Abstract
Adipose tissue–derived mesenchymal stem cells (ADMSCs) are multipotent cells. Here we examined whether human ADMSCs (hADMSCs) could differentiate into cardiomyoblast-like cells (CLCs) by induction with dimethylsulfoxide and whether the cells would be utilized to treat cardiac dysfunction. Dimethylsulfoxide induced the expression of various cardiac markers in hADMSCs, such as α-cardiac actin, cardiac myosin light chain, and myosin heavy chain; none of which were detected in noncommitted hADMSCs. The induced cells were thus designated as hADMSC-derived CLCs (hCLCs). To confirm their beneficial effect on cardiac function, hCLC patches were transplanted onto the Nude rat myocardial infarction model, and compared with noncommitted hADMSC patch transplants and sham operations. Echocardiography demonstrated significant short-term improvement of cardiac function in both the patch-transplanted groups. However, long-term follow-up showed rescue and maintenance of cardiac function in the hCLC patch-transplanted group only, but not in the noncommitted hADMSC patch-transplanted animals. The hCLCs, but not the hADMSCs, engrafted into the scarred myocardium and differentiated into human cardiac troponin I-positive cells, and thus regarded as cardiomyocytes. Transplantation of the hCLC patches also resulted in recovery of cardiac function and improvement of long-term survival rate. Thus, transplantation of hCLC patches is a potentially effective therapeutic strategy for future cardiac tissue regeneration.
Introduction
Cellular cardiomyoplasty was developed as a new approach to restore impaired heart function,3,4 using a variety of cell types, with encouraging initial results.3–5 Mesenchymal stem cells (MSCs) seem particularly advantageous for cellular therapy in general because they are multipotent, potentially immune privileged, 6 and expand easily ex vivo. MSCs also proliferate rapidly, induce angiogenesis, and can differentiate into cardiomyogenic cells.7–10 An MSC population was recently isolated from human adipose tissue, which is abundantly available and can be resected easily and safely in most patients.11,12 These adipose tissue-derived cell lineages showed cardiomyocytic differentiation and rescued cardiac dysfunction in a myocardial infarction (MI) animal model. Thus, the adipose tissue is a convenient and preferred source of stem cell recovery for cardiac therapy. Recently, transplantation of monolayered adipose tissue-derived MSCs (ADMSCs) into MI rats reversed wall thinning in the scarred area and improved cardiac function in a short term, with the engrafted sheet of cells forming a thick stratum containing newly formed vessels and scattered cardiomyocytes derived from the implanted cells. 13 As patients with severe heart failure desire sustained and long-term recovery of cardiac function after treatment rather than short-term improvement, continued efforts should be made to develop cell transplants from ADMSCs that survive and differentiate into cardiomyocytes in vivo for subsequent engraftment onto scarred myocardium.
This study investigated the differentiation of human ADMSCs (hADMSCs) into cardiomyoblast-like cells (CLCs) in vitro, analyzed the functional and histological regeneration of damaged myocardium after transplantation of CLCs in vivo, and examined the effects of such transplantation on long-term patient survival.
Materials and Methods
Adipose tissues from human subjects
Excess omental adipose tissues were resected from the gastro-omental artery during coronary artery bypass graft surgery and gastrectomy in 10 subjects [4 men and 6 women; age, 55 ± 5 years, mean ± standard error of mean (SEM); range, 40–60 years]. All subjects provided informed consent, and the Review Board for Human Research of Osaka University Graduate School of Medicine approved all protocols. All subjects fasted for at least 10 h before surgery and none was on steroid therapy at the time of surgery. Ten to 50 grams of adipose tissue was obtained from each subject.
Isolation of hADMSCs and differentiation into CLCs
hADMSCs were obtained as reported previously, with modification.11,14 Briefly, the resected excess adipose tissue was minced and then digested at 37°C for 1 h in Hank's balanced salt solution (Gibco–Invitrogen, Grand Island, NY) containing 0.075% collagenase type II (Sigma–Aldrich, St. Louis, MO). Digests were filtered through a cell strainer (BD Bioscience, San Jose, CA) and centrifuged at 800 g for 10 min. Red blood cells were excluded using density gradient centrifugation with Lymphoprep (d = 1.077; Nycomed, Oslo, Norway), and the remaining cells were cultured in Dulbecco's modified Eagle's medium (Gibco–Invitrogen) with 10% defined fetal bovine serum (Hyclone, Logan, UT) for 24 h at 37°C. Following incubation, the adherent cells were washed extensively and then treated with 0.2 g/L ethylenediaminetetraacetate solution (Nacalai Tesque, Kyoto, Japan). The resulting suspended cells were replated at a density of 10,000cells/cm2 on human fibronectin-coated dishes (BD BioCoat, Franklin Lakes, NJ) in 60% Dulbecco's modified Eagle's medium-low glucose, 40% MCDB-201 medium (Sigma–Aldrich), 1 × insulin-transferring selenium (Gibco–Invitrogen), 1 nM dexamethasone (Sigma–Aldrich), 100 μM ascorbic acid 2-phosphate (Sigma–Aldrich), 10 ng/mL epidermal growth factor (PeproTec, Rocky Hill, NJ), and 5% fetal bovine serum. After passaging five to six times in the same medium, the hADMSCs were used for transplantation. Cardiomyocytic differentiation was achieved by inducing hADMSCs with 0.1% dimethylsulfoxide (DMSO) for 48 h, resulting in a population named CLCs.
Reverse transcriptase–polymerase chain reaction
Total RNA was isolated from hADMSCs and cardiomyoblasts using an RNAeasy kit (Qiagen, Hilden, Germany). As a control, excess human myocardium was resected during maze surgery from 10 matched subjects (4 men and 6 women; age, 55 ± 5 years, mean ± SEM; range, 40–60 years) with informed consent. Control subjects also fasted for at least 10 h before surgery, and none was taking steroids. Approximately 1 g of myocardium was obtained from each subject, and the same protocol was performed to obtain total RNA. After treatment with DNase, cDNA was synthesized from 500 ng total RNA using Superscript III reverse transcriptase RNase H minus (Invitrogen, Carlsbad, CA). The absence of DNA contamination in RNA samples was confirmed with polymerase chain reaction (PCR) primers flanking an intron. Primers and the reaction conditions are described in Table 1. The PCR products were fractionated by 2% agarose gel electrophoresis.
Model animals for MI
The left anterior descending coronary artery of rats with severe combined immunodeficiency was ligated. In brief, rats were anesthetized with nembutal (40 mg/kg), before being intubated and ventilated at a rate of 60 cycles/min with a tidal volume of 5 mL under room air supplemented with oxygen (2 L/min). The hearts were exposed through the fifth left-intercostal space and the left anterior descending was ligated. After 4 weeks, the hearts were again exposed through the fifth left-intercostal space, and the infarct area was identified visually based on surface scarring and abnormal wall motion. Cell sheets were subsequently implanted onto the infarcted myocardium. The control group was treated similarly, but no cell sheets were implanted. The Osaka University Graduate School of Medicine Standing Committee on Animals approved all experimental protocols.
Preparation of monolayered cell sheets
After four to five passages, the hADMSCs were trypsinized and then replated onto 35-mm temperature-responsive dishes (CellSeed, Tokyo, Japan) in 2 mL of expansion medium at 1 × 106 cells per dish. After culture at 37°C for 2 days, 0.1% DMSO was added to the medium on half of the dishes to differentiate the hADMSCs into cardiomyoblasts. After 2 days of culture, the cells were incubated again at 20°C. Within 20 min, the hADMSCs and CLC sheets detached spontaneously and floated up into the medium for use as monolayered cell grafts.13,15–17
Assessment of rat cardiac function
Cardiac ultrasound studies were performed before ligation, before implantation, and at 2, 4, 8, 10, 12, 14, and 16 weeks after implantation using a SONOS 7500 (Philips Medical Systems, Andover, MA). Plasma atrial natriuretic protein (ANP) level was analyzed using an ANP ELISA system (Phoenix Pharmaceuticals, Burlingame, CA) by following the instructions supplied by the manufacturer.
Histological analyses
The rat hearts were dissected out and immediately fixed overnight in 4% paraformaldehyde, washed in 70% alcohol, dehydrated through a graded ethanol series, cleared in xylene, and finally processed for embedding in paraffin wax. Paraffin sections were cut at 5 μm thickness, delineated on the microscope slide using a Dako pen (Dako, Glostrup, Denmark), deparaffinized in xylene, and then rehydrated through a graded ethanol series into distilled water. The sections were then immersed in Target Retrieval Solution (Dako) in distilled water and boiled, followed by cooling at room temperature for 20 min. The sections were then washed in two changes of Tris-buffered saline (TBS), pH 7.4, followed by 1% polyoxyethylene sorbitan monolaurate (Tween 20) in TBS (TBS-T), and then an overnight incubation with 10% Blocking One® (Nacalai Tesque) in TBS-T. The sections were then incubated in a humid chamber for 16 h at 4°C with mouse monoclonal antibodies to α-cardiac actin (α-CA) and human troponin I, diluted in the blocking solution, followed by Alexa Fluor 546-labeled donkey anti-goat IgG (Molecular Probes, Eugene, OR). The stained slides were viewed on a BioZero laser scanning microscope (Keyence, Osaka, Japan).
Statistical analysis
All data were expressed as mean ± SEM. Differences between groups were analyzed for statistical significance by the Student's t-test using SPSS Statistics 17.0 (SPSS, Inc., Chicago, IL). A p-value less than 0.05 denoted a statistically significant difference. Survival curves were constructed by the Kaplan–Meier method and survival among groups was compared using the Log-Rank test (StatMate III for Windows; Atoms, Tokyo, Japan).
Results
Cardiac differentiation of hADMSCs into CLCs
The potential for hADMSCs to differentiate into CLCs was evaluated from the mRNA expression of several cardiac differentiation markers by reverse transcriptase-PCR before and after DMSO induction, as follows: islet-1 is a cardiac stem cell marker; Nkx2.5 and GATA-4 are transcription factors required for subsequent cardiac differentiation; and α-CA, myosin light chain, and myosin heavy chain (MHC) are markers of cardiac differentiation (Fig. 1A). Preinduced hADMSCs expressed islet-1 and Nkx2.5 mRNA, but not that of GATA-4, α-CA, myosin light chain, or MHC. After induction by DMSO for 48 h, hADMSCs expressed all markers, indicating that DMSO treatment successfully differentiated hADMSCs into cells of the cardiac lineage, and these induced cells were named CLCs.
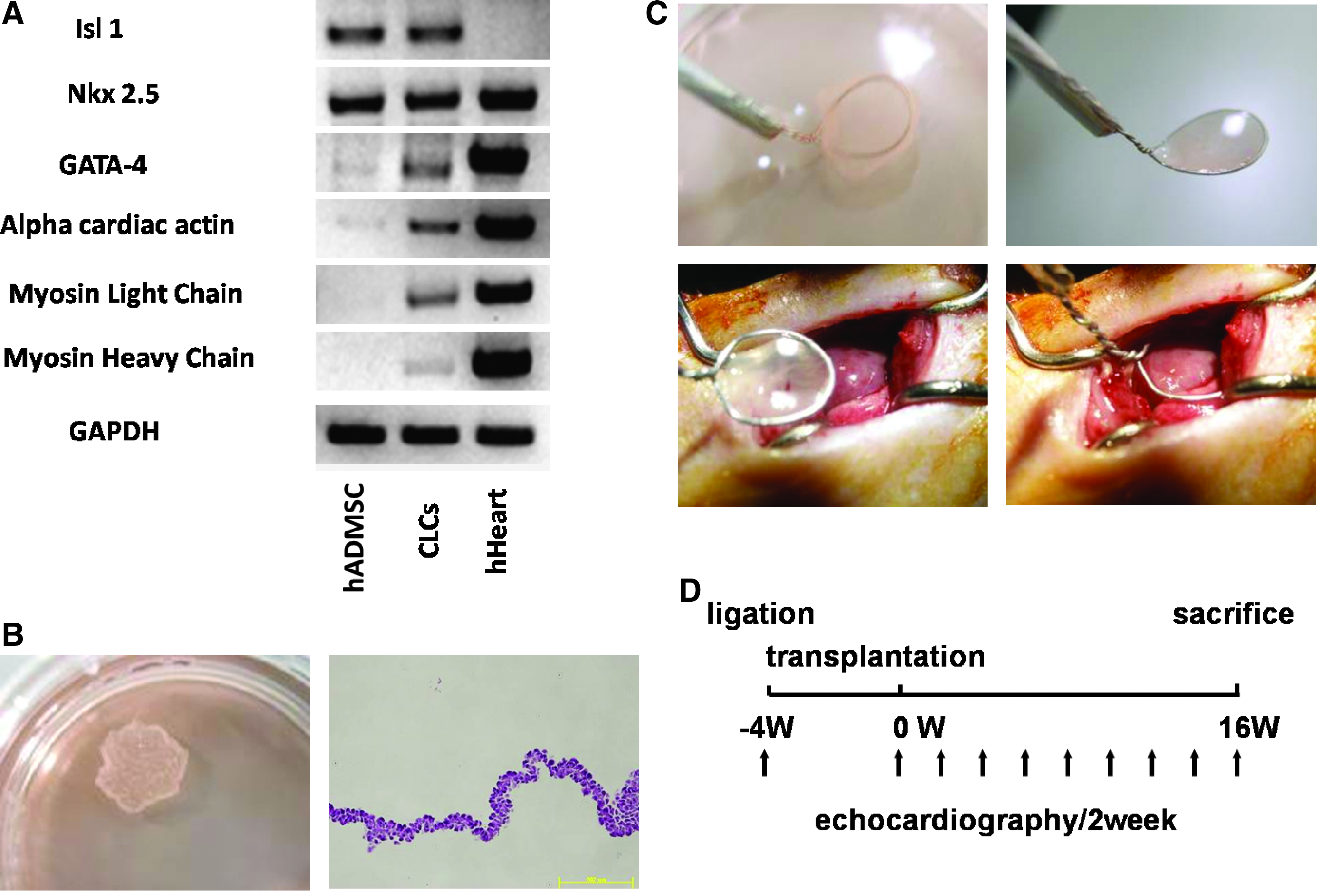
Preparation and transplantation of human adipose tissue-derived mesenchymal stem cell (hADMSC)-derived cardiomyoblast-like cell (hCLC) patches. (
Preparation and transplantation of hADMSC-derived CLC patches
To evaluate the potential therapeutic usefulness of CLCs, we designed an experimental rat model of coronary ligated infarction to assess cardiac function after transplantation of CLC patches. CLC and control hADMSC patches were prepared from cell sheets, as described earlier (Fig. 1B). These patches were transplanted onto the scarred area of the left ventricular wall in the MI model Nude rats, whose left anterior descending artery had been ligated 4 weeks before graft implantation (Fig. 1C, D). Sham transplantations were also performed.
Effects of CLC transplantation on cardiac function and survival rate
Cardiac function was assessed by echocardiography at preligation, pretransplantation, and every 2 weeks after transplantation (Fig. 1D). Sixteen weeks after transplantation, the treated animals were sacrificed and cardiac tissues prepared for histological examination. Four weeks after graft implantation, wall motion was improved in both control and CLC patch-implanted hearts. However, the wall motion of control and noncommitted hADMSC patch-transplanted heart tissue was exacerbated at 16 weeks after transplantation, while improved motion was maintained with the CLC patch transplants (Fig. 2A). In the early phases of the post-transplantation period, left ventricular diastolic dimension was significantly reduced in both the transplanted groups, but by 8 weeks after implantation this parameter increased in the control hADMSC patch-transplanted group, whereas it remained unchanged in those animals that received CLC patch transplants (Fig. 2B). Likewise, left ventricular ejection fractions improved in both the implanted groups until 8 weeks, after which time it worsened only in the group transplanted with noncommitted hADMSC patches (Fig. 2B).
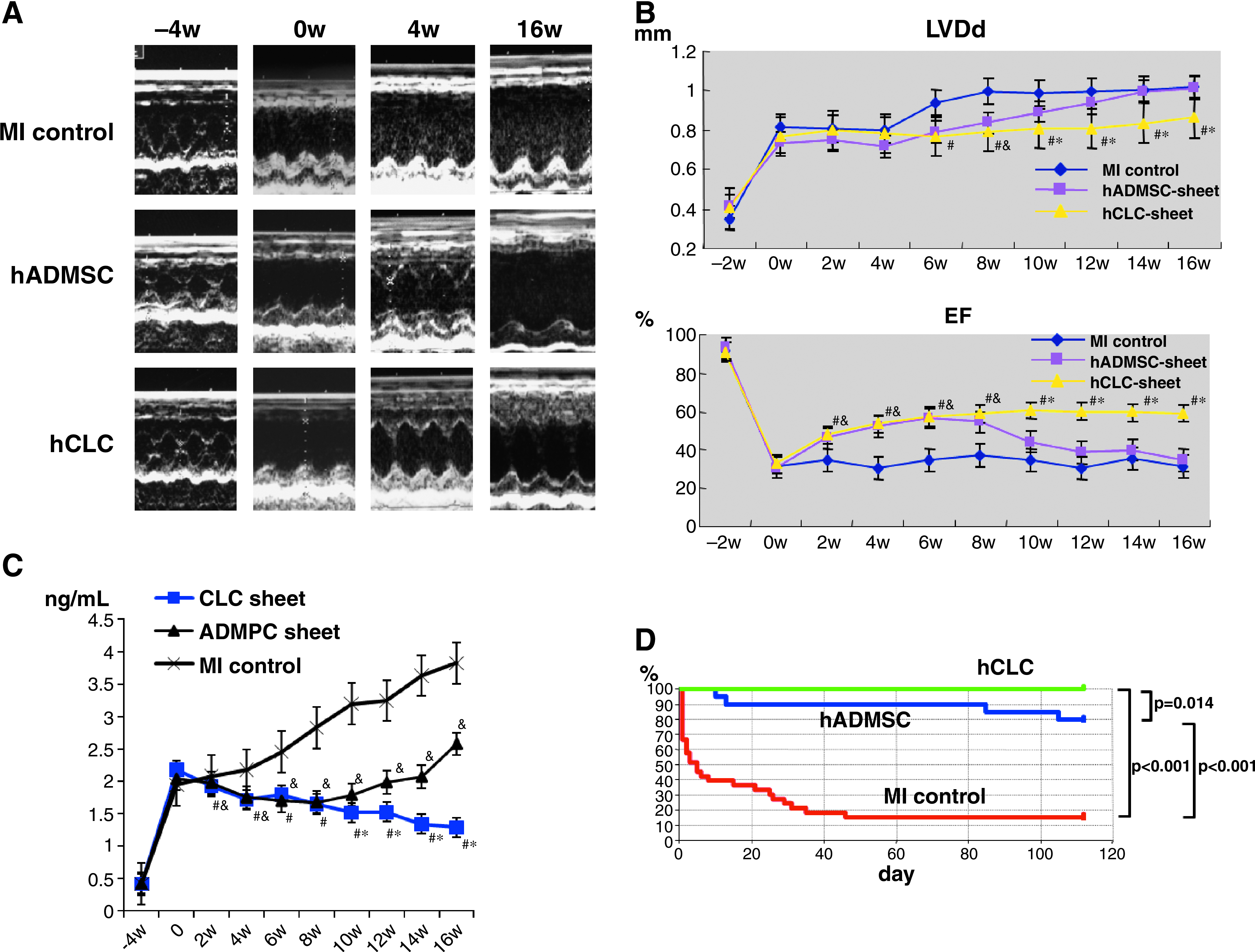
Effects of CLC patch transplantation on cardiac function and long-term survival. (
ANP was then measured to confirm that chronic heart failure due to MI could be treated by CLC patch transplantation (Fig. 2C). The ANP levels were significantly increased after MI in all groups (Fig. 2C). The sham-operated MI control group showed incremental increases in plasma ANP over the time course of examination, whereas both CLC patch- and hADMSC patch-transplanted animals had low ANP levels until 8 weeks after treatment. However, ANP levels increased subsequently in the hADMSC patch-transplanted group, whereas the CLC patch-transplanted group maintained the improvement in ANP levels.
The Kaplan–Meier survival curve showed higher long-term survival rates in cell patch-transplanted groups than in sham-operated MI controls (Fig. 2D). Notably, no rat died after transplantation of an hADMSC-derived CLC patch. Survival at 16 weeks after surgery was 100% for the CLC group, 80% for the hADMSC group, and 16% for the sham-operated group, with a significant difference between the two transplanted groups. These results suggest that transplantation of hADMSC-derived CLCs has beneficial effect in rats with heart failure induced by MI.
Effects of CLC transplantation on cardiac structure
Cardiac structure was next examined histologically to analyze further the difference between CLC patch- and noncommitted hADMSC patch-transplanted animals in the longer term (Fig. 3). On hematoxylin and eosin and Masson trichrome staining, the sham-transplanted MI control rats showed only a thin layer of cardiac muscle and fibrotic tissues in the scarred anterior left ventricular wall (Fig. 3A, B). Rats implanted with noncommitted hADMSCs showed small patches of cardiac muscles over that seen in the control MI rats (Fig. 3C, D). On the other hand, the rats transplanted with CLC patches showed significant reversal of the infracted myocardium and a full cardiac muscle layer overlaying the transplanted area (Fig. 3E, F, arrowheads).
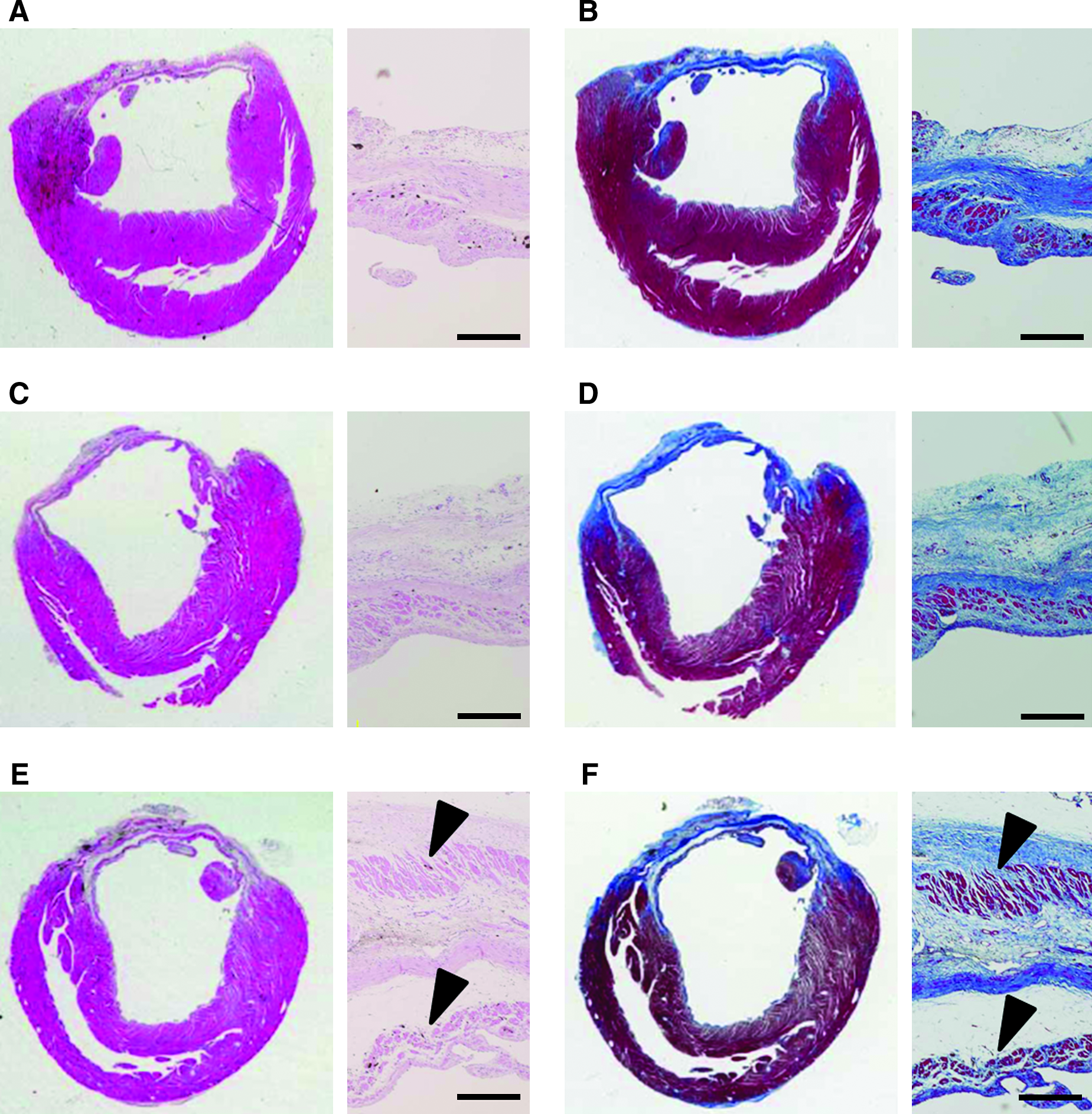
Effects of CLC transplantation on cardiac structure. Photomicrographs showing representative myocardial sections stained with hematoxylin and eosin (
CLCs differentiate into cardiac muscle in situ
The in situ differentiation capacity of the implanted cell sheets into cardiomyocytes after grafting onto the scarred myocardium was assessed by immunohistochemical staining for α-CA and human troponin I (Fig. 4). Thin layers of α-CA-positive cells were observed on the scarred myocardium of sham-operated MI control rats (Fig. 4A). A similar but thicker layer of α-CA-positive cells was apparent in the tissues from noncommitted hADMSC-transplanted rats (Fig. 4A, C), whereas the CLC patch-transplanted group showed two cardiac muscle layers positive for α-CA (Fig. 4E, arrow and arrowhead). There were no human troponin I-positive cells in the sham-operated MI control group (Fig. 4B), but some were observed in the noncommitted hADMSC patch-transplanted group (Fig. 4D). As shown in Figure 4F, large amounts of human troponin I-positive myocardium was observed in the CLC-transplanted animals (arrow) in addition to some human troponin I-negative but α-CA-positive myocardium in the internal myocardial layer (Fig. 4E, F, arrowhead). These results indicated that CLCs can efficiently differentiate into cardiomyocytes in situ.
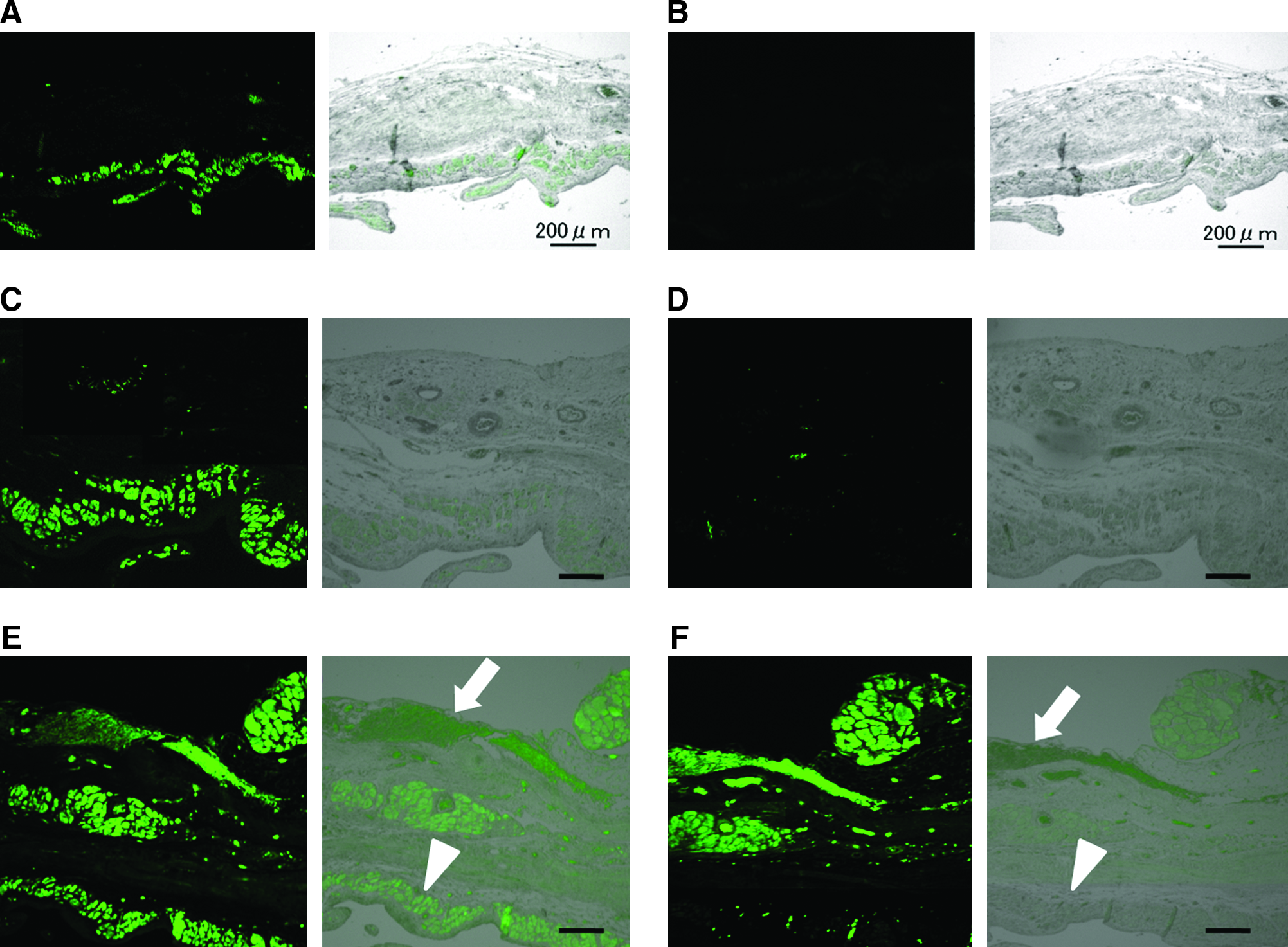
CLCs differentiate into cardiac muscles in situ. Immunofluorescence with anti-α-CA (
Discussion
There are several advantages to hADMSC-derived CLC patch transplantation for regeneration therapy. First, the source of adipose-derived cells is easily and safely accessible and the cells can be obtained in large quantities, without serious ethical issues. Second, hADMSCs differentiate into CLCs by induction with DMSO, which is available in current good manufacturing practice grade. Third, hADMSC-derived CLCs can differentiate into cardiomyocytes in vivo within the myocardial milieu, resulting in increment of myocardial muscle force. Finally, reconstruction of thick myocardial tissue rescued cardiac dysfunction after MI and improved long-term survival.
The choice of cell source is critical for realizing success in cellular therapy. 18 Liposuction surgeries yield from 100 mL to >3 L of lipoaspirate tissue. 19 The initial isolation of cells from adipose tissue was described by Bjorntorp et al. 14 This procedure was since modified to isolate cells from human adipose tissue specimens.20–22 In this context, Zuk et al. 11 reported that the preadipocytes exhibited stem cell features as MSCs, currently known as ADMSCs. Because of the above-stated advantages of procuring cells for therapy from adipose tissues, hADMSCs present a potential and promising source for cellular therapy, even in patients with post-MI severe heart failure.
The in vitro differentiation of ADMSCs is now well reported, and experimental findings in recent years suggested considerable therapeutic potential for cellular replacement in the context of acute MI and chronic progressive cardiac disease.23–27 Stem cells are differentiated into a cardiomyocyte lineage by treatment with 5-azacytidie, retinoic acid, oxytocin, and many other reagents.28–32 We proposed that DMSO could differentiate hADMSCs into CLCs, based on the differentiation of P19 embryonic stems cells into cardiomyocytes with DMSO.31–33 It was notable that DMSO is also available in current good manufacturing practice grade. Unfortunately, DMSO-treated hADMSCs did not show spontaneous beating as their terminal differentiation function, but the cells did express the mature markers α-CA, myosin light chain, and myosin heavy chain to a lesser extent. There are no reports of the use of DMSO to commit ADMSCs to a cardiomyocytic lineage. The mechanism by which DMSO elicits its effect on differentiation remains unclear. It is possible that DMSO increases intracellular calcium ion concentration, thereby elevating phosphatidylethanolamine levels in the cells and controlling the distribution of protein kinase C to commit the P19 stem cells.33–36 These mechanisms should be investigated further in the near future.
The in vitro differentiation of ADMSCs has been well reported,23–27 although only a few reports relate to the differentiation of these cells into cardiomyocytes in vivo. Recently, Miyahara et al. 13 reported the use of monolayered ADMSCs for myocardial repair. In their study, rat ADMSCs were isolated and grown as intact monolayer sheets using temperature-responsive culture dishes. Placement of the ADMSC sheets onto a scarred myocardium in rats resulted in diminished scarring and enhanced cardiac structure and function. Histological analysis demonstrated that the engrafted ADMSC sheets grew to form a thickened layer over the infarcted muscle that included newly formed vessels and a few cardiomyocytes. In our study, hADMSC-derived CLCs differentiated into cardiomyocytes in a myocardial milieu, indicated by the immunohistological results in which transplanted cells expressed human troponin I in vivo. Newly developed myocardium might augment cardiac function, and thus hADMSC patch transplantation was performed as a control. Cardiac dysfunction was rescued in a short term, although the numbers of cardiomyocytes derived from transplanted cells were low. In this context, Gimble et al. 19 suggested that hADMSCs might secrete angiogenic factors and/or antiapoptotic factors.
Transplantation of the hADMSC-derived CLC regenerated the thick myocardial tissues, rescued cardiac dysfunction after MI, and improved long-term survival rate compared with the noncommitted hADMSCs and sham-operated MI controls. The existing literature suggests that ADMSCs can be engrafted and survive within an infarcted myocardial milieu, acquire phenotypic markers consistent with cardiomyocytic and vascular-related lineages, and have a positive impact on structural and functional endpoints.19,23–27 These are desirable outcomes for cardiac function and survival. However, few reports have applied long-term observation of the transplanted animals. Our study therefore observed the three rat groups for 16 weeks after transplantation. Only CLC transplantation provided the desired outcome at the experimental endpoint. Despite these encouraging results, much progress is needed to realize the hope of cell therapies for myocardial damage. First, delivery of the cell sheets to patients should be optimized for each given disease. Second, the issue of vascularization should be considered in the infracted or affected tissues after transplantation, because many small CLC patches would be necessary for a clinical cure. Finally, the value and impact of CLC patch transplantation should be confirmed in large animal models before embarking on clinical applications.
In conclusion, we showed that the phenotype of hADMSCs could be changed to that of CLCs by induction with DMSO. These hADMSC-derived CLCs engrafted into a scarred myocardium and differentiated into cardiomyocytes. The CLC patch transplantation also resulted in recovery of cardiac function and improved survival rate. Thus, transplantation of hADMSC-derived CLC patches in heart patients might be a potentially effective therapeutic strategy for cardiac tissue regeneration in the near future.
Footnotes
Acknowledgments
This report was supported in part by a grant-in-aid for Yoshiki Sawa from the New Energy and Industrial Technology Development Organization of Japan and in part by a grant-in-aid for Akifumi Matsuyama from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Disclosure Statement
No competing financial interests exist.
