Abstract
It is well known that the biomechanical and tribological performance of articular cartilage is inextricably linked to its extracellular matrix (ECM) structure and zonal heterogeneity. Furthermore, it is understood that the presence of native ECM components, such as collagen II and aggrecan, promote healthy homeostasis in the resident chondrocytes. What is less frequently discussed is how chondrocyte metabolism is related to the extracellular mechanical environment, at both the macro and microscale. The chondrocyte is in immediate contact with the pericellular matrix of the chondron, which acts as a mechanocoupler, transmitting external applied loads from the ECM to the chondrocyte. Therefore, components of the pericellular matrix also play essential roles in chondrocyte mechanotransduction and metabolism. Recreating the biomechanical environment through tuning material properties of a scaffold and/or the use of external cyclic loading can induce biosynthetic responses in chondrocytes. Decellularized scaffolds, which retain the native tissue macro- and microstructure also represent an effective means of recapitulating such an environment. The use of such techniques in tissue engineering applications can ensure the regeneration of skeletally mature articular cartilage with appropriate biomechanical and tribological properties to restore joint function. Despite the pivotal role in graft maturation and performance, biomechanical and tribological properties of such interventions is often underrepresented. This review outlines the role of biomechanics in relation to native cartilage performance and chondrocyte metabolism, and how application of this theory can enhance the future development and successful translation of biomechanically relevant tissue engineering interventions.
Impact statement
Physiological cartilage function is a key criterion in the success of a cartilage tissue engineering solution. The in situ performance is dependent on the initial scaffold design as well as extracellular matrix deposition by endogenous or exogenous cells. Both biological and biomechanical stimuli serve as key regulators of cartilage homeostasis and maturation of the resulting tissue-engineered graft. An improved understanding of the influence of biomechanics on cellular function and consideration of the final biomechanical and tribological performance will help in the successful development and translation of tissue-engineered grafts to restore natural joint function postcartilage trauma or osteoarthritic degeneration, delaying the requirement for prosthetic intervention.
Introduction
Articular cartilage is a highly specialized connective tissue found lining the end of bones in synovial joints, where it provides a low friction and low wear surface for joint articulation, permitting tissue longevity over tens of millions of loading cycles over a lifetime. 1 It also serves to withstand and redistribute the compressive, tensile, and shear forces imposed during joint articulation. 2 These functions of cartilage derive from its unique extracellular matrix (ECM) structure and composition. However, the tissue is susceptible to disruption through age-related degeneration and acute trauma, accompanied by poor healing due to the avascular nature of articular cartilage.3,4 Inflammation mediates further cartilage degradation, 5 beginning a positive feedback loop resulting in significant pain, immobility, and emotional distress for patients who develop osteoarthritis (OA). 6
Total knee arthroplasty in younger patients with active lifestyles can lead to high failure rates,7,8 therefore restorative techniques such as mosaicplasty, osteochondral allo/autograft transfer, microfracture, and autologous chondrocyte implantation (ACI) are more commonly used in first-line treatment. Despite pioneering the use of cell therapy for cartilage repair,9–12 there are concerns surrounding the limited biomechanical function prior to regeneration with ACI, as well as cell leakage from the site of implantation 13 and fibrocartilage formation.14–16
Cartilage tissue engineering has the potential to address these limitations and allows the possibility of tailoring the biological, biomechanical, and tribological properties of a scaffold to provide a biocompatible environment that is most suitable for delivery with therapeutic cells or to support endogenous cell repopulation. However, the role of biomechanics in the development and performance of these interventions currently is underrepresented in the literature. In this review, we will outline the complex biomechanical and tribological requirements of articular cartilage and detail the importance of considering these in the design of scaffolds for cartilage repair. We propose that recapitulation of the chondrocyte micromechanical environment and cartilage biomechanical function will result in improved outcomes for patients undergoing ACI and future tissue engineering interventions.
Zonal Structure and Biomechanical Function of Articular Cartilage
Articular cartilage is described as a biphasic material due to the presence of both fluid and solid ECM phases. 17 Interstitial fluid makes up ∼80% (w/w) of the wet weight of the tissue. The solid ECM phase is represented most prevalently by collagen (10–20%) and proteoglycans (5–10%). 18 The tissue is anisotropic, and can be divided into three principal zones, the superficial, middle, and deep zones (Fig. 1). The heterogeneity is present in both density and organization of the collagen fibers and proteoglycan aggregates, as well as in the fluid and cellular composition (Table 1).
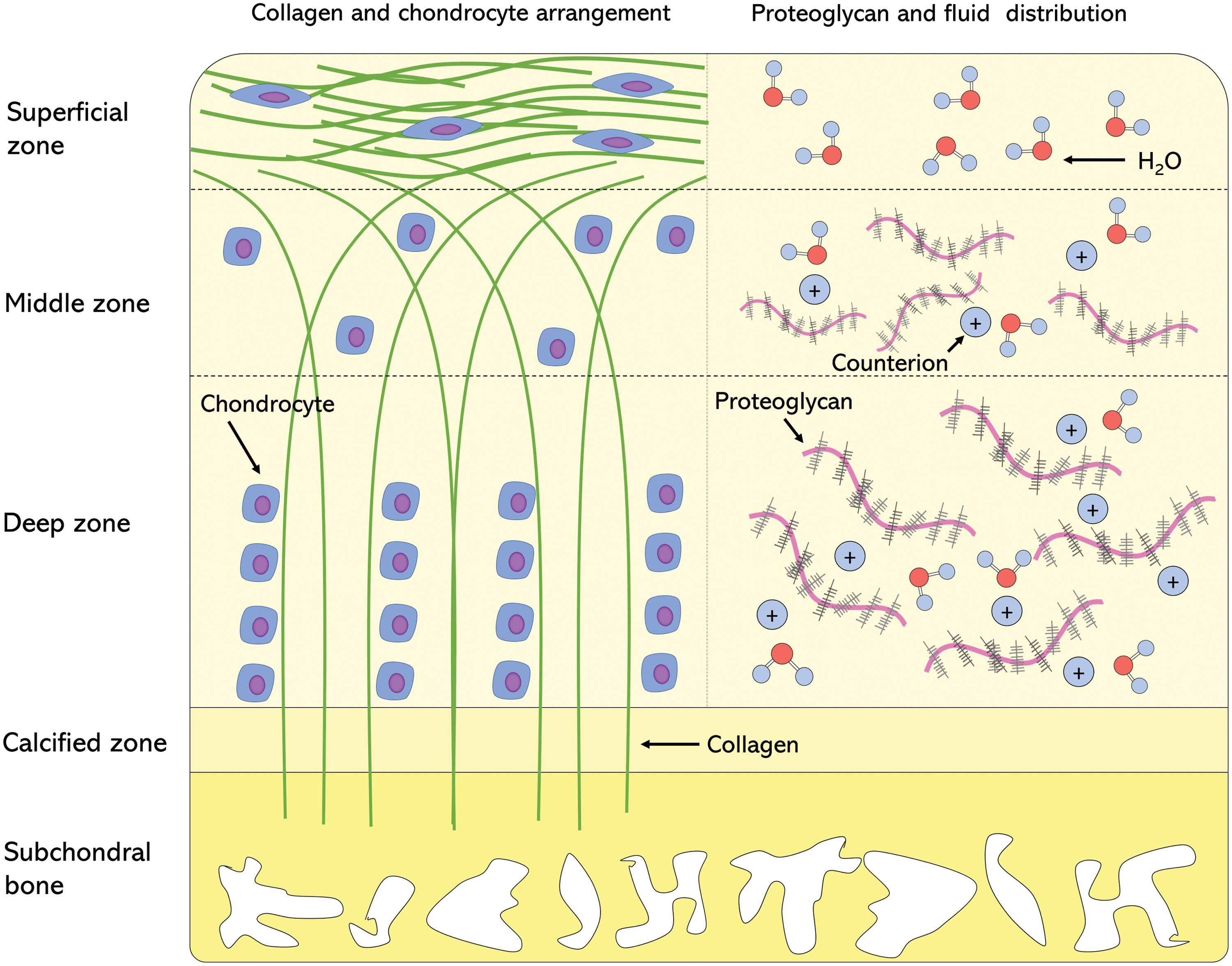
Arrangement of key ECM components and chondrocyte distribution. Left: collagen fiber structure and zone-dependent chondrocyte morphology and arrangement. Right: zonal distribution of proteoglycans, associated counterions, and water molecules. ECM, extracellular matrix. Color images are available online.
Anisotropic Distribution and Function of Extracellular Matrix Components, Interstitial Fluid, and Chondrocytes in Chondrocyte Biological and Biomechanical/Tribological Function
DZ, deep zone; ECM, extracellular matrix; FCD, fixed charge density; GAGs, glycosaminoglycans; MZ, middle zone; PCM, pericellular matrix; PG, proteoglycans; SZ, superficial zone.
This heterogeneic structure of cartilage is intrinsically linked to its function. In the superficial zone, the highly aligned collagen fibers provide a smooth, congruous surface for movement of the knee joint as well as a resistance to the shear and tensile stresses of articulation. 19 The tensile stiffness of this zone has been measured to be fivefold higher compared with the deep zone of cartilage. 20 However, in the deep zone, the perpendicular fiber alignment provides a means of integration between the cartilage ECM and the underlying subchondral bone.20,21 Both functions are a result of the ability of collagen to resist tensile forces in the direction of fiber alignment. This property is also evident in the middle zone, where the alignment and tensile resistance of collagen acts to provide shear resistance, upon deformation in the direction of shear. 4 The stiffness of collagen in tension is not matched in compression, a phenomenon referred to as tension–compression nonlinearity. 33
Proteoglycans and glycosaminoglycans (GAGs) play an important role in the load-bearing capacity of articular cartilage. The density of proteoglycans varies throughout the zones (Table 1). Aggrecan is tightly packed between the collagen fibers, and the close proximity of the negatively charged GAG side chains creates a fixed charge density in the ECM.25,34 To maintain electroneutrality, there is a cation influx, which creates a swelling pressure known as Donnan osmotic pressure. 26 This swelling pressure may have a contribution of 50–60% toward the total tissue stiffness.27,28 On compression, the fixed charge density becomes greater, and hence the permeability decreases, and the osmotic pressure increases. This pressurized interstitial fluid can support up to 90% of the compressive load, 30 reducing the stress on the solid ECM matrix. Due to this contribution of the bulk and physicochemical properties of proteoglycans to the compressive stiffness of articular cartilage, their differential distribution leads to anisotropic compressive properties throughout the tissue. The compressive modulus is lowest in the superficial zone and increases with proteoglycan content into the deep zone of the tissue.25,27
Interstitial fluid support is also important in the low friction and low wear function of cartilage, 2 being critical to the mechanism of biphasic lubrication. 35 Upon loading the interstitial fluid is pressurized and bears most of the load, contributing to low friction at the articulating surfaces. 23 Further lubrication mechanisms also exist, such as boundary and fluid film lubrication, which also enable low friction articulation (reviewed in Ateshian, 23 Katta et al., 36 and Daniel 37 ).
These biphasic and zone-dependent mechanical properties are essential in healthy articular cartilage function and are important to recapitulate in cartilage tissue engineering intervention to restore native joint function.
Biomechanical microenvironment of the chondrocyte
The ECM components provide a scaffold for the resident chondrocyte cells. As described in Table 1, chondrocyte morphology and properties vary throughout articular cartilage, a result of the differential experience of mechanical force, which is accompanied by a zone-dependent metabolic profile with higher GAG production from middle and deep zone chondrocytes. 38 From a cartilage tissue engineering perspective, harnessing and maximizing the chondrocytes' healthy metabolic activity is an imperative. Such anabolic and catabolic activities of chondrocytes are regulated by both the biological and biomechanical stimuli experienced at a macro (tissue) and micro (chondron) level.
As well as the depth-dependent ECM structure, there are also concentric structural differences in the tissue structure surrounding the cell. The interterritorial region is the largest of three matrix regions and describes the region often referred to as the ECM and makes up the greatest proportion of the tissue structure. 39 Closer to the cell, there is a zone of fine collagen fibrils, which form a mesh surrounding the cell called the territorial matrix. It is this layer which is hypothesized to contribute toward the cell's ability to withstand deformation. 40 The innermost zone is the pericellular matrix (PCM), which is a 2–4 μM zone composed of biomolecules, such as collagen VI, collagen IX, perlecan, decorin, biglycan, aggrecan, and hyaluronic acid (HA) 41 (Fig. 2). The PCM is hypothesized to function as a supportive niche for healthy chondrocyte growth, demonstrated by studies showing that isolated chondrons grow larger in pellet culture compared with chondrocytes alone. 42
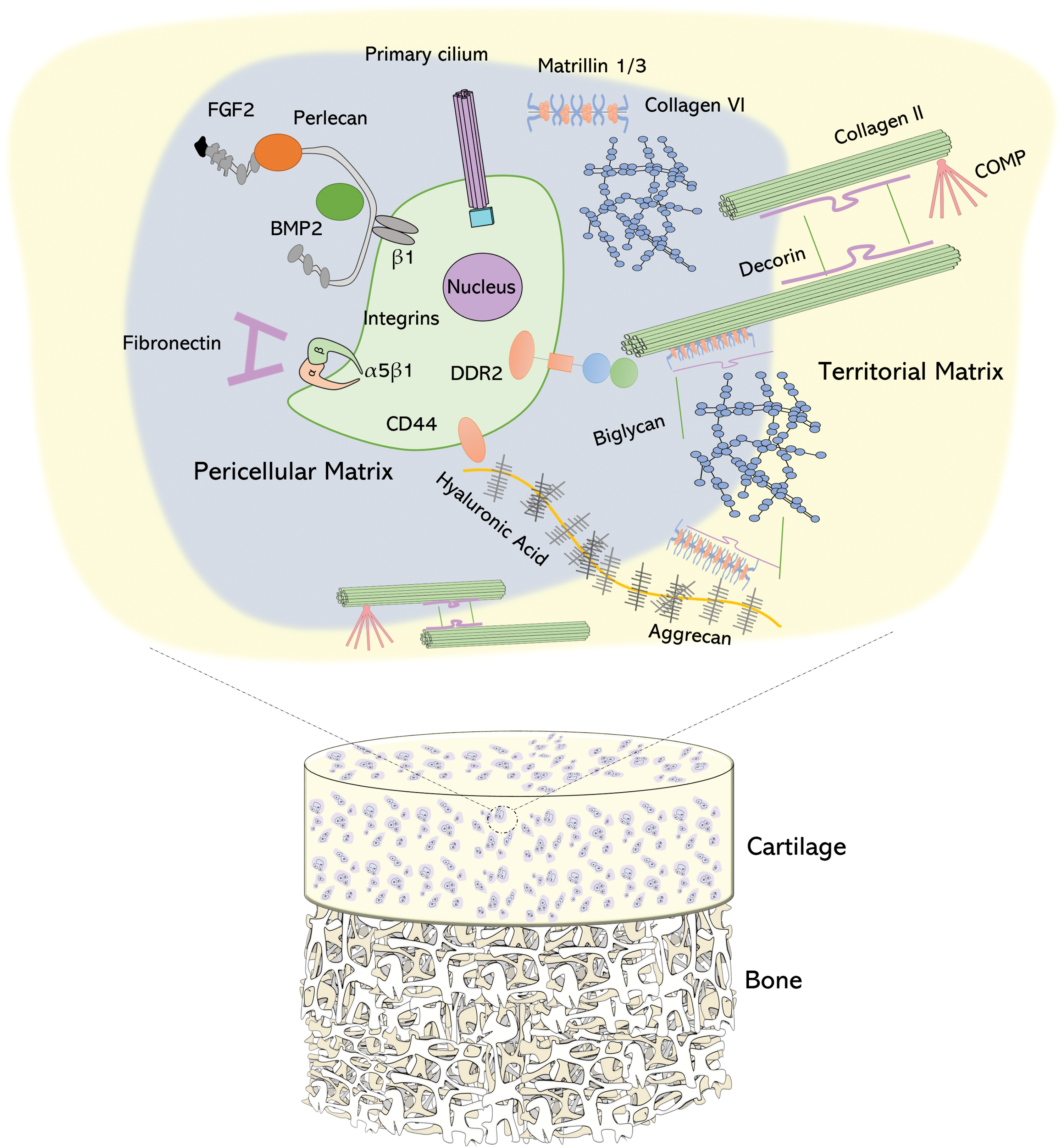
Pericellular matrix arrangement in relation to the osteochondral unit. Magnified view demonstrates the pericellular matrix and territorial matrix components. Collagen II fibrils, linked by decorin/biglycan, from the ECM can interact directly with chondrocytes through DDR2 receptors on the surface. CD44 also links the chondrocyte to hyaluronic acid, which in turn can associate with collagen VI and II through matrilin 1/3 and biglycans. Cell surface integrins also permit interaction with fibronectin and perlecan, which itself acts as a biomolecule repository of FGF-2 and BMP-2. Primary cilia are also located on the cell surface membrane, which deform in response to mechanical loading. BMP, bone morphogenetic protein; CD, cluster of differentiation; DDR2, discoidin domain-containing receptor 2; FGF, fibroblast growth factor. Color images are available online.
As the immediate buffer between the chondrocyte and the ECM, the PCM is hypothesized to both protect and amplify loads experienced by the chondrocyte during loading cycles. This effect is zone dependent, with protection offered in the superficial zone and amplification deeper in the tissue. 43 The chondrocyte has been well documented as a mechanosensitive cell44,45 and it is understood that interaction with the surrounding ECM influences chondrocyte cell signaling pathways and therefore cartilage homeostasis (Fig. 3). The response of the cell to mechanical loads is dependent on the magnitude and frequency of the load, as well as the strain rate and nature (static or dynamic) of the applied load. Strain below 20%, and stress below 18 MPa in vivo are considered physiological. 46 The PCM is thought to play a pivotal role in translating these macromechanical forces experienced on a tissue scale to the chondrocyte, resulting in changes to cell biosynthesis. This process is known as mechanotransduction and is an essential concept in cartilage homeostasis. 47
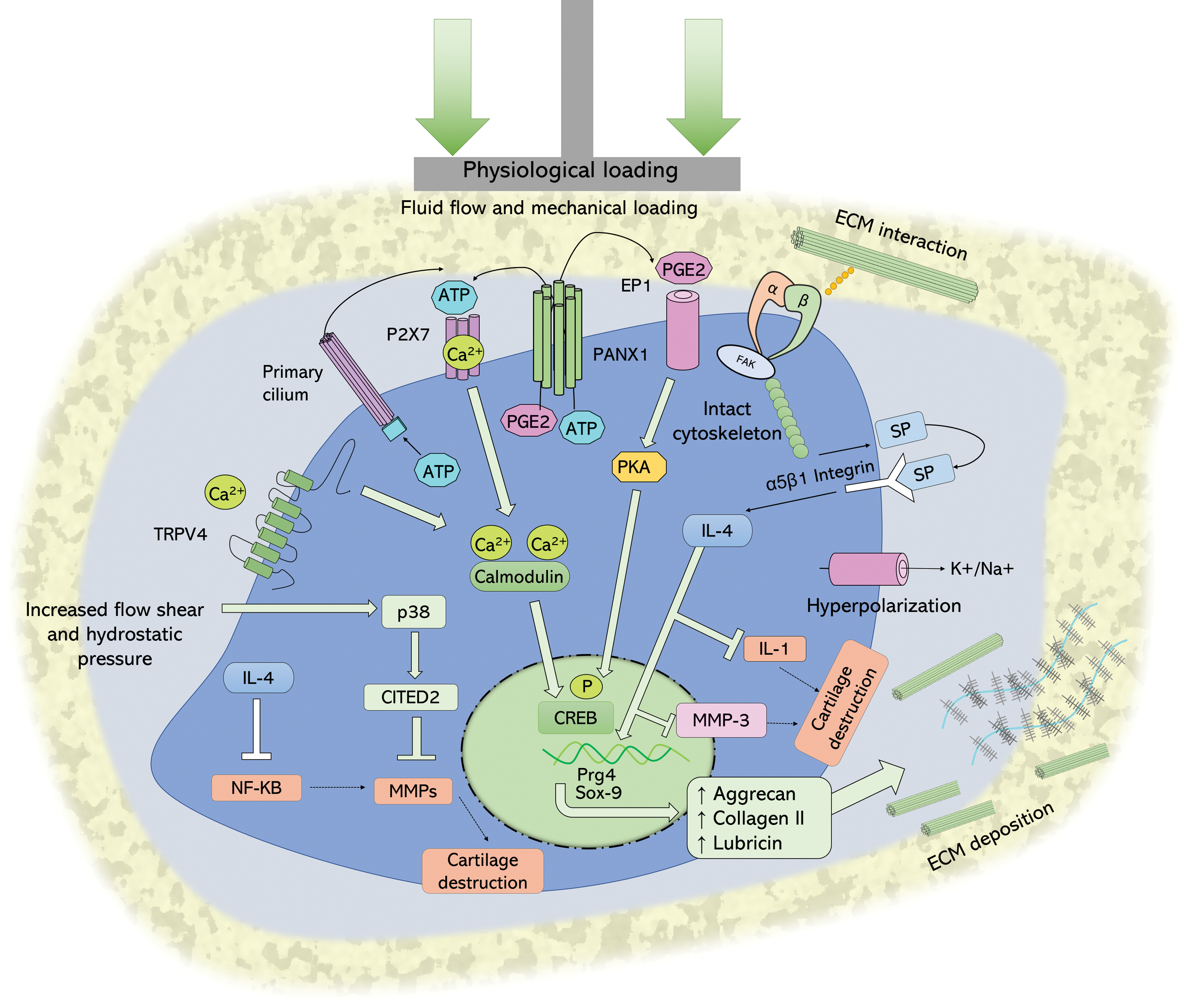
Chondrocyte cell signaling response to physiological mechanical loads. Mechanical stimuli cause interaction of ECM with cell surface integrins, allowing membrane hyperpolarization through cation efflux and interaction with the intact cytoskeleton. The result is SP secretion, which interacts with NK1 channel to activate intracellular IL-4 activity, increasing anabolic aggrecan production and inhibiting IL-1 and expression of MMP-3. Fluid flow promotes efflux of PGE2 and ATP through PANX1 channel. Extracellular ATP can interact with P2X 7 channels to increase intracellular [Ca2+]. PGE2 interacts with EP1 G-protein-coupled receptor, to activate PKA, which in turn phosphorylates CREB on serine-133, allowing for transcription of Pgr4 and lubricin production. Mechanosensitive TRPV4 receptors respond to osmotic pressure to mediate Ca2+ influx, which interact with many downstream signaling pathways, such as calmodulin, CREB to increase transcription of anabolic genes, such as Sox-9, MAPK to activate CITED-2, which can inhibit the NF-κB-mediated MMP production and cartilage destruction. ATP, adenosine triphosphate; CITED, Cbp/p300-interacting transactivator; CREB, cAMP response element-binding protein; IL, interleukin; MAPK, p38 mitogen-activating protein kinase; MMP, matrix metalloproteinase; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; P2X, purinergic type 2X receptor; PANX1, pannexin 1; PGE2, prostaglandin E2; PKA, protein kinase A; Sox, sex-determining region Y-box; SP, substrate P; TRPV4, transient receptor potential vanilloid subtype 4. Color images are available online.
The chondrocyte is anchored to the PCM through surface integrins and collagen VI. Collagen VI is a PCM hallmark biomolecule and has also been shown to tether the chondrocyte to territorial and ECM molecules such as collagen II and hyaluronan through biglycans and decorin. 48 This connection is hypothesized to facilitate mechanotransduction, and the presence of PCM components has been shown to promote chondrocyte responsiveness to static and cyclic loading. 49
The cell surface receptors present on the chondrocyte such as discoidin domain-containing receptor 2 (DDR2) 50 and cluster of differentiation (CD)-44 51 allow direct or indirect interaction with ECM molecules such as collagen II and HA, respectively, facilitating a continuous connection with the extracellular environment.
Deformation of the tissue can be detected by cell primary cilia, which protrude from the chondrocyte into the PCM and act as principal mechanosensors. 52 Transgenic mice studies have shown that without cilia, chondrocytes produce cartilage with significantly reduced mechanical properties. 53 Cilia act as an intermediary between the intracellular Golgi apparatus and the ECM, 54 and under compression, release adenosine triphosphate (ATP) allowing for the activation of ATP-gated Ca2+ channels.55,56 The Ca2+ influx activates the transcription of chondrogenic genes. 53 Deformation is also experienced at a cellular level, which activates stretch-activated cation channels and membrane hyperpolarization. This mechanism has been associated with the activation of anti-inflammatory interleukin (IL)-4, and the subsequent inhibition of matrix metalloproteinase (MMP)-3, as well as increased aggrecan production.44,57,58
Studies have also shown that the laminar and nonpulsatile shear stress applied by fluid flow can also activate chondrocyte signaling through increasing ATP and prostaglandin efflux. Upon interaction of these molecules with cell surface receptors, intracellular protein kinase A (PKA) cell signaling cascades result in the production of lubricin from the superficial zone. 59 The p38 mitogen-activating protein kinase (MAPK) is also activated through fluid flow shear, stimulating Cbp/p300-interacting transactivator (CITED)-2-mediated inhibition of MMP activity. 60 Interstitial fluid pressure has also been shown to activate the mechanosensitive transient receptor potential vanilloid subtype 4 (TRPV4) Ca2+ channel. 45 Activation of this channel with small-molecule treatment in the absence of mechanical loading has been shown to increase aggrecan deposition. 45
The anabolic response of the chondrocyte is partially governed by the mechanical stimuli it receives from the ECM and PCM. To maximize the regenerative response of a tissue engineering intervention, consideration of the extracellular environment and how it will translate stimulation to the cell is an important design consideration.
Functional and metabolic consequences of cartilage damage
Thus far, we have provided an overview of the intricacies of articular cartilage structure and function, as well as the delicately balanced homeostatic condition. Maintenance of this balance favors cartilage longevity, however, age-related changes or mechanical insult can skew this balance toward inappropriate catabolism, beginning a vicious cycle of degradation and loss of function.
Upon mechanical insult, resident chondrocytes secrete IL-1-β. 61 This triggers a cascade of proinflammatory events culminating in the upregulation of MMP expression and the subsequent degradation of damaged cartilage. 62 At this point, chondrocytes begin to express transforming growth factor (TGF)-β to stimulate both chondrogenesis and regeneration of the damaged joint. 63 As articular cartilage is an avascular tissue, chondrocytes have a low turnover rate in terms of ECM secretion. Therefore, if the damage is greater than the chondrocyte's natural capacity to regenerate, the damage will persist, triggering a vicious loop of damage, inflammation, and degradation. Furthermore, the resident chondrocytes can undergo phenotypic switch toward a degradative phenotype, characterized by aberrant expression of catabolic enzymes, such as MMP-13. 64 Studies have suggested that upon damage to cartilage, collagen II exposure is detected by DDR2 domains on the chondrocyte surface triggering an up regulation of Runx-2, which drives chondrocytes into a hypertrophic state, characterized by MMP-13 expression.65,66
Focal cartilage lesions disrupt the tissues' integrity, and the size of the defect affects the outcome of this initial damage. There is a threshold known as a critical defect size, which dictates a defect size which is capable of resolution, above this, however, there are progressive degenerative changes. Focal cartilage defects have been shown to result in increased stress at the rim of the defect, resulting in hyperphysiological loads experienced by the cartilage and chondrocytes in this area. 67 Such damage and/or degeneration is also causative in changes to the bulk mechanical environment such as a reduction of the compressive stiffness,68,69 and both shear and elastic moduli.70,71 Through osteoarthritic mechanisms, cartilage proteoglycans can become depleted, affecting the interstitial fluid pressurization and biphasic lubrication mechanism. 72
There are also alterations to the micromechanical environment of cartilage, specifically in the PCM mechanical properties. Chondrons isolated from human OA cartilage exhibited 40% lower Young's moduli and a two to three times higher permeability compared with chondrons isolated from healthy samples.12,47 These changes in bulk and micromechanical properties increase the exposure of chondrocytes to hyperphysiological loads. 73 Such loads are applied dynamically and acutely, as in an injury setting, and can cause resident cell death and the promotion of inflammation, which triggers increased catabolic activities.74,75
There are several mechanisms by which these loads can trigger catabolic signaling pathways, which are illustrated in Figure 4. Disruption to the cytoskeleton from mechanical trauma is detected by integrin receptors, activating MAPK signaling, and catabolic gene expression of MMPs and A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS). Damaged collagen exposes intracellular domains to DDR2 receptors, activating MMP-13 expression. 76 Hyperphysiological strain (>50%) can also increase membrane tension activating the transport of Ca2+ ions, through Piezo channels into the cell, culminating in chondrocyte cell death through Caspase activity. 77 Strains of 20% have also been shown to decrease primary cilium length, which activates Indian hedgehog (Ihh)-mediated chondrocyte hypertrophy.78,79
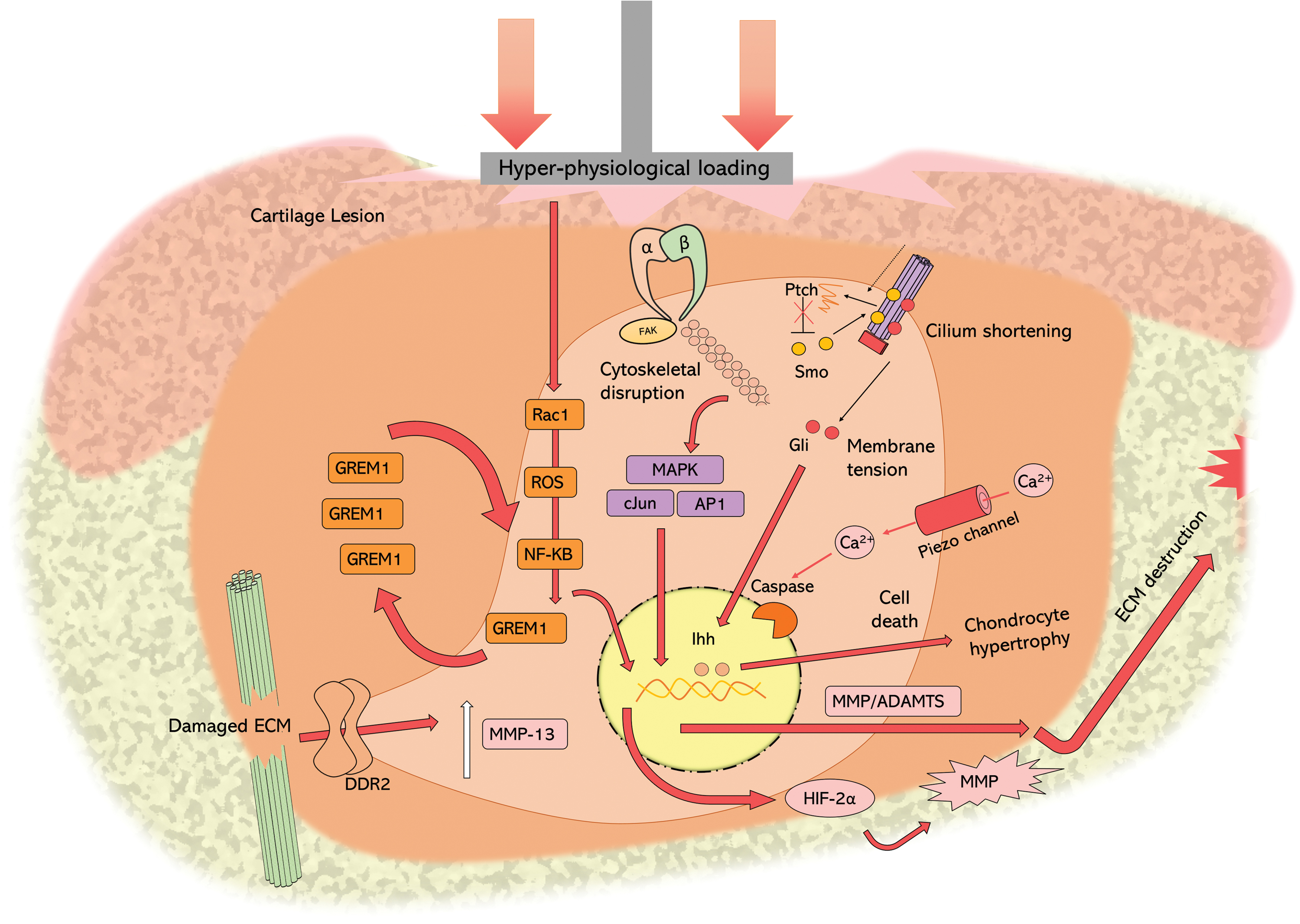
Chondrocyte cell signaling response to hyperphysiological loads. Upon exposure to hyperphysiological loads, integrins interact with the disrupted cytoskeleton, activating MAPK signaling, and catabolic gene expression of MMPs and ADAMTS. This membrane tension activates the transport of Ca2+ ions, through Piezo channels, into the cell which activates Caspase activity, resulting in chondrocyte cell death. High loads decrease primary cilium length through HDAC-6 causing release of Ptch, relieving inhibition of Smo, which can activate Gil to express Ihh and downstream ADAMTS5 and chondrocyte hypertrophy. Rac1 is activated in response to high mechanical loads, which stimulates ROS-mediated activation of NF-κβ, triggering a positive feedback loop with GREM-1, further increasing NF-κβ expression of HIF-2α. High loading also causes damage to the ECM surrounding the chondrocyte. Damaged collagen exposes intracellular domains to DDR2 receptors, activating MMP-13 expression. ADAMTS, A disintegrin and metalloproteinase with thrombospondin motifs; GREM, Gremlin; HDAC, histone deacetylase; HIF, hypoxia-induced factor; Ihh, Indian hedgehog; ROS, reactive oxygen species. Color images are available online.
Finally, loads of 20 MPa have been used in loading regimes as models of hyperphysiological loading. Such magnitudes of stress can stimulate intracellular reactive oxygen species signaling, followed by a nuclear factor kappa-light-chain-enhancer of activated B cell (NF-κβ)-positive feedback loop and expression of hypoxia-induced factor (HIF)-2α and MMPs. 80 It is therefore of paramount importance that any intervention aiming to treat cartilage lesions, acts to restore biomechanical function at a bulk, and micromechanical level to protect chondrocytes, breaking the cycle of catabolic cell signaling to restore a healthy cell phenotype.
It is clear that mechanotransduction is important for cartilage homeostasis and should be considered to deliver successful tissue engineering repair therapies.
Cartilage Tissue Engineering
Despite pioneering the field of cell therapy for tissue repair 81 and reports of long-term success in generating hyaline-like cartilage, ACI in its simplest form disregards many of the elements that contribute to the functional re-engineering of a specialized connective tissue such as articular cartilage. This often results in suboptimal mechanical properties, 82 requiring long postoperative rehabilitation times (10–21 months) until the tissue is of suitable biomechanical strength. 14
Chondrocytes are very sensitive to changes in their microenvironment. Therefore, the removal from this niche and expansion in two-dimensional tissue culture conditions can cause dedifferentiation of chondrocytes, 83 characterized by an increased collagen I expression, which favors fibrocartilage repair, resulting in a repair tissue with inferior tribological and biomechanical properties. 84
Fortunately, dedifferentiation is not only preventable but reversible, through the use of a three-dimensional (3D) scaffold it is possible to redifferentiate chondrocytes back to a chondrogenic phenotype. 85 Cartilage tissue engineering offers the possibility of generating such 3D scaffolds, which can accurately represent the biomechanical and tribological behavior of native articular cartilage.
Table 2 gives an overview of the range of tribological and biomechanical parameters that have been observed in native cartilage and provide a range of values to aim for in the fabrication of tissue-engineered cartilage constructs. However, such properties are merely an example of bulk properties observed, and can vary greatly depending on species, location, zone of cartilage, as well as the test setup. It is also important to emphasize that bulk biomechanical properties are only part of the solution to developing functional cartilage tissue engineering interventions. The in situ scaffold performance is also highly dependent on graft tribological properties, the depth-dependent structure and mechanical properties, as well as the stability of the graft in the site of implantation. Determination of such parameters requires more physiologically relevant testing methodologies (reviewed in Bowland et al. 86 ).
Bulk Biomechanical and Tribological Properties of Human and Animal Articular Cartilage Obtained Through Defined Testing Regimes
The following sections will focus on four aspects of reproducing the biomechanical environment of the chondrocyte: (1) the use of external biomechanical stimuli, (2) the rational design of mechanically relevant scaffolds, (3) engineering chondron structure, and (4) the use of decellularized cartilage scaffolds.
Mechanical stimuli for functional tissue engineering
Mechanical stimuli have been shown to prevent dedifferentiation. Through tuning scaffold mechanical properties Li et al. 96 demonstrated that chondrocytes cultured in a stiffer hydrogel matrix (29.9 kPa vs. 17.1 and 3.8 kPa) were more likely to maintain a chondrogenic phenotype, characterized by increased collagen II and aggrecan expression as well as GAG secretion. Chondrocytes respond to these regions of higher moduli with increased aggrecan deposition. Interestingly, blocking mechanosensation with the small molecule blebbistatin reverses this effect. 97 Research in this area gave rise to the term functional tissue engineering, 98 which encompasses the use of mechanical stimuli into the existing paradigm, 99 recognizing their role in the development of mature tissue-engineered grafts (Table 3).
Chondrocyte Metabolic Response to Various Loading Regimes for Cartilage Tissue Engineering Constructs Seeded with Chondrocytes or Mesenchymal Stem Cells
C, continuous; I, intermittent; MMP, matrix metalloproteinase; mRNA, messenger RNA; TGF, transforming growth factor.
Compressive stimuli are a staple in the maintenance of balanced metabolic activity in chondrocytes and hence the structural integrity of articular cartilage. 111 Moderate exercise regimes that load cartilage within its physiological limit improve GAG content and reduce the risk of developing OA. 112 Conversely, once afflicted with OA, joint distraction has been shown to reduce disease progression. However, this is suspected to be a result of decreasing hyperphysiological loading and OA mediators such as MMP-13. 113
Dynamic in vitro loading regimes, which aim to mimic in vivo loading at frequencies of 0.1–1 Hz, have been shown to increase proteoglycan 114 and cartilage oligomeric matrix protein (COMP) synthesis 115 in cartilage explant models. Kisiday et al. 116 utilized this paradigm in a self-assembling peptide hydrogel model. Through application of a dynamic compression loading regime (2.5% strain and 1.0 Hz frequency), an increased GAG accumulation and accompanied increase in stiffness and equilibrium modulus of the scaffold was observed against the free swelling control. 116 Additionally, as shown in Table 3, a combination of compression and shear can enhance biosynthesis over either stimulus alone. This response to loading can be enhanced through supplementation with growth factors, such as TGF-β and insulin growth factor (IGF)-1, as well as through emulation of the hypoxic environment, which naturally occurs in articular cartilage. 102 However, loading regimes that are either static,109,110 high-velocity impact, 106 or high strain106,108 are shown to have negative effects on ECM deposition and cell viability.
Another key challenge in cartilage tissue engineering is the production of an ECM, which represents skeletally mature tissue. Luo et al. 117 state that many tissue-engineered scaffolds more commonly represent immature cartilage tissue, characterized by a lack of concentric and depth-dependent structure, which ultimately impacts their mechanical performance 118 and the load experienced by the cells. Middendorf et al.119,120 have also indicated that inconsistent aggrecan deposition and distribution can result in construct buckling, and cell death as a result of high local strains. The importance of the presence of a skeletally mature tissue on chondrocyte metabolism has also been shown in bovine cartilage models. When subject to cyclic stress (1–5 MPa), chondrocytes within immature cartilage (4–8 weeks old) were much more susceptible to injury compared with chondrocytes embedded in mature cartilage (1.5–2 years old). The authors observed less-intense collagen VI staining as well as the presence of flattened chondrocytes in the immature samples, indicating a role of the skeletally mature structure as well as the PCM in the protection of chondrocytes from injury inducing mechanical loads. Although beyond the scope of this review, Khan et al. 121 have shown that it is possible to biochemically induce the maturation of immature cartilage, defined by maturation-dependent changes in stiffness, collagen alignment, and the presence of distinct concentric territorial, interterritorial, and pericellular domains, through the use of fibroblast growth factor (FGF)-2 and TGF-β.
The knowledge of how loading regimes can affect chondrocyte metabolism and construct maturation, allows for the use of tailored regimes to produce constructs with more appropriate biomechanical and tribological properties. For example, Mauck et al. 122 achieved a sixfold increase in the equilibrium aggregate modulus (from 0.015 to 0.1 MPa) when loading bovine chondrocytes within an agarose hydrogel construct, utilizing an intermittent (1 h on/off) loading cycle at 1 Hz for 28 days. This demonstrates the importance of utilizing a pause in compressive dynamic loading regimes to enhance de novo cartilage synthesis. 123 Furthermore, using static hydrostatic pressure (10 MPa) alone, Elder and Athanasiou 124 improved the aggregate modulus of bovine chondrocyte-seeded agarose constructs up to 0.248 Mpa.
While the majority of research focuses on compression, loading regimes have been investigated, which can enhance the tensile properties of constructs, through improvements to the ultimate tensile strength (UTS). Lee et al. 125 demonstrated, using a tension stimulation device to impart 15% tensile strain for 1 h a day over to a course of days 10–14 during a 28-day culture cycle, that it is possible to achieve an UTS of 3.3 MPa when used in conjunction with TGF-β and lysyl oxidase-like protein 2 with copper sulfate and hydroxylysine (LOXL2). This value was 3.3 times higher than the untreated control value. Furthermore, loading regimes can also encourage improvements in the tribological properties of cartilage. Through application of a combination of dynamic compression and sliding motion, Bian et al. 126 engineered an agarose hydrogel scaffold seeded with chondrocytes to yield an equilibrium friction coefficient of ∼0.08, which was significantly lower than the constructs loading under compression alone.
Engineering biomechanically functional constructs
In recognition of the need for a biomechanically functional environment as well as structural support for chondrocytes, the ACI technique has evolved into a matrix-assisted chondrocyte implantation (MACI). This is referred to as third-generation ACI, whereby chondrocytes (0.5–1 × 106 cells/cm2) are seeded into a collagen I/III matrix before implantation. 127 In a large cohort (827 patients) improved results with MACI versus ACI were shown. 128 Furthermore, from a clinical perspective, MACI offers an implantation time 20 min faster than ACI 129 and an accelerated rehabilitation program of just 6 weeks has been reported. 130
Despite promising indications, there are still key limitations in the mechanical strength of the repair material. Compression testing of MACI-repaired cartilage only achieved 15% of the aggregate modulus of native tissue in a canine model. 131 Furthermore, an equine model demonstrated no difference in mechanical properties in repair tissue from an empty defect versus a MACI implant-treated defect. 132 This is common in tissue-engineered grafts due to the lower concentrations and different distributions of both collagen and aggrecan, resulting in local tissue weakness and construct buckling. 120 Interestingly, Griffin et al. 133 report aggregate moduli up to 70% of the native tissue. However, they suspect this is due to the use of a long-term implant and a large animal model.
Beyond this, there is a 4th generation of MACI, which encompasses more innovative approaches such as the implantation of allogeneic mesenchymal stem cells (MSCs) with intact whole chondrons. The key benefit being the retained presence of the PCM, which has been shown to cause greater production of proteoglycans and collagen II. 42
The use of synthetic materials offers the possibility of finely tuning mechanical properties of a scaffold. McCullen et al. 134 used electrospinning to create a scaffold with the same zonal fiber orientation as articular cartilage using electrospun poly(ɛ-caprolactone) (PCL). They achieved an increased tensile modulus compared with articular cartilage and a comparable compressive modulus at 10% strain. 134 The use of aligned fibers was found to improve the resistance to tension and damage arising from shear when tested in a multiaxial testing rig. 135 Using a polyethylene terephthalate (PET) scaffold, another group achieved compressive moduli values of 2.8 MPa at 15% strain with compressive culture with chondrocytes over an 84-day period, which was only slightly higher than reported native value of 1.8 MPa. 136 While only one article demonstrates any tribological analysis and neither of these articles details in vivo performance data, both show potential strategies of replicating the native mechanical properties of cartilage with a biocompatible material.
Despite this potential replication of biomechanical properties, there are several problems frequently observed with synthetic biomaterial scaffolds for tissue engineering applications. Many lack natural binding motifs and hydration, are hydrophobic, and often fail to precisely recapitulate the bioarchitecture and biphasic properties found in cartilage, which the chondrocytes are sensitive to.
The biocompatibility of synthetic scaffolds can be improved through hybrid fabrication methods. Owida et al. 137 incorporated polylactic acid (PLA) nanofiber mesh within HA hydrogels. The fiber alignment matched native cartilage, and as a result, chondrocytes exhibited zone-dependent morphology and distribution, and the repair tissue had higher GAG content in the deeper zones accompanied by a higher compressive modulus. 137 Nguyen et al. 138 used poly(ethylene glycol) (PEG)-based hydrogels with incorporated chondroitin sulfate in the top and middle zones, and PEG:HA in the deep zones. MSCs cultured within the matrices deposited collagen II in the superficial zone and collagen X and proteoglycan in the deep zone, resulting in respective compressive moduli of 0.5 and 1.7 MPa 138 which were consistent with their literature reference for native cartilage. 139
Despite a simplified cartilage structure, Wang et al.
140
fabricated a tricomposite osteochondral scaffold from poly(
Means et al. 142 developed a “double network hydrogel” scaffold composed of a poly(2-acrylamido-2-methylpropanesulfonic acid) (PAMPS) network and poly(N-isopropylacrylamide-co-acrylamide) [P(NIPAAm-co-AAm)]. The scaffold exhibited a low coefficient of friction, a compressive modulus >1 MPa, a compressive strength of 25 MPa and demonstrated a similar creep response profile to native cartilage. The group showed cytocompatibility compared with the tissue culture plastic control but are yet to investigate whether cells can be successfully encapsulated. However, this study demonstrates the potential of successfully recapitulating multiple biomechanical and tribological properties for restoration of cartilage function.
Engineering the chondron
The presence of PCM has been shown to be beneficial for chondrocytes in terms of mechanocoupling and in promoting the deposition of cartilage matrix molecules. 143 One approach to engineer constructs containing chondrons, is using scaffold-free cartilage constructs, using the technique of self-assembly of chondrocytes within nonadherent agarose wells. 144 Jeon et al. 143 also have demonstrated that culturing chondrocytes for 2 weeks before further treatment with compression and saw an increase in collagen VI production and the formation of chondron structures within the agarose gels.
It has been shown that scaffold morphology has an influence over the formation of chondron structures. Using a HA polymer (HYAFF11), Fraser et al. 145 demonstrated that bovine chondrocytes seeded onto a nonwoven scaffold formed as chondron-like collagen VI structures in the neotissue, whereas those onto a sponge scaffold lacked distinct chondron formation.
Stoddart et al. 123 have shown that in response to compressive loading with pauses, distinct chondron-like structures are formed that integrate with the surrounding agarose hydrogel. Steward et al. 146 demonstrated chondrogenesis of MSCs seeded into agarose hydrogels and found that MSCs produced a denser and stiffer PCM in the presence of a stiffer hydrogel. Furthermore, gene expression analysis of chondrocytes cultured in a 3D collagen sponge demonstrated higher expression of the PCM component matrilin in response to cyclic loading. 147
Few studies have investigated the production of PCM biomolecules or structures through mechanical loading regimes. This may be a result of multiple studies demonstrating PCM production in tissue-engineered scaffold without the requirement for loading.143,148 Interestingly, the aforementioned study by Jeon et al. 143 also observed that following the 2-week preculture period, which allowed for the production of PCM structure in their construct, increased the cellular response to mechanical loading. This suggests that recapitulating the PCM biomechanical niche may be key to successful tissue engineering, and also elucidates a potential strategy to optimize preimplantation loading regimes for tissue-engineered constructs.
Overall, there appears a disparity in the literature between research advocating the benefits of an intact PCM in cartilage tissue engineering and the rational design of scaffolds with chondron-like structure or with a structure primed for the formation of such structures. 123
Decellularized cartilage tissue scaffolds
So far, this review has indicated a variety of strategies for engineering functional cartilage constructs. One approach, which encompasses all of these elements (native composition, biomechanical properties, and depth-dependent/concentric ECM structure) is decellularization, a process that can remove cells from a range of human and xenogeneic biological tissues while preserving the macro and microstructure of the native tissue, as well as the ECM composition, 149 producing a fully functional regenerative tissue scaffold. These tissues have been shown to induce favorable cell responses such as chemotaxis, differentiation and induce tissue regeneration (reviewed in: Badylak 150 ). This technique has been applied successfully to generate decellularized cartilage and osteochondral scaffolds.151–153 These scaffolds have taken many forms summarized below in Table 4.
Summary of Methods for Producing Decellularized/Acellular Cartilage Scaffolds from Both Human and Xenogeneic Cartilage Sources
ASCs, adipose stem cells; BMSCs, bone marrow mesenchymal stem cells; MSCs, mesenchymal stem cells; PBS, phosphate-buffered saline; SDS, sodium dodecyl sulfate.
The biological response to decellularized cartilage has been extensively shown, using animal models. Li et al. 157 report significant improvement in macroscopic ICRS and histological O'Driscoll scores, accompanied by the integration of the scaffold with the surrounding tissue and a lack of collagen I expression in the neocartilage. Xue et al. 161 showed similar repair after 6 months in vivo implantation of acellular cartilage sheets seeded with bone marrow mesenchymal stem cells. Histology showed good integration at the cartilage interface and lack of fibrosis in the repair tissue, and mechanical testing saw Young's moduli and maximum compressive strength of regenerated cartilage approaching that of native cartilage. Due to the retention of native ECM structure, as well as chondrogenic growth factors, such as TGF-β, IGF-1, and bone morphogenetic protein (BMP)-2, these scaffolds have been shown to direct chondrogenesis. 162
A key observation in the performance of decellularized grafts is the improved histological and functional outcomes when seeded with cells versus scaffold-alone controls. This is likely due to the immobility of chondrocytes and the low porosity of cartilage, resulting in low endogenous cell migration into the scaffold. Furthermore, cartilage is an avascular tissue with low cell density, 4 therefore acellular grafts without either large porosity or biomolecular homing signals are unlikely to undergo mass endogenous cell repopulation.
The structural composition of decellularized cartilage, in terms of collagen alignment and composition has been shown to match that of the native tissue. 117 However, initially postdecellularization, there is a reduction in GAG content, which translates in increases in deformation 152 and decreases in Young's moduli under compression compared with native cartilage. 157 Despite the reduction from native values, instances where the compressive modulus is measured, values above 1 MPa are reported. 163 However, several studies show the recovery of both GAG content and mechanical properties postrecellularization. Li et al. 157 report that decellularized scaffolds seeded with autologous chondrocytes recover the native Young's modulus and increase DNA and GAG content with respect to the native controls after 8 weeks of in vivo implantation in a rabbit defect model. Using decellularized cartilage microfilaments, Kang et al. 164 created a lyophilized ECM-derived scaffold, showed a similar recovery up to 83% of the stiffness of native cartilage after 6 months in vivo implantation.
A recent study by Luo et al., 117 exemplifies many of the strategies suggested in this review. Mature and immature cartilage was decellularized and seeded with fat pad-derived stem cells for 4 weeks. The regenerated tissue mimicked the zonal collagen arrangement of the surrounding decellularized scaffold, and displayed depth-dependent compressive moduli in the mature tissue but not the immature tissue. 117 This study provides an example of how scaffolds designed to accurately recreate tissue structure can influence cell behavior toward appropriate neotissue formation with depth-dependent mechanical properties.
Conclusion
This review has aimed to emphasize how the complex heterogeneic structure and anisotropic properties of cartilage governs tissue biomechanical and tribological function in vivo, and how these functions are much more complex than simply an ability to withstand compression.
As well as permitting healthy joint function, appropriate biomechanical properties are essential for healthy cellular function. As the intermediate material between applied loads and the cells, the ECM and PCM, directly influence how the chondrocyte experiences the load and the response. 47 Harnessing these concepts in the scaffold design, alongside application of the correct mechanical and biological cues should yield an environment primed for graft maturation and the deposition of functional cartilage to restore joint function.
Decellularized cartilage tissues offer an ideal scaffold for MACI, due to the retention of much of the skeletally mature cartilage macro/microstructure and biochemical cues to promote healthy homeostasis. The provision of an immediate restoration of functional performance may allow for faster rehabilitation times for patients and improve long-term graft performance. 165
Footnotes
Disclosure Statement
No competing financial interests exist.
Funding Information
P.S. holds a Center of Doctoral Training in Tissue Engineering and Regenerative Medicine studentship, funded by grant EP/L014823/1 from the Engineering and Physical Sciences Research Council.
