Abstract
Tissue Engineering (TE) is a branch of Regenerative Medicine (RM) that combines stem cells and biomaterial scaffolds to create living tissue constructs to restore patients' organs after injury or disease. Over the last decade, emerging technologies such as 3D bioprinting, biofabrication, supramolecular materials, induced pluripotent stem cells, and organoids have entered the field. While this rapidly evolving field is expected to have great therapeutic potential, its development from bench to bedside presents several ethical and societal challenges. To make sure TE will reach its ultimate goal of improving patient welfare, these challenges should be mapped out and evaluated. Therefore, we performed a systematic review of the ethical implications of the development and application of TE for regenerative purposes, as mentioned in the academic literature. A search query in PubMed, Embase, Scopus, and PhilPapers yielded 2451 unique articles. After systematic screening, 237 relevant ethical and biomedical articles published between 2008 and 2021 were included in our review. We identified a broad range of ethical implications that could be categorized under 10 themes. Seven themes trace the development from bench to bedside: (1) animal experimentation, (2) handling human tissue, (3) informed consent, (4) therapeutic potential, (5) risk and safety, (6) clinical translation, and (7) societal impact. Three themes represent ethical safeguards relevant to all developmental phases: (8) scientific integrity, (9) regulation, and (10) patient and public involvement. This review reveals that since 2008 a significant body of literature has emerged on how to design clinical trials for TE in a responsible manner. However, several topics remain in need of more attention. These include the acceptability of alternative translational pathways outside clinical trials, soft impacts on society and questions of ownership over engineered tissues. Overall, this overview of the ethical and societal implications of the field will help promote responsible development of new interventions in TE and RM. It can also serve as a valuable resource and educational tool for scientists, engineers, and clinicians in the field by providing an overview of the ethical considerations relevant to their work.
Impact statement
To our knowledge, this is the first time that the ethical implications of Tissue Engineering (TE) have been reviewed systematically. By gathering existing scholarly work and identifying knowledge gaps, this review facilitates further research into the ethical and societal implications of TE and Regenerative Medicine (RM) and other emerging biomedical technologies. Moreover, it will serve as a valuable resource and educational tool for scientists, engineers, and clinicians in the field by providing an overview of the ethical considerations relevant to their work. As such, our review may promote successful and responsible development of new strategies in TE and RM.
Introduction
Tissue Engineering (TE) uses biomaterials, (stem) cells and bioactive molecules to construct functional, living three-dimensional (3D) tissues in a laboratory environment (in vitro) or directly in a patient's body (in situ). Examples include tissue engineered skin, 3D bioprinted bone, and liver organoids for transplantation. TE is one of three pillars of the broader field of Regenerative Medicine (RM), which aims to “replace or regenerate human cells, tissue, or organs to restore or establish normal function.” 1 Stem cell (SC) transplantation and gene transfer (GT) constitute the other two pillars.
As a rapidly evolving medical technology that has been argued to be potentially disruptive 2 and even paradigm-changing, 3 TE holds great promise for novel cures, but also requires attention to the ethical implications of its development, clinical translation, and future impact on individuals and society. Addressing such ethical implications early on and embedding societal values into the design of TE products will help TE to reach its ultimate goal of improving patient welfare. 4
The TE field has been maturing over the last two decades and ethical issues have received attention of ethicists, social scientists, and ethics-minded biomedical scientists and engineers. This body of literature has last been reviewed comprehensively in 2008, at which time the field was still in its infancy. 5 Since then, the first applications have reached the stage of clinical application.6,7 Emerging technologies such as 3D bioprinting, biofabrication, supramolecular materials, induced pluripotent stem cells (iPSCs), and organoids have entered the TE field, and a shift has taken place toward strategies that mobilize the inherent regenerative capacity of the human body–and its endogenous cell populations–to a greater extent than before (in situ TE).2,8
Therefore, we have performed a systematic review (SR) of the ethical implications of the development and application of TE for regenerative purposes, as mentioned in the academic literature. While TE might have applications outside RM (e.g., as a drug screening platform or for in vitro meat production), we have focused on the clinical application of TE for tissue regeneration. To our knowledge, this is the first time that the ethical implications of this field have been reviewed in a systematic manner. An SR has the advantage of being comprehensive, methodologically rigorous and transparent, and of reducing (selection) bias. 9
Method
We performed an SR to identify the ethical implications of the development and application of TE for regenerative purposes. The reviewing process was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for SRs 10 and the adapted PRISMA ethics guidelines by Kahrass et al. 9 Ethical implications were broadly understood to be expressions of an ethical or societal issue or consideration, which may but need not take the form of a fully argument-based reason. 11
Search strategy
Four databases (Embase, PubMed, Scopus, and PhilPapers) were searched for relevant English language articles using a search string combining search terms for (a) TE, RM, or bioprinting with (b) ethics or human rights, covering the period January 01, 2008 until October 05, 2021. An experienced librarian was consulted regarding the design of the search strings and choice of databases to ensure that relevant keywords were included, and that a comprehensive area of biomedical, bioengineering, and ethics literature was covered. The original search was performed on March 30, 2020 and updated on October 05, 2021. See Supplementary Data S1 for the complete search strategy of all databases.
Inclusion and exclusion criteria
Articles were screened for eligibility by A.J.K based on title, abstract, and keywords, and next, if applicable, based on the full text, using Rayyan software (Fig. 1). Inconclusive screening decisions were resolved through consultation with K.R.J. and A.L.B.
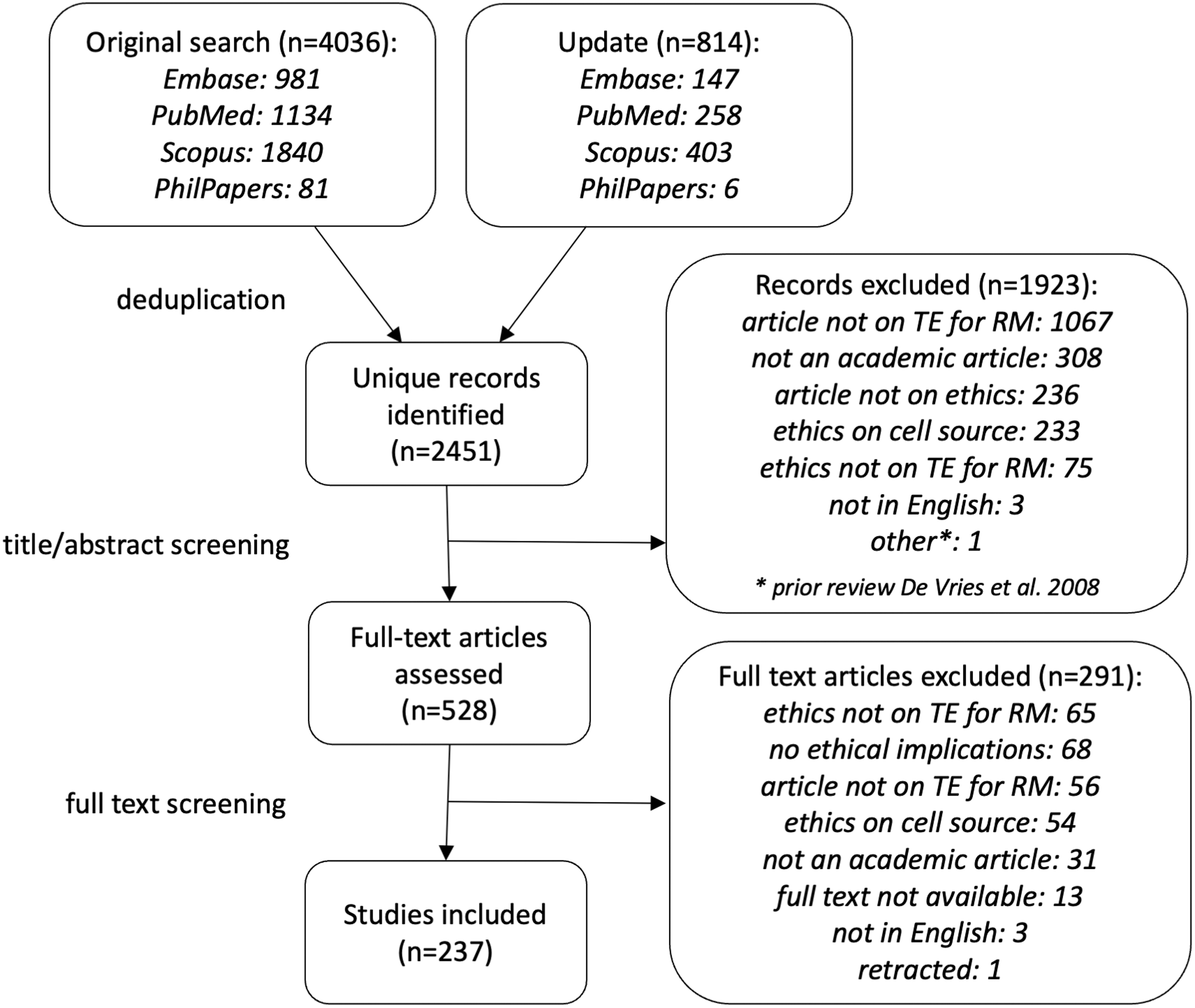
Overview of the number of articles identified and included throughout the several phases of the screening process, depicted according to the PRISMA guidelines.
Articles (e.g., original research, editorials, opinion articles, letters) published in English in a peer-reviewed academic journal in the period January 01, 2008 to October 05, 2021, which discussed an ethical implication of the development or application of TE for regenerative purposes, were eligible for inclusion. Also included were articles that discussed as their main topic ethical or societal implications of RM more generally, provided that these implications were applicable to TE as well.
Excluded were book chapters, conference articles, and news articles, in addition to articles on RM that discussed SC transplantation/research or GT instead of TE. Also excluded were ethical implications related to the source of (stem) cells and the use of embryonic tissues, which have been discussed in depth elsewhere.12–14 Supplementary Data S1 for a more detailed description of the inclusion and exclusion criteria.
As a minimal standard for quality assessment, articles had to be published in a peer-reviewed journal.
Data extraction and analysis
Full texts of the included articles were analyzed systematically using a data extraction sheet (Supplementary Data S1) (A.J.K.). For each article, information was extracted about publication type, publication year, and about the topic and aim, substantiated by relevant quotes. Moreover, one-sentence descriptions of the relevant ethical implications mentioned in each article were extracted (referred to as an implication mention), substantiated by one or more quotes. Quote lengths diverged from one sentence up to page-length. Additional quotes were extracted to preserve contextual information relevant to the ethical implications where necessary.
A thematic analysis was conducted to synthesize the data, using data synthesis sheets (Supplementary Data S1). 15 Implication mentions were compared across articles, and combined into implication types based on close similarity (compare “reason mention” and “reason type” as described by Strech and Sofaer) 16 (A.J.K.). Implication types were then thematized using a coding tree of four hierarchical levels (i.e., with primary, secondary, and tertiary sub themes under each main theme) (A.J.K.). Each implication type was assigned to one sub theme. If more than one sub theme was applicable, the most suitable one was selected. The formulation of the implication types and the coding tree was then evaluated by all authors and optimized through an iterative process of discussion and adaptation until agreement on a meaningful thematization was reached between all authors (A.J.K., K.R.J., M.C.V., and A.L.B.).
Results
The database search resulted in the identification of 2451 unique articles. Of these, 237 articles were considered eligible and were included in this review (Fig. 1).
Characteristics of included articles
The included articles encompassed reviews in natural sciences (n = 61/237) and humanities (n = 19/237), original research in natural sciences (n = 13/237) and humanities (n = 56/237, of which were 8 empirical and 48 theoretical studies), perspectives (n = 68/237), and other publication types (n = 20/237, of which were 10 workgroup and project reports, 5 interviews, 3 methods articles, 1 case report, and 1 book review) (see Supplementary Data S2). The articles discussed TE for regenerative purposes as a main topic (n = 115/237, including 21 on bioprinted implants and 4 on organoid transplantation) or subtopic (n = 65/237, including as a main topic: 9 on Advanced Therapy Medicinal Products (ATMP), 8 on SC transplantation, 6 on RM, and 6 on in vitro TE), or discussed RM, broadly yet in a manner relevant to TE, as their main topic (n = 51/237) or subtopic (n = 6/237) (Supplementary Data S2). The articles were published in 2008 (n = 10), 2009 (n = 17), 2010 (n = 13), 2011(n = 9), 2012 (n = 10), 2013 (n = 18), 2014 (n = 22), 2015 (n = 33), 2016 (n = 16), 2017 (n = 23), 2018 (n = 14), 2019 (n = 24), 2020 (n = 21), and 2021 (n = 18).
Synthesis of the ethical implications of TE for regenerative purposes
A range of ethical implications of TE for regenerative purposes was identified in the included literature. These were categorized into 10 main themes. The first seven themes cluster phases of TE development from bench to bedside:
animal welfare handling human tissue informed consent therapeutic potential risk and safety clinical translation societal impact.
The remaining three themes group overarching ethical domains for safeguarding responsible development:
scientific integrity
regulation
patient and public involvement.
The ethical implications (as “implication types”) identified in the literature are discussed below per main theme and sub theme, and are also summarized in Tables 1–10. Supplementary Data S3 for one-sentence descriptions of the “implication mentions” per article corresponding to each aggregated “implication type”.
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Animal Welfare
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Animal welfare
The literature discusses ethical implications related to animal welfare that can be subdivided between conditions for preclinical testing on animals and concerns related to animal-derived TE components (Table 1 and Supplementary Data S3).
Animal experimentation
Animal welfare should be balanced with the scientific need and expected human benefit of animal models.17,18 The design should therefore take the predictive value of a particular animal model for humans into account19–24 and the experiments should be performed in accordance with the 3R principles (replace, reduce, and refine).25–29
To replace animals for biomaterial testing or to study regenerative processes, the use of computational modeling 30 or in vitro models31–33 are advocated. These include SCs, 34 organoids, 35 decellularized tissue, 36 and organs-on-a-chip.37,38 Ex vivo tissue culture systems39–44 and the chorioallantoic membrane assay45–48 are proposed as means of partial replacement. To reduce the number of animals per experiment, continuous monitoring of each animal,49–51 testing of multiple biomaterials per animal, 52 and skipping small animal models in favor of one large animal model 53 are suggested. Last, a preference is expressed not to use companion animals,18,27,50,54,55 such as dogs, or primates 56 as large animal models, and, if primates have to be used, to refine the experiments by minimizing the suffering caused.57,58
Animal-derived TE components
Animal-derived decellularized scaffolds and culture medium components might raise religious and moral objections related to xenotransplantation and animal welfare, especially among religious or vegan patients.22,59–62 Autologous, 63 allogeneic,64–66 or plant-based components67–69 are proposed as alternatives.
Handling human tissue
The literature identifies ethical considerations regarding how to handle human tissues. These can be grouped in two sub themes: first, the collection, storage, and use of cells and tissues from human donors for research and clinical applications, and second, ownership over and commodification of donated and engineered tissues (Table 2 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Handling Human Tissue
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Tissue collection, storage, and use
Tissues should be collected through non-invasive methods that do not cause harm or pain to the donor.17,22 Consent of the donor or their family is required for collection and storage of tissues,22,23,58,70–72 which should be explicit19,73 and voluntary.74,75 Some suggest that consent should be specific to the intended use 17 while others argue that broad consent suffices.20,76 As a prerequisite for consent, donors should be provided relevant information, including information on present and foreseeable use of the collected tissues,7,77–81 on the need to forego financial rights over the tissue, 82 and on policies outlining the return of findings83,84 and the possibility to withdraw.85,86
To safeguard the confidentiality and privacy of the donor of collected and stored samples,19,22,58,80,82,86,87 some suggest that sample anonymization is desirable,7,17,77 while others point out that this would limit clinical value 73 and the possibility of return of findings 23 and of dynamic (ongoing) consent. 88 Particularly, safeguarding privacy and confidentiality of the (medical) data and digital models required to create personalized 3D bioprinted tissues is warranted.75,78,89,90
A dispute exists whether private (commercial) biobanking is acceptable 91 or that public banking should be preferred 74 for tissue storage. Specifically, private banking is said to be based on a (false) promise of yet-to-be-developed RM treatments,88,92 which could erode public trust.85,93 Yet public banks would fail in providing a supply that is inclusive to all. 84 A hybrid model of public-private biobanking is therefore advocated that allows privately stored tissues to become publicly available, increasing the publicly available supplies94–96 and accelerating research. 73
Finally, moral and religious objections could be raised to the use of (decellularized) human tissues, especially cadaveric tissues, as scaffold components. Synthetic scaffolds, 97 animal-derived scaffolds,72,98 or (decellularized) tissues from living donors99,100 would therefore be preferred.
Ownership and commodification
The question is raised which parties may claim ownership over donated7,22,23 and engineered17,75,79,88,90,101–104 tissues: the tissue donor, the patient receiving the implant, or the parties involved in producing the organ? Moreover, it is debated whether and under what conditions intellectual property rights over TE, for example, by patenting the technology or the product, is desirable. While patenting could foster innovation, 105 ensure quality control, and prevent misuse,17,101 it might also hinder open science and research82,106 and equal access to the fruits of the technology.74,107,108 According to some, notwithstanding property interests of other parties, patients should retain a right to control certain uses and access to the organ.73,109 Finally, TE creates a paradoxical and potentially undesirable situation in which altruistically donated tissues are turned into profitable materials for which donors have no right to financial gain.71–74,80,84,93,110 This appears at odds with human dignity 111 and the ban on the commodification of human body parts, 112 and might lead to donor exploitation or coercion.88,113–115
Informed consent
The literature mentions informed consent as a necessary precondition for several phases of TE development: for tissue donation (as discussed under ‘Tissue collection, storage, and use’), participation in clinical trials, and clinical application, whether as innovative therapy, compassionate use, or routine treatment. A distinction can be made between challenges and conditions for obtaining valid, informed consent (Table 3 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Informed Consent
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Challenges to consent
Consent might be challenged if the patient's capacity to consent is impaired, for example, in emergency situations 116 or when treated for brain conditions. 75 Second, patients' hope or desperation for a cure might lead them to misunderstand the risky and experimental nature of a TE trial (therapeutic misconception) and affect the validity of their consent.19,23,37,74,115,117–119 Third, patients might feel pressured to participate by family members or researchers.22,120 Fourth, possible positive bias and conflicting financial or professional interest of the physician-researcher involved in an experimental TE treatment might affect the neutrality of the information provided.119,121–123 And last, uncertainty about possible complications due to the novel, 81 complex,22,119,124 invasive,20,118 and irreversible 125 nature of TE makes it difficult to provide accurate information about the risk-benefit ratio of the intervention.23,71,88,104,112
Conditions for consent
Due to these challenges, medical professionals should provide, and carefully explain, all relevant information, including information on the characteristics of the TE intervention itself, the source of cells and other components, foreseeable risks and benefits, safety measures, alternative treatment options, and a disclosure of interests.37,119,126–128 Yet, even if high quality information is provided, consent should always be accompanied by oversight mechanisms to prevent patients from unacceptable risks. 74 Moreover, information should be provided in understandable language,17,129 and may be supported by the use of educational materials 119 and consultation of an independent medical professional.130,131
Therapeutic potential
One of the main ethical opportunities and key drivers of TE is its therapeutic potential. The literature elaborately discusses its therapeutic promise, and briefly its objectives related to broader wellbeing (Table 4 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Therapeutic Potential
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Therapeutic promise
TE is proposed to address an unmet clinical need, having the potential to treat diseases for which currently no curative therapy is available75,106,116,132–134 or for which current therapies have risky side effects, 135 and to improve the quality of life of elderly patients. 82 Most often, TE is presented as an alternative to donor organ transplantation. As such it would remove the need for immunosuppression,133,136 improve length and quality of life60,74,79,88,103,128,137 and psychosocial outcomes (for face TE),138,139 provide patient-tailored75,81,89,125,140 and/or off-the-shelf available78,141,142 treatment options, and reduce the cost of care.23,143 In the long run it might solve organ shortages,102,117,144–148 thus saving lives and avoiding complexities related to organ donation, including the surgical risks for donors,149,150 the need for (prior or proxy) consent,112,151,152 and the acceptability of directed donation.17,101 Moreover, compared to other RM strategies such as SC injections or GT, cell scaffolding provides TE the benefit of excision if needed, thus allowing partial reversibility.81,125
Objectives for wellbeing
Finally, TE should not merely be aimed at treating disease, but also at improving overall quality of life 20 and promoting human flourishing. 153
Risk and safety
Notwithstanding the therapeutic potential of TE, the literature discusses several risks of harm for the patients or trial participants, along with safety measures (Table 5 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Risk and Safety
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Risks to participant
Risks are associated with different aspects of TE constructs: The presence of living cells poses risks of immunogenicity,70,88,154 mutagenesis (especially for bioprinting),89,101,124,155 undirected differentiation,81,125,156 and teratoma and tumor formation,22,23,75,79,86,141 which are compounded by bioprinting forces. TE components such as scaffolds and bioactive factors pose risks of contamination and introduction of pathogens,22,23,79,124,155,157 potential immunogenicity,72,158 and cytotoxicity (especially of degradation products).81,116,125,156
The complexity of the TE constructs themselves pose risks that are foreseeable but hard to quantify, that is, risks associated with inherent variability of the constructs,78,79,119 unpredictable dynamic interactions with the bodily environment,25,80,124,132 and the irreversibility of the surgical implantation and regeneration process.3,102,115,122 The latter particularly impedes patient's opportunity to withdraw from the trial.70,75,81 In addition to the known risks, TE involves inherent uncertainties, because of the complexity and manufacturing variability of the construct,23,71,159 and because the predictive value of preclinical animal studies is limited.3,153 As a result it is challenging to provide a realistic risk-benefit analysis,88,104,160 and to decide when evidence suffices to start first-in-human trials.80,161
Safety measures
Trials might be justified if adequate safety measures are taken to minimize risks131,132,162 and if the benefits to the individual (compared to available therapies)3,126,161 and society (in terms of scientific value)23,37,73,158 are maximized. The following safety measures are described: To understand the biological mechanisms underlying TE126,154 and to evaluate safety and efficacy of the technology, thorough in vitro 163 and in vivo26,129,131,161 preclinical research is required and must be optimized to minimize translational distance.29,37,120,124 To monitor adverse events and the success of regeneration, there is a need for long-term follow-up of trial participants,37,58,154,164–166 and pediatric patients in particular.120,124 Artificial organs should be identifiable in case emergency situations arise. 102 However, safety measures should not be unnecessarily burdensome131,132 or hamper patient access.165,167
Clinical translation
The pathway of TE treatments from experimental design toward clinical application can roughly take three routes: through clinical trials (i.e., traditional pathway, aimed at evidence collection for research), as innovative therapy (i.e., non-research pathways aimed at improving patient access, including off-label use, compassionate use, and hospital exemption), or interventions can simply reach the market before evidence of efficacy and safety is complete (i.e., unproven commercial interventions). The literature discusses ethical implications related to each of these pathways (Table 6 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Clinical Translation
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Clinical trial design
Several suggestions are provided on how to optimize the design of clinical trials for TE. First, while randomized controlled trials (RCT's) should be used where possible,168,169 this might be ethically challenging, especially if no standard of care is available.167,170–172 Sham surgery may be required as a comparator, but is justifiable only if surgery is minimally invasive and if anticipated social value outweighs harm to the control group.131,161,173 Moreover, adequate and fair participant selection is challenging,19,73 because fewer patients can be enrolled than are eligible, 74 little is known about comorbidities, 174 and end-stage patients are especially vulnerable to therapeutic misconception.23,37
Still, consensus seems to exist that for high-risk TE, performing first-in-human phase I trials on healthy volunteers or stable patients is unacceptable. 3 Patients with mid- or end-stage disease should be included instead,70,81,124,161,168,175 because they might benefit most and risk losing least. Regarding outcome measures, first-in-human trials may involve efficacy endpoints in addition to safety endpoints,73,81 to maximize the scientific value. Efficacy endpoints should include objective outcomes, such as overall survival 167 and success of tissue regeneration,126,164 evaluation of which should preferably not rely on invasive biopsies,21,176 and subjective outcomes related to patient wellbeing.37,131,160,168 For personalized (3D bioprinted) implants, heterogeneous outcomes are to be expected and limit the possibility to extrapolate the results.70,75,125,155,156
Finally, curated registries should be set up and used to collect long-term data from clinical trials and follow-up studies regarding safety and efficacy of TE interventions.3,17,37,74,86,101,120,126,167 This would allow to trace adverse events, 177 help build a solid evidence-base, and allow to evaluate when the intervention needs no longer be considered experimental.117,131 Moreover, in combination with general non-biased and transparent publishing practices and the use of scientifically valid and rigorous research methods,71,74,125,127,163,164,178,179 this could promote reproducibility and foster translation of TE products to the clinic.
Innovative therapy pathway
Because the use of traditional clinical trials for TE is faced by several challenges, alternatives are needed. One option is to allow patients to access experimental RM and TE treatments as innovative therapy outside clinical trials. This would improve patients access74,122,180 and circumvent the need for sham surgery.23,37 However, such access might be at tension with the need to protect patients from considerable risk,7,121,178 and the need to generate conclusive evidence regarding safety and efficacy of the product123,181,182 and could therefore harm public trust. Publishing outcomes and adverse events in open registries might partially overcome these concerns.19,131,166,183
Unproven commercial interventions
Commercial clinics also offer SC-based RM interventions (which could include TE interventions) without sufficient proof of efficacy and safety. Authors warn that such unproven interventions operate outside existing regulations and are based on false promises,123,180,184,185 thus exposing vulnerable patients to the risk of physical harm and financial exploitation,19,74,156,178,186 and harming public trust in the RM field.120,122 Both researchers and physicians are responsible to inform patients and protect them against these risks.7,18,120,183,187,188
Societal impact
The literature discusses several societal impacts of TE and RM development. These include so-called “soft” impacts related to human values, experiences, identity, relations, and perceptions. The literature covers challenges to distributive justice, the impact on health care and market, biosecurity, longevity and enhancement, and human experience and identity (Table 7 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Societal Impact
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Distributive justice
Concern exists that the expected high costs of TE interventions will limit its accessibility. If only those who can afford to pay will benefit in terms of quality and length of life,17,75,140,189–191 this may exacerbate socioeconomic health disparities19,22,23,70,107,158,192,193 both locally 102 and globally.79,88,104,180,194,195 However, if, in the long run, the costs of TE will decrease, treatments could become cost effective.89,112,166,196 Efforts should be made toward that end so as to improve accessibility.25,37,74,90,124,155,170,197,198 Additionally, to ensure equal access to TE treatments, reimbursement will be necessary154,182 and reimbursement decisions should be based on evidence of clinical benefit, 123 patient preferences, 167 and initial and lifetime costs102,167,178 compared to current treatments options.
Moreover, while there is an imperative to fund RM and TE research,199,200 this might draw resources away from existing health care solutions, exposing a value trade-off between the future fruits of innovation and the demand of patients in need today.71,74,88,104 Resources should therefore be allocated based on good science rather than hype137,146,169 and arrangements for benefit-sharing (such as price limits or revenue sharing) should be put in place.158,201,202
Impact on health care and markets
TE might provide an alternative to several ethically contentious health care practices: TE organs might circumvent personal identity issues related to organ donation, 203 and naturalness objections against xenotransplantation. 140 Transplantable bioartificial uteri might provide an alternative to gestational surrogate motherhood149,204,205 and could possibly contribute to the realization of human ectogenesis on the long run. 133 TE organs could also help ending illegal organ trade,79,141,206 provided that regulations are adopted to prevent new black markets in TE organs.90,140
Biosecurity
Concern is raised that TE technology, and particularly 3D bioprinting because of its relative accessibility and affordability, might lead to unregulated home-use, which could facilitate bioterrorism.17,81,107,125,190,207
Longevity and enhancement
TE might help to prolong the average human life span or even “cure aging”, which raises the question whether longevity is socially desirable193,208,209 or that ageing should be viewed as a normal part of life.88,103,192 In particular, an expanded life span might impact the retirement system 210 and could decrease solidarity with older generations.110,211 If TE tissues and organs would become widely available, this could also affect social norms: unhealthy and reckless lifestyles might be normalizsed17,88,101,108,192 and support for organ donation might decrease. 212 Moreover, TE could be used for cosmetic purposes or potentially to enhance physical or cognitive abilities. While the pursuit of enhancement is deeply embedded in human culture23,75,112 and could help to pursue the good life, 88 it raises concerns about potential harm, 108 increasing inequalities,70,158 fair resource distribution,20,110 and the legitimate goals of medicine.180,190,213,214
Human experience and identity
TE presents the body as malleable110,189,215 or machine-like. 104 This raises the question whether the human body or life itself is something that should be engineered,17,115,180,216 and might challenge our understanding of what it means to be human,210,217,218 and our perception of the self219,220 and of others. 101 The engineering ideal driving TE might obscure the existential and embodied experiences of patients.88,221–223 Last, TE organs blur the boundaries between the corporal and the technical,88,224 life and non-life,140,225 and natural and engineered. While its social acceptance might depend on whether it will be perceived as the one or the other, 17 TE is best presented as engineered. 223
Scientific integrity
The literature identifies issues of scientific integrity related to individual conduct and to transdisciplinary collaboration (Table 8 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Scientific Integrity
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Responsible conduct
TE, SC and organoid research, and organ bioprinting are surrounded by hype and high public expectations.88,180,194 To prevent disillusionment and promote public trust, researchers, scientific journals, and popular media should set realistic expectations,140,146,169,178,191,226 by making modest claims and refraining from overstated promises.23,85,112,137,156 The TE field has been faced with a serious case of scientific misconduct and research fraud by the thoracic surgeon Paolo Macchiarini. This indicates the need for a more benign research environment and better oversight mechanisms within TE, to promote integrity of conduct and to restore trust in the field.129,169,227,228 Responsible conduct requires that scientists express scientific virtues such as openness, honesty, and accountability74,107 and that they anticipate how their choices affect the final TE product and its beneficiaries.3,37,102 They should therefore be trained to become aware of and be able to engage in discourse on the societal and ethical implications of TE.25,168,192,229–231
Transdisciplinary collaboration
Successful TE development co-depends on transdisciplinary collaboration between engineers, scientists, and clinicians102,137,232 in a globally inclusive manner182,233; adequate training of physician-researchers to gain necessary expertise knowledge and skills to implement TE safely in clinical trials37,121,131,234,235; infrastructures of knowledge exchange 106 ; and mutual collaboration and relationships of trust between academia, industry, and regulators.92,125,177,236
However, industry involvement and commercial pressure might steer the research agenda, 106 incentivize selection of trial sites in areas with permissive regulations,177,180,237 and lead to premature clinical testing and commercialization of TE products.20,115,118,174,178,194 To deal with potential conflicts of interest and to maintain trust from physicians and the public, independent oversight mechanisms are needed,80,89,168 full disclosure of interests should be provided by those involved in clinical trials67,131 and trials sponsored by governments rather than industry might be preferred.17,162
Finally, ethics, humanities, and social science scholars should be involved in the innovation process to take ethical, legal, and societal considerations into account, to manage potential conflicts of interest and to promote successful and responsible translation of TE to the clinic.17,38,106,140,181,238,239
Regulation
The need for adequate regulatory frameworks for TE is widely discussed in the literature. The relevant ethical implications can be divided in the need for appropriate guidelines and oversight, and liability issues (Table 9 and Supplementary Data S3).
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Regulation
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Need for guidelines and oversight
The regulatory frameworks guiding TE development should be revaluated.103,120,181 Particularly, given regional disparities in TE regulations, many authors call for international harmonization70,154,157,178,240 or, at minimum, agreement on an international code of conduct.180,234 In particular, guidelines and adequate oversight should be established for the collection, storage, and use of human tissues84,93,128,136,162,241 and for ownership rights over such tissues.85,165 Moreover, there is a need to set uniform safety, quality, and efficacy standards for cell culture,120,126 biomaterials,23,25,242,243 manufacturing processes,75,108,124 and TE end products106,171,195,244,245 and to establish ethical oversight127,132 or regulatory controls174,179 to guide risk assessment. Finally, global regulations to guide the application of experimental TE and RM treatments as innovative therapy outside clinical trials are needed.19,122,153
A particular challenge for TE regulation is fostering a balance between the product safety and speed of product development.3,20 While regulations should prevent commercial pressure from impeding good patient care, 102 regulations should not be unnecessarily risk-averse, so as not to slow down progress,135,144,246 drive up costs, or impede patient access.74,111 This relates to the question when clinical evidence suffices to introduce TE products into regular clinical practice.22,80 Another regulatory challenge for TE and 3D-bioprinted products is that–because they combine features of drugs, devices, and biologicals and because of their personalized nature–they fall in between most currently existing classificatory categories.78,217 These classifications should therefore be urgently revised to provide clarity.17,79,81,89,101,126,224
Liability
Considering the multidisciplinary nature of TE development, it should be determined who (regulator, engineer, manufacturer, or clinician) is responsible for quality assurance 112 and long-time maintenance 102 of TE implants and who should be held liable in case serious complications arise.20,75,83,101 This is especially challenging in case a patient decides to withdraw from a trial. 126 While one author warns that currently patient protection lags behind because companies can avoid liability for TE product failure too easily, 217 another argues that physicians should be better protected from negligence claims if innovative therapies turn out harmful, in order not to stifle innovation. 121
Patient and public involvement
The literature shows a general interest in involving patients and the public in a dialog on TE development, to promote public trust and to enable scientific citizenship (Table 10 and Supplementary Data S3). As a start, the public should be educated on the opportunities and hurdles, along with ethical and societal aspects of TE. This requires scientists to communicate clearly about their research, and provide accessible and reliable information sources,23,82,88,108,131,147,158,180,181,245,247 or even set up counseling services to counter misinformation. 185
Overview of the Ethical Implications of Tissue Engineering for Regenerative Purposes, as Identified in Relation to Patient and Public Involvement
The ethical implications are presented as aggregated “implication types”, categorized into sub themes. See Supplementary Data S3 for one-sentence descriptions of every ethical implication as mentioned in each relevant article (i.e., “implication mentions”).
Next, a wide range of stakeholders, including lay people and patients, should be consulted on their values, needs, expectations, and concerns regarding TE development.74,82,88,195,233,248 In particular, such consultation could probe the acceptability of risks,20,237 approaches to deal with uncertainties, 71 and fair distribution of health care resources.175,230 Patient advocates are especially well-positioned to articulate patient needs.181,249 One author group points out that to foster truly inclusive deliberation, scientists should be attentive to a wide variety of stakeholders and empower them to make informed and meaningful contributions. 107
Finally, (bio)artists are particularly well positioned to explain or visualize scientific work, 38 to critically question the TE field, its implications and assumptions215,250 and to provide alternative places for discourse on the intersection of art and bioethics.222,251
Discussion
In this SR of 237 academic peer-reviewed articles, we identified a range of ethical implications of TE for regenerative purposes. These stretch over 10 overarching themes, representing phases of TE development and implementation (animal welfare, handling human tissue, informed consent, therapeutic potential, risk and safety, clinical translation, and societal impact), and safeguards necessary for responsible development (scientific integrity, regulation, and public and patient involvement).
A prior literature review performed in 2008, De Vries et al noted that until then the literature on the ethics of TE and RM mainly focused on considerations relating to the source and collection of SCs in preclinical research and that the ethical evaluation of TE clinical trials lacked short. 5 Our SR reveals that, since then, ethical reflection in academic discourse has followed the pace of TE development, shifting focus from preclinical to clinical translation, and providing several concrete recommendations on how this process should take place.
Our SR indicates several characteristics of TE that are specific to the ethical implications of the product. That is, (a) TE is highly innovative 23 and has a novel method and aim,3,161 that is, tissue regeneration rather than “mere” restoration of function; (b) TE products are increasingly personalised, 23 for example, bioprinted from patient-derived cells after a patient-specific shape; (c) TE products deeply invade 161 in and dynamically interact3,126 with the human body and finally disappear 88 ; and (d) TE is multidisciplinary38,119,140 in nature and relies on state-of-the-art technologies such as 3D (bio)printing, iPSCs, and organoid technology. Several ethical implications related to these characteristics are touched upon in this review but remain in need of further ethical reflection.
First, the novel aim and personalized character of TE challenge traditional clinical trial pathways and guidance is needed on how to proceed with clinical translation of TE. The possibility to perform traditional RCTs or to extrapolate the results from one patient to another is limited because suitable comparators are lacking, and TE interventions are patient specific. N-of-1 trials have been proposed as an alternative,252,253 but guidelines are needed on how to reduce risks for individuals while maximizing the scientific value of the outcomes. Alternatively, patients could be provided access to unproven interventions as innovative therapies outside clinical trials when individual benefit can be reasonably expected.23,74 This may be beneficial provided that a learning component, including registration and reporting, is maintained.122,254
Second, TE constructs invade and integrate deeply into the human body, which may have implications for how the technology is perceived and experienced. For example, TE may impact embodiment and perceptions of (un)naturalness. While these so-called “soft” impacts on society are often indirect and intangible, their effect may be significant.4,255 Therefore, we agree with others88,192,256 that more efforts should be made to anticipate and evaluate the soft mid- and long-term impacts of TE and RM on society at large.
Third, the multidisciplinary nature and commercial incentives in the TE field raise questions to whom ownership rights and responsibilities regarding TE products should be ascribed. While this could be approached as a strictly legal question on the patentability of tissue products and processes,105,109 a broader ethical analysis is needed that takes into account how individuals perceive ownership over TE implants. It has been shown that intuitions about ownership over tissues derived from one's own body tend to deviate from ownership rights as laid down in existing regulations. 257 Both the value attached to cells and tissue contructs,84,258,259 and the invasive character of TE implants that integrate deeply into the body could and should affect what degree of ownership and control is attributed and to whom. This will have implications for who will gain financially, and how much, and could therefore also impact the accessibility of TE interventions. Broadening the notion of ownership could help justify benefit sharing schemes73,201,202 to improve access and to let patients and the public share in the benefits of TE and RM innovations.
Methodological choices, strengths, and limitations
Because the focus of the TE debate has moved toward settling conditions for responsible development and anticipating future implications, we opted for an SR of “ethical implications” rather than “reasons for/against” as is sometimes done for ethics literature.11,16 We broadly interpreted “ethical implications” to include considerations of a philosophical, existential, artistic, legal, regulatory, and anticipatory nature and questions of governance, public and patient involvement, and (scientific) misconduct.
We restricted the scope of our SR to “TE for regenerative purposes” because of its clinical significance and because we expected that at least some of the ethical implications would be related to the particular characteristics and purpose of TE thus defined. However, demarcating “TE for regenerative purposes” as distinct from other forms of RM was challenging because in the literature different definitions of “TE” and “RM” are used, 260 but often not explicated. Moreover, in practice, TE, SC interventions, and GT often overlap or are combined. A detailed list of inclusion and exclusion criteria that was used to guide this demarcation is provided in Supplementary Data S1.
Overall, we present a comprehensive review that covers over two hundred articles, indicating a broad set of ethical considerations related to all phases of TE development, including potential future societal impact. Three possible limitations should be mentioned. First, by excluding book chapters and conference articles we may have missed a small number of ethical implications. Second, both phases of literature screening (title/abstract screening and full text screening) were performed by one reviewer only, a method for which support exists 261 but that might be more fallible than traditional double-blind screening. However, as has been argued elsewhere, 262 we may have captured all (or most) relevant ethical implications, even if a small number of articles was missed. Third, while we touch upon several regulatory considerations, we did not discuss existing regulatory regimes in detail, as this is outside the scope of our review. However, others have recently compared ATMP and bioprinting regulations across jurisdictions.79,263
Concluding Remarks
Overall, as TE and RM shift toward clinical translation, it raises ethical questions regarding fundamental and clinical research, translation to the patient, and its impact on society at large. To our knowledge, this is the first time that the ethical implications of this field have been reviewed systematically. By gathering existing scholarly work and identifying knowledge gaps, this SR facilitates further research into the ethical and societal implications of TE and RM. Moreover, it will serve as a valuable resource and educational tool for TE and RM scientists, engineers, and clinicians by providing an overview of the ethical considerations relevant to their work. As such, our review will help promote successful and responsible development of new interventions in TE and RM in a manner attentive of the current and future ethical and societal implications encountered along the way. Given its broad and comprehensive scope, it may also serve as a steppingstone for the ethical analysis of other emerging biomedical technologies.
Footnotes
Acknowledgments
We thank Paulien Wiersma of the Utrecht University Library for revising the database search strategy. We would like to thank our colleagues from the Medical Humanities Department for their valuable comments on earlier versions of this work.
Authors' Contributions
All authors contributed to conceptualization and writing of this article.
Disclosure Statement
Annelien Bredenoord is a member of the Dutch Senate. She also serves as a member of the Ethics Committee of the ISSCR and the Ethics Advisory Board of IQVIA. The other authors have no conflicts of interest to declare.
Funding Information
This research was financially supported by the Gravitation Program “Materials Driven Regeneration,” funded by the Netherlands Organization for Scientific Research (024.003.013).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
