Na+-dependent bile acid transporter; ATP, adenosine triphosphate; CLF, cholyl-lysyl-fluorescein; EpCAM, epithelial cell adhesion molecule; FBS, fetal bovine serum; FOXM1β, forkhead (fox) factor M1B; FP, foregut progenitor; GH, growth hormone; HES, hairy and enhancer of split; HEY, hairy/enhancer of split related with YRPW motif protein; HNF, hepatic nuclear factor; IL, interleukin; InsP3R III, inositol 1,4,5-trisphosphate receptor type 3; JAG1, Jagged1; K, keratin gene; KDR, kinase insert domain receptor; KRT, keratin protein; LY294002, phosphoinositide 3-kinase inhibitor; MDR, multidrug resistance protein; NCAM, neural cell adhesion molecule; NICD, notch intracellular domain; OPN, osteopontin; P2RY1, purinergic receptor P2Y G-protein coupled 1; PKD2, polycystin-2; PROX1, prospero homeobox protein-1; RA, retinoic acid; SALL4, sal-like protein 4; SB-431542, transforming growth factor-β signaling inhibitor; SOX9, sex determining region Y (SRY)-box 9; SSTR, somatostatin receptor; STH, sodium taurocholate hydrate; TBX3, T-box transcription factor 3; TDO2, tryptophan 2,3-dioxygenase; TGF-β, transforming growth factor-β; TGR5, G protein-coupled bile acid receptor 1; ZO-1, zonula occludens-1.
Hepatocyte Nuclear Factor
Leading HNFs that are required for biliary specification are HNF1β and HNF6.91,92 An insufficient presence of HNF1β in human cholangiocytes resulted in the lack of primary cilia on the apical membrane. 93 More recently, bile duct cysts were observed in extrahepatic bile ducts of patients with mutations in the HNF1β gene. 94 Hnf6-overexpressing mice showed increased hepatocyte proliferation due to induced transcription of genes involved in cell cycle regulation. 95 The role of HNF1β or HNF6 to control in vitro human cholangiocyte cultures has not been addressed.
Members of the HNF3 subfamily, also known as FOXA proteins, fulfil a species-specific role in cholangiocytes. Although their presence is mainly important during initial liver specification in mice, 96 some FOXA proteins remain specifically present in human adult cholangiocytes. 97 Inhibited expression of Foxa1 and Foxa2 in cholangiocytes resulted in enhanced bile duct proliferation in mice, however, this increased proliferation was also correlated to cholangiopathies in vivo. 98 Future work should investigate the potential to target these TFs for controlled human cholangiocyte behavior, as demonstrated for hepatocytes. 99
Other interesting TFs that could possibly be used for TF delivery are sal-like protein 4 (SALL4) and SOX9. SALL4 is known to regulate cholangiocyte differentiation in early stages of liver development, initially shown in mouse livers. 100 Overexpression of SALL4 in embryonic hepatoblasts resulted in lineage commitment toward cholangiocytes, while inhibiting hepatocyte differentiation. This effect was partly mediated via increased gene expression of Notch signaling ligands and receptors. In human tissue, SALL4 suggested an oncofetal protein expressed in fetal hepatic tissue and in HCCs during the adult state. 101 However, SALL4 gene expression has been observed in adult cholangiocytes, 13 encouraging further investigations of this TF in differentiation of hepatoblasts.
SOX9 belongs to the SOX family of TFs that plays a pivotal role in liver organogenesis. During mouse liver development, intrahepatic bile duct development is controlled by SOX9. Inactivation of Sox9 in mice cause a delay in the maturation of the asymmetric primary ductal structures into bile ducts. 102 SOX4 also controls the timing of cholangiocyte differentiation. SOX4 and SOX9 TFs cooperatively regulate the bile duct development through activating signaling pathways (e.g., TGF-β, Notch, and Hippo-Yap signaling). Both SOX factors also support apicobasal polarity in the developing cholangiocytes. 103 Moreover, the Sox9 gene is a dose-sensitive modifier of bile duct paucity and liver phenotypes in Jag1+/− mice. 104 Mouse studies implicated a function for SOX9 in biliary specification of hepatoblasts,105,106 expression of SOX9 is observed in differentiated human CLCs.13,14,48,87,107 This warrants further investigations onto SALL4 and SOX9 to guide biliary differentiation in vitro.
ECM molecules
Decellularized liver tissue is capable of initiating the differentiation of HPCs, 60 illustrating the importance of ECM components. Seeding of PSCs on Matrigel-coated surfaces improves hepatic differentiation13,14,67 and supports long-term liver organoid cultures. 64 Matrigel is a gelatinous mouse-derived protein mixture composed of many ECM molecules, including collagen type IV, laminin, and entactin, and an array of growth factors. 108 Investigations into these individual ECM components, which are also present in the basement membrane in intrahepatic cholangiocytes, showed the importance of laminins on PSC differentiation into hepatocytes 109 and cholangiocytes. 87 The laminin-integrin interaction as shown for laminin 411 and 511 improves the differentiation of human iPSCs toward cholangiocytes. 87 Additional to ECM-components, ECM-stiffness properties influence cholangiocyte differentiation. Both collagen IV and fibronectin support cholangiocyte differentiation of liver progenitors on 30 kPa stiff substrates while only collagen IV does so on 4 kPa soft substrates. 110 Cell-matrix interactions should be investigated in-depth before being applied in therapeutic tissue engineering.
Scaffolds
Scaffolds are 3D biocompatible structures aiming to advance cell attachment and stimulate tissue regeneration in vivo or in vitro. Various sources of hydrogel or other scaffold materials in liver tissue engineering have been tested.111,112 For instance, recent advances in 3D bioprinting technologies have been applied to biocompatible materials that create biliary tree constructs using decellularized ECM. 113 Compared to 2D, in 3D scaffolds, cholangiocytes showed a ductal phenotype in Matrigel, 114 whereas functional polymers accelerated HepaRG cell differentiated into mature hepatocytes and improved metabolic functions. 115 HepaRG in nanofibrillar cellulose and hyaluronan-gelatin hydrogels enhanced albumin expression along with CYP3A4 expression and efflux transporter functions, 116 resulting in cells closer to native tissue than 2D cultures. 117
A large variety of synthetic and natural materials can be selected to manufacture (bio-)artificial scaffolds.
118
For tissue engineering purposes, different material properties need to be fine-tuned to optimize cell adherence and regeneration.119,120 For example, 3D formation of small hepatocytes stacked morphology was influenced by the pore size of poly(
Natural materials have superb biocompatibility to stimulate cell adhesion and growth, however, their use is limited by physical and mechanical stability. 126 Sampaziotis et al. established a collagen tube scaffold populated with primary human ECOs that maintained expression of biliary markers such as K7, K9, HNF1β, SOX9, cystic fibrosis transmembrane conductance regulator (CFTR), alpha-fetoprotein, and gamma glutamyl transpeptidase enzymatic activity. 26 Synthetic material, on the contrary, with defined structures can be better controlled but often lack the necessary biocompatibility and absorbability. 127 Human biliary epithelial cells on polyglycolic acid fiber mesh scaffolds collapsed after 3 to 4 weeks. To improve the stability of the constructs, they developed a polycaprolactone-stabilized polyglycolic acid scaffold that allows cells to form spheroidal aggregates while supporting long-term proliferation and phenotypic stability. 29
We recently combined synthetic and natural materials by coating polyethersulfone hollow fiber membranes (HFM) with
Functional Characterization of Differentiated Cholangiocytes
The cholangiocytes are epithelial cells that line a complex system of interconnecting bile ducts extending from the canals of Hering to the hepatic ducts. The bile is first secreted by hepatocytes into the bile canaliculi and then flows through the bile ducts for the final bile composition (Fig. 3). To evaluate cholangiocyte differentiation, their heterogeneity in vivo must be addressed. Differences between small and large cholangiocytes are summarized in Table 5. These phenotypic differences include for instance, villi or cilia presence, sensitivity to secretin, GABA-responsiveness, and cellular plasticity. An overview of relevant markers is given in the next paragraphs.
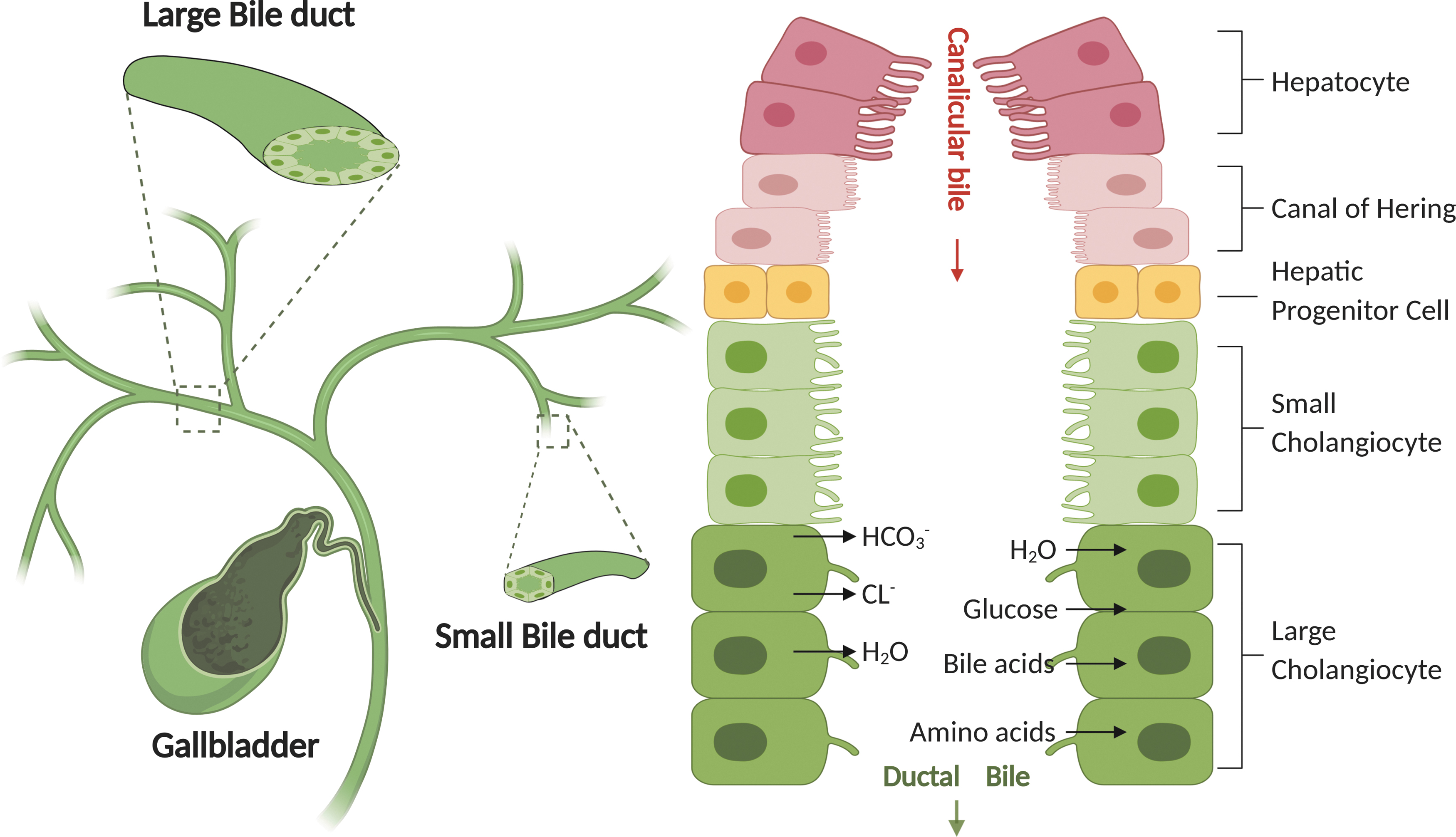
Biliary tree, bile duct, cholangiocytes, and duct bile formation. Bile is secreted by hepatocytes (i.e., primary or hepatic bile) and subsequently delivered to the bile ducts. The canals of Hering provide the continuum between the hepatocyte canaliculus and the ductules or cholangioles. HPCs are bipotential cells that reside in the canals of Hering. Due to the heterogeneous of bile ducts, hepatic bile is modified by the large cholangiocytes which are lining the large bile ducts. Large cholangiocytes mainly modify the hepatic bile through secretory (e.g., HCO3−, Cl−, H2O) and absorptive (e.g., H2O, glucose, bile acids, amino acids) processes, thus resulting in modified ductal bile.
Known Differences Between Small and Large Cholangiocytes
ALP, alkaline phosphatase; Cl−/HCO3− AE2, chloride bicarbonate anion exchanger 2; γ-GT, γ-glutamyl transpeptidase.
Cytoskeleton markers
Specific for cholangiocytes is the coexpression of K7 and K19 to provide cellular integrity and mechanical stability. 129 Staining of cytokines is used to distinguish between bile duct cells (K7/K19) and hepatocytes (K8/K18). 130 However, expression is not necessarily exclusive to these cells, as the expression of K7 and K19 is also shown in HPCs and intermediate CLCs; 131 that makes this marker combination not valid for discriminating between various stages of cholangiocyte maturation, let alone to distinguish large from small cholangiocytes. For this latter distinction, acetylated α-tubulins form highly stable microtubules, 132 which are required for the formation of a single, functional primary cilium on the apical membrane of large cholangiocytes. 21
Transporters and channels
Various ion channels and transporters are present on mature large cholangiocytes, including Cl−/HCO3− anion exchanger 2 (AE2), CFTR, aquaporin 1 (AQP1), and apical Na+-dependent bile acid transporter (ASBT). Although not cholangiocyte-specific, the presence of these channels in combination with their polarized expression is commonly used to prove the cholangiocyte-like state of cells in vitro. For example, AE2, ASBT, and CFTR are present on the apical membrane, while AQP1 is detected on both the apical and basolateral membrane. 2
Transport of fluorescent dyes provides information on transport activity. For example, rhodamine 123 is transported via the MDR1 gene product, P-glycoprotein, into the cholangioid lumen 133 and cholyl-lysyl-fluorescein uptake reveals ASBT activity. 14 Furthermore, changes in intracellular calcium levels represent acetylcholine and secretin receptor (SCTR) activity on cholangiocytes. 134 Finally, forskolin addition leads to cyst swelling, indicative of CFTR function in cholangiocytes, 13 as initially proven in intestinal organoids. 135
Ion channel activity can also be evaluated in relationship to diseases modeling in vitro. For example, cystic fibrosis (CF) patient-derived cells were used to create cholangiocytes to test for drug-restoring CFTR activity. 136 Further, overexpression of microRNA-506 in cholangiocytes inhibits AE2 and type III inositol 1,4,5-trisphosphate receptor (Ins3PR3), mimicking biliary cholangitis-like features. 137 In addition, a cholangiocyte responds to a high bile flow by activation of adenosine triphosphate-activated P2Y receptors on the apical membrane. This increases the intracellular Ca2+ concentration, which drives Ca2+-activated Cl− channels to secrete chloride ions, enabling the exchange of ions by AE2.138,139 Within this process, all channels are required to facilitate the complete mechanosensitive secretory pathway, pointing to the complexity to acquire properly functioning cholangiocytes in vitro.
G-protein-coupled receptors
An important group of membrane receptors needed for cholangiocyte function are the G-protein-coupled receptors (GPCRs). G-protein-coupled bile acid receptor 1 (GPBAR1, also known as TGR5) is one of these essential GPCRs, reviewed in detail elsewhere. 140 In addition, TGR5 is an important mediator of bile acid-induced cholangiocytes proliferation and it protects cholangiocytes from death-receptor-mediated apoptosis. 141
Other GPCRs that can be used for cholangiocyte classification include secretin receptor (SCTR), somatostatin receptor (SSTR), and muscarinic acetylcholine receptor (AChR M3). The SCTR and SSTR counteract each other by increasing and decreasing intracellular cAMP levels after activation, respectively, thereby influencing cholangiocyte secretion function. Together with AChR M3, these receptors regulate cAMP and Ca2+ activated Cl− channels. Besides the secretory function, SCTR is an important regulator of cholangiocyte proliferation 142 and involved in inflammatory cholangiocyte communication. 143 Together GPCR function is required for a definitive characterization of cholangiocytes.
Nuclear receptors
Nuclear receptors (NRs), shuttling between plasma membrane and nucleus, act like TFs. 144 Ligands for cholangiocyte NRs include bile acids, vitamins, and steroid hormones. For example, farnesoid X receptor, pregnane X receptor, and vitamin D receptor bind intracellular bile salts, leading to increased gene expression of genes involved in bile acid hydroxylation, detoxification, and secretion. 145 Because these NRs are not exclusively expressed in human cholangiocytes, they are not suitable for distinguishing between hepatocytes and cholangiocytes. However, their presence can be used to classify the functionality of cultured cholangiocytes and can be therapeutic targets in liver diseases. 146
Polarity
The characterization of cholangiocytes can include cellular polarity. In native hepatic tissue, cholangiocytes are polarized with the apical side facing toward the luminal space of the bile duct. A frequently used marker for polarity is the tight junction marker zonula occludens-1 present in apical junctions. Recently, the apical orientation of the Golgi system was used to determine polarity in cholangioids, using an antibody against the Golgi protein GM130. 18 Together with the basolateral markers β-catenin and integrin α6, they are used to indicate apical-basal polarity in cholangiocytes. 58
Taking into account the various characteristics described above, it is at present difficult to rationally design optimal cholangiocyte differentiation protocols. We believe that aiming for an open comparison and consensus on terminology/characterization is instrumental and will greatly accelerate the steps to optimize cholangiocyte culture systems and their application in future studies.
Modeling Cholangiopathy in Bioengineered Bile Ducts
Different biliary disease models have been established, both in vivo and in vitro, to improve the current therapeutic strategies. 147 For instance, Mdr2 (Abcb4) knockout mice develop sclerosing cholangitis, 148 and rats present a human primary sclerosing cholangitis phenotype upon intrabiliary injection of 2,4,6-trinitrobenzenesulfonic acid. 149 Chronic feeding of DDC (3,5-diethoxycarbonyl-1,4-dihydrocollidine) in mice should serve as the small bile duct cholestatic model. 150 Primary sclerosing cholangitis is a chronic liver disease, characterized by inflammation and scarring of the bile ducts that ultimately lead to cirrhosis. 151 Interestingly, the frequently observed lymphocytic cholangitis in privately owned cats is similar to primary sclerosing cholangitis based on histology, the contribution of microbiological drivers, gender predisposition, and the involvement of Notch-signaling.152–155 Preclinical (cats) and fundamental (rodents) models for cholangiopathies are taking the complexity of organ interactions into account. Other typical in vivo animal models to model cholangiopathy have been thoroughly reviewed elsewhere.147,156 Although these models can significantly increase our basic mechanistic understanding of cholangiopathies, they are limited in use. In vitro models have a great advantage in maintaining cells in controlled experimental conditions. Thus, research groups developed cholangiocyte disease models to model in vivo cholangiopathies. Various in vitro models have been developed for cholangiopathies, including gene editing 14 and chemically induced 157 models. Here, we describe the current understanding of the disease modeling of cholangiopathies in 3D in vitro cultures.
Primary sclerosing cholangitis is a chronic liver disease, predominantly manifested in human, characterized by inflammation and scarring of the bile ducts that ultimately lead to cirrhosis. Loarca et al. reported a novel 3D model that mimics several features of primary sclerosing cholangitis and can be used to study pathogenesis and to identify potential therapeutic targets. 18
Primary liver cancer is one of the most lethal malignancies worldwide, with the majority classified into HCC and CCA. 158 Laura et al. created organoids from HCC, CCA, and combined HCC/CCA with preserved histological architecture, gene expression, and genomic landscape of the original tumor 159 and, therefore, these organoids can be applied to better understand liver cancer biology even at a personalized level.
Cystic fibrosis (CF) is a systemic disease caused by mutations in the CFTR gene. 160 CFTR mutations in cholangiocytes influence chloride secretion, bile viscosity, and focal biliary cirrhosis secondary to bile plugs occluding the intrahepatic bile ducts. Sampaziotis et al. developed a biliary CF in vitro model by generating hiPSCs and hiPSCs-derived CLCs from patients with the common CF mutation ΔF508 to test the CF drug VX809's ability to rescue the disease phenotype in vitro. 14
Future Outlook
The heterogeneous etiology of cholangiopathies explains the different responses to therapy and emphasizes the need to develop representative models and alternatives for liver transplantation. Tissue engineering as a new strategy provides a potential addition (or even alternative) for organ transplantations. Challenges not addressed include the use of Matrigel, feeder cells, or animal-derived supplements, each of these aspects render them impossible to implement in human clinical practice. Second, the effect of the in vivo transplantation site on the cells must be investigated.
An important need for future studies is to determine the specific effects of the in vivo environment. The delicate interplay between hepatocytes, cholangiocytes, and other cells present in hepatic tissue needs a deeper clarification. Tissue engineering allows studying the cellular interplay between different cell types. A 3D biomimetic patterning was demonstrated, 161 using rapid 3D bioprinting of human HPCs and complementary layered supporting cells, which had improved in vitro hepatocyte differentiation. Moreover, 3D hepatic lobule-like constructs were assembled by gear-like microstructures encapsulating multiple cell types. The 3D integrations constructs exhibited hepatic lobule-like morphology and a built-in lumen, replicating in vivo liver architecture and function. This construct showed a high proliferation rate and spreading during long-term coculture. 162
Microfluidic devices provide precise control concentration gradient, spatial distribution of cells, and relatively easy to control flow systems to mimic the in vivo environment. Bile duct-on-chip is under development. Our group successfully bioengineered mouse CLCs on HFM, which acquired polarity and mimicked the native bile duct transport activity in a 3D-printed microfluidic device. 128 Others demonstrated that mouse cholangiocytes in a microfluidic device become monolayered, polarized, and formed tight junctions. This device allowed proof-of-concept toxicity studies with the biliary toxin, biliatresone, and specific bile acids. 24
Although major improvement of tissue engineering strategies is still required, these bile duct-on-chip captured some essential features and functions of native bile duct, which makes them feasible for disease modeling and drug screening and allows studying interactions between biliary tract and other systems. Bioengineered bile ducts have been successfully transplanted into mice. 26 These bioengineered bile ducts could be considered as a possible therapy for cholangiopathies in human. Nevertheless, cell or tissue therapy for liver disease has a clear requirement for unlimited cell sources with native function, phenotypic stability, and without risk of tumorigenicity. HPCs have unlimited availability, indefinite proliferation, and less safety concerns, which are vital when considering cell-based therapies. Advanced therapy medical products (ATMPs) are innovative therapies that involve gene therapy, somatic cell therapy, and tissue-engineered products. 163 Legal and regulatory framework for ATMPs still needs to be established and applied to liver cell therapy.
Footnotes
Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the funding for PHD students from China Scholarship Council (No. 201808620130).
