Abstract
Neuromuscular junctions (NMJs) are specialized synapses responsible for signal transduction between motor neurons (MNs) and skeletal muscle tissue. Malfunction at this site can result from developmental disorders, toxic environmental exposures, and neurodegenerative diseases leading to severe neurological dysfunction. Exploring these conditions in human or animal subjects is restricted by ethical concerns and confounding environmental factors. Therefore, in vitro NMJ models provide exciting opportunities for advancements in tissue engineering. In the last two decades, multiple NMJ prototypes and platforms have been reported, and each model system design is strongly tied to a specific application: exploring developmental physiology, disease modeling, or high-throughput screening. Directing the differentiation of stem cells into mature MNs and/or skeletal muscle for NMJ modeling has provided critical cues to recapitulate early-stage development. Patient-derived inducible pluripotent stem cells provide a personalized approach to investigating NMJ disease, especially when disease etiology cannot be resolved down to a specific gene mutation. Having reproducible NMJ culture replicates is useful for high-throughput screening to evaluate drug toxicity and determine the impact of environmental threat exposures. Cutting-edge bioengineering techniques have propelled this field forward with innovative microfabrication and design approaches allowing both two-dimensional and three-dimensional NMJ culture models. Many of these NMJ systems require further validation for broader application by regulatory agencies, pharmaceutical companies, and the general research community. In this summary, we present a comprehensive review on the current state-of-art research in NMJ models and discuss their ability to provide valuable insight into cell and tissue interactions.
Impact statement
In vitro neuromuscular junction (NMJ) models reveal the specialized mechanisms of communication between neurons and muscle tissue. This site can be disrupted by developmental disorders, toxic environmental exposures, or neurodegenerative diseases, which often lead to fatal outcomes and is therefore of critical importance to the medical community. Many bioengineering approaches for in vitro NMJ modeling have been designed to mimic development and disease; other approaches include in vitro NMJ models for high-throughput toxicology screening, providing a platform to limit or replace animal testing. This review describes various NMJ applications and the bioengineering advancements allowing for human NMJ characteristics to be more accurately recapitulated. While the extensive range of NMJ device structures has hindered standardization attempts, there is still a need to harmonize these devices for broader application and to continue advancing the field of NMJ modeling.
Introduction
Malfunction of the specialized components of the neuromuscular junction (NMJ) can result from developmental disorders, toxic environmental exposures, and neurodegenerative diseases. Population trends indicate an increase in neuromuscular disease diagnosis since the early 2000s 1 and the majority of these diseases are chronic and disabling, resulting in a significant economic burden. 2
There is increasing evidence linking environmental exposures to diseases of the NMJ. 3 While the effects and mechanisms of botulinum toxin (BT) and organophosphate (OP) exposure on the NMJ have been established for decades,4–8 compounds like lithium, corticosteroids, antibiotics, and beta blockers can potentiate effects of local anaesthesia, 9 induce phenotypic characteristics of NMJ disease, 10 and mechanisms are not always well understood.9,11 Increased incidence of NMJ disease from known and unknown causes along with technological advances in bioengineering and disease modeling continues to propel the investigation of NMJ development, function, and potential repair mechanisms.
Neuromuscular development and physiological function were initially investigated using animal NMJ models, including rodents, zebrafish, and drosophila. There is dissonance between animal and human NMJ anatomy and physiology 12 ; animal models are restricted in their ability to accurately capture human genetic and epigenetic variation 13 and pharmacokinetics differ between animals and humans. This limits the predictive ability of animal models to determine the clinical outcome of therapeutic success in humans. 14 In addition, anesthesia can introduce undesirable artifacts into animal NMJ studies. 15 Thus, there has been a significant push to reduce, refine, and replace animal NMJ models with alternative in vitro models.
Engineered microscale in vitro platforms, termed microphysiological systems (MPSs), have been developed to study the NMJ. These systems aim to recapitulate structural, electrical, biochemical, and mechanical cues needed to support NMJ formation, maturation, and function. When studied in isolation, in vitro NMJs can provide a deeper understanding of the system independent from the influence of other organ systems. There are a variety of MPS platforms spanning from monocultures, co-cultures of multiple cell types, spheroid cell formations, and maintenance of explants derived from tissues. These engineered systems have advanced our ability to identify tissue-specific mechanisms and molecular and cellular signaling, and provide a platform for testing chemicals.
In vitro models of NMJs have advanced in recent years to allow for precise control of important extrinsic factors like fluid shear force, concentration gradients, and mechanical stress. 16 Development of novel cell culture technologies, biocompatible materials, and microfluidics has resulted in the progression from inflexible two-dimensional (2D) cultures to multicellular three-dimensional (3D) NMJ models.
Previous reviews have discussed NMJ platforms in the contexts of engineering design evolution,17–20 cell types,21,22 and creation of disease models.23,24 This review assesses NMJ models as they relate to three major applications: (1) developmental studies; (2) understanding underlying mechanisms in disease; and (3) screening for drug discovery and toxicity testing. The engineering designs and experimental goals of NMJ culture systems need to be strongly tied to a specific scientific objective and will differ significantly depending upon the desired application. 13 Often an MPS can only be fit for a specific aim, and significant modifications and relevant data sets need to be generated to repurpose an MPS for other scientific applications.
Anatomy and Physiology of the NMJ
The simplest components of a NMJ model include the end bulb of a neuron, the synaptic cleft, and the motor end plate of a muscle fiber (Fig. 1). Motor neurons (MNs) end in roughly branched nerve terminals, which have specialized regions called active zones containing critical signaling molecules. Synaptic vesicles released from MNs contain acetylcholine (ACh), the active signaling molecule responsible for neurtransmission at the NMJ. Synaptosomal nerve-associated protein 25 (SNAP25) is required for ACh release and exposure to BT inhibits this neurotransmitter exocytosis by cleaving SNAP25.25–28 The synaptic cleft, the space between the nerve terminal and the muscle fiber, contains acetylcholine esterase (AChE). The motor end plate itself contains junctional folds, which increase the surface area where the ACh can interact with the muscle.
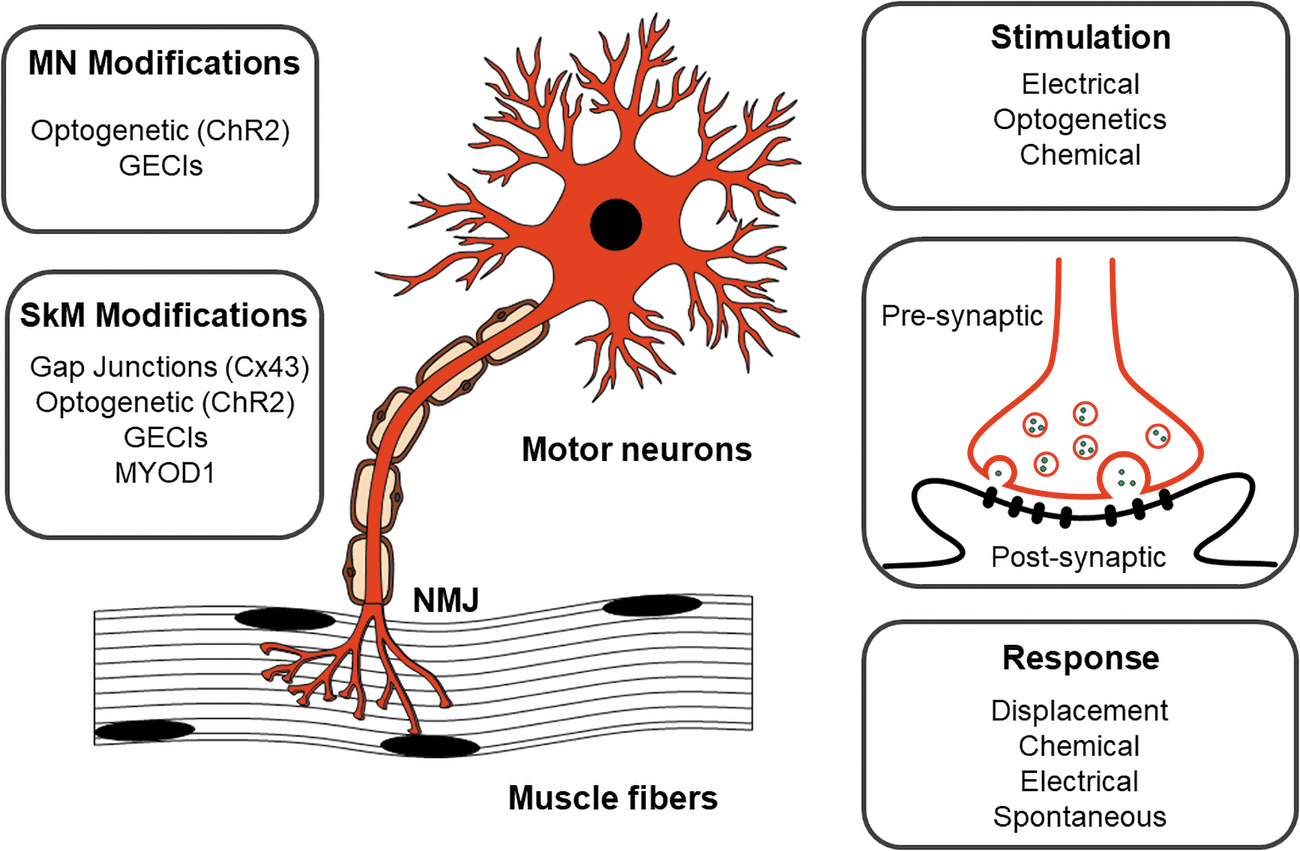
In vitro co-culture NMJ components. (Left) Genetic modifications to improve NMJ formation and signal transduction are inserted in NMJ models for both neurons and muscles. (Right) Multiple options for stimulation and muscle response are utilized in different NMJ models based upon the intended use of the system. ChR2, channelrhodpsin-2; Cx43, connexin-43; GECIs, genetically encoded calcium indicators; MN, motor neuron; MYOD1, myoblast determination protein 1; NMJ, neuromuscular junction; SkM, skeletal muscle.
Key cellular components of in vitro NMJ models—MNs and skeletal muscle—(Table 1) are typically sourced from primary biopsies, commercial cell lines, induced pluripotent stem cells (iPSCs), or embryonic stem cells (ESCs). MNs and skeletal muscles are terminally differentiated, unable to undergo further mitosis, and therefore defined as postmitotic. Mature primary MNs and skeletal muscles, or their myoblasts, can be harvested from human donors to fully capture genetic backgrounds and variations. Due to their limited proliferative capability and low collection number, primary cells can be challenging to culture after harvesting. Established cell lines have been derived from a variety of species and offer predictable and reproducible outcomes, as observed with the NSC-34 hybridoma cell line 29 and the N2A neural crest-derived cell line. 30
Summary of Species-Specific Neuromuscular Junction Models, Including Source of Motor Neuron and Muscle Cells
Optogenetic modification in NMJ models.
Patient-derived cells.
C2C12, immortalized mouse myoblasts; ESC, embryonic stem cell; iPSC, induced pluripotent stem cell; MN, motor neuron; NMJ, neuromuscular junction; NSC, neuronal stem cell; SkM, skeletal muscle.
There are limitations and variability as reported for the NSC-34 cells; this fusion of mouse embryonic spinal cord cells with a neuroblastoma creates a culture containing multiple cell types, which express morphological and physiological properties of MN-like cells, but they are not capable of all glutamatergic responses. 31 Also, proliferative neuronal cells have different uptake and sensitivity to SOD1-induced toxicity, a component of amyotrophic lateral sclerosis (ALS) degeneration, compared to terminally differentiated MNs. 32
iPSCs and ESCs can be obtained commercially or derived from a specific donor, and multiple protocols have been developed to directly reprogram stem cells.33–36 ESCs from transgenic mice lacking neural cell adhesion molecules can be differentiated into MNs; when cultured with myotubes, these NMJ models allow for definitive investigation of synaptic pathology in a targeted manner. 37
However, it remains challenging to optimize these protocols as they can be time consuming, unreliable, and produce mixed populations of cells. 38 Patient-derived iPSCs allow for a personalized approach,39–41 which is valuable when disease etiology is not fully known, yet remains labor-intensive.
As reflected in Table 1, NMJ systems have been created using a range of cell types and genomic editing strategies. Mouse and human NMJs have been designed along with mixed species culture systems.37,76,84 While there are species-specific NMJ morphologies and functions, cross-species NMJs have utility as models of human NMJs. 12
Advances in gene editing allow for insertion of genetically encoded calcium indicators (GECIs) into neurons to visualize calcium signaling.57,85 iPSCs have been modified by inserting exogenous copies of myoblast determination protein 1 [MYOD1] to drive skeletal muscle differentiation.65,86 These genetic modifications enable precise control and measurement of cellular activity to more accurately model NMJ disease phenotypes. Other genetic modifications of iPSCs have been pursued to rapidly develop mature lineages. 47 Since iPSCs have broad plasticity, they can also be differentiated into Schwann cells65,87,88 to incorporate a myelin sheath.
2D NMJ co-cultures are ideal for microscopy analysis and can provide insight into the cellular and molecular markers. While 2D NMJ co-cultures are short lived, they are easy to manipulate and analyze (Fig. 2). Modification to the 2D culture surface by extracellular matrix (ECM) coating or microfabrication techniques can improve cell attachment, viability, and morphology.48,57,58,62,82
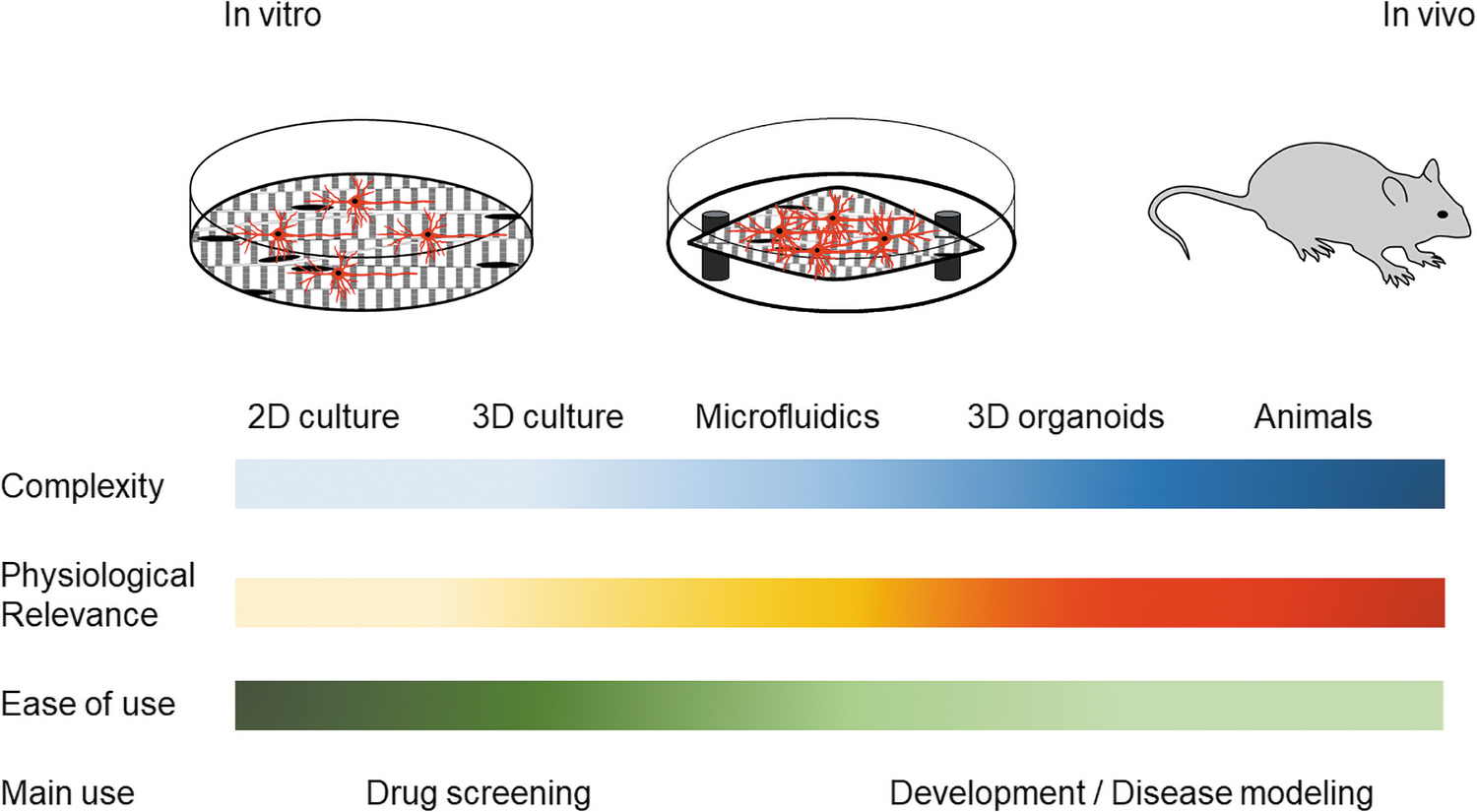
Types and characteristics of NMJ models. Each characteristic bar (left) represents application to the presented type of NMJ model (top). Darker bar color corresponds to higher value of presented characteristics (e.g., complexity of the NMJ models increases from 2D culture to in vivo animal models). 2D, two dimensional.
3D designs have more variation in their structure and size ranging from encapsulating muscle in ECM to provide softness and support 48 to using pillars or attachment points to drive muscle fiber elongation and alignment (Fig. 2).43,45,78,81 Myotube alignment is a priority in these NMJ models as it more accurately recapitulates in vivo skeletal muscle fiber orientation and physiological function.48,89 Subsequently, MNs are added to the 3D muscle cultures, either directly to the culture54,57,78,79 or in an adjoining compartment,39,45,59,61,69,73,75 to allow the formation of NMJs. These cell culturing technologies, newly engineered biocompatible materials, and microfluidic devices have resulted in the progression from inflexible 2D cultures to multicellular 3D NMJ tissue models (Table 2).
Neuromuscular Junction Models and Their Application
Segregation of NMJ models based on their intended application and model complexity.
Cell separation.
Cell separation and active fluid exchange.
2D, two dimensional; 3D, three dimensional; HTS, high-throughput screening.
The success of NMJ formation in 2D and 3D cultures can be assessed by visualizing synaptic organization or by functional connectivity. Pre synaptic and postsynaptic markers can be stained to indicate the aggregation and co-localization of key receptors.90–93 NMJ formation can also be qualitatively assessed by their functional activity. The traditional, gold standard method for measuring NMJ function is to perform patch clamp experiments,76,94,95 which can stimulate MNs or record muscle contraction.
When applied to 2D cultures, this method allows for direct electrical activation and measurement of intracellular responses, but it is highly invasive and destructive to the tissue.37,44,82 Alternatively, 2D NMJs can be cultured on noninvasive multielectrode arrays (MEAs), which detect spontaneous or stimulated extracellular electrical responses. MEAs allow for increased throughput, but have reduced sensitivity.34,96–99 The advantage of using this method is that the same NMJ culture can be followed over a time course, revealing the maturation of tissues as spontaneous action potentials increase in frequency and synchronization.64,67 Multiplexing of MEA platforms allows for monitoring of functional changes following drug or toxin exposure and potential recovery. 100
This can reveal important insights into dose responses, drug efficacy, and toxicity studies. Other indirect methods have also been developed to stimulate 2D NMJ cultures. For example, exposing the cultures to neurotransmitters (e.g., glutamate or ACh signaling blockers) or genetically incorporating light-sensitive ion channels into MNs (optogenetics) allows for controlled NMJ stimulation.39,42,57,60,62 Following MN stimulation, muscle responses can be monitored by electrical, cell imaging, or displacement techniques. Chemical responses can be monitored using calcium influx,50,52,57,72 intracellular signaling and neurotransmitter release from the presynaptic neuron to the muscle. It is possible to measure NMJ function in 3D cultures, despite the added complexity of muscle tissue organizing into dense fibers. In these cultures, muscle contractions can be monitored by measuring cantilever movement or pillar deflexion.39,42,45,81
Quantifying muscle displacement requires extensive imaging and analysis, which are labor-intensive. 85 Ongoing research endeavors are being investigated to assess the functionality and strength of NMJ in 3D cultures. For example, 3D MEAs have been created which allow for muscle to form around a 3D electrode. 99 Other groups have incorporated biocompatible conductive material that project into the 3D tissue cultures to measure electrical activity.101,102 All of these NMJ components and design options should be taken into consideration when applying their use to specific applications, which are detailed below (Fig. 2).
MPSs by Application
Development
Developmental models of NMJs provide insights into neurological dysfunction associated with aging and repair mechanisms critical for treating neuronal injuries. Complexity has been built into these models incrementally to more accurately reflect human development. NMJ models of development have significant design flexibility, as some provide more extensive physical stimulation (stretch, strain, and construction) while others capture complex cellular signaling, revealing the critical mechanisms of tissue maturation.
Many NMJ systems are optimized for 2D culturing due the ease of manipulating these simple designs. Studies implementing these models report the following: (1) the necessary concentration gradient of secreted cellular trophic factors to enhance maturation53,65; (2) the importance of substrate and media composition to enhance cell viability and synaptogenesis76,106–108; and (3) the critical mechanical and electrical cues needed for cellular maturation.47,48
A compartment culturing approach directs the growth of neurites outwards toward muscle, while preventing the movement of the neuronal cell body,50,53,80,109 and a version of this design is commercially available. 71 This can be accomplished using microfabrication of polydimethylsiloxane (PDMS) into microtunnels and has been used to study spatial mimicry of cellular interactions, 80 trauma recovery, 49 axonal regeneration, 110 and the stiffness modulation effect. 48
The role of electrical and physiochemical stimulation on NMJ development has also been explored. The positive effects of electrical stimulation have been reported for both MN and muscle maturation as measured by upregulated gene expression and neuronal synaptogenesis formation.47,111 In addition, the expression of optogenetic modifications allows for stimulation by light,60,112 which has been shown to support NMJ maturation.
These findings have been applied to 3D culturing designs to further capture the complexity of mature NMJ interactions. For instance, 3D models contain embedded muscle in a compartment that allows for efficient innervation when MNs are added to the culture. In these designs, mechanical structures are not required, as secreted trophic factors from muscle are sufficient to induce positive chemotaxis directing neurite extension—allowing for the force of muscle contraction to be investigated—45,75 and these 3D devices are also commercially available. 113
However, it remains challenging to model NMJ aging. Since many developmental models are seeded with stem cells, they best recapitulate early-stage development and are limited in their ability to resemble NMJ aging. This has been addressed by seeding these models with iPSCs expressing age-related mutations, adding external stressors to the cultures to induce faster aging of the tissues, 114 or transdifferentiating older cells to maintain the age-associated features of donor cells. 115 While challenges establishing long-term cultures currently limit some age-related developmental studies, bioengineering advancements continue to enhance the viability and longevity of NMJ cultures.
Disease
Disorders and diseases of the NMJ can cause communication deficits, limited mobility, and respiratory failure, and often have fatal outcomes. These disorders can be categorized by their causative agent: genetic, immune mediated, or toxic exposure.
In vitro NMJ models of neurological disease give insights into disease progression, potential biomarkers for early detection, and the therapeutic potential of pharmacology, gene therapy, and stem cell therapy. 116 Modeling NMJ diseases is challenging due to the fact that few neurological diseases can be resolved down to specific genetic mutations; instead a variety of genetic mutations are responsible for NMJ disease phenotypes. 14
Primary cells from patients with neurological disease are a valuable, but limited, resource to investigate pathology responsible for NMJ disruption. Undifferentiated stem cells can be gene edited to introduce mutations to better model these disease states. 67 The use of patient-derived iPSCs can further improve these models, capturing the entire genetic background even when the etiology is unknown.39,40,73 Cultures established using iPSCs can capture interactions at the cellular level as well as the unique way in which an individual's own cells respond to a stimulus or treatment.40,117
Spinal muscular atrophy (SMA) patient-derived iPSCs have been used to screen and identify drugs that can restore normal NMJ function. 41 An SMA NMJ model has also been created from a single iPSC source that is differentiated into both neural and muscle tissue. 65 There are challenges to using iPSCs: the differentiation protocols for iPSCs may result in a heterogeneous population of cells; the differentiated cells may not be functionally mature; and mutations may accumulate as cells are repetitively passaged. 118 This is especially relevant for iPSC-derived NMJ models of neuromuscular diseases associated with aging.
Technological advancements in personalized medicine continue to propel the study of NMJ diseases.22,39 The role of autoantibodies on disease pathology can be investigated. Incubating IgG from myasthenia gravis patients in an NMJ model has been reported to mimic the characteristic loss of muscle strength observed in patients. 60 The added complexity of 3D NMJ co-culturing can more accurately reflect the in vivo environment45,73 and thus can be better reflective of disease pathology. Major advancements have been reported in NMJ disease modeling of ALS. iPSC-derived muscles and optogenetic MNs cultured in a 3D model have been shown to elucidate the pathogenesis of ALS and enable screening of drug candidates. 39
Models that incorporate mesenchymal stem cells as support cells have shown these cells are responsible for releasing extracellular vesicles, which may reveal novel strategies for ALS treatment. 116 MNs derived from ALS-patient iPSCs expressing disease-specific mutations were developed into NMJ models, which reproduced pathological phenotypes, allowing for specific treatments to be screened to determine ALS etiological investigation and patient stratification for drug testing. 40
The influence of environmental pollutants on the development of ALS has been observed in various case studies3,119 and can be recapitulated in in vitro NMJ models. Environmental neurotoxin exposures, in particular OP exposure, are associated with various NMJ diseases, 120 such as a higher incidence of ALS among Gulf War veterans. 121 OPs, and other nerve agents, inhibit AChE and acute exposure can cause a cholinergic crisis.
Exposure to high doses of OPs can cause Organophosphate-Induced Delayed Neuropathy (OPIDN) resulting in nonlethal, reversible, ALS-like motor impairment and paralysis. 122 Occupational, chronic, or repeated exposure to low OPs levels, which do not cause a cholinergic crisis, is associated with neurodevelopmental disorders and diseases,123–125 but a cause–effect relationship is difficult to establish in humans. Better monitoring of exposure levels and timing in in vitro NMJ models can help establish noncholinergic mechanisms of OP neurotoxicity and the complex aspects of environment-gene interactions.
High-throughput screening
There are distinctive needs of NMJ high-throughput screening (HTS) models, including drug toxicity and efficacy testing. OP and toxin testing (e.g., BT) cannot ethically be evaluated in humans due to the irreversible nature of their binding, which can lead to morbidity and mortality. Animal models have therefore been widely used to gain insight into toxin exposure and to evaluate potential therapeutic interventions.126,127 NMJ HTS systems have been developed for in vitro toxin potency testing, with the expectation to replace the currently used mouse lethality bioassays.25,27,128–130
NMJ HTS models are specialized in their ability to be used in pharmacological settings. These models are formatted to accommodate standardized plate layouts, which fit robotic liquid handling devices, sensitive detectors, and large-scale data processing systems.131,132 This allows for mass production, easy scalability, and user-friendly handling. 2D NMJ models are predominately used because they can be easily scaled and applied for HTS. Using a compartmented design, it is possible to determine precise cell-type dosing due to the physical segregation of MNs and muscle.
A multiplex approach has been reported for an NMJ HTS model where 126 compartmentalized replicates were designed on a platform compatible with automated liquid handling and image analysis. In this system, rapid identification and quantification of functional NMJs are explored using viral vectors for neuronal tracing, which has the potential to be automated. 62 3D models are rarely compatible with HTS; however, they can be multiplexed into a 24-well format. 69 A custom bioengineered plate layout has been used for 3D muscle culturing and could be adapted as an NMJ model by adding neurons. 133 While these approaches increase the throughput, additional modifications need to be implemented to allow for automation and commercialization.
The automated process of data collection and analysis remains a particularly challenging area of HTS NMJ development. There have been efforts to develop automated processors, such as the “NMJ-Analyzer,” to screen NMJ morphology and determine the innervation status. 134 Cell-based assays that measure NMJ responses by muscle displacement, spontaneous or synchronized electrical activity, or chemical release (Fig. 1) require significant user input and intervention. However, HTS models can capture electrical action potentials following NMJ activation using automation ready techniques, like an MEA, which removes user subjectivity and increases data processing.34,42,58,67 Ongoing work, in our laboratory and others, is enabling multiplexing of MEA recordings.
Another major hurdle in implementing NMJ HTS models for pharmacological studies is the lack of comparative validation studies. 131 Even the selection of cell sources, whether obtained or created in bulk, needs careful consideration to limit variability in these systems. 135 While several studies have investigated the effect of specific drugs on NMJ systems—either targeting muscle, neuron, or synaptic transmission34,47,71,136—these results have not been directly correlated to in vivo studies. This can be due to a lack of comparison of in vivo to in vitro data, emphasizing the need for harmonizing benchmark validation studies.
Several large-scale efforts are being pursued to standardize MPS for drug testing. In the United States, the National Institute of Health has awarded funding to teams at multiple locations utilizing MPS models and replicating results with selected compounds. They aim to promote MPS use at regulatory agencies and pharmaceutical companies, and at a greater frequency within the general research community. Their work assists in determining if these models can be used to assess reproducibility, efficacy, and safety in ways that can replace some current animal models.137,138
Recent publications highlight the use of quantitative pharmacology 139 and species comparative IC50 culturing 140 for improvements in research to preclinical translation. However, there are challenges adapting HTS for drug development and screening among the worldwide research community.141–145 There are several global networking organizations whose mission is to drive the development and application of MPS, which is further discussed below. These efforts, while focused on the broader MPS community, support the improvements necessary for creating guidelines for NMJ drug and toxicity testing. 146
Conclusions and Future Directions
Over the last 20 years, novel bioengineering and cellular technologies have advanced the field for NMJ culture in the laboratory and multiple teams have demonstrated the utility of a co-culture system compared to interrogation of single neurons or muscle cultures. For example, incorporating disease-specific neurons into these systems can shed light onto disorders such as ALS or myasthenia gravis. Functional NMJ co-cultures can also look at recovery of the system as a whole from an environmental insult like OP exposure along with therapeutic countermeasures. Even patient demographics can be considered in building these models. However, each model in itself must be evaluated for its own specified uses.
The overarching goal of these systems, regardless of model type, is to provide sound data to understand complex cell and tissue interactions and physiology. In some cases, these data may ultimately be desirable for use in decision making within a regulatory environment if the system and data can be characterized as specific for the job intended, reliable, and sensitivity benchmarked as reproducible. While these co-culture systems provide physiological complexity and potential relevance, understanding what can confidently be inferred from each model is a nontrivial task that requires multiple sets of data for each type of insult, toxin, or disease state.
The biological end-goal or use of the data should be kept in mind when designing or choosing an NMJ model. Some models may not transition easily to providing answers for regulatory questions. If the intention of the model is for regulatory use, new alternative toxicological testbeds and methods must be shown to meet the needs of federal or international agencies.147–149 To help teams across the globe align testing efforts, several commissions have been formed to increase collaboration. The T4 Translatlantic Think Tank for Toxicology and the European Commission directed Organ-on-Chip in Development (ORCHID) Project have developed roadmaps for relevant stakeholders to align MPS technology.
This includes evaluating the technology and identifying unmet needs, establishing standards for regulatory implementation, and developing a guide to assist with the required R&D efforts. The work cites elimination of animal testing as a major driver; however, they also seek to boost accuracy and shorten timelines for standard cell culture methods. 150 Understanding the translational implications of results and cataloging of similarities and differences in pharmacokinetics and physiological processes in the system compared to animal models and humans will be required, as well as computational toxicological models to extrapolate the data.
Other efforts underway with the US National Institute of Standards and Technology to standardize multiple aspects of the MPS field from ECM to microfluidic parameters will help the field produce more aligned and comparable data sets from all types of MPS, including NMJ models. 151 There is outstanding research being performed the world over and finding ways to encourage and support the globalization of MPS translational research will continue to propel this field forward.
As NMJ models become more user friendly and easily accessible to general or commercial laboratory use, it is expected that they will accelerate and enhance pipelines for the study of NMJ development, disease research, and pharmaceutical discovery by providing a platform to gain a deeper understanding of the tissue as a whole.
Footnotes
Acknowledgments
Motifolio images were used to design the included figures. Approved by Los Alamos National Lab for Unlimited Release: LA-UR-21-29377.
Authors’ Contributions
Conceptualization: E.A.S. Formal analysis: J.B.S., E.A.S., K.D.-A., and J.F.H. Data curation: J.B.S. and E.A.S. Writing—original draft: J.B.S. and E.A.S. Writing—review and editing: J.B.S., E.A.S., K.D.-A., J.F.H., and S.M.-V. Visualization: J.B.S., E.A.S., and K.D.-A. Supervision: J.F.H. and R.I. Funding acquisition: S.T., J.F.H., and R.I.
Disclosure Statement
No competing financial interests exist.
Funding Information
This review was supported by the Defense Threat Reduction Agency (DTRA) interagency Agreements HDTRA-1620298, HDTRA-1138382, and by the Laboratory Directed Research and Development program of Los Alamos National Laboratory under project number 20190167.
