Abstract
Bioreactors have been widely acknowledged as valuable tools to provide a growth environment for engineering tissues and to investigate the effect of physical forces on cells and cell-scaffold constructs. However, evaluation of the bioreactor environment during culture is critical to defining outcomes. In this study, the performance of a hydrostatic force bioreactor was examined by experimental measurements of changes in dissolved oxygen (O2), carbon dioxide (CO2), and pH after mechanical stimulation and the determination of physical forces (pressure and stress) in the bioreactor through mathematical modeling and numerical simulation. To determine the effect of hydrostatic pressure on bone formation, chick femur skeletal cell-seeded hydrogels were subjected to cyclic hydrostatic pressure at 0–270 kPa and 1 Hz for 1 h daily (5 days per week) over a period of 14 days. At the start of mechanical stimulation, dissolved O2 and CO2 in the medium increased and the pH of the medium decreased, but remained within human physiological ranges. Changes in physiological parameters (O2, CO2, and pH) were reversible when medium samples were placed in a standard cell culture incubator. In addition, computational modeling showed that the distribution and magnitude of physical forces depends on the shape and position of the cell-hydrogel constructs in the tissue culture format. Finally, hydrostatic pressure was seen to enhance mineralization of chick femur skeletal cell-seeded hydrogels.
Introduction
B
Hydrostatic pressure is present in vivo in a number of different regions and magnitudes.4,5 Osteocytes in the canalicula-lacuna network of load-bearing bones are subjected to physiological pressures of approximately 270 kPa, 6 which was the maximum hydrostatic pressure that was applied in this study to investigate its effect on bone formation. Moreover, the heartbeat of chick embryos delivers a dynamic pressure of 4 kPa, 7 the blood pressure is usually between 8–24 kPa, 8 the hydrostatic pressure in the cerebrospinal fluid is around 1.2 kPa 9 and the interstital fluid pressure is around 0.27 kPa. 10 In addition, chronically elevated hydrostatic pressures can also be found in disorders and injuries such as glaucoma, chronic nerve compression injury, traumatic brain injury, and spinal cord injury.11–14
Although the use of bioreactors for the production of tissue-engineered constructs is promising, the aims and anticipations for their development and application have only begun to be met. 1 To manufacture implantable tissue substitutes, bioreactors need to provide the appropriate environmental and growth parameters (e.g., dissolved oxygen [O2] and carbon dioxide [CO2], pH, nutrient transport, and waste removal) as well as mechanical forces (e.g., hydrostatic pressure, shear forces) during culture. In addition, for clinical relevance, bioreactors should provide the possibility for scale up, enabling the mechanical stimulation and preconditioning of a larger number of tissue constructs. To date, this has not always been achieved, because environmental parameters and the effect of different mechanical stimulation regimes are not fully understood and optimized for their specific application. 3
Among the most important factors within the bioreactor are mass transfer (e.g., O2 and CO2 supply) and mechanical stimulation (e.g., hydrostatic pressure, fluid flow, and stress). The application of hydrostatic pressure during tissue formation, for example, could result in enhanced transfer of small molecules, such as oxygen and CO2, into the tissue matrix and also provide physical forces to cells and tissues. 3
Both the concentration of dissolved oxygen and the pH of the medium are essential variables in biomedical engineering. pH and dissolved oxygen have been shown to affect cellular mechanisms such as inter-cellular signaling, cell proliferation, and cell differentiation.15,16 They also influence cell cycle, apoptosis, and protein synthesis.17–22 Moreover, the measurement of pH provides information about the metabolic activity of the cells and tissues. 21 The effects of increased dissolved CO2 concentrations on mammalian cells have been widely investigated.23–25 Since CO2 is able to diffuse across cell membranes, it decreases the intracellular pH, resulting in alterations of the cell metabolism and enzyme activities.26,27 Studies have shown that high levels of dissolved CO2 reduce the growth rate and protein production in mammalian cells.23,25,28 Hence, it is essential to monitor and control the levels of dissolved CO2 during mammalian cell culture. 28
Despite their importance, limited information seems to be available about the changes of environmental parameters in cell culture medium within closed culture systems. 19 In closed cell culture environments, such as incubators or bioreactors, the pH of the cell culture medium is a result of bicarbonate concentration in the medium, the CO2 level in the incubator, the type of buffer system, and accumulation of lactic acid. 22 Sodium bicarbonate and HEPES are commonly used buffers that maintain a physiological pH of the cell culture medium. 29 The bicarbonate buffer system enables the adjustment of pH under acidic conditions and is able to neutralize acid by eliminating dissolved CO2 through dissociation of carbonic acid. 29 However, bicarbonate is a base and, therefore, non-bicarbonate buffers such as HEPES might be required to reduce alkalization when the medium is supplemented with bicarbonate. 30 In addition, in order to avoid fluctuations in pH due to variations in dissolved CO2 in the medium, the HEPES buffer system is used, which is a suitable buffer for both alkaline and acidic conditions. A combined effect of bicarbonate and HEPES might, therefore, provide a better adjustment of the pH during cell culture. 29
To investigate the effects of both mechanical forces and physiological parameters on cells and tissue constructs, mathematical modeling and numerical simulation can be used to complement experimental studies.31–37 Mathematical modeling can be used to determine the local mechanical and biochemical environment experienced by cells in culture, and how this environment depends on experimentally controllable parameters such as applied external pressure, hydrogel properties. In turn, once validated, such models can aid in determining favorable operating conditions for tissue formation. These combinational approaches aid investigation into local conditions that cells and cell constructs are exposed to during mechanical stimulation in bioreactors. 3
This study aims at evaluating the growth environment and mechanical stimulation that the HF bioreactor provides for cells, cell-scaffold constructs, and tissues. Measurements of physiological parameters, that is, dissolved O2, dissolved CO2, and pH, using optical sensors in combination with the development and solution of a mathematical model to determine physical forces during stimulation of a cell-seeded hydrogel were undertaken. In addition, the effect of hydrostatic pressure on chick fetal femur skeletal cell-seeded matrigel hydrogels was studied.
Materials and Methods
Bioreactor design
The HF bioreactor utilized in this study was developed in collaboration between Keele University (United Kingdom) and Instron TGT (USA) to facilitate easy handling and scale up and has previously been described by Henstock et al.. 5 A schematic of the bioreactor system and further digital images are shown in Figure 1. Briefly, the bioreactor chamber is a sealed aluminum chamber fitting standard tissue culture multi-well plates. In order to apply hydrostatic pressure to samples within the bioreactor chamber, incubator air is compressed using a continuously running scroll compressor and, subsequently, pumped into the bioreactor chamber via a temperature controller that maintains the temperature of the compressed air at 37°C, creating a gas–liquid interface with a pressurized gas phase on top of a liquid medium phase. This principle of operation makes it necessary to determine changes in dissolved O2, dissolved CO2, and pH of the media. A vacuum is generated to remove the gas phase from the bioreactor chamber and direct the air back into the incubator where it is reused for the stimulation process. The stimulation regime is controlled by system accompanying software (GrowthWorks, Instron TGT) that enables the application of sinusoidal waveforms with frequencies between 0.0001 and 2 Hz and a pressure range between 0 and 280 kPa. In addition, this software also enables continuous stimulation regimes.
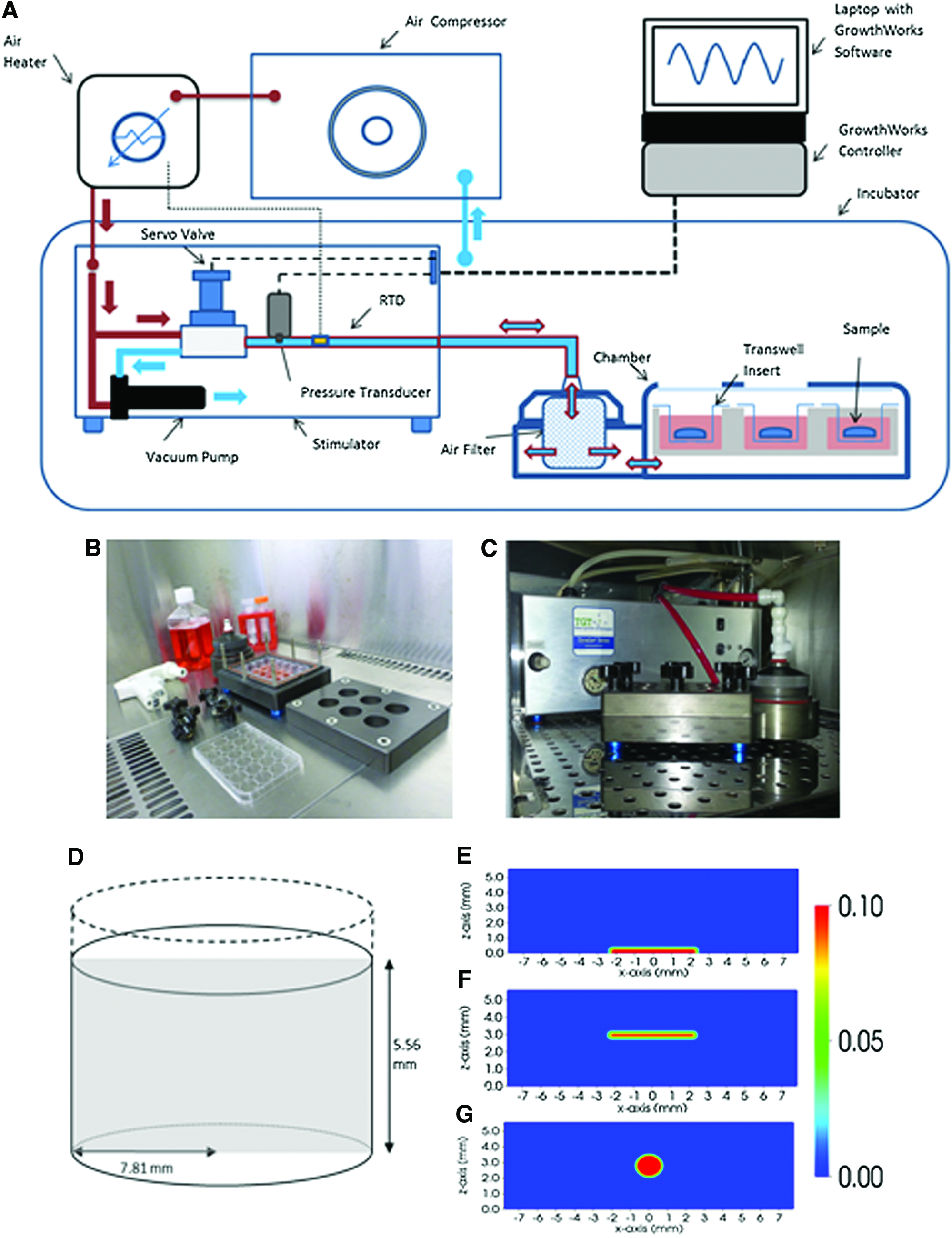
Schematics of the hydrostatic force bioreactor of experimental setup that inspired the theoretical setups considered for the mathematical model. The bioreactor system
Measurements of physiological parameters in the medium surrounding the hydrogels
Changes of physiological parameters in cell culture medium were measured at various time points before and after mechanical stimulation. Briefly, media samples were placed in a standard cell culture incubator for 24 hours at 37°C and 5% CO2 before mechanical stimulation for equibrilation. At the beginning of each experiment, dissolved O2, CO2, and pH were measured at 0, 10, and 20 min before mechanical stimulation. Samples were then subjected to various magnitudes of cyclic hydrostatic pressures (0 [control], 0–90, 0–180, and 0–270 kPa) at a frequency of 1 Hz. Dissolved O2 and CO2 were measured every 20 min (pH every 30 min) immediately after mechanical stimulation for samples placed in the HF bioreactor and left in a cell culture hood at room temperature. A needle-type oxygen microsensor (PreSens) and a chemical optical CO2 sensor dipping probe (PreSens) were used to measure the partial concentration of dissolved O2 and CO2 in the medium. The pH of the cell culture medium was determined using a pH meter (Orion 3 Star pH Benchtop; Thermo Scientific). Calibration of sensors and pH meter was carried out before measurements according to the manufacturer's instructions. For pH measurements, bicarbonate-buffered Dulbecco's Modified Eagle Medium (DMEM) was further supplemented with 25 mM HEPES. Furthermore, changes in pH between bicarbonate-buffered and HEPES-buffered medium were compared. To investigate the reproducibility of the changes in O2, CO2, and pH, mechanical stimulation was carried out for 60 min every other day over an 8 day period. Measurements were performed directly before and after mechanical stimulation at each time point. The temperature within the bioreactor was allowed to adjust to 37°C before mechanical stimulation was performed.
Assessment of physical forces in the bioreactor
To gain an understanding of the velocity fields, stress fields, and pressure distribution within the well plate, a multiphase model based on Osborne et al. was formulated, which provides a framework to investigate the interactions between the constituents in the biological tissue.38,39 The model is based on the idea that the tissue may be represented by a mixture of “continua,” which are able to occupy the same region of space (here cells, extracellular fluid, and substrate). Interactions between the different phases are specified via mass and force balance equations, along with appropriate constituting equations. The cell and fluid phases are assumed to behave as viscous fluids, and the substrate is assumed to be a stationary solid. The dependent variables are the cell and fluid volume fractions (defined to be the proportion of the total volume occupied by the respective phase at each spatial point), the cell and fluid velocities, and the hydrodynamic pressure. In the model, cells experience forces due to the gradient of the hydrodynamic pressure (which drives cells from high to low pressure regions), viscous drag between the cells and the fluid and solid phases, and active forces that model the tendency of cells to aggregate at a low cell volume fraction and repel at a high cell volume fraction. In the absence of inertial effects, these forces are in equilibrium. The initial in vitro experiment examined the setup of a 65.15 mm3 cell-seeded collagen gel placed on the bottom of the well and submerged in 1 mL of cell culture medium. The gel was made by seeding 25,000 cells in 300 mm3 collagen suspension at a concentration of 2.5 mg/mm3. This was allowed to contract for 24 h, by which time it had become 65.16 mm3 in volume. Assuming the well is cylindrical in shape with a radius of 7.81 mm, as obtained from the Corning Catalogue, the total height of the gel and fluid was calculated to be 5.56 mm. The cylindrical-shaped well in the absence of any gel is shown in Figure 1D. The two-dimensional cross-section of this domain, as illustrated by the gray region, is considered for numerical simulation and was derived from a real experiment.
The three theoretical setups that were investigated are shown in Figure 1E–G. The mathematical model enables any size and shape of the gel to be specified and the resulting environment to be investigated. In Setup 1, a gel, with a smaller width than that of the well, is placed centrally on the base of the well plate (Fig. 1E). In Setup 2, this gel is held in position by a transwell insert, so that fluid can flow around the gel (Fig. 1F). Finally, in Setup 3, a spherical-shaped gel is held in position by a transwell insert in the center of the culture medium (Fig. 1G). All three setups were chosen so that the same quantity of cells and hydrogel is considered in each of the setups, where the volume ratio of cell to hydrogel is 2:3. This ratio was calculated from the initial experimental data as follows. It was assumed that the the hMSC are spherical in suspension with a radius of 15.25 μm and that the cell volume does not change when in suspension or adhered to the gel. 40 Further, using the density of dry collagen as 1.35 mg/cm3, the initial volume of space occupied by collagen was calculated. 41 These findings gave a volume fraction ratio of cells to collagen as 2:3.
Functions of hyperbolic tangents are used to prescribe smooth, continuous cell and gel conditions. No-flux and no-slip boundary conditions are imposed on the walls and the base of the well plate, and traction boundary conditions are imposed on the surface of the liquid to model the force applied by the bioreactor. All parameters were given elsewhere 38 except for the viscosity of the culture medium phase, which was assumed to be the same as water, and set to be 6.914×10−4 Pa·s, 42 and the cell phase viscosity, which was set at 5290 Pa·s. 43 Numerical methods were implemented in a similar manner to those described elsewhere. 44
Isolation and characterization of chick fetal femur skeletal cells
All reagents were purchased from Lonza unless otherwise stated. Chick fetal femur skeletal cells were isolated from explant cultures of femoral bone fragments removed from day 11 chick embryos. Cells were cultured in DMEM supplemented with 2 mM L-glutamine, 10% fetal bovine serum, and 5× antibiotics/antimycotics (100 U/mL penicillin, 0.1 mg/mL streptomycin, and 0.25 μg/mL amphotercin B) and incubated at 37°C and 5% CO2.
In order to investigate the capability of the chick fetal femur-derived skeletal cells to form bone or cartilage, cells were characterized by the expression of stemness' marker SOX2, osteogenic and chondrogenic transcription factors SOX9 and RUNX2, type-I collagen, type-II collagen, alkaline phosphatase, as well as CD44 using quantitative polymerase chain reaction (PCR). Primers and SYBR Green were obtained from Qiagen. The expression values for each sample were normalized against the expression of GAPDH.
Preparation of cell-seeded hydrogels
Cells (passage 2) were cultured until confluent, then trypsinized, and counted. For the preparation of hydrogels, 1×104 cells were added to 100 μL matrigel (Englebreth–Holm–Swarm mouse sarcoma basement membrane matrix; BD Biosciences). The cell-hydrogel mix was added to transwell membrane inserts and allowed to set at 37°C and 5% CO2. Cell-hydrogel constructs were cultured for 48 h before starting the mechanical stimulation to enable cells to adhere to the hydrogel. On the commencement of mechanical stimulation, gels were cultured in osteogenic medium for a period of approximately 14 days. Osteogenic medium was prepared from DMEM medium supplemented with 100 nM Dexamethasone (Sigma), 0.05 mM L-ascorbic acid-2-phosphate (AnalaR), and 10 mM beta-glycerophosphate (Sigma).
Mechanical stimulation of cell-seeded hydrogels
In order to investigate the effect of HF on cell-seeded hydrogels cultured in transwell inserts in osteogenic medium, hydrogels were subjected to a cyclic hydrostatic pressure of 0–279 kPa for 60 min at 1 Hz over 14 days (5 consecutive days per week).5,45
Micro-X-ray computed tomography
Micro computed tomography (MicroCT) analysis of the stimulated and non-stimulated control hydrogels was performed after 14 days using a desktop micro-computed tomograph (Scanco μCT40) with a beam energy of 55 kV, a beam intensity of 145 μA, 200 ms integration time, and 10 μm spatial resolution. To quantify the total size, volume, and average density of each gel and to determine the volume of the denser, mineralized matrix, and its average density, the gels were analyzed at two density thresholds (50/1000 and 80/1000). The lower threshold was chosen for the segmentation of the hydrogel content, and the higher threshold was applied for the analysis of mineralization. Three-dimensional (3D) images were generated using Scanco accompanying software. Student's t-test between the stimulated and hydrogels' unstimulated control (n=5) was performed for statistical analysis.
Histological staining and imaging
The hydrogels were fixed in 4% paraformaldehyde. Subsequently, gels were embedded in optimal cutting temperature compound and cut in 10 μm sections using a microtome cryostat. Sections were stained for calcium using 1% alizarin Red S solution. Images were taken using a dark-field microscope.
Results
Measurements of physiological parameters
Measurements of dissolved oxygen concentration in the medium are shown in Figure 2. Samples were initially placed in a standard cell culture incubator, where the oxygen concentration in the medium remained constant over 20 min. Subsequently, one set of samples was transferred to the HF bioreactor and stimulated at 0–270 kPa and 1 Hz (Fig. 2A). Every 20 min, the dissolved O2 in the medium was measured. On cyclic mechanical stimulation, the dissolved oxygen concentration initially increased to around 30% and then decreased to around 23% dissolved O2 after stimulation for 80 min. To determine the time it takes for the medium to reach initial oxygen levels, samples were placed back to the incubator. Around 120 min, post-stimulation oxygen levels reached initial levels of 20%.
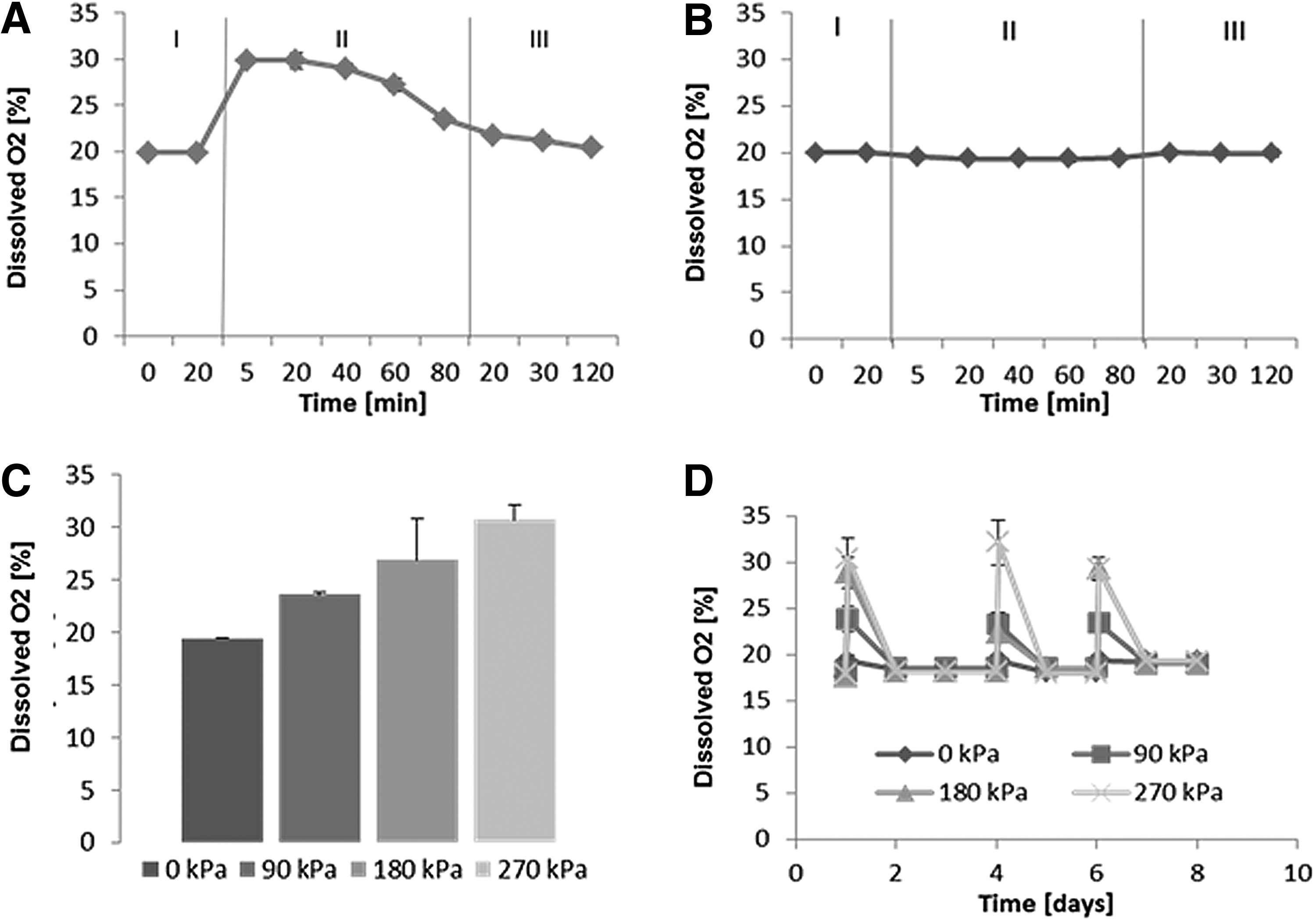
Changes in dissolved O2 of cell culture medium on application of hydrostatic force. An optical sensor was used to measure O2 in the cell culture medium when exposed to cyclic stimulation at 270 kPa
The second set of media samples was left outside the bioreactor chamber at room temperature in order to investigate changes in O2 that might occur during the cell culture processes, such as medium change or cell culture assays (Fig. 2B). Oxygen concentration was measured every 20 min over a period of 80 min, after which samples were placed back in the incubator. Throughout the whole experiment, the oxygen concentration remained between 19% and 21%.
O2 measurements for samples stimulated at different pressures, including 0, 0–90, 0–180, and 0–270 kPa, were taken immediately after mechanical stimulation and have shown that the higher the applied pressure, the higher the oxygen concentration in the medium (Fig. 2C). In addition, oxygen concentrations obtained after mechanical stimulation at various time points over a period of 8 days showed that the stimulation process is reproducible (Fig. 2D).
For the determination of dissolved CO2 and pH of the medium, the same experimental procedure was carried out. Dissolved CO2 concentrations in the medium are shown in Figure 3. Dissolved CO2 concentration remained constant at around 2% when samples were placed in the incubator. On mechanical stimulation, dissolved CO2 increased to ∼4% and then remained stable (Fig. 3A). CO2 concentration decreased to initial levels 50 minutes after the medium samples were transferred back into the incubator. Samples kept on the bench outside of the bioreactor chamber showed a decrease in CO2 concentration to 1% after 60 min, which then remained constant. When tissue culture well plates were transferred to the incubator after mechanical stimulation, levels of dissolved CO2 in the medium increased again to 2% within 50 min. CO2 measurements for samples stimulated at 0, 0–90, 0–180, and 0–270 kPa indicated that the higher the applied pressure, the higher the CO2 concentration in the medium after stimulation.
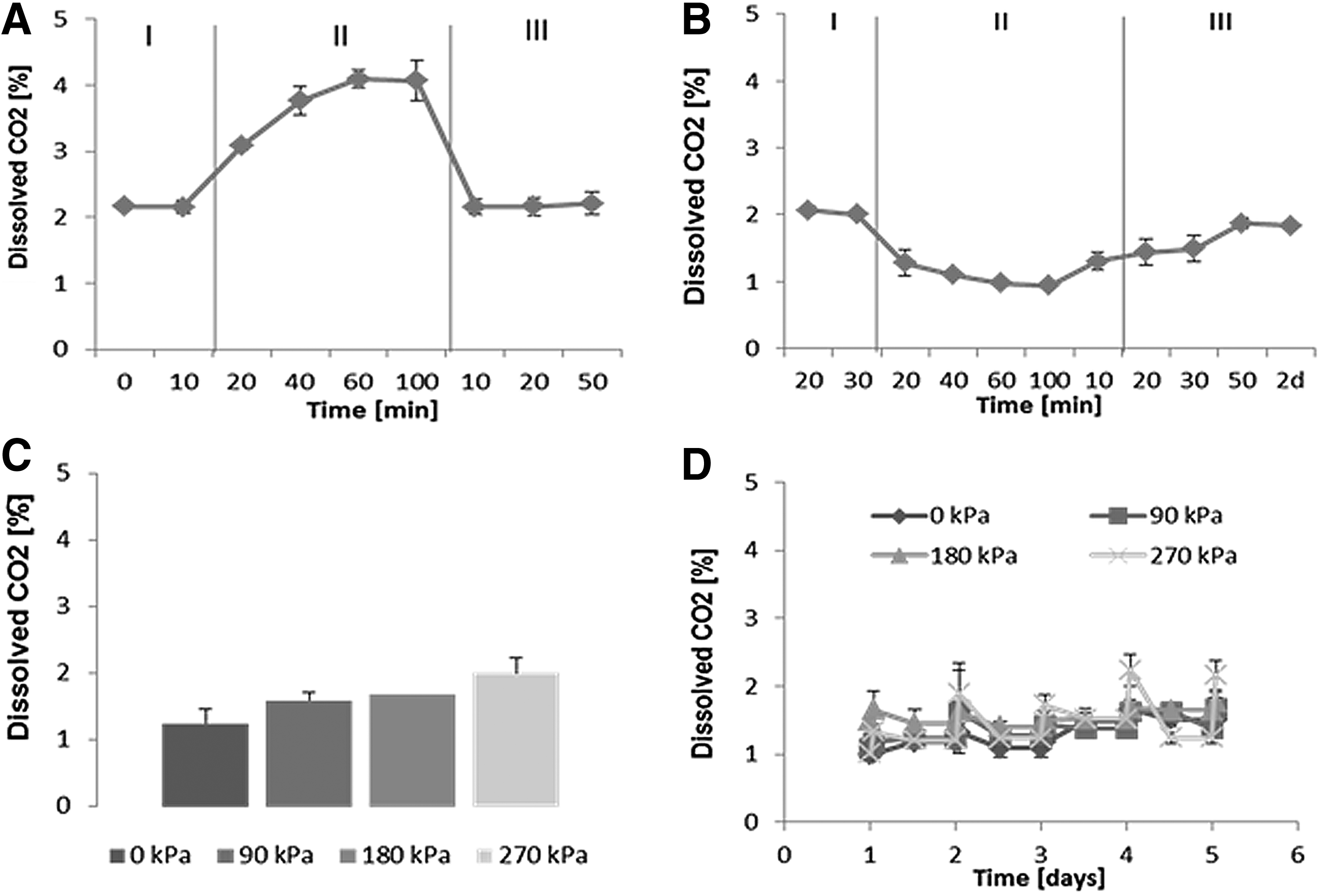
Changes of dissolved carbon dioxide concentration in cell culture medium on application of hydrostatic force. Dissolved CO2 in the cell culture medium was measured using an optical sensor. Measurements were carried out when samples were exposed to mechanical stimulation at 270 kPa
Data obtained from pH measurements of bicarbonate-buffered and HEPES-buffered (bicarbonate-buffered medium supplemented with 25 mM HEPES) medium are shown in Figure 4. The pH of the samples placed in the incubator remained stable. On mechanical stimulation, the pH decreased to 7.7 (Bicarbonate) and 7.5 (HEPES) and then remained constant. After placing the samples back into the incubator, the pH increased to initial values after around 30 min (Fig. 4A). The pH of samples maintained at room temperature increased to 8.0 (HEPES) and 8.5 (Bicarbonate), as shown in Figure 4B. Changes in pH reversed when tissue culture well plates were placed back in the incubator at norm-cell culture conditions (O2 21%, CO2 5%). Same trends were obtained for HEPES-buffered and bicarbonate-buffered media. pH fluctuations for HEPES-buffered medium were within a narrower range. Samples stimulated at various pressures show that the higher the applied pressures, the lower the pH of the bicarbonate-buffered (Fig. 4C) and HEPES-buffered medium (Fig. 4D). One-way analysis of variance test was performed in order to perform statistical analysis. Changes in pH for bicarbonate-buffered medium at different pressures were not found to be significantly different (p>0.05, p=0.1049). Changes in pH for HEPES-buffered medium at different pressures were not found to be significantly different (p>0.05, p=0.0628).
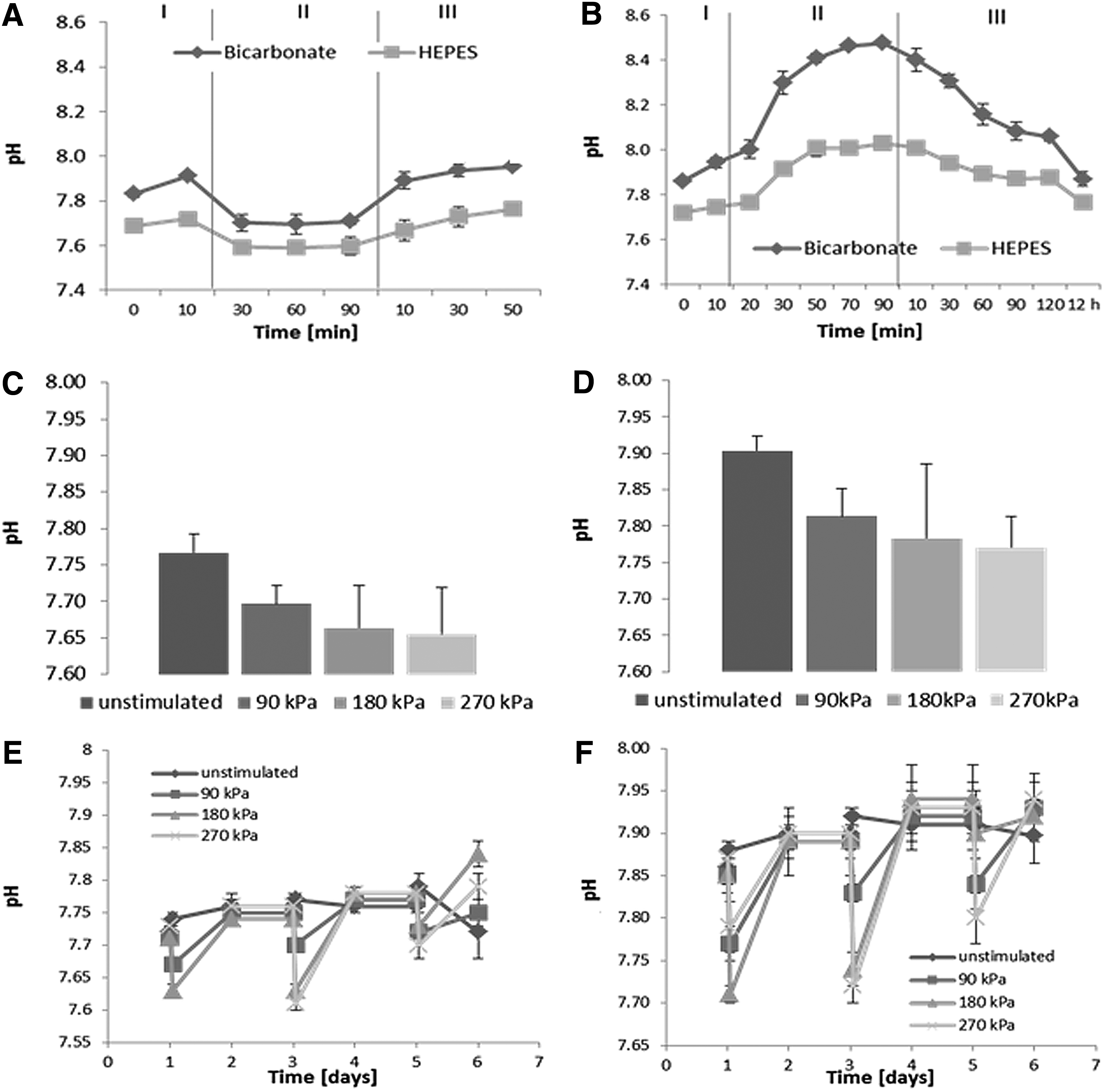
Changes of pH in cell culture medium on application of hydrostatic force. pH of HEPES-
Modeling the mechanical environment in the bioreactor chamber
In order to simulate mechanical forces and fluid flows within the bioreactor, a time-independent mathematical model of the bioreactor as previously described was solved numerically 44 for the three different theoretical setups (Fig. 1E–G).
Results for the initial environment within the gel with a given applied pressure are presented in Figure 5. The applied pressure could represent both oscillating and constant applied stimuli by adjusting the stimulus later in the simulation. In the absence of any gel, that is, the wells of the tissue culture plates are filled with culture medium only (as shown in Fig. 1D), fluid, and cell velocity fields; the associated stresses are zero; and the pressure in the well is equal to the pressure applied by the bioreactor (data not shown). When a gel is present, non-zero pressure and stress fields are present (Fig. 5). Since all changes occur in the gel and its vicinity, only the gel and surrounding region are plotted (Fig. 5A–C). Figure 5 shows the deviation of the pressure field from the applied pressure (Fig. 5D–F) and the maximal stress distribution (Fig. 5G–I) for the three different theoretical setups. For all three setups, pressure and stress was increased throughout the construct area, being maximal at the center of the constructs. This is especially clear in the case of a spherical construct as seen in Figure 5F and I. The numerical results for the three experimental setups were simulated for applied hydrostatic pressures of 0 kPa. Increasing the applied pressures did not result in a difference in the distribution of stresses and velocities, and simply added a constant, determined by the increase in applied pressure, to the pressure distribution. Cell-seeded hydrogels are exposed to maximum stresses of 2.36×10−5 Pa (setup 1), 1.15×10−4 Pa (setup 2), and 9.92×10−5 Pa (setup 3) during mechanical stimulation. The fluid flow and associated stresses and pressure fields were highly dependent on the configuration of the gels and their positioning within the wells of the tissue culture formats. The theoretical framework used is flexible, enabling the pressure and stress fields to be determined for any given shape and position of cell-hydrogel construct. Furthermore, the applied pressure can be varied to model the chosen experimental loading regime. The results shown in Figure 5 are representative plots and the numerical solutions presented are indicative of the nature of the flows, stresses, and pressure distributions found when a cell-hydrogel construct is cultured within the HF bioreactor.
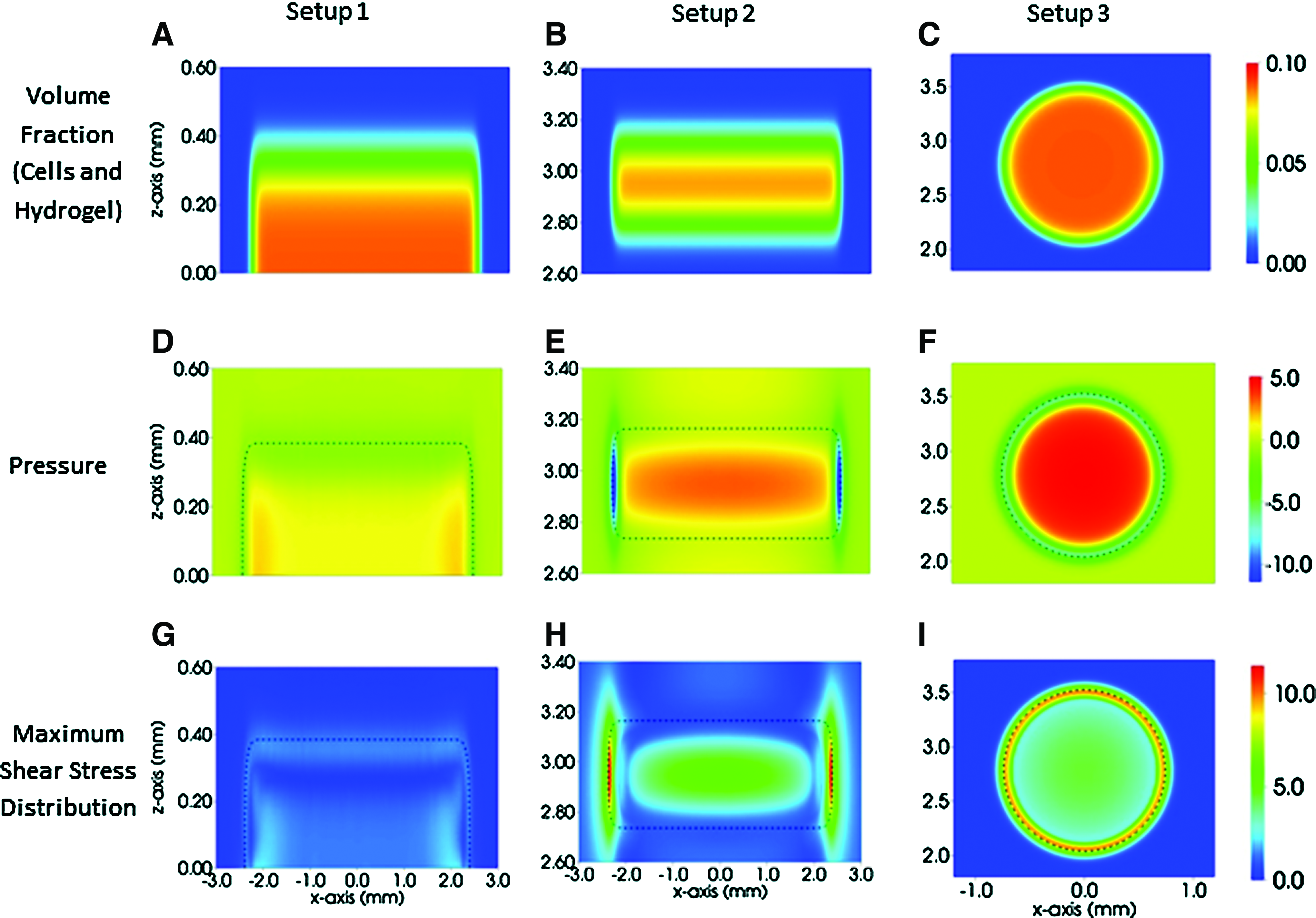
Mechanical forces and velocity fields subjected onto 3D hydrogels during mechanical stimulation at atmospheric pressure. The spatial distributions of the volume fractions of the gel (cells and hydrogel)
Conditioning of tissue-engineered constructs
Finally, the combined effect of cyclic hydrostatic pressure and chemical cues onto the cell-seeded hydrogels was investigated. Gels were set in transwell inserts and cultured for 14 days, both with and without mechanical stimulation. The morphology of the hydrogels was similar for both mechanically stimulated and non-stimulated control gels. Cells contracted the hydrogel to approximately 10% of the original 100 μL volume within 48 h. Therefore, the mechanical forces' cell constructs experienced in this study are represented by experimental setup 3 (Fig. 1F). Cross-sections through the reconstruction of a stimulated gel showed that the outer layer or cortex of the hydrogel appeared to be denser and more continuous than the inner porous structure (Fig. 6). Histological sections revealed that this outer region stains strongly for calcium, indicating enhanced mineralization in this area and hence supporting the microtomographic analysis (Fig. 6C). Quantitative MicroCT analysis showed that stimulated gels were significantly denser than unstimulated control gels (p<0.001) (Fig. 6D). A comparison of the mineralized volume with the total gel volume revealed that regions within the stimulated gels were mineralized and hence showed an increased density (p=0.08) (Fig. 6E).
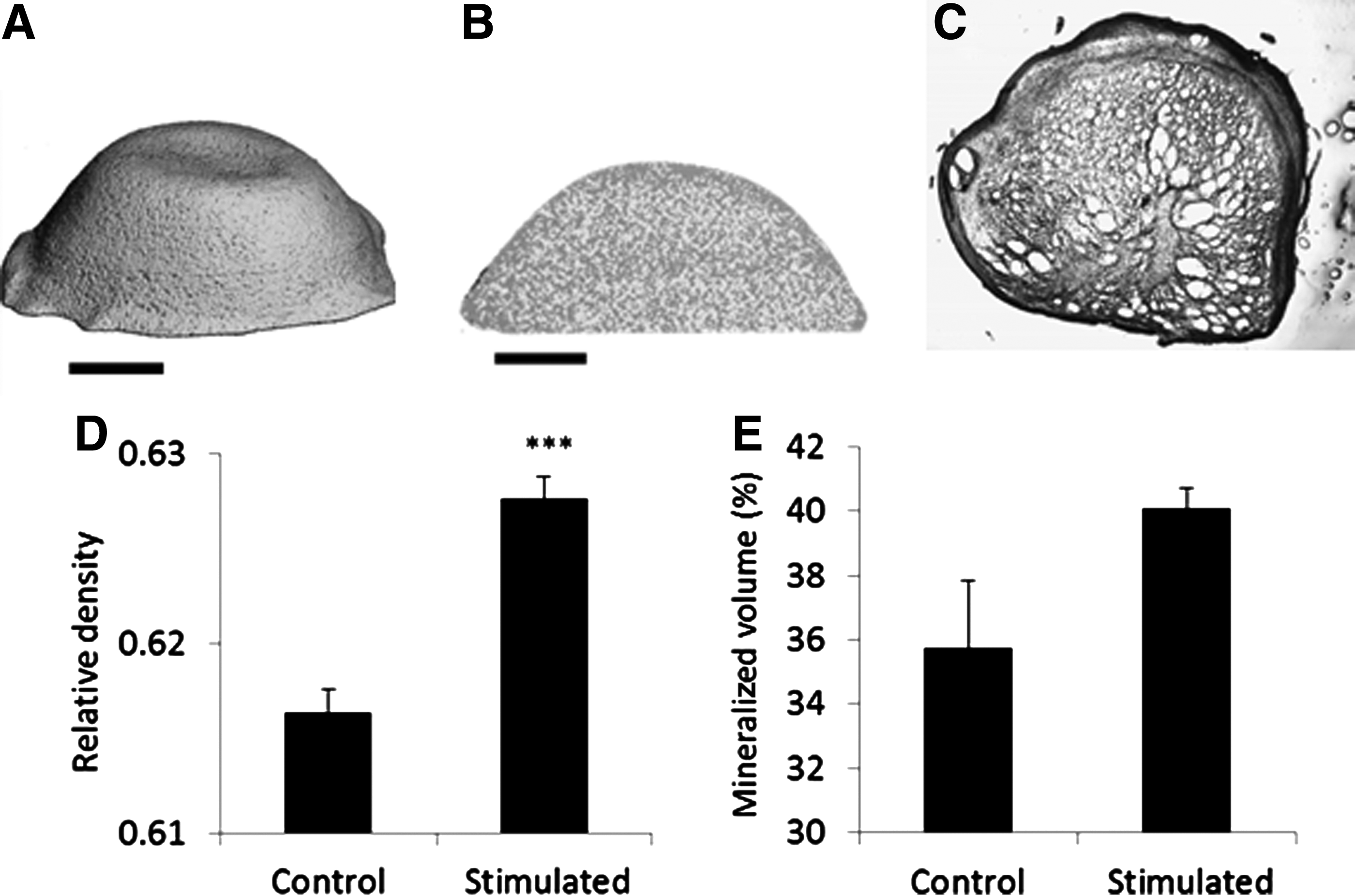
MicroCT analysis and image analysis of cell-seeded hydrogels. MicroCT reconstructions of hydrostatically stimulated hydrogels
Characterization of chick fetal femur skeletal cell on monolayer culture
Chick femur-derived skeletal cells were characterized by their morphology and ability to form calcium deposits in a monolayer. The cells were found to have a mesenchymal-like morphology (Fig. 7A). Cells were further characterized by their expression of markers, including type-I collagen, type-II collagen, alkaline phosphatase, RUNX2, SOX2, SOX9, and CD44 using quantitative PCR in comparison to GAPDH (Fig. 7B). The pluripotency marker SOX2 was expressed in these cells, as were chondrogenic (SOX9) and osteogenic (RUNX2) transcription factors (SOX9/RUNX2=4.2). Both Type-I and Type-II collagen were expressed at similar levels (COL2/COL1=0.98). Alkaline phosphatase (ALP) was also expressed and found to be actively able to dephosphorylate the substrate 4-MUP into a fluorescent product (data not shown). Furthermore, CD44 was expressed at high levels, indicating cell matrix, particularly in association with bone matrix proteins.
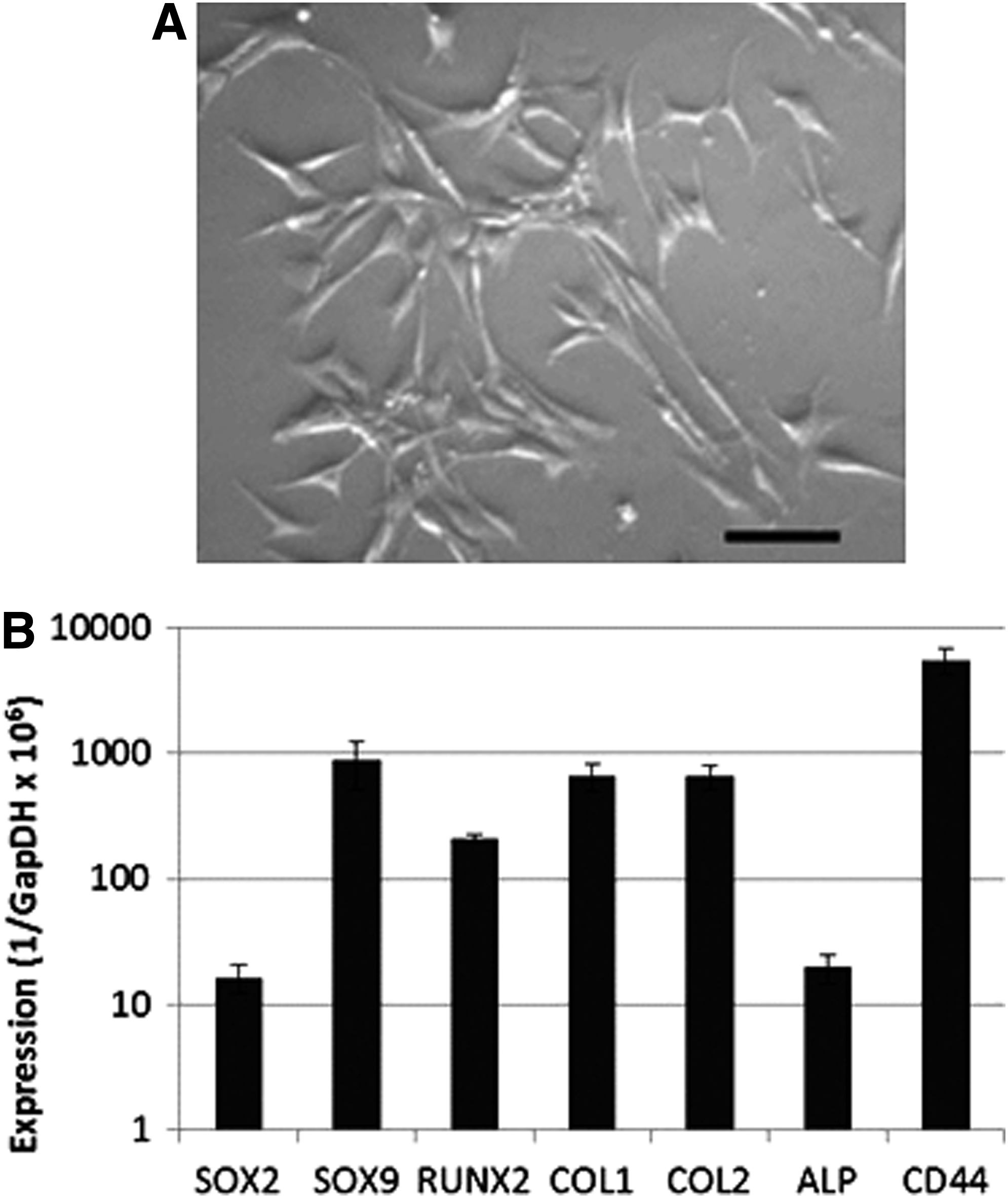
Chick femur-derived stem cells cultured on tissue culture plastic after 2 days have the morphological characteristics of mesenchymal cells
Discussion
This study was carried out to investigate the growth environment cells experience in the HF bioreactor. The results have shown how physiological parameters such as pH, dissolved O2, and dissolved CO2 changed for different applied hydrostatic pressures over time in a bioreactor system with liquid–gas interface. In addition, computational modeling was used to demonstrate the presence of non-zero velocity, pressure, and stress fields at the onset of the experiment when cell-seeded hydrogels were placed in the bioreactor, thereby providing spatial information about the mechanical environment that cells experience within the bioreactor, information which cannot be obtained experimentally.
The concentration of dissolved oxygen and CO2 in the medium remained constant in the cell culture incubator before and after stimulation in the bioreactor. Once cyclic hydrostatic pressure was applied, dissolved O2 and CO2 in the medium increased as shown in Figures 2 and 3. Pressurizing a closed system with a gas–liquid interface by applying hydrostatic pressure leads to an increase in the solubility of CO2 and O2 and, hence, resulted in higher concentrations of dissolved gases in the medium.4,46 Frieboes and Gupta reported similar results for the investigation of changes in dissolved oxygen concentration in a custom-built HF bioreactor. 4 The concentrations of dissolved gases in the medium are directly proportional to the applied pressure in the transient phase. A linear relationship between pressure and dissolved gases was found in the steady state, which is consistent with Henry's Law, 4 which describes the relationship between the concentrations of a gases dissolved in the medium and their partial pressure.
Dissolved O2 and CO2 concentrations decreased when samples were returned to the cell culture incubator after mechanical stimulation. This decrease is due to pressure differences between the bioreactor and the ambient partial pressures in the incubator (21% O2 and 5% CO2). Hence, the solubility of gases changes and gases diffuse out of the culture medium. When samples were placed on the bench outside of the bioreactor chamber, dissolved oxygen concentration remained stable. Dissolved CO2 concentration decreased, due to the differences between CO2 levels in the incubator (5% CO2) and atmospheric CO2 (0.03% CO2). CO2 diffused out of the medium, causing the pH of the medium to increase 47 as shown in Figure 4. Since CO2 dissociates into HCO3− in the medium, the pH of the medium will increase when CO2 decreases, due to a decrease in H+ concentration. 48 pH ranges during cell culture processes are between 6.00 and 7.40. 22 Here, the pH of the media samples in the incubator remained stable. On mechanical stimulation, the pH of the medium decreased and when samples were placed on the bench, the pH increased. Changes in pH did not occur beyond physiological relevant ranges.
It is widely accepted that O2, CO2, and pH affect cellular responses. Dissolved oxygen tension and pH are known to influence cell proliferation and cell differentiation15,16 as well as apoptosis.17,18 In the bioreactor system described here, air from the incubator is compressed to 2.7 times the atmospheric pressure before it is pumped into the bioreactor chamber. Therefore, air at hyperbaric pressure and hence elevated oxygen and CO2 levels are applied to the samples within the bioreactor chamber.
Several researchers have shown that the treatment of osteoblasts and bone-like cells in vitro with hyperbaric oxygen (HBO) resulted in increased cell proliferation and differentiation as well as enhanced ALP activity and collagen type I production. HBO treatment also led to enhanced osteogenesis. 48 On the other hand, research showed that low oxygen tension led to the reduction of osteoblast proliferation and differentiation as well as to diminished bone-forming ability of osteoblasts.49,50
An increase in dissolved CO2 in the cell culture medium resulted in a decrease in pH. Arnett and colleagues have shown that a lower pH resulted in the reduction of bone mass and increased bone resorption. 51 Moreover, reduced mineralization and bone nodule formation by primary rat osteoblasts were observed. 52
Increased CO2 concentration also leads to changes in H+ transport across the cell membrane and since it is able to diffuse across the cell membrane, it alters the intracellular pH of cells, changing the formation of carbonic acid by carbonic anhydrase.53,54 The extra-and intra-cellular pH is dependent on the dissolved CO2 concentration. Since CO2 is able to diffuse across the cell membrane, it causes the intracellular pH to change and, hence, leads to alterations in cell metabolism.26,27
Oxygen tension and pH in vivo strongly vary between different tissue types. Generally, in the brain, 12–14% O2, 50 3.9–12% O2 in liver 55 and 3.6–6.4% O2 in bone marrow 56 are found. In the duodenum, the pH is around 8.5; whereas the pH of blood is 7.35–7.45. The physiological relevant CO2 level ranges are between 4 and 7%. 28 In comparison, standard cell culture is carried out at around 21% O2 (atmospheric oxygen levels) and 5% CO2 (cell culture incubator) and a pH between 6.00 and 7.8 depending on cell types.
The optimum oxygen and CO2 concentrations for chick fetal femur skeletal cells are assumed to be in the range of O2 and CO2 in chick femurs during development. Dawes and Simkiss described an increase in the CO2 tension during the embryonic chick development from 19.1 mmHg (2.5% CO2) on day 11 to 57 mmHg (7.5% CO2) on day 20. 57 It was further reported that during embryonic chick limb development, a gradient of metabolites (including oxygen) along the developing limb in the chick embryo determines cell differentiation. 58 Non-vascularized (hypoxic) regions at the periphery of the limbs are observed until stage 20 (incubation day 6) when vascular patterning results in the spreading of blood vessels and, hence, an increase in oxygen levels in the surrounding tissues. 58 The exposure to elevated oxygen levels is crucial during embryonic chick development. 59 Moreover, the expansion of bone-like cells in hypoxic conditions and subsequent differentiation in hyperoxic environments was found optimum for enhanced bone formation. 60
Hence, it is assumed that the optimum CO2 concentration for culturing the chick fetal femur skeletal cells will be between 2.5% and 7.5% CO2, and optimum O2 concentration for the chick fetal skeletal cells is in the range of norm oxygen or hyperoxic for cell differentiation. In this study, chick fetal skeletal cells are cultured under standard culture conditions (21% pO2 and 5% pCO2). During mechanical stimulation in the HF, bioreactor cells are then exposed to elevated levels of oxygen during mechanical stimulation and to CO2 concentrations below 7%, simulating optimum gas concentrations for this cell type.
In this study, the oxygen tension and CO2 tension measured were about 9% above and 2% below norm cell culture ranges, respectively. The pH was within the range of cell culture standards. Since the pH of the cell culture medium also depends on the CO2 level in the incubator, 22 a slightly lower concentration of dissolved CO2 in the medium and hence a higher pH might also have been due to the ratio between the CO2 level in the incubator and the amount of bicarbonate in the medium. In order to increase the dissolved CO2 in the medium when no hydrostatic pressure is applied, it might be possible to adjust the ratio between buffer concentration and incubator CO2 level. When hydrostatic pressure is applied, CO2 level increases to physiological relevant ranges.
HEPES and sodium bicarbonate are commonly used buffer systems for cell culture media. Bicarbonate is a base that enables the neutralization of dissolved CO2 by dissociation into carbonic acid and buffers the medium under acidic conditions. 29 HEPES, on the other hand, prevents the alkalization of the medium when sodium bicarbonate is added. 30 It further reduces variations in pH due to changes in dissolved CO2 concentration in the medium. This is in correlation with the data obtained in this study, which showed that variations in pH for HEPES-buffered medium were smaller. Therefore, HEPES might be more suitable for buffering the pH in the physiological range of pH 7.2–7.6. 61 However, in contrast to bicarbonate and CO2, HEPES is not able to diffuse across the cell membrane and hence cannot contribute to the adjustment of the cell internal pH. As a consequence, the extracellular pH might remain stable; however, the ability to regulate the intracellular pH might be limited and, hence, the cytosolic pH could change. 62 Waldman et al. proposed that a combination of HEPES and bicarbonate buffer provides a more effective control over pH during cell culture. 29 For this reason, cell culture media used in this study were buffered using either bicarbonate on its own or bicarbonate-buffered medium supplemented with HEPES.
The bioreactor used in this study enables the transduction of mechanical force from the gas (air) into liquid (medium) phase through gas–liquid interface culture. 5 In order to assess the physical forces that cells and cell constructs are subjected to during mechanical stimulation, computational modeling at the initial time point was performed. Data shown in Figure 5 revealed that the centers of the cell-seeded hydrogels were subjected to lower fluid flow and stress compared with the gel peripheries. Similar observations were made in Hasel et al., who investigated the effect of cyclic hydrostatic pressure on human coronary artery smooth muscle cells, seeded in collagen-coated tissue culture well plates. 63 The precise nature of the fluid velocity, stress, and pressure fields is determined by the relative volume fractions of the cells and hydrogel, the construct shape, and its location within the culture well. Modification of these conditions will change the numerical solutions for stresses and fluid velocities. The results in Figure 5 are representative plots for given theoretical setups, and they indicate the nature of the stresses and pressure distributions that the cells and cell constructs are exposed to.
Seeding chick femur-derived skeletal cells into hydrogels enabled the investigation of the effect of cyclic hydrostatic pressure on cells cultured in a 3D environment that provides attachment sites and matrix proteins, which are known to influence cell proliferation, differentiation, and metabolic activity. Mechanically stimulated and non-stimulated cell-seeded hydrogels contracted to about 10% of their initial volume and when provided with a mechanical stimulus from the bioreactor, the cell-seeded constructs were found to be more regular in shape, significantly denser, and with a greater proportion of the gel mineralized compared with the non-stimulated (control) gels.
The contraction of hydrogels, once cells were incorporated, has been previously reported by Bell et al. and Liu et al., who investigated fibroblast-seeded collagen gels.64,65 The results suggested that gel contraction was due to the interaction of cells with the hydrogel fibers which along with the production of extracellular matrix resulted in the remodeling of fibrous gel structure.64–66 When cells adhered onto the hydrogel fibers through integrin-adhesion complexes, tensile forces are created between cells and the surrounding matrix. 65 Once cells migrate within the hydrogel, fibers are rearranged, resulting in gel contraction. 64 Liu et al. further assumed that hydrogel contraction is a consequence of changes in cell shape. 65 It is assumed that similar mechanisms are responsible for the contraction of chick fetal femur cell-seeded hydrogels in this study.
The cross-sections of both the gels and the microCT reconstructions showed that the exterior of the gel formed a denser cortex-like layer, while the interior remained porous. A denser, mineralized layer at the surface of cell constructs is a common observation in tissue engineering.67–70
It is hypothesized in this study that the greater degree of organization in the hydrostatically stimulated gels at the gel surfaces, which corresponds to the region predicted theoretically where the cells will be subjected to higher stress, is a cell-mediated response. A possible cell response was described by Brighton et al., who showed that hydrostatic pressure increased inositol triphosphate, one of the mediators of the bone cells to respond to mechanical stimulation. Inositol triphosphate is known to increase the release of intracellular calcium from bone cells. 71 Higher stress and fluid velocity at the gel surfaces might have triggered cells to deposit more calcium at the gel periphery through increased inositol triphosphate release and, hence, promoting the formation of a denser cortex-like layer at the hydrogel surface. Computational analysis has shown that higher stresses were found at the hydrogel periphery compared with its center where less calcium was found. Wong and Carter carried out a finite element analysis of an experimental study performed by Klein-Nulend et al., where fetal mouse bones were isolated and hydrostatically stimulated over 5 days, resulting in enhanced bone formation in stimulated compared with non-stimulated samples.72,73 They postulated that the mineralization was a result of stresses caused by the externally applied hydrostatic pressure. 73
In addition, the applied hydrostatic pressure of around 270 kPa is a pressure that osteocytes are subjected to in the canalicula-lacuna network of load-bearing bones, 6 and, hence, the applied hydrostatic pressure is of physiological importance for natural occurring bone formation. Previous studies have also shown that the application of various magnitudes of cyclic hydrostatic pressure to isolated bone cells, cell constructs, and ex-vivo organotypically cultured bone fragments resulted in enhanced calcium deposition and bone formation.5,72
Besides a possible cell response through physical forces exerted onto the hydrogels by the application of hydrostatic pressure, another explanation for the enhanced mineralization could be the changes in physiological parameters. 74 Physiological changes of the cell culture medium are known to affect the mineralization process and cell activity when cells are subjected to alterations in biophysical environments. 75 Urban postulated that since the hydrostatic pressure was applied through a compressed gas phase on top of a liquid phase, mineralization might have also been stimulated by fluctuations in physiological parameters.74,75
To independently investigate the effects of changes in physiological parameters and mechanical forces on cells, cell constructs, and tissues, further experiments are required. Furthermore, future work will focus on the possibility to scale up the number of samples that are mechanically pre-conditioned simultaneously for clinical application as well as the effect of HBO on cell proliferation and its influence on mineralization.
Conclusion
A novel HF bioreactor was developed in collaboration between Keele University and Instron TGT that enables the application of HF for gas–liquid interface culture at physiological relevant pressures and provides a growth environment for engineered tissues.
The application of hydrostatic pressure resulted in fluctuations of dissolved O2, CO2, and pH of the medium, which remained within the human physiological range. These changes in environmental parameters were dependent on the applied hydrostatic pressure, which means that the higher the applied hydrostatic pressure, the higher the measured dissolved oxygen and CO2 concentration and the lower the pH in the cell culture medium. Changes in O2, CO2 levels and pH were reversible. Further investigation is necessary to investigate the effect of these changes on potential cell fate and tissue development.
Mathematical modeling and numerical simulation can give the spatial distribution of the dependent variables that are impossible to be obtained experimentally, such as fluid velocity, stresses, and pressure. Here, a snapshot of the initial behavior of three theoretical setups has been shown, and it has been demonstrated that non-zero spatial distributions in pressure and stress are predicted by the model. Cells and cell constructs are subjected to various magnitudes of stresses and fluid velocities depending on the position of the cell-hydrogel construct in the tissue culture well plate rather than the magnitude of applied pressures. Moreover, these distributions suggest that stress is larger toward the periphery of the gel, indicating that stimulation of cell proliferation and the deposition of extra cellular matrix may be increased there. A time-dependent model can also be simulated in order to indicate how the dynamics alter over the course of the experiment. Once the mathematical model has been validated, it can be used to predict system behavior, enabling optimization and control of the experimental system.
The results of this study suggest that forces and fluid velocities experienced by cells at the periphery of the hydrogel trigger a bone cell response, causing the formation of a denser mineralized cortex-like layer at the hydrogel periphery.
Footnotes
Acknowledgments
The author would like to thank the EPSRC Center for Innovative Manufacturing in Regenerative Medicine (ECP007/1010) and Instron TGT, Ltd. for funding. KHLL would like to acknowledge the support of the EPSRC through a Systems Biology Doctoral Training Centre studentship (Grant No EP/G03706X/1) and JMO would like to acknowledge the support of the EPSRC and Microsoft Research through a 2020 Science Research Fellowship.
Disclosure Statement
No competing financial interests exist.
