Abstract
The large number of animal models used in spinal cord injury (SCI) research complicates the objective selection of the most appropriate model to investigate the efficacy of biomaterial-based therapies. This systematic review aims to identify a list of relevant animal models of SCI by evaluating the confirmation of SCI and animal survival in all published SCI models used in biomaterials research up until April 2021. A search in PubMed and Embase based on “spinal cord injury,” “animal models,” and “biomaterials” yielded 4606 papers, 393 of which were further evaluated. A total of 404 individual animal experiments were identified based on type of SCI, level of SCI, and the sex, species, and strain of the animals used. Finally, a total of 149 unique animal models were comparatively evaluated, which led to the generation of an evidence-based list of well-documented mid-thoracic rat models of SCI. These models were used most often, clearly confirmed SCI, and had relatively high survival rates, and therefore could serve as a future starting point for studying novel biomaterial-based therapies for SCI. Furthermore, the review discusses (1) the possible risk of bias in SCI animal models, (2) the difficulty in replication of such experiments due to frequent poor reporting of the methods and results, and (3) the clinical relevance of the currently utilized models.
Systematic review registration
: The study was prospectively registered in PROSPERO, registration number CRD42019141162.
Impact statement
Studies on biomaterial-based therapies within the field of spinal cord injury (SCI) research show a large inconsistency concerning the selection of animal models. This review goes beyond summarizing the existing gaps between experimental and clinical SCI by systematically evaluating all animal models used within this field. The models identified by this work were used most often, clearly confirmed SCI, and had a relatively high survival rate. This evidence-based list of well-documented animal models will serve as a practical guideline in future research on innovative biomaterial-based therapies for SCI.
Introduction
Between 250.000
Recent advances in biological and engineering strategies have led to the emergence of innovative biomaterials for use in SCI treatment strategies.2,3 Biomaterial-based applications hold great potential to restore the anatomical structure and augment function after SCI. 3 Biomaterials can provide an environment to facilitate the survival, differentiation, and growth of grafted cells or residual axons across lesions to improve functional recovery.2,3
Still, despite encouraging results emerging from preclinical research, clinical treatment options have hardly improved during the past decades.8–12 In fact, many randomized clinical trials on biomaterial-based therapies failed to reproduce the therapeutic efficacies that were reported in preceding animal studies.13–17 This discrepancy between preclinical and clinical research on SCI stresses the urgent need for the animal models to simulate the clinical scenario as closely as possible. 18
To date, an extensive amount of different animal models of SCI has been described in the literature. However, clinical translation is severely complicated by cross-species differences and highly standardized injuries, which do not resemble the heterogeneity of clinical SCI. 6 In case of testing biomaterial-based interventions, the tendency of researchers to select transection and hemisection defects further limits the predictive value of these experimental SCI models, since these injuries are a rarity in clinics.19–23
Moreover, a recent review showed that SCI treatment strategies were significantly affected by the geographical location of the research performed. 22 These findings raise the question to what extent animal models are differentially adopted based on the biomaterial application or geographical location of the publishing research group. The correlation between experimental and clinical injuries should be investigated in detail to facilitate the objective selection of the most adequate animal model of SCI and augment successful translation of novel biomaterial-based therapies for SCI.
To create uniformity within the field of biomaterial-based experimental SCI, the current systematic review aims to go beyond reviewing the current plethora of model parameters, which has been done previously.20,22 First, the distribution of type and level of SCI will be provided, as well as the sex, species, and strain of the animals used, to address the gaps between experimental and clinical SCI. In addition, the methodological quality of experimental SCI research, such as the reporting of randomization and blinding procedures and animal welfare standards, will be assessed. Finally, the identified animal models will be evaluated based on the confirmation of SCI and animal survival. The resulting evidence-based list of relevant animal models of SCI can serve as a starting point for future research studying biomaterial-based interventions for the treatment of SCI.
Materials and Methods
Study design
The aim of this systematic review was to identify and evaluate the animal models that were used to simulate SCI to investigate biomaterial-mediated repair of the injured spinal cord. This evaluation was performed according to a recently reported strategy that was used to systematically summarize and compare the preclinical models for chemotherapy-induced peripheral neuropathy. 24 The review methodology was specified in advance in PROSPERO (ID: CRD42019141162) according to SYRCLE's systematic review protocol for animal intervention studies. 25
The following amendments were made: (1) to provide more insight into model selection and animal welfare, additional data on biomaterial application, geographical distribution of research, and reporting of ethical statements and humane endpoints were extracted, and descriptive statistics of these characteristics were performed; (2) animal survival was included as part of model evaluation, since the severe effects of experimental SCI on animal welfare render this parameter an essential part of model analysis. Not including animal survival in the original protocol was an oversight on behalf of the authors. This review was reported according to the PRISMA guidelines, 26 presented in Supplementary Data File S1.
Literature search strategy
A comprehensive systematic search was conducted in Medline through the PubMed and Embase interfaces, to identify animal models used to simulate SCI with the purpose of biomaterial implantation. The full search strategy was based on the search components “spinal cord injury,” “animal models,” 27 and “biomaterials,” and is presented in Supplementary Data File S2. Search results from both databases were combined, followed by the removal of duplicates (Fig. 1). The initial search was performed on February 18, 2020, followed by a search update conducted on April 2, 2021.
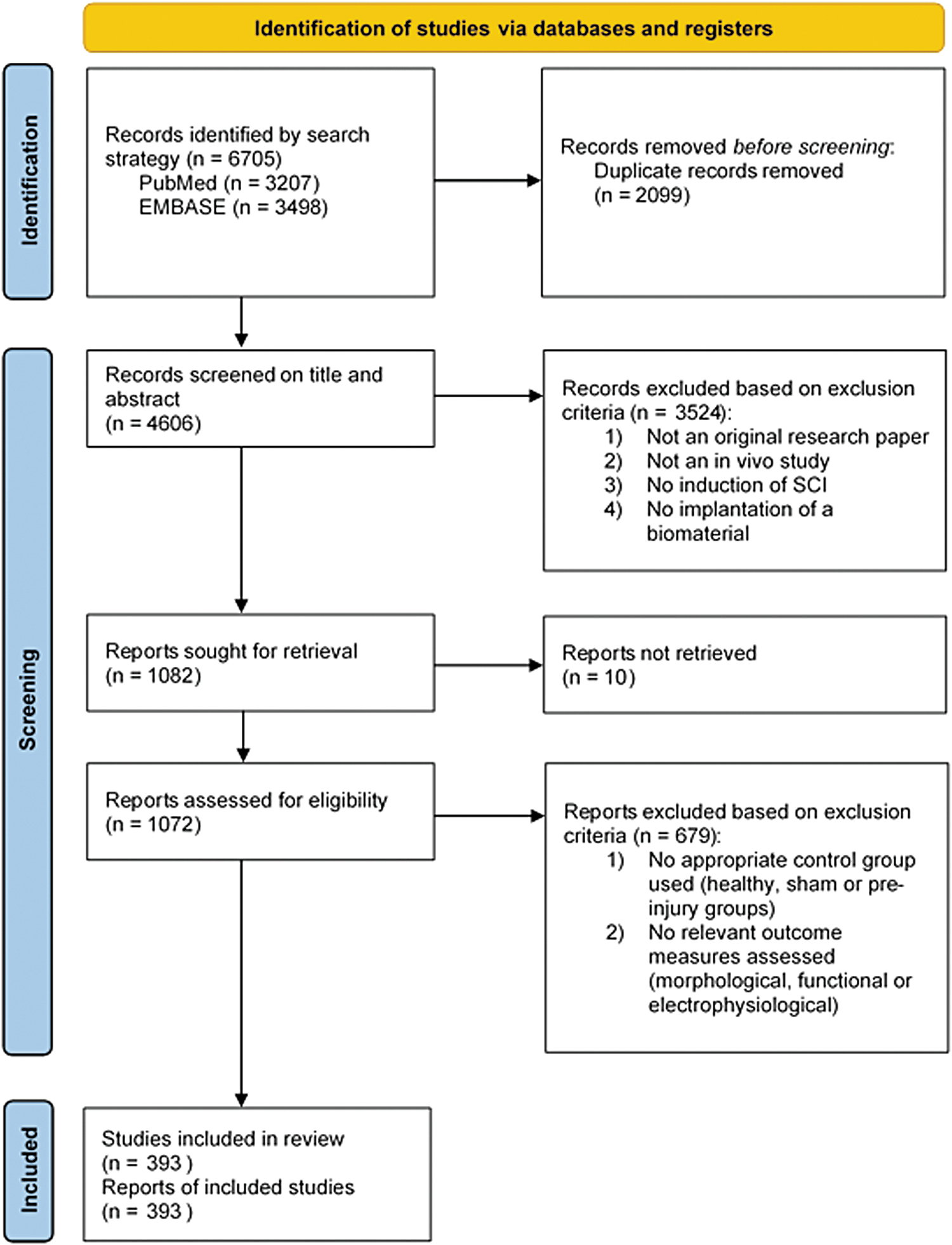
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analysis) flow chart of the systematic search of literature. Color images are available online.
Study selection
Search results were imported in an online reference management tool (Rayyan Systems, Inc.). 28 After the removal of duplicates, studies were first screened based on title and abstract independently by two reviewers (K.V. and R.A.). Studies were excluded if (1) they did not involve an original full-length research article; (2) they were not an in vivo animal study; (3) SCI was not induced; or (4) no biomaterial was implanted after the induction of SCI. Biomaterials were defined as biological or synthetic constructs, with or without the inclusion of cells, cytokines, or drugs.
Following the title and abstract screening, full-text documents of included references were additionally screened, and studies were excluded if (5) they did not include an appropriate control group (healthy animals without SCI, animals undergoing a sham procedure, or pre-SCI baseline measurements); or (6) they did not report on the following outcome measures: anatomical/histological data, functional recovery, or electrophysiological recordings.
No language or publication date restrictions were applied. If necessary, publications in languages other than English were translated by a native speaker for that particular language. Corresponding authors of full-text documents that could not be retrieved online were contacted by mail. Authors who did not respond to the initial email were sent a reminder 2 weeks later. If authors did not respond to this reminder within 2 weeks, the reference was excluded. Disagreements during screening were solved by discussion or by consulting a third party (C.H.). The complete list of included references is provided in Supplementary Data File S3.
Data extraction
Bibliographic details including author, journal, year of publication, and original location of research groups were extracted from each publication. In addition, data concerning one of the following study characteristics were extracted: type of injury (transection, hemisection, contusion, compression); level of injury; animal sex (male, female, both), species and strain; type of outcome measure (morphological, functional, electrophysiological); data on animal survival (absolute numbers, survival rates, or percentages), if provided by the authors; biomaterial application (type of biomaterial, method of application, timing of intervention, timing of final outcome measurement relative to the induction of SCI).
Preclinical models were classified as transection injuries when it involved a complete transverse excision of spinal cord tissue resulting in two stumps or defined as a hemisection in case part of the spinal cord remained intact after excision of tissue. Injuries caused by the impact from weight-drop methods or impactors were classified as contusion injuries. Applying prolonged pressure to the spinal cord, for instance clipping of the spinal cord using a vascular clip, was classified as compression injury. The location of the lesion as stated by the authors was defined as the level of injury.
Definition of animal experiments and animal models
Within the current review, it is important to note the distinction between animal experiments and animal models. Animal experiments were individually extracted from the included studies and identified based on five individual parameters: type of injury, level of injury, and the sex, species, and strain of the animals used. Unique animal models consisted of a combination of these parameters; if one (or more) of these parameters differed from another preclinical model, it was defined as a unique animal model. For instance, three separate studies reporting a transection at T10 in female Sprague–Dawley rats would result in the identification of three individual animal experiments. However, based on the definition stated above, this would lead to the identification of one unique animal model.
Assessment of SCI confirmation and animal survival
Animal experiments were assessed by analyzing the number of times induction of SCI was confirmed based on either morphological (i.e., histology), functional (i.e., Basso, Beattie, Bresnahan locomotor rating scale), and electrophysiological (i.e., motor and somatosensory evoked potentials [MEPs and SSEPs]) analyses defined as relevant outcome measures. Induction of SCI was confirmed when at least one of the relevant outcome measures significantly differed (based on the authors' own significance testing) from values derived from the appropriate control group, defined as either healthy animals, sham-operated animals, or pre-injury baseline measurements.
Animal survival, if provided by the authors, was determined for every uniquely identified animal model and defined as the number of animals that remained alive after induction of SCI and throughout the remainder of the study, divided by the number of animals at the start of the experiment. This approach thus generates a ratio between 0, meaning no animals survived the procedures, and 1, indicating all animals survived the procedures. Animal survival was graded as 1 in case authors reported no exact data but indirectly, in text, indicated all animals survived the procedure. Animal survival was graded as “Unknown” if authors did not provide data on animal survival and no indirect statements concerning survival were made. Animal survival rates were presented as percentages for final interpretation.
Model analysis
The analysis of uniquely defined animal models consisted of three separate phases, and a schematic representation is provided in Supplementary Fig. S1. First, animal models were divided based on the type of injury, since it is difficult to compare the effect of different injury paradigms on the relevant outcome measures. Second, models were classified based on the following parameter hierarchy: level of injury, species, sex, and strain. Animal models were required to meet the following criteria: (1) the model was reproduced at least five times within the dataset, and (2) data on SCI confirmation or animal survival were reported in at least three separate model repetitions. In case a specific model met these predefined criteria during model analysis, it was regarded as a well-documented and promising animal model of SCI.
For the sake of clarity, the following hypothetical models serve as an example:
Transection at T10 in female Sprague–Dawley rats (n = 11); SCI confirmed based on morphological (n = 6), functional (n = 8), and electrophysiological (n = 4) outcomes; animal survival reported (n = 7). This model would be included in the analysis as both criteria were met. Transection at T10 in female Wistar rats (n = 4); SCI confirmed based on morphological (n = 2), functional (n = 3), and electrophysiological (n = 1) outcomes; animal survival reported (n = 3). This model failed to meet the reproducibility number and would be excluded from analysis. Transection at T10 in male Wistar rats (n = 5); SCI confirmed based on morphological (n = 1), functional (n = 2), and electrophysiological (n = 1) outcomes; animal survival reported (n = 2). This model would be excluded from analysis as no reported outcome measure was reported at least three times.
Sensitivity analysis
Animal models of SCI should produce consistent and reproducible results, independent of the level of injury or the species, sex, and strain of the animals used. Therefore, model robustness was assessed in the third phase of model analysis by prioritizing the model-specific parameters in alternating order (i.e., level—species—sex; level—sex—species; species—level—sex; species—sex—level; sex—level—species; sex—species—level). Strain was disregarded in this analysis, since it was completely dependent on the parameter species. In case a specific animal model was not excluded, even with a different parameter hierarchy, it was regarded as a robust animal model.
Methodological quality and animal welfare
The methodological quality of a representative selection (25%) of included references was assessed using SYRCLE's Risk of Bias (RoB) tool. 29 First, an online random generator (Random Integer Generator, random.org) was used to produce a list of random integers that were directly linked to the list of references in alphabetical order based on the author's last name. In brief, 25% of included references were requested following both the initial search (minimum: 1, maximum: 333, both inclusive, performed on 16th of February 2021) and the second search (minimum: 1, maximum: 60, both inclusive, performed on 3rd of May 2021). Identified references, only those written in English, were screened (L.D.) for systematic risks of bias, covering selection, performance, detection, attrition, and reporting bias.
The presence of bias was scored as “Yes,” indicating high risk of bias, “No,” indicating low risk of bias, or “Unclear,” in case information required to address potential bias was not reported. Animal welfare was additionally scored in the same representative sample of references, based on a self-established scoring system to assess the reporting of (1) inclusion of an ethical statement, (2) discussion of humane endpoints, and (3) presentation of data on animal survival. Each component was scored with a “Yes” or “No,” indicating whether authors provided the relevant information.
Results
Study selection
The electronic search strategy retrieved 3207 records from PubMed and 3498 records from Embase (Fig. 1). Following the screening of the title and abstract of 4606 unique references, 1082 studies were included for full-text review. Of these 1082 reports, a total of 393 references were included in the final analysis. The references of the included studies can be found in Supplementary Data File S3.
Study characteristics
Animal models used in the literature were identified based on the type of injury, level of injury, sex, species, and strain. Information regarding the biomaterial application was extracted from these animal experiments, including the type of biomaterial and the method of delivery, along with the timing of intervention and assessment. The country in which the animal model was performed was extracted as the geographical location of research, along with the publication date. Of the 393 references, a total of 404 separate animal experiments were identified, as some research groups reported utilization of multiple experiments.
Distribution of animal characteristics
Transection (n = 158, 39.1%) and hemisection (n = 132, 32.7%) injuries constituted the most prevalent method to induce injury, in contrast to contusion (n = 62, 15.3%) and compression injuries (n = 50, 12.4%) (Fig. 2A). The type of injury was not reported in two studies (0.50%).
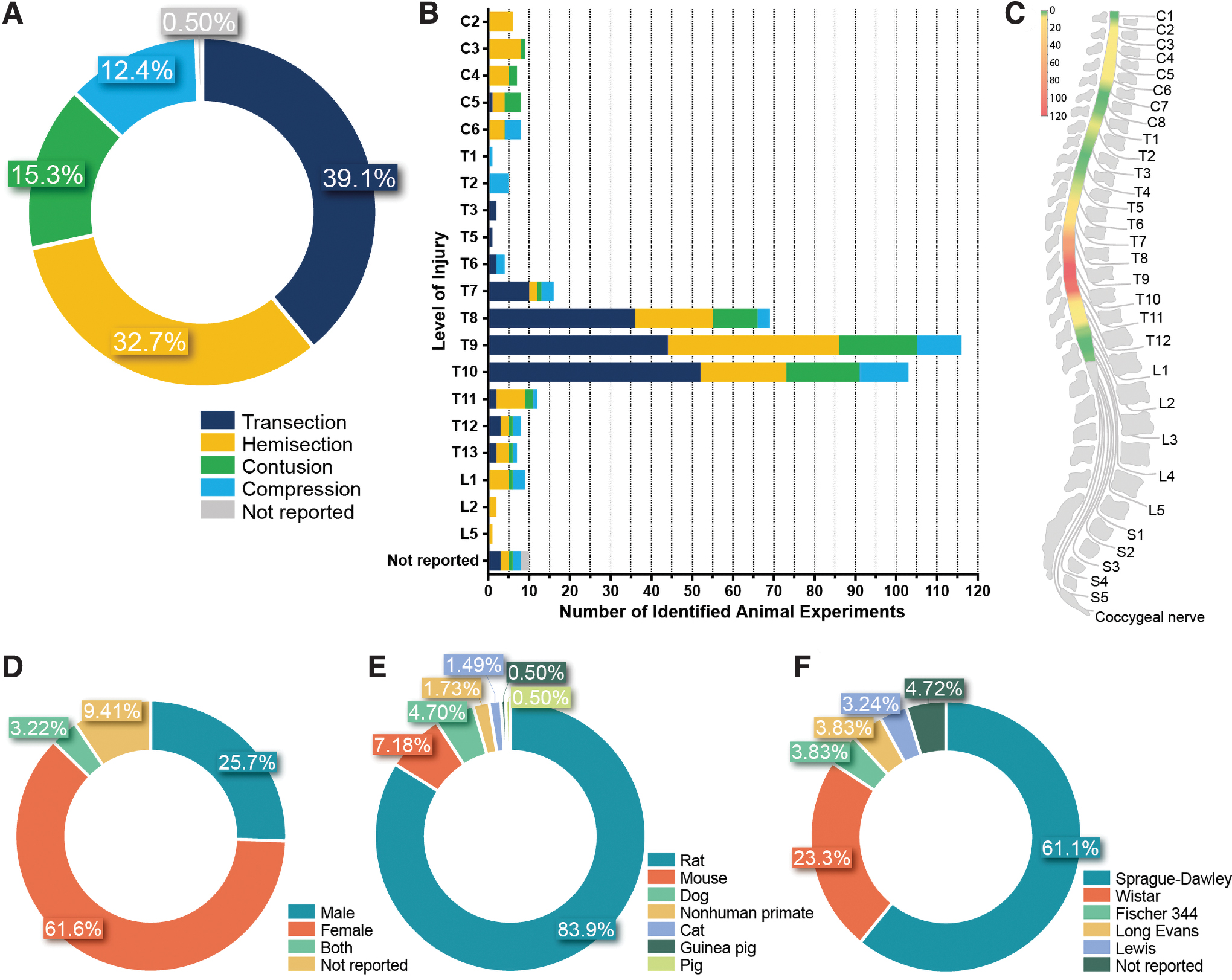
Distribution of model-specific parameters over the 404 separate animal experiments. Type of injury
The thoracic area of the spinal cord was injured most often (n = 344, 85.1%) (Fig. 2B), with T10 as the most prevalent SCI location (n = 96, 23.8%), as shown in Fig. 2C. A total of 38 animal experiments (9.41%) involved an injury inflicted at the cervical area, while lumbar injuries were induced in 12 animal experiments (2.97%). Animal experiments that did not include the exact location of SCI constituted the remaining part (n = 10, 2.48%).
Female animals were more frequently used (n = 249, 61.6%) compared with male animals (n = 104, 25.7%) (Fig. 2D). Only a small number of studies were performed using both sexes both sexes (n = 13, 3.22%), while 38 experiments (9.41%) did not contain any information about the sex of the animals used.
A total of 9 different animal species have been used (Fig. 2E). Rats dominated the dataset with 339 experiments (83.9%), while 16.1% of the experiments were performed in other species: mice (n = 29), dogs (n = 19), cats (n = 6), guinea pigs (n = 2), and pigs (n = 2). Research focusing on nonhuman primates involved rhesus monkey (n = 4), African green monkey (n = 2), and cynomolgus monkey (n = 1).
Sprague–Dawley was the most prevalent strain used in rat-based research (n = 207, 61.1%; Fig. 2F). Other strains of rats used were Wistar (n = 79, 23.3%), Fischer 344 (n = 13, 3.83%), Long Evans (n = 13, 3.83%), and Lewis rats (n = 11, 3.24%). A total of 16 experiments did not report the strain of the rats (4.72%). Concerning experiments in mice, the strain C57BL/6 (n = 22) was most often selected. In the 19 experiments involving dogs, beagles (n = 14) were most often used.
Confirmation of SCI and animal survival
Before model analysis, all 404 animal experiments were assessed with respect to the fact if the induction of SCI was confirmed through morphological, functional, and electrophysiological testing (Supplementary Fig. S2). Injuries were most often confirmed by a combination of morphological and functional outcome measures (n = 222, 55.0%), while all three assessment techniques were used to confirm the injury in 101 animal experiments (25.0%). Only a small number of experiments included a combination of electrophysiological testing with either morphological (n = 12, 2.97%) or functional (n = 9, 2.23%) analyses. Studies reporting just one outcome measure to confirm the induction of injury were based on either morphological (n = 40, 9.90%), functional (n = 13, 3.22%), or electrophysiological (n = 7, 1.73%) assessments.
Data on animal survival could be extracted from about half of the animal experiments. Animal survival after transection (88.9%; Supplementary Fig. S3) and hemisection (90.4%; Supplementary Fig. S4) was found to be lower compared with contusion (93.3%; Supplementary Fig. S5) and compression injuries (97.0%; Supplementary Fig. S6).
Model analysis
Of the 404 identified animal experiments, a total of 149 unique animal models were defined. Supplementary Figures S3–S6 show the results of model analysis regarding transection, hemisection, contusion, and compression models, respectively. Alternating the hierarchy of model-specific parameters did not result in the exclusion of models. Table 1 summarizes the preclinical models that were identified after model analysis. Further specification for each type of injury indicated that the reporting accuracy regarding SCI confirmation and animal survival was relatively high for (1) transection at T10 in female Sprague–Dawley rats, (2) hemisection at T10 in male Sprague–Dawley rats, (3) contusion at T10 in female Sprague–Dawley rats, and (4) compression at T9 in female Sprague–Dawley rats.
List of Most Promising Preclinical Models Following Model Analysis
Shown are all preclinical models that met the predefined criteria (ntotal ≥5; n ≥ 3 for at least one outcome measure) following the three phases of model analysis: stratification based on the type of injury, division based on parameter hierarchy, and the sensitivity analysis. Models are presented based on the type of injury, level of injury, species, sex, and strain used along with the following values: number of confirmed SCIs (based on morphological (M), functional (F), or electrophysiological (E) assessments), animal survival (AS; %), number of times AS was reported, and the total number of times the specific model was repeated. For each specific type of injury, the model with the highest level of SCI confirmation and (reporting of) animal survival is presented in bold text.
SCI, spinal cord injury.
Biomaterial intervention
The type of surgical biomaterial delivery was subsequently categorized to identify potential correlations between the selected type of injury and the type of biomaterial delivery. After the transection of the spinal cord, biomaterials were either implanted into the resulting defect (n = 137, 86.7%) or injected as a gelling solution (n = 16, 10.1%), which applied to hemisection injuries as well (n = 98, 74.2% for biomaterial implantation; n = 22, 16.7% regarding injections to fill the cavity) (Supplementary Fig. S7A).
Most biomaterial delivery modes after contusion and compression injuries involved an intraspinal injection (n = 34, 54.8% and n = 24, 48.0%, respectively), using biomaterials such as cell suspension matrices or hydrogels. Solid premade scaffolds were applied in these injury paradigms as well, including topical applications over the injury site (n = 16, 25.8% and n = 11, 22.0%, after contusion and compression injuries, respectively), such as patch-like applications of hydrogels or membrane films.
Furthermore, both the timing of intervention and assessment were assessed to clarify how biomaterial therapies are applied within this field. Supplementary Figure S7B shows that biomaterial therapies were predominantly applied acutely after the induction of injury (n = 329, 78.1%), while in 6.65% (n = 28) of the identified animal experiments the injuries were treated 1 week postinjury. The exact timepoint of assessment of these 329 acute biomaterial applications is shown in Supplementary Fig. S7C, which demonstrated a strong preference for analyzing these therapies at 4 (n = 59), 8 (n = 85), or 12 weeks (n = 48).
Geographical distribution of research
The geographical location of research was extracted to assess whether there were region-specific differences in model selection. The majority of the 404 experiments was performed in either Asia (n = 231, 57.2%), Northern America (n = 94, 23.3%), or Europe (n = 58, 14.4%). The utilization of preclinical models of SCI substantially increased over the recent years, specifically in Asia (Supplementary Fig. S8A), Northern America (Supplementary Fig. S8B), and Europe (Supplementary Fig. S8C). The selection of a certain type of injury showed contrasting trends over time within each region of interest. For instance, the yearly cumulative percentage of transection models increased in Asia (Supplementary Fig. S8D), in contrast to Northern America (Supplementary Fig. S8E) and Europe (Supplementary Fig. S8F) where this type of injury was decreasingly selected.
On the other hand, utilization of contusion and compression models steadily increased over time in Northern America and Europe, in contrast to research based in Asia where a decreasing or stabilizing trend for contusion and compression models was observed, respectively. In view of these differing selection preferences, each region presented different characteristics regarding the type of injury. In Asia, transection (n = 122, 52.8%) and hemisection (n = 59, 25.5%) methods were preferred over contusion (n = 23, 9.96%) and compression (n = 25, 10.8%) models (Supplementary Fig. S8G).
Animal experiments conducted in Northern America showed a more frequent use of contusion (n = 23, 24.5%) and compression (n = 14, 14.9%) models, although transection (n = 25, 26.6%) and hemisection (n = 32, 34.0%) injuries still comprised a large part of research performed within this region (Supplementary Fig. S8H). Europe-based research showed a strong preference for hemisection (n = 34, 58.6%) models (Supplementary Fig. S8I), while only a minor part involved the complete transection of the spinal cord (n = 5, 8.62%). Contusion (n = 9, 15.5%) and compression (n = 10, 17.2%) models comprised the rest of the experiments conducted in Europe.
Methodological quality and animal welfare
The risks of bias (selection, performance, detection, attrition, and reporting bias) and animal welfare reporting standards were assessed in a representative sample (n = 97) of included studies. The risk of bias assessment (Fig. 3A) showed that essential details were not provided with respect to allocation sequence generation, allocation concealment, random housing, and random outcome assessment. Animals were often reported to be randomly distributed over experimental groups, but authors failed to describe the actual method of randomization, for instance by referring to a random number generator. 29
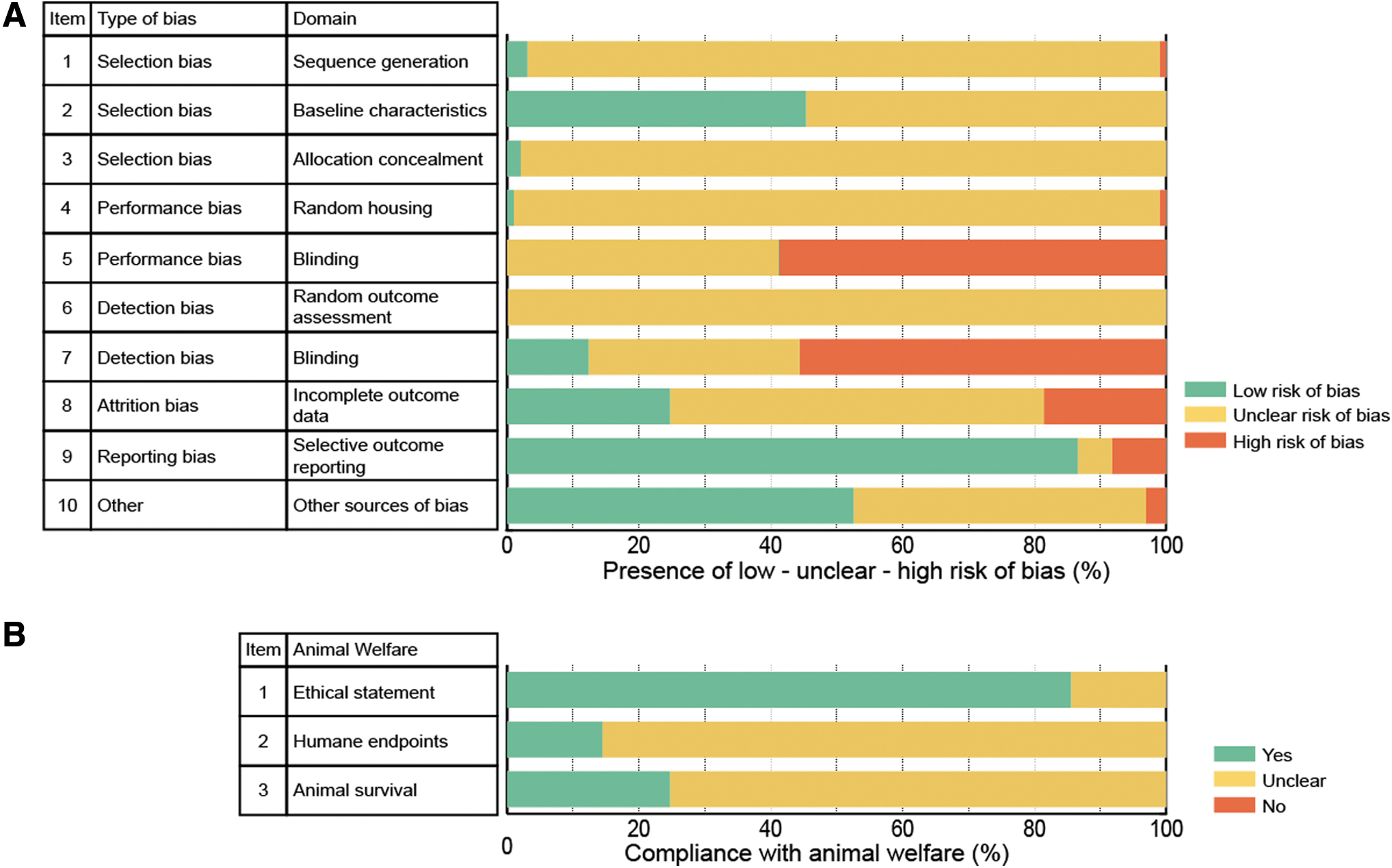
SYRCLE's risk of bias analysis
This limits the assessment's accuracy of the randomization method and therefore contributes to the risk of selection bias. Baseline characteristics, including the age, sex, and weight of the animals in addition to surgical procedures during SCI induction, were evenly distributed over experimental groups in 45.4% (n = 44) of the selected studies. Since these studies often compare experimental groups with control groups such as healthy or sham-operated animals, blinding of caregivers and investigators during the experiments as well as blinded outcome assessment suffered from high risks of bias (58.8% and 55.7%, respectively). Still, 12.4% of the selected studies included details regarding blinding during outcome assessment.
Furthermore, a large amount of the selected studies (n = 55, 56.7%) did not adequately describe outcome data, as animal numbers in the Results section often differed from the Methods section, resulting in an unclear risk of attrition bias. In contrast to these discrepancies between methods and results, the selected manuscript was generally (86.6%) free of selective outcome reporting. Compliance with animal welfare (Fig. 3B) was reported in an ethical statement in 85.6% (n = 83) of the included studies. On the contrary, humane endpoints (n = 14, 14.4%) and animal survival (n = 24, 24.7%) were only marginally reported.
Discussion
The aim of the current systematic review was to create uniformity within the field of biomaterial-based experimental research on SCI by providing an evidence-based list of well-documented animal models. The evaluation of 393 studies, including 404 individual animal experiments, led to the identification of the following animal models: (1) transection at T10 in female Sprague–Dawley rats, (2) hemisection at T10 in male Sprague–Dawley rats, (3) contusion at T10 in female Sprague–Dawley rats, and (4) compression at T9 in female Sprague–Dawley rats (Table 1). These models were used most often, clearly confirmed the presence of SCI, and had a low number of unexpected animal deaths.
This list of models could serve as a principal starting point for future research studying the therapeutic efficacy of biomaterial-based interventions to facilitate successful clinical translation. However, the translational value of promising biomaterial-based therapies depends on the resemblance of these models to the clinical situation. The current review revealed gaps in experimental SCI research that should be addressed to improve the translational value of biomaterial-based treatment strategies, which will be discussed below.
Concerning the types of defects, biomaterial-based therapies are predominantly investigated in hemisection and transection models, contrasting the injuries typically observed in the clinic. 21 Since these injuries involve precise cuts into well-characterized spinal cord tracts, they can be valuable in finding novel therapies to enhance axonal regeneration and anatomical reorganization in addition to evaluating overall tissue response after biomaterial implantation. 30 Furthermore, insights gained from these types of animal models could be valuable for treatment of patients suffering from chronic SCI. Necrotic tissue could possibly be excised from the lesion site, followed by the implantation of a scaffold into the created cavity. 9
However, models that involve resection of spinal cord tissue generally fail to reproduce many of the features of SCI as observed in the clinic.2,9 In fact, the mechanisms responsible for functional recovery after contusion injuries are different from those observed after precise cuts, 30 hampering intermodel translation. On the contrary, contusion injuries are highly variable in pathway interruption, severity, and functional outcomes. 30 Comparative studies utilizing multiple types of models, which are rarely applied to date, should be implemented to allow for effective intermodel assessments and enhance clinical translation.
Human SCI predominantly involves cervical defects,4,31 while experimental SCI is mainly induced in the thoracic areas of the spinal cord. Severely reduced animal distress and overall better outcomes on animal survival, along with enhanced accessibility of thoracic areas during surgery, could contribute toward motivating the choice for this anatomical site in animal models. 32 Despite these practical reasons, there exist site-specific differences regarding anatomy and pathophysiology. 6
Moreover, the receptivity to a certain treatment could be greatly affected by the exact lesion location. 22 In addition, the low number of cervical models, which are often accompanied by large outcome variance, limits the assessment and subsequent comparison to thoracic models. 22 Therefore, cervical models should also be considered to better mimic the clinical situation and investigate the potential differences in biomaterial effect sizes between the two areas of the spinal cord.
Importantly, most studies are focused on the lesion epicenter to confirm the induction of SCI and assess treatment outcomes. However, SCI could affect segments extending both rostrally and caudally from the injury site all along the spinal cord and even different areas of the brain. 33 To fully evaluate the effect of the lesion and the efficiency of biomaterial therapies, it is critical to concentrate more efforts on areas distant from the injury site. 34
Although translational differences exist among species, rat models of SCI are currently regarded as adequate experimental rodent models since they closely resemble human SCI.35,36 Moreover, rats are cheaper, more abundantly available, easier to handle within an animal research facility, and to a lesser extent associated with ethical concerns (3R principle) compared with larger sized animal models.18,37 However, the rodent and human spinal cord substantially differ regarding size, anatomy, function, and inflammatory response. 38
This should be taken into account when developing regenerative therapies, as functional recovery in rodents could result from axonal regeneration covering a few millimeters, while human SCI could involve several centimeters of injured tissue. 38 This review clearly demonstrated the current lack of larger sized animal models, such as dogs, pigs, or nonhuman primates, all of which more closely resemble the size and neuroanatomy of humans. 39 Experimental canine models or even spontaneous canine SCI, which recapitulates many of the heterogeneous injury characteristics observed in human SCI, could prove to be valuable intermediate animal models.40,41
Furthermore, studies including multiple species are required to allow for more effective clinical translation.8,42–44 For instance, biomaterial implantation after resection procedures improved neural regeneration and functional recovery in rats and canines.8,42,43 Indeed, identical biomaterial treatments applied in pigs after contusion injuries only partially replicated the recovery observed in rats, 44 demonstrating the need to thoroughly investigate the underlying injury and regeneration mechanisms between multiple species.
Traumatic SCI occurs more often in male patients (79.8%) than in females (20.2%). 31 In contrast, animal experiments were more often conducted in female animals (61.6%) than in male animals (25.7%). There appears to exist a gender-based improvement in recovery favoring female animals in experimental SCI primarily due to the neuroprotective effect of 17β-estradiol,45,46 which could in turn lead to an overestimation of the therapeutic efficacy of biomaterial-based therapies.
Currently, it is unclear whether this gender-based difference also exists in the clinic. 46 The preference for female animals is probably caused by the lower tendency to develop urinary tract infections (one of the most abundant and severe complications after SCI) and easier handling.47,48 Promising results emerging from research performed solely in female animals should be carefully interpreted and validated in their male counterparts to increase the predictive value of biomaterial-based therapies.
Clinical resemblance does not seem to be the main driving force for model selection to study the therapeutic efficacy of biomaterials after SCI. This is demonstrated by the over-representation of acute therapies. The pathophysiology of acute, subacute, intermediate, and chronic phases of SCI differs considerably, and given the fact that millions of patients are currently facing the chronic consequences of SCI, the need for effective interventions for treatment of such chronic deficits is evident.4,6
Moreover, data presented in this systematic review pointed to a correlation between model selection and subsequent biomaterial application. This could support the hypothesis that researchers actually select a certain animal model based on the type of biomaterial that needs to be tested, rather than from clinical motives. For instance, it would be more convenient to use models that involve resection of tissue in case of a solid scaffold or hydrogel.
These findings can also be attributed to previous experience of researchers, which is supported by the region-specific model selection over time regarding Asia, Northern America, and Europe (Supplementary Fig. S8D–F). Specifically, the use of transection models is steadily increasing in Asia, while research based in either Northern America or Europe seems to focus on the other types of defects. These findings can partially be attributed to cultural differences with respect to ethical concerns. 32
Furthermore, the geographical location of publishing research group was previously shown to affect SCI treatment effect sizes, which was suggested to be caused by attrition bias. 22 Extensive research focusing on research groups is needed to investigate whether animal models are selected to maximize effect sizes. Since biomaterial-based therapies are less likely to be reproduced by unrelated research groups, data sharing and international cooperation are required to explain the underlying differences and increase the chances of translational success.
Low methodological quality was previously shown to result in overestimated effect sizes of experimental SCI treatment strategies. 22 In the current review, the assessment of the actual risk of selection bias was hampered due to the generally poor reporting standards of the experimental design. To decrease bias in future biomaterial-based research, it is strongly encouraged to report on especially (1) randomization procedures, (2) allocation concealment, and (3) sample size calculations, since these factors were rarely discussed in the included studies.
Furthermore, blinding between experimental groups and control groups is practically impossible due to the obvious differences in locomotor function. Because of the lack of blinding of caregivers and outcome assessors, performance and detection bias may have been introduced, which detracts from the overall validity of the confirmation of SCI induction.49,50 Exclusion of animals during the experimental procedures should be reported, for example by providing survival rates or percentages, along with transparent reporting of reasons why animals were excluded. To minimize study variability, it is strongly emphasized that future research should report the experimental design and compliance with animal welfare according to international standardized protocols, 32 such as the Minimum Information About a Spinal Cord Injury Experiment 51 or the ARRIVE guidelines. 52
Conclusions
In conclusion, this review identified the following set of principal experimental models of SCI: (1) transection at T10 in female Sprague–Dawley rats, (2) hemisection at T10 in male Sprague–Dawley rats, (3) contusion at T10 in female Sprague–Dawley rats, and (4) compression at T9 in female Sprague–Dawley rats. These models could serve as a starting point for future research investigating the therapeutic efficacy of biomaterials after experimental SCI.
To allow for more effective clinical translation and bridge the gap between experimental and clinical SCI, it is necessary to (1) develop comparative studies, including various types of injuries, more cervical models, multiple species, and both sexes, to address intermodel and interspecies differences; (2) intensify data sharing to enhance international cooperation within the field of experimental SCI and increase the adoption of a clinic-based selection of animal models before biomaterial interventions; and finally (3) improve the reporting of essential details regarding experimental design by including randomization and blinding procedures, provide sample size calculations, and present animal welfare and survival in a transparent manner.
Footnotes
Acknowledgments
The authors express their appreciation to the colleagues of NeuroStimSpinal for valuable input and critiques concerning this research work.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Authors' Contributions
K.V., R.A., C.H., X.F.W., R.B., and K.W. contributed to conceptualization; K.V., R.A., C.H., and K.W. contributed to constructing search strategy; K.V., R.A., and L.D. performed investigation; K.V. and R.A. contributed to data acquisition; K.V. performed visualization; K.V., R.A., and A.K. contributed to writing of the original draft; K.V., R.A., A.K., C.H., X.F.W., R.B., and S.L. contributed to writing—review and editing; X.F.W., C.H., R.B., and S.L. performed supervision; R.A. contributed to project administration; X.F.W. and R.B. performed funding acquisition. All authors read and approved the final article.
Disclosure Statement
The authors declare to have no competing interests.
Funding Information
This study was funded by the European Union's Horizon 2020 research and innovation programme under grant agreement No 829060 and the Netherlands Organisation for Health Research and Development (ZonMW; Project number 114024132; received by X.F.W. and R.B.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
