Abstract
Background and Aims:
Cell therapy approaches to treating chronic liver disease provide only transient improvements, mainly due to loss of hepatocytes after infusion. Microencapsulation in alginate has been shown to protect transplanted cells from physical stress and rejection, but the poor biocompatibility of alginate can lead to graft failure. This study aimed to evaluate a biocompatible poly(vinyl alcohol) (PVA)-based microcapsule against standard alginate for improved transplantation outcome of liver spheroids.
Materials and Methods:
Human hepatocyte spheroids were microencapsulated in alginate or PVA hydrogel microspheres. Viability and function (albumin secretion and CYP activity) of the encapsulated spheroids were assessed in vitro at 3, 10, and 30 days postencapsulation and compared with unencapsulated spheroids. Spheroids were implanted intraperitoneally into immunodeficient mice, and human albumin levels in serum were monitored over 30 days. Cell-free microspheres were implanted in immune-competent mice to assess material biocompatibility.
Results:
Unencapsulated spheroids aggregated extensively beyond 10 days, precluding day 30 assessment. At day 30, PVA spheroids showed significantly higher CYP1A1 induction, albumin secretion, and metabolic activity compared with alginate. Mice receiving PVA spheroids had significantly higher serum albumin after 30 days compared with alginate and unencapsulated spheroids. Empty PVA microspheres showed less evidence of foreign body response in vivo, whereas thicker regions of inflamed tissue were observed in the alginate group.
Conclusions:
PVA-encapsulated hepatocyte spheroids maintained better overall viability, metabolic activity, and function compared with alginate-encapsulated cells both in vitro and in vivo. Both encapsulated groups demonstrated substantially improved outcomes compared with unencapsulated cells.
Impact Statement
This work utilizes the use of a novel microsphere production method that utilizes poly(vinyl alcohol) (PVA) to encapsulate human hepatocyte liver spheroids. The work compares the functionality of liver spheroids encapsulated in PVA, both in vitro and in vivo, and compares it with commonly used alginate. The work presented shows enhanced functionality of cells encapsulated in PVA and opens the door for further clinical study using the material for transplants of cells to treat liver disease.
Introduction
Liver disease comes in many highly prevalent forms, including cirrhosis, steatohepatitis due to nonalcoholic liver disease, and infections by various virus strains, including hepatitis B and C. 1 Nonalcoholic fatty liver disease is a major cause of liver dysfunction and is estimated to have a global prevalence of 25% of the population with an overall mortality of 11.77 per 1000 person-years. 2 In fact, liver disease ranks as the 11th highest cause of global mortality, 3 and the prevalence is expected to expand with increased obesity worldwide.
Although liver transplantation has been used as a primary form of treatment for chronic liver disease, there are various obstacles that make it necessary to explore alternative treatments including cell therapies. 4 Initial studies on cell therapies for liver disease used primary hepatocytes as the cell source and were based on the concept of cell “homing,” assuming that an infusion of cells remained in the liver and functioned correctly. In fact, multiple studies have shown a lack of functioning cells retained even when injected directly into the liver. 5 It is estimated that within the first month after the infusion of hepatocytes, 70% of the injected cells are lost. Much of this loss is attributed to the immediate blood-mediated immune reaction and phagocytosis of the injected hepatocytes, even when the recipients are systemically immunosuppressed.6,7 The clinical result is a transient improvement in blood profile with no durable effect.5,8 Only one clinical trial report using bone marrow stem cells showed a durable improvement past 30-month postimplantation. 9 In addition, there has been some success in placing the cells in lymph nodes with a recent seminal clinical trial initiated.10,11
Providing the cells with an optimized environment using scaffolds may protect them from immune attack and improve the clinical outcome. Some commonly used, naturally occurring scaffold materials include fibrin, alginate, and Matrigel, whereas synthetic materials include poly(vinyl alcohol) (PVA) and polyethylene glycol (PEG). Animal studies with alginate-encapsulated cells show mixed results, with some successful protection of the cells and transient optimization of their function,12,13 whereas others identified strong immune responses with pericapsular fibrous overgrowth, leading to cell necrosis. 14 In a human clinical trial, alginate induced a strong foreign body response (FBR) that resulted in the U.S. Food and Drug Administration placing the trial on hold. 15 However, even with mixed results, alginate is still the most widely utilized cell encapsulant.16–18 This is partially due to the ease of manufacturing because alginate cures rapidly with exposure to calcium, whereas other hydrogels may take minutes to cure.19–21 Because of this limitation, the traditional manufacturing method of droplet generation is not appropriate for nonalginate hydrogel polymers.
In contrast to alginate, biocompatible polymers, such as PVA, PEG, and hyaluronic acid, can be used as coatings for microencapsulation of cells.20,21 Such materials do not elicit an FBR, commonly noted with alginate, and can be produced in a durable microencapsulation format. PVA is of particular interest because it is highly biocompatible and has a high water content.22–24 Since the 1980s, PVA has been used successfully as an embolic agent 25 and later was developed as microspheres for the delivery of drugs and cells.26–28
In the current report, we compared the encapsulation of liver spheroids in alginate with PVA. First, we examined the vitro viability, metabolism, and function of unencapsulated cells compared with cells encapsulated in alginate or PVA microspheres. The results demonstrate that cell metabolic levels were statistically higher in PVA compared with alginate; albumin secretion was greater in PVA compared with alginate at 3, 10, and 30 days following encapsulation; and CYP activity was equal to or better in PVA compared with alginate. Furthermore, in vivo data in mice illustrated a greater FBR and loss of function of alginate-encapsulated spheroids, which was avoided with PVA. Our results show that encapsulation in PVA results in better functional outcomes and provides the basis for future clinical trials.
Materials and Methods
Spheroid formation and culture
Cryopreserved human hepatocytes (HepaRG® cell line) obtained from BioPredic International (Differentiated HepaRG Cells Cat No. HPR116) were thawed and plated into AggreWell® plates (StemCell Technologies, Vancouver, BC) at 305 cells per microwell and a final volume of 1 mL per well. The plate was centrifuged at 1300 RCF for 5 min and incubated at 37°C with 5% carbon dioxide (CO2), allowing the cells to self-aggregate. After 5–6 days, the spheroids were collected, transferred into bulk culture in anti-adherence-treated petri dishes (Nunc, Thermo Fisher Scientific, Waltham, MA) at a density of 1200–2500 spheroids/mL, and returned to incubation in fresh medium. After 24–48 h, aggregated spheroids were ready for either encapsulation or long-term culture, which consisted of incubation in standard conditions with media changes every other day, using HepaRG+ culture medium (100 mL William Medium E [Gibco No. A12176-01], 1 mL Glutagro supplement [Corning No. 25-015-Cl], 14 mL HepaRG Thawing Supplement [Biopredic International No. ADD670], and 578 µL 10% Pluronic® F-68).
Encapsulation of hepatocyte spheroids
Encapsulation of hepatocyte spheroids was accomplished in either alginate or PVA using previously published methods. For alginate microencapsulation, a 1.5% alginate solution was mixed with HepaRG spheroids at ∼20,000 spheroids/mL. 29 The mixture was extruded dropwise into a calcium chloride bath using a commercial droplet generator (Buchi Corporation, New Castle, DE). Once the droplets entered the bath, calcium in the bath diffused into the droplets, resulting in crosslinked alginate microspheres containing hepatocyte spheroids.
Encapsulation of hepatocyte spheroids in PVA utilized Likarda’s proprietary Core-Shell Spherification technology.20,30 Briefly, a custom acrylamide functionalized PVA hydrogel precursor solution containing calcium chloride, 4-(2-hydroxyethyl)-1-piperasineethanesulfonic acid (HEPES) buffer, and Lithium Phenyl-2,4,6-trimethylbenzoylphosphinate (LAP; a photoinitiator) was mixed with the hepatocyte spheroids at ∼20,000 spheroids/mL. The mixture was extruded dropwise into an alginate bath using Likarda’s proprietary Core Shell Spherification (CSS) instrument. Once the droplets entered the bath, calcium reacted with the alginate, resulting in spherical uncrosslinked core- and crosslinked alginate-shell constructs. The core was crosslinked by activation of the photoinitiator with ultraviolet light within the CSS instrument. Subsequently, the crosslinked alginate shell surrounding the polymerized core was dissolved with citrate, leaving PVA hydrogel microspheres containing hepatocyte spheroids. Encapsulated spheroids were imaged using brightfield microscopy and calcein AM staining (2 µM).
Hepatocyte viability
The viability of hepatocyte spheroids was measured using multiplexed dye staining conducted on six replicates of 40–75 spheroids over a 2-h incubation at 37°C with 5% CO2, using our published protocol.
31
Viability calculations of the spheroids were calculated after exposure to ethidium homodimer-1 (2 µM) and Hoechst 33342 (10 µM). The Hoechst nuclear stain verified the location and border of the spheroids in the brightfield images. Following rinsing, the spheroids were imaged at 4× or 10× magnification, in brightfield and fluorescent channels, using a Biotek Cytation 5 Imaging Reader. Excitation/emission wavelengths used were 377/447 (blue) and 586/647 (red). Viability results were calculated for each spheroid as follows:
Hepatocyte metabolic activity
Metabolism was monitored using PrestoBlue (10% v/v). During incubation, the spheroids were imaged in brightfield at 4× magnification using a Biotek Cytation 5 Imaging Reader to count the number of spheroids in each replicate for normalization of the PrestoBlue fluorescence signal. After the 2-h incubation period, the PrestoBlue fluorescent signal was read, and the reagent was rinsed from the spheroid suspensions with Dulbecco’s phosphate-buffered saline solution. Metabolic activity was reported as relative fluorescence units (RFU) per spheroid per minute.
Secretion of albumin
Albumin secretion from HepaRG spheroids was quantified using an Albuwell Hu Enzyme-Linked Immunosorbent Assay (ELISA) kit (Ethos Biosciences, Logan Township, NJ). Six replicates of 40–75 spheroids were incubated in 200 µL fresh culture medium for 24 h at 37°C with 5% CO2. Samples of supernatant were removed from each replicate, transferred into low protein binding microcentrifuge tubes, and frozen at −80°C until testing, which was performed according to the manufacturer’s instructions. Samples were diluted 1:1 with Bovein Serum Albumin plus NHE inhibitor diluent. Albumin secretion levels were normalized to spheroid number and reported as picograms of albumin per spheroid per hour.
Basal and activated CYP activity
Basal and induced activities of hepatocyte spheroids were quantified using P450-Glo CYP1A1 and P450-Glo CYP3A4 Assay kits Promega Corporation, (Madison, WI, USA). CYP1A1 induction was accomplished by incubating in omeprazole (10 µM) for 24 h. 32 CYP3A4 induction was accomplished by incubating the spheroids in rifampicin (10 µM) for 24 h. 33 P450-Glo luminescence for both Cytochrome 450 (CYP) enzyme assays was measured using a Biotek Cytation 5 imaging plate reader. The relative luminometer units (RLU) were normalized to spheroid number and fold induction of each enzyme, calculated as the ratio of RLU/spheroid of induced to uninduced spheroids.
In vivo albumin secretion
Immune-compromised mice obtained from Harlan labs were randomized into three groups: unencapsulated spheroid, alginate-encapsulated spheroid, and PVA-encapsulated spheroid (four mice per group). Mouse studies were conducted at the University of Kansas Medical Center’s Laboratory Animal Facility under the Animal Protocol 2021-2611. Mice were injected intraperitoneally (IP) using an aseptic technique. Briefly, mice were anesthetized using isoflurane, and the lower quadrant of the abdomen was aseptically prepared. Alginate- or PVA-encapsulated and unencapsulated spheroids were injected into the intraperitoneal cavity using a 16G catheter needle attached to a 5 mL syringe, with each animal receiving approximately 5 M HepaRG cells (∼16.3 k spheroids). The injection site was sealed using tissue glue, and mice were placed on a warm bed until recovery. After transplantation, mice were monitored for general health and weight for 60 days. Blood draws for human albumin were conducted before transplantation and again at days 3, 10, and 30 posttransplantation. Pretransplant human albumin levels in the mouse serum were subtracted from posttransplant levels to account for possible cross-reactivity of mouse albumin with the human ELISA assay, using the manufacturer’s instructions. Sixty days after transplantation, the animals were euthanized, and the tissues were sectioned and stained for histological analysis. This time point was chosen to match the same length of time for testing of biocompatibility in the immune-competent animals described below.
Biocompatibility
Immune-intact C57/BL mice were implanted IP with ∼1.5 mL (loosely packed volume in Dulbecco's Phosphate Buffered Saline) empty microspheres composed of either alginate or PVA using the methods described above. At necropsy (60 days after implantation), the IP cavity was examined for the presence and distribution of the microspheres, and tissue samples were collected for histology analyses. Sixty days mark an important time point after material implantation when the chronic phase of Foreign Body Response (FBR) is active. 34
For histological analysis, various abdominal tissues including the omentum/gastrointestinal tissues, urogenital tissues, and muscles were collected. Tissues were fixed in 10% nonbuffered formalin for 24 h and then stored in Phosphate Buffered Saline. Tissues were processed and stained for H&E at the University of Kansas Histology Core Lab using routine histological methods. Histological images were captured using the BioTek imaging software (Cytation 5 Imaging Reader, Agilent, Santa Clara, CA, USA). Analysis of the images was completed using previously published methods to quantify the thickness of the fibrotic capsule around microspheres and the level of inflammation in the surrounding tissue. 21
Statistical analysis
Statistical analysis, including one-way analysis of variance (ANOVA) and repeated-measures ANOVA, was performed on data using the SigmaPlot 13.0 software when appropriate. Dunn’s test was used to make pairwise comparisons. For analysis of the levels of fibrosis and inflammation in the tissue samples, hierarchical statistical analysis was performed with levels defined as hydrogel groups, animals, and tissue sections. Statistical significance was defined as p < 0.05. Error bars, where shown, denote the standard error of the mean.
Results
Effect of encapsulation on hepatocyte spheroid viability
Hepatocytes (HepaRG cells) spontaneously form large clusters referred to as spheroids. As early as 2 days into the self-assembly process, the hepatocytes aggregated into multispheroid clusters, termed mega-aggregates. Figure 1A illustrates the type of mega-aggregates that formed after 3 days in culture. The formation of mega-aggregates resulted in significant cell death in the core of the aggregates that were not present in small spheroids as shown by the calcein/ethidium staining in Figure 1B. In contrast to the formation of mega-aggregates in the unencapsulated cells, when the spheroids were encapsulated on day 0 of the study, the formation of mega-aggregates was avoided, as shown in Figure 1C and D. Alginate-encapsulated cells are shown in Figure 1C, and PVA-encapsulated cells are shown in Figure 1D.
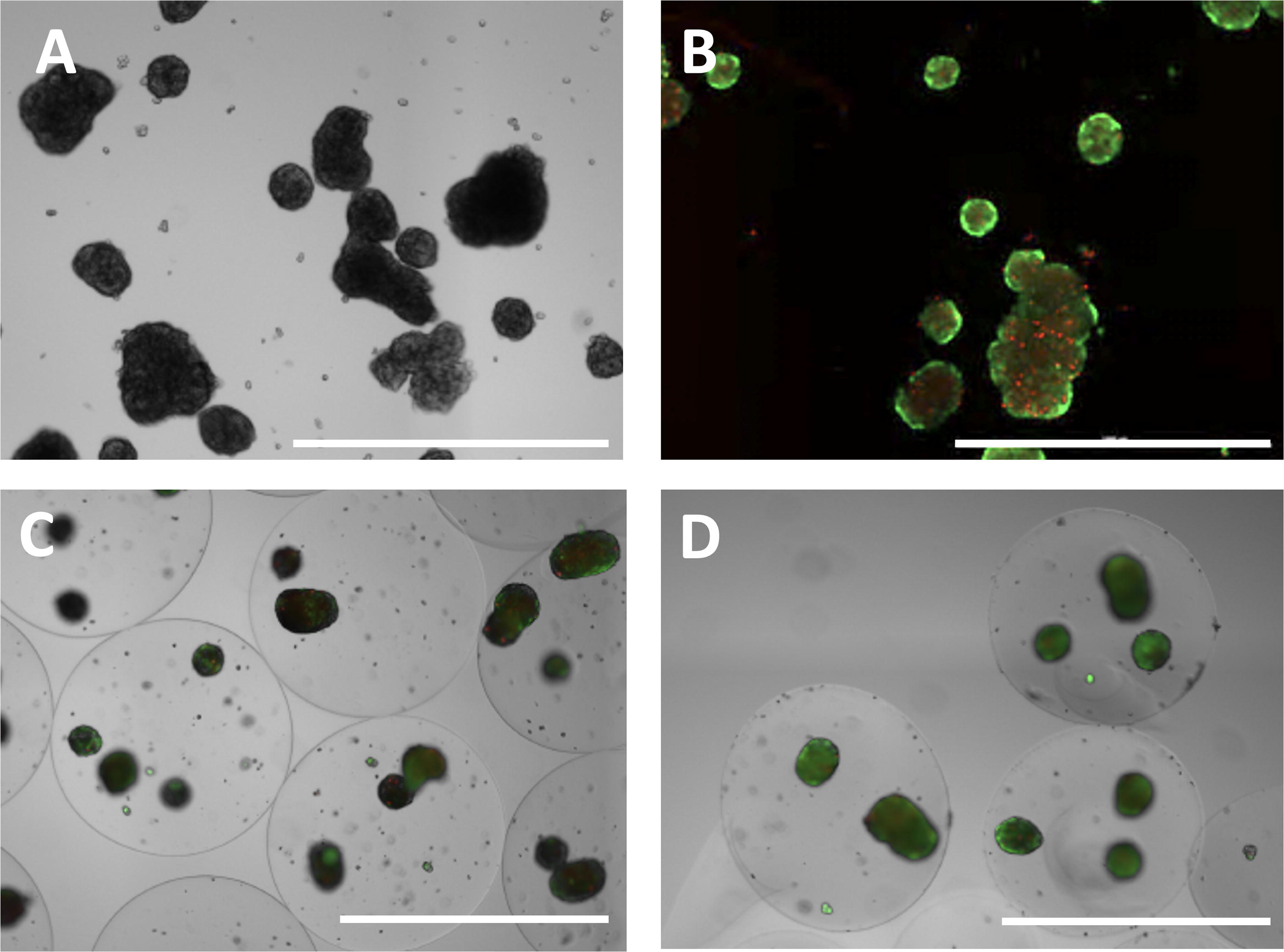
Cultured hepatocytes form mega-aggregates.
With the objective of determining the long-term effect of hydrogel encapsulation on the cells, in vitro assays were conducted at 3, 10, and 30 days after previous publications.35–37 To maintain the long-term viability of the unencapsulated hepatocyte spheroids, daily trituration and filtration through 200–225 µm diameter stainless steel meshes were required to remove mega-aggregates from the culture. The harvest losses associated with mega-aggregation became increasingly significant with extended time in culture so that by day 14, no functional spheroids were left.
Encapsulation of the spheroids resulted in microspheres characterized in Table 1. The average diameter for the alginate microspheres was slightly larger than the PVA microspheres and those diameters did not change statistically over the course of the study. Likewise, there was no statistical difference in the number of spheroids encapsulated per microsphere. Due to the differences in viscosity of the precursors, there was a difference in the mass fraction between the two hydrogels. The viability of spheroids was quantified by staining with the fluorophore ethidium bromide to identify dead cells (ethidium bromide) and Hoechst and brightfield to determine the location and pixel area of the entire spheroids and compared between unencapsulated and encapsulated conditions (Fig. 2A). Initial analysis illustrated high viability in both encapsulated and unencapsulated spheroids, with all three groups showing over 90% viability on day 3. There was a slight decline in viability for all three groups on day 10, which was not statistically different. As explained previously, the unencapsulated spheroids formed mega-clusters that did not survive in culture for 30 days. Therefore, there was no 30-day viability data for the unencapsulated group (Fig. 2B). There was no statistical difference across the groups at any time point.
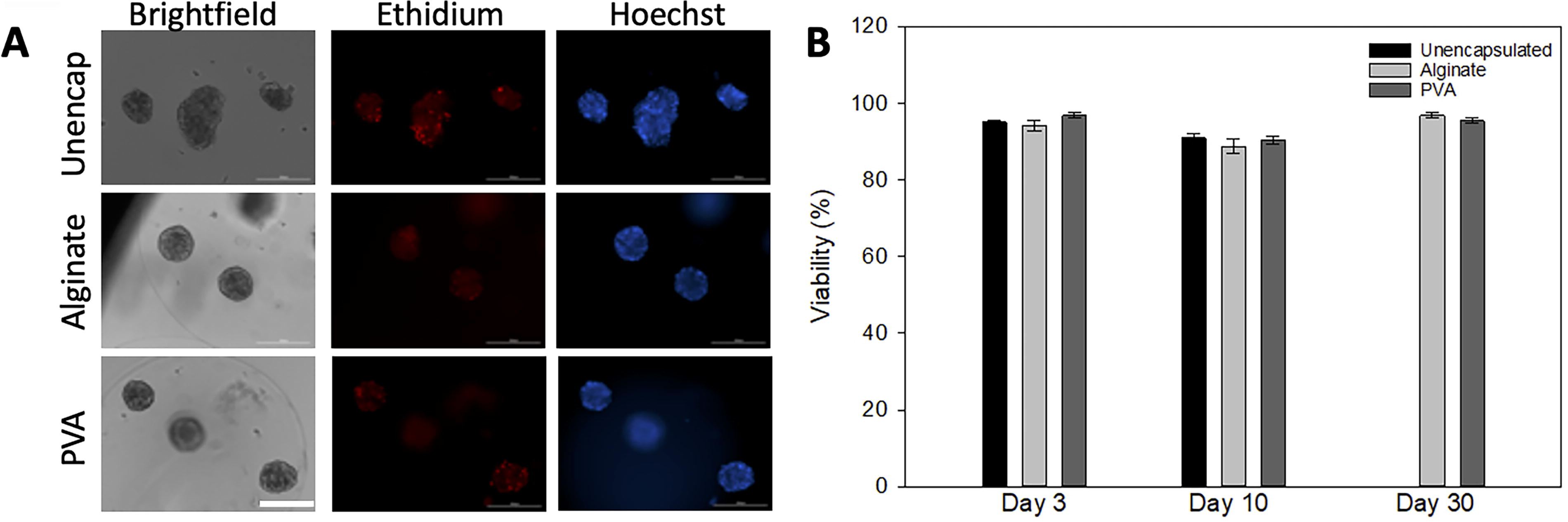
Viability of unencapsulated and encapsulated hepatocyte spheroids.
Description of Microbeads
PVA, poly(vinyl alcohol).
Effect of encapsulation on hepatocyte spheroid metabolism
To assess metabolic activity, the same three groups were compared (Fig. 3). All experimental groups exhibited comparable metabolic activities on day 3. By day 10, the unencapsulated spheroids showed twice the activity of the encapsulated groups. However, by day 30, the mega-aggregation of the unencapsulated cells resulted in losses that depleted the entire culture. The alginate- and PVA-encapsulated spheroids exhibited similar metabolic activity on day 10, whereas PVA-encapsulated spheroids exhibited significantly elevated activity by day 30 (p < 0.001). Since the aggregated spheroids were not the same size, data were also normalized to spheroid area (Table 2) in addition to spheroid number (Fig. 3). Whether normalized to area or spheroid number, the statistical significance shown in the figure was the same with the unencapsulated group statistically different at 10 days and the PVA group statistically higher on day 30.
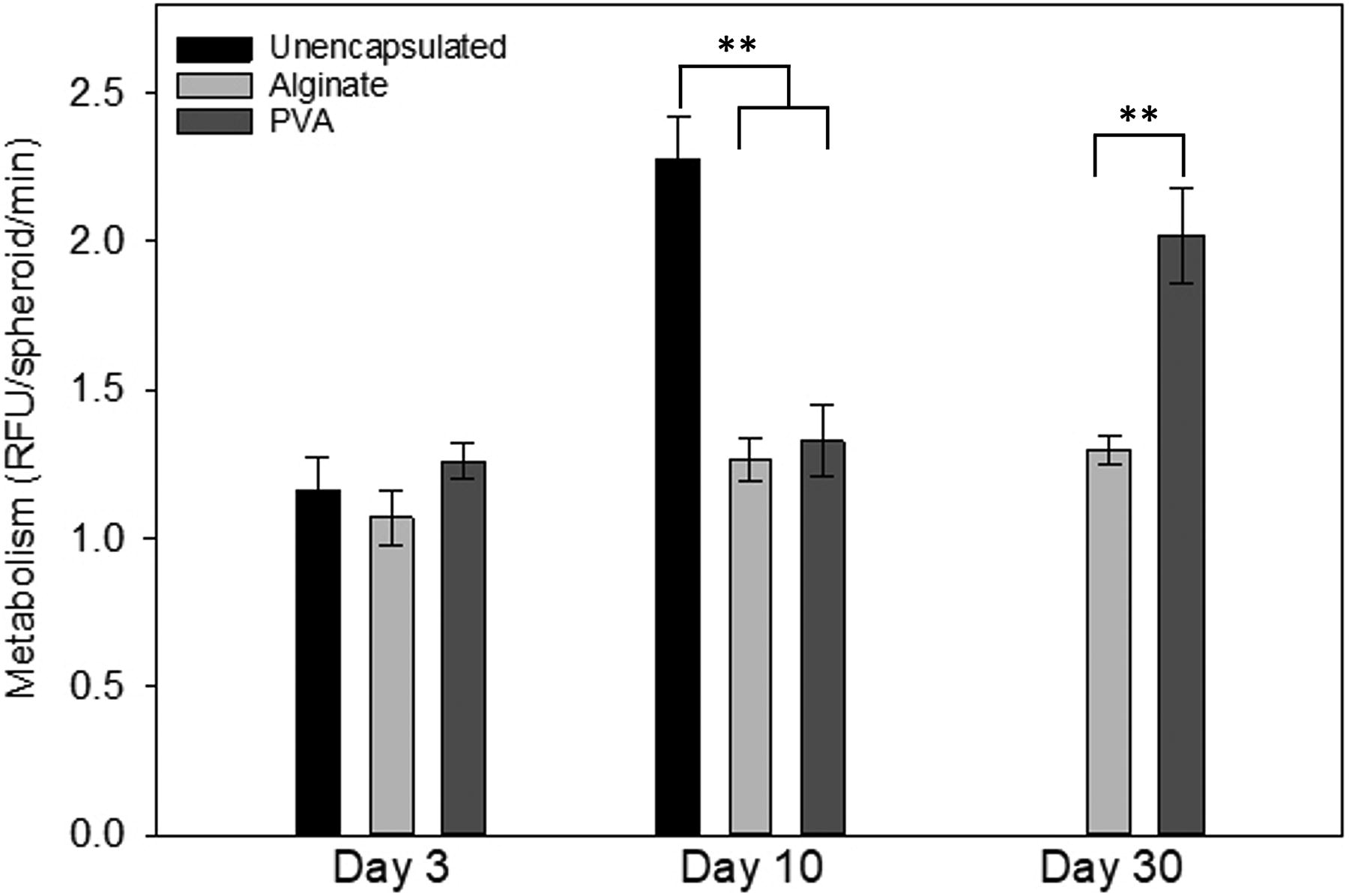
Metabolic activity of unencapsulated and encapsulated hepatocyte spheroids. Metabolic activity, measured by PrestoBlue, showed that by day 30, PVA maintained metabolic function better than the other two groups. On day 3, there were no statistical differences between groups. However, by day 10, there was a statistical difference between both encapsulated groups and the naked spheroids. On day 30, there was a statistical difference between the PVA and alginate groups with PVA performing better. ** indicates p < 0.001.
Comparison of Metabolic Activity Between Groups
Functional assays
Functionality of the cells was determined by albumin secretion and CYP activity. The secretion of albumin from the hepatocytes is shown in Figure 4. At all time points, secretion of albumin was highest from PVA-encapsulated spheroids, although day 30 data for unencapsulated spheroids are unavailable because unencapsulated spheroids could not be maintained for 30 days. Specifically, on day 3, the amount of albumin secreted from PVA was statistically higher than either the alginate group or the unencapsulated cells (p < 0.001). At day 10, the PVA was again higher than both of the other groups and, at day 30, was statistically greater than alginate.
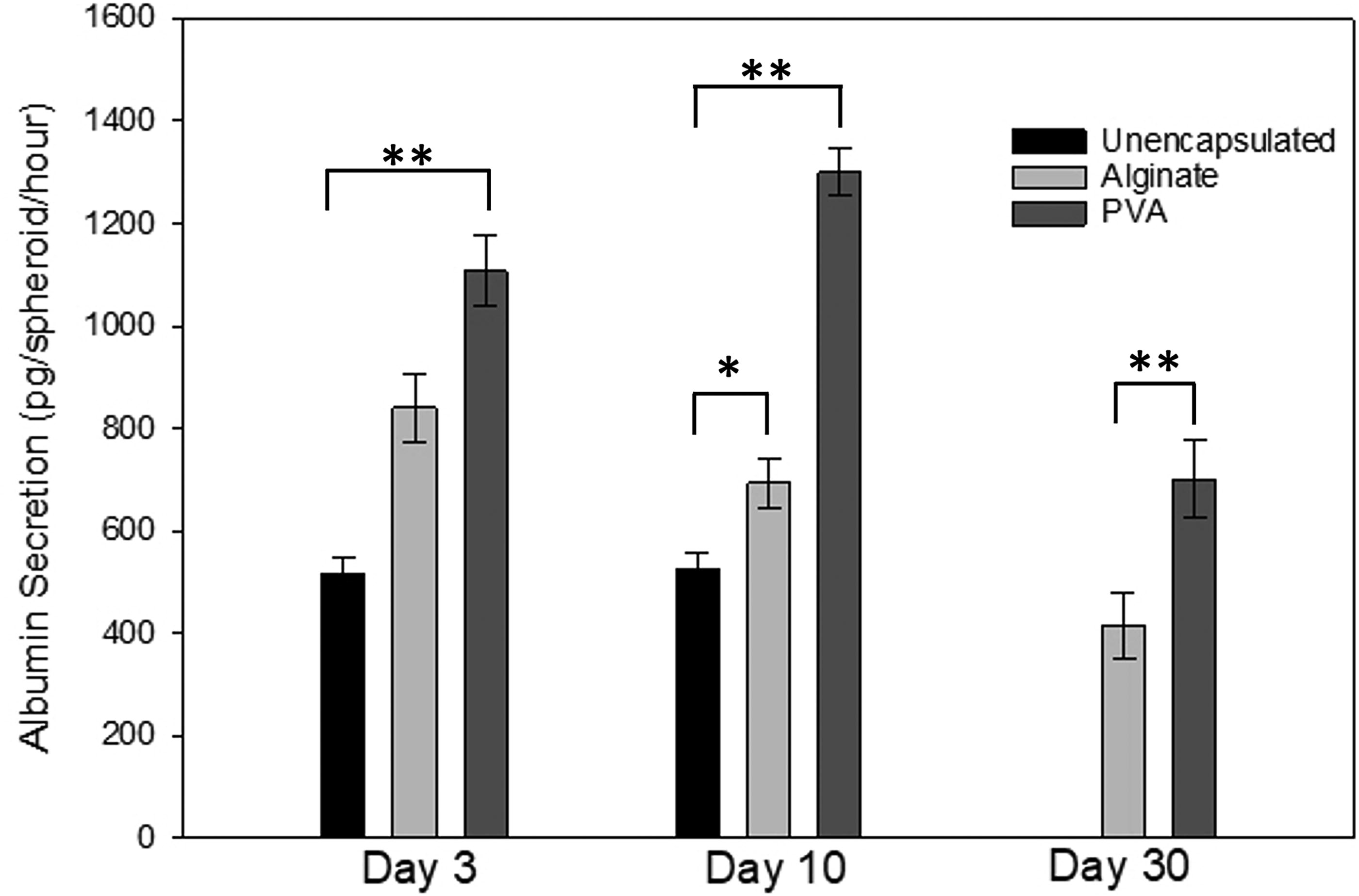
Albumin secretion from unencapsulated and encapsulated hepatocyte spheroids. Secretion of albumin was measured using ELISA and normalized to the number of spheroids per hour. At all three time points, there was a statistical difference between the groups with the PVA showing the highest level of secretion at each time. * indicates p < 0.01, ** indicates p < 0.001.
Cytochrome P450 enzymes comprise a family of hepatic enzymes that play a large role in drug metabolism by the liver. The assays for two members of the family (CYP1A1 and CYP3A4) provided another measure of hepatocyte function. Basal CYP1A1 activity was used to calculate the fold induction of the enzyme (Table 3). Following induction by omeprazole, CYP1A1 activity for both encapsulated groups followed a trend of increased activity through day 30 (Fig. 5A). In contrast, induced CYP3A4 activity of unencapsulated spheroids decreased with time from days 3 to 10 (Fig. 5B). Normalization by either spheroid number or spheroid area did not alter the results. All comparisons were statistically significantly different except the day 10 comparison among all three groups.
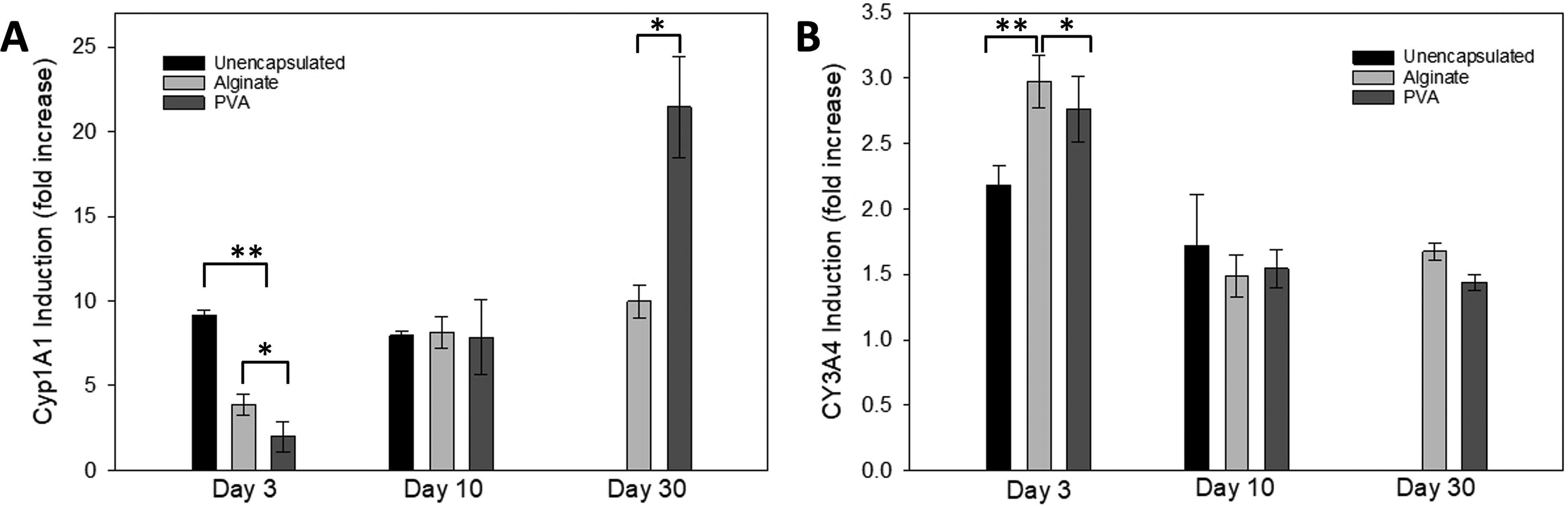
CYP activity from unencapsulated and encapsulated hepatocyte spheroids.
Basal CYP3A4 activity was relatively constant over time for all three groups (Table 3). Again, the unencapsulated spheroids had the lowest basal CYP activity. Like CYP1A1, there were no statistical differences between CYP3A4 activity on day 10. Normalization by either spheroid number or spheroid area did not alter the results.
In vivo cell function
Severe Combined Immunodeficiency (SCID) mice were implanted IP with hepatocyte spheroids microencapsulated in either alginate or PVA. A third group of mice was implanted with unencapsulated cells. The mice were observed and weighed weekly. One mouse in the PVA group died before the implantation of the encapsulated spheroids; thus, the death was not associated with the treatment. All other mice in the study remained healthy and continued to gain weight during the study (Table 4). Mice with unencapsulated cells gained an average of 12% body weight over the course of the study. Mice implanted with PVA-encapsulated cells gained 13% body weight, whereas mice with alginate-encapsulated cells gained 10%. The general health was assessed using Activity and Coat Appearance Scores, and all animals in the three groups obtained the highest score (9/9), demonstrating good health.
Blood was obtained from the submandibular vein before the transplantation and again at 3, 10, and 30 days after the transplantation. No human albumin was detected before transplantation in any of the animals (data not shown). After the implantation of the spheroids, human albumin was detected in all three groups, but the two encapsulated groups demonstrated higher levels (Fig. 6A). Due to the high level of variability, there was no statistical difference between groups on day 3. However, from days 3 through 30, the albumin secreted by the unencapsulated spheroids declined to slightly above the detection level. In contrast, the two encapsulated groups showed steady albumin secretion through day 10. By day 30, only the PVA group maintained high levels of albumin secretion, whereas the albumin group lost significant levels of serum albumin. Repeated-measures ANOVA determined that the outcome for the PVA group was statistically different from the other two groups.
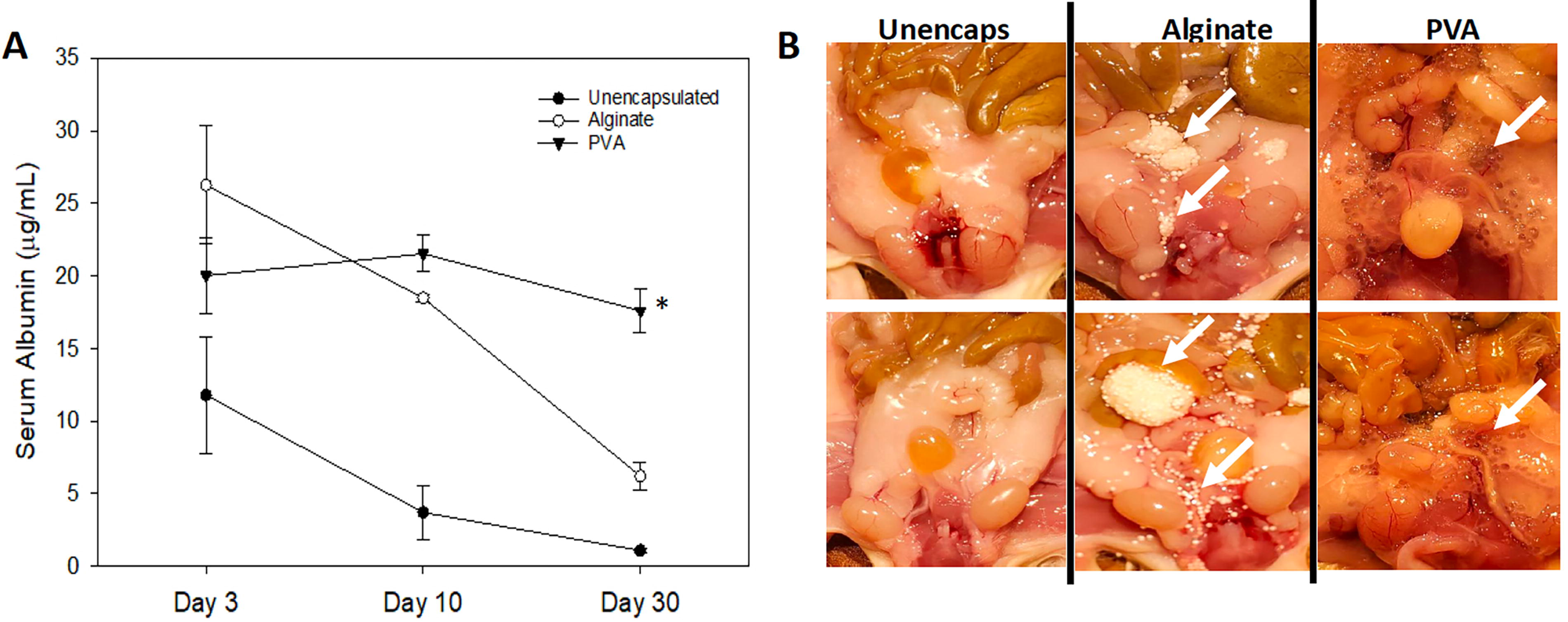
Immune-compromised mice—response to transplants of unencapsulated and encapsulated hepatocyte spheroids.
At the completion of the study (60 days), necropsy revealed a stark difference between the three groups. Figure 6B shows the IP region at the time of the necropsy. As expected, no microspheres were found in the unencapsulated cell group. In the animals receiving alginate microspheres, they were white and in large clusters throughout the peritoneum. Figure 6B shows the locations (white arrows). In contrast, the PVA microspheres were greater in number, spread more evenly throughout the tissue, and still translucent in appearance without signs of fibrosis.
The explanted microspheres were stained for viability, and fewer dead cells (red) were found in the PVA-containing microspheres. Figure 7 shows clear PVA spheres with predominantly live encapsulated cells (few red areas). In contrast, the microspheres made of alginate were often encompassed in dead cells (red stain). It should be noted though that the red ethidium homodimer-1 stain around the alginate microbeads was the result of dead host tissue adhered to the beads, which was evident in the alginate but not in the PVA beads.
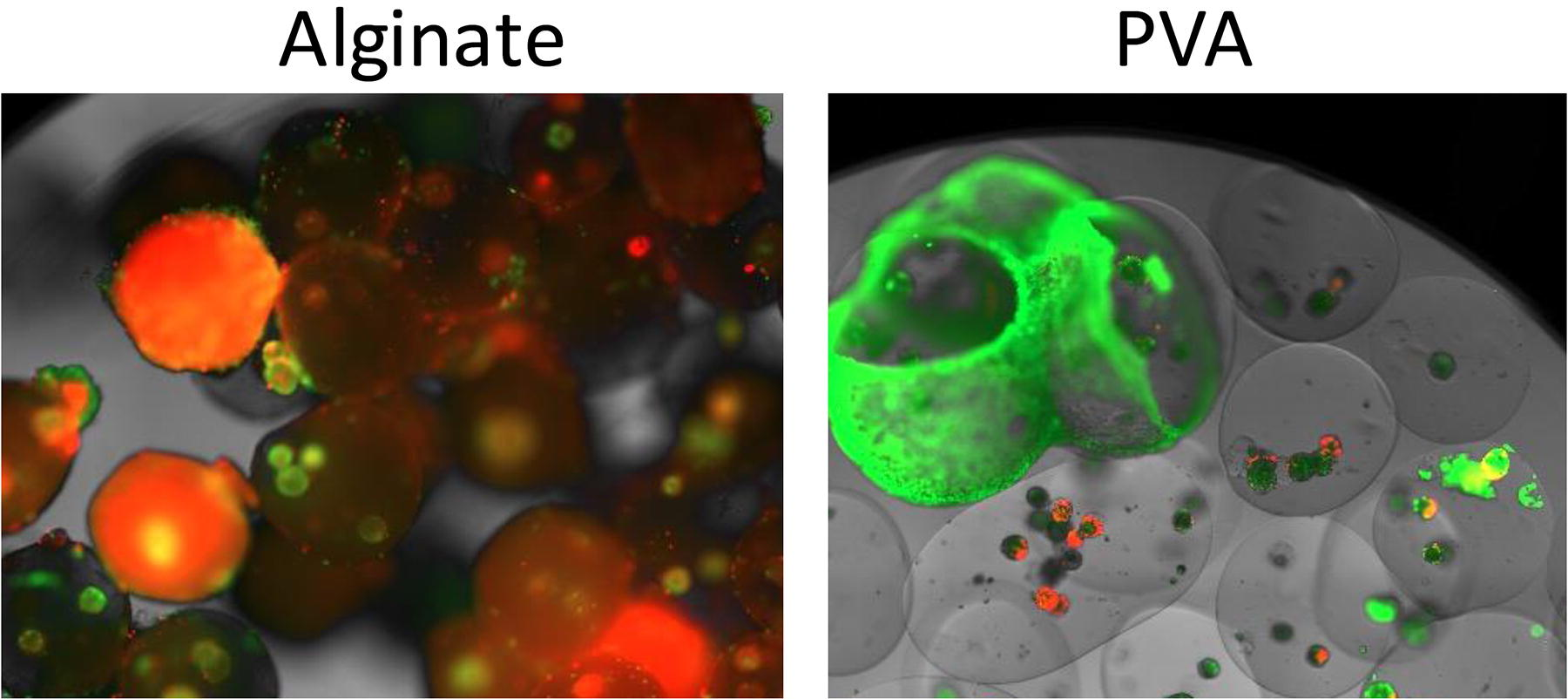
Histology of microspheres from immunocompromised mice. The images illustrate liver spheroids retrieved at the time of necropsy and stained with calcein and ethidium bromide. The explants of the alginate microbeads were covered with material that stained for dead cells. In contrast, the PVA microspheres did not show signs of overgrowth.
In vivo biocompatibility in immune-competent mice
Observing what appeared to be immune rejection to the alginate group in immune-compromised mice led to the question of whether the response was to the hydrogels or the xenografted human cells. A second animal study utilizing immunocompetent C57 mice was undertaken with empty (no cells) alginate or PVA microspheres implanted IP into the mice. In this manner, the potential for a xenogeneic immune response to secretions from human-derived hepatocytes could be ruled out. The mice were observed and weighed weekly during the period, and they showed good health and normal weight gain (Table 5). In addition, the Activity and Coat Appearance Scores were demonstrated that the mice were in good health.
Sixty days later the study ended, and mouse necropsy was performed. Figure 8 illustrates the differences in the appearance of the microspheres at the time of necropsy consistent with the observations made in the NOD mice (Fig. 7). The alginate microspheres were distinctly opaque white in appearance and tended to aggregate in clusters (white arrows). In contrast, the PVA microspheres remained translucent and were found scattered throughout the peritoneum.
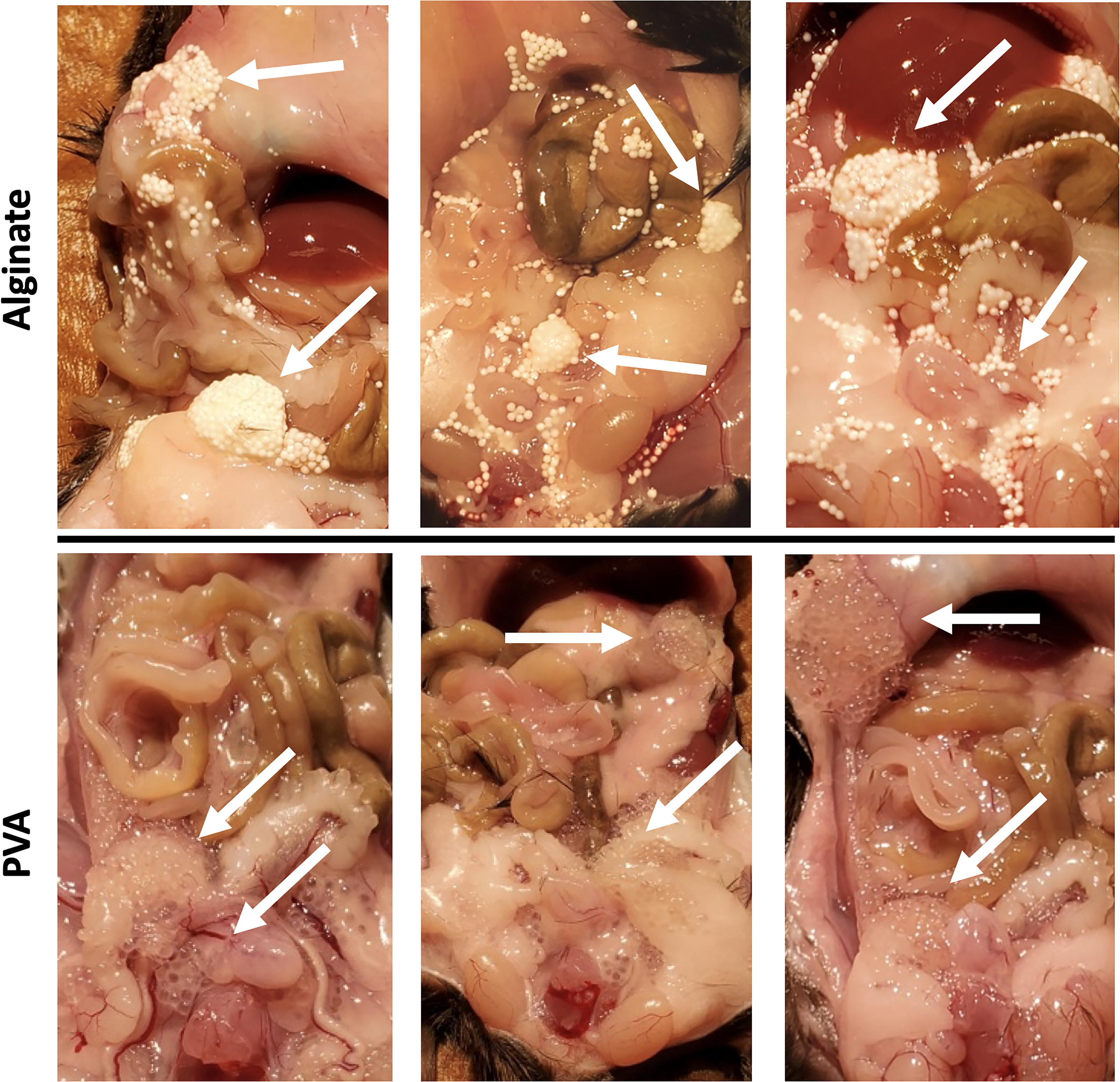
Necropsy of immune-competent mice after transplant of empty microspheres. Examples of alginate microspheres (without cells) were found in the peritoneum of immune-competent mice, showing opaque microspheres clustered together within the peritoneum (arrows, upper panel). PVA microspheres (arrows, lower panel) were translucent without clustering.
H&E staining of the tissues surrounding the microspheres revealed a strong FBR in the animals that received the alginate microspheres (Fig. 9). The images in Figure 9D show large clusters of alginate microspheres with extensive inflammatory cells surrounding the spheres and rings of fibrotic material. In contrast, PVA microspheres (Fig. 9E) showed little inflammation in the surrounding omentum with small fibrotic perimeters were found surrounding the PVA microspheres. Using previously published methods, the thickness of the inflammatory band and the fibrotic capsule surrounding the individual microspheres was measured. 21 The graph (Fig. 9F) summarizes the results. The tissue with alginate microspheres had significantly more inflammatory regions than the PVA group. Likewise, the thickness of the fibrotic ring surrounding each microsphere was greater in the alginate group compared with PVA (Fig. 9F).
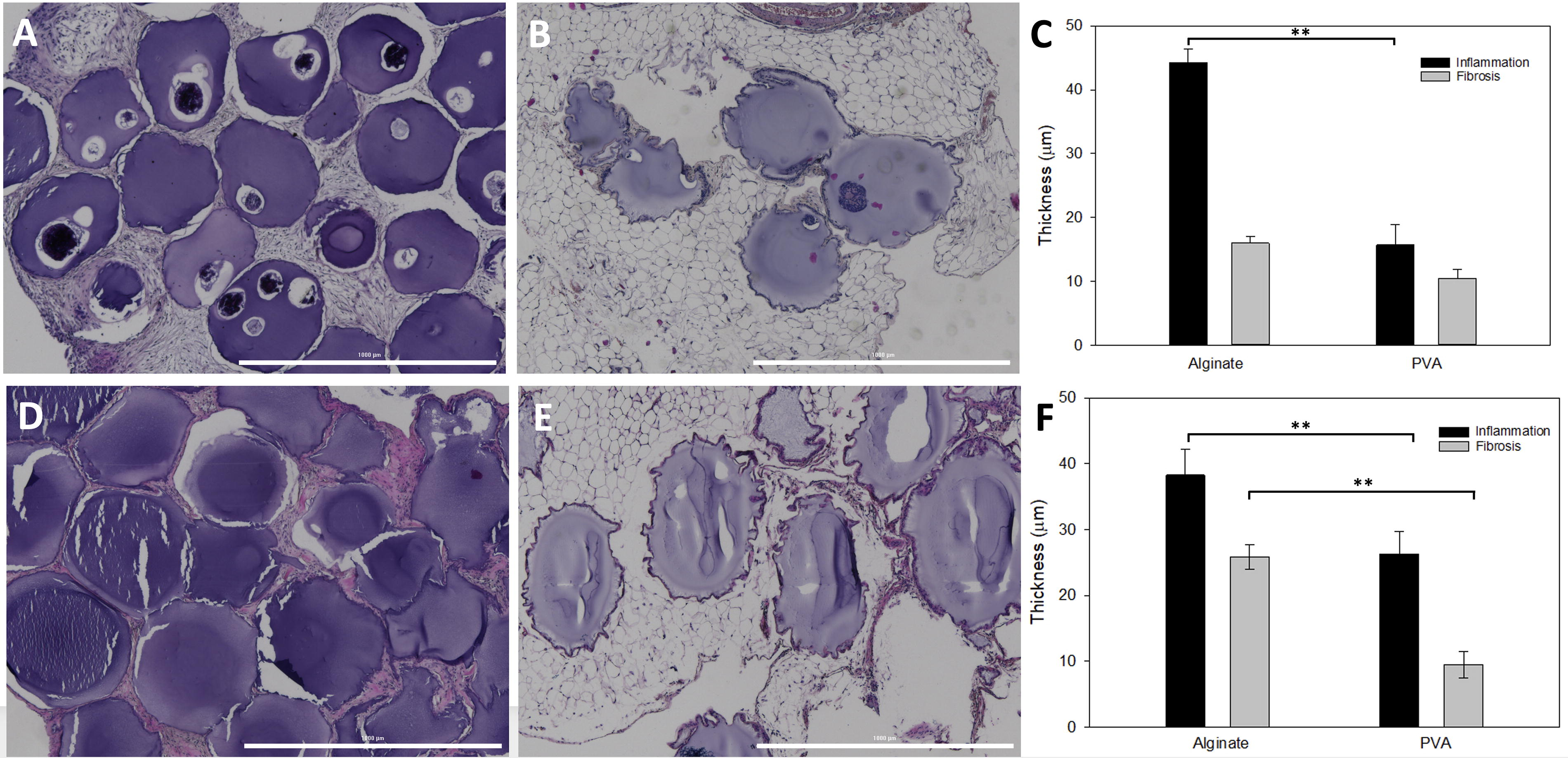
Histology of tissue surrounding empty microspheres.
Both the immune-compromised and immune-competent mice showed signs of fibrosis surrounding the alginate microspheres at necropsy (Figs. 6 and 8); thus, we conducted the same biocompatibility measurements on tissue sections from the NOD-SCID mice described above. Again, at 60 days, the same trend was noted with more inflammation surrounding the alginate microspheres (Fig. 9A) compared with the PVA (Fig. 9B). The graph (Fig. 9C) illustrates the statistical difference in the level of inflammation with the alginate group.
Discussion
Liver transplantation has been the treatment option for patients with end-stage liver disease and acute liver failure. However, transplantation has severe limitations including limited availability of donor livers, difficulties and limitations in isolating high-quality cells, lack of suitable cryopreservation methods, poor engraftment and proliferation, and immune rejection.38–40 Due to these challenges, alternative forms of treatment are direly needed, with recent reports showing that transplantation with healthy cultured hepatocytes may be feasible alternatives.41–43 The current study was designed to determine whether encapsulation of HepaRG spheroids could improve transplant outcomes by comparing alginate, a commonly used encapsulant, with a more potentially biocompatible polymer, PVA.
The unencapsulated cells quickly formed mega-aggregates in culture, resulting in core cell death. Although great effort expended to maintain the unencapsulated hepatocyte spheroids in culture, including daily trituration and filtration, there were no functional, viable spheroids left by day 30 in culture. In contrast, immobilization of spheroids in a hydrogel prevented the formation of mega-aggregates. Our results demonstrate the potential benefits of maintaining viable and functional long-term spheroid cell cultures by encapsulating them in hydrogels. Similar to earlier reports using HepG2 cells, there was no evidence of cell proliferation within the hydrogels. 44
The results of this study indicate that hydrogel encapsulation assists the long-term culture of HepaRGs by providing a physical barrier that prevents unwanted aggregation over time. The in vitro viability of the spheroids encapsulated in alginate or PVA was comparable over the study period. The in vitro secretion of albumin from the hepatocyte spheroids encapsulated in alginate and PVA was also comparable, with PVA-encapsulated spheroids showing the greatest albumin secretion at 30 days. Basal CYP activity of the spheroids was unaffected by encapsulation, whereas induced CYP activity was similar between the two types of encapsulated spheroids, but divergent from unencapsulated cells. Thus, at the conclusion of the in vitro tests, there appeared to be improvement in cell viability and function when encapsulated in either alginate or PVA, with PVA performing better.
However, significant differences between alginate and PVA encapsulation were apparent with the in vivo tests. Three days after implantation into immunocompromised mice, human albumin secretion was detected in all three groups, with the two encapsulated groups demonstrating the highest levels of albumin release. However, over the course of the study, albumin secretion from the unencapsulated and alginate groups fell dramatically, whereas the PVA group continued to show a release of albumin that was statistically similar on day 30 as it was on day 3 of the study. Examination of the microbeads at the time of necropsy showed liver spheroids maintained in the PVA microbeads, whereas the alginate microbeads were covered with dead cells. This 60-day time point for both the immune-competent and incompetent animals relates to a state of chronic FBS. For the animals in this study, fibrosis and indicators of inflammation were both higher in the alginate group. This illustrates that the PVA microbeads were better able to support the functionality of the spheroids in vivo, unlike the alginate and the unencapsulated spheroids.
The placement of the microspheres in the peritoneum was important as it allowed ample uptake of the secreted albumin for distribution through the bloodstream. The peritoneum has been utilized previously as a site to deliver cells for secretion of proteins and factors. 30 It represents a large absorptive surface that provides an optimal location for therapies that require the uptake of molecules by the vasculature, 45 which is one of the reasons it can be utilized for dialysis. 46 Figures 6 and 8 of this study, so the stark differences in the distribution of the microspheres between alginate and PVA, even though both were administered using the same techniques and assessed with the same methods. The alginate microspheres were clustered together in large masses that were not optimal for the secretion of albumin. In contrast, the PVA microspheres remained evenly distributed throughout the peritoneum at both 30 and 60 days.
To better examine the potential causes of the failure of the alginate microbeads, empty (without cells) microbeads were implanted into the peritoneum of immune-competent mice. Two months later, the microspheres were recovered along with the surrounding tissue to examine the possibility of a chronic FBR. The recovered empty alginate microbeads were covered with solid white fibrotic material, whereas the PVA microbeads were clear without gross signs of fibrosis. Histological analysis of tissues revealed a clear FBR to the alginate microbeads that was less evident in the PVA sections. At 60 days, the FBR is characterized as chronic, which is when a transition from inflammatory reaction to fibrotic occurs. Chronic FBR involves a deposit of fibrous material surrounding each microsphere that acts as a barrier between the hydrogel and the surrounding tissue. 34 The collagen barrier can clearly be visualized with H&E staining, is clearly greater in the alginate group, and may explain why there was a loss of albumin secretion in the mice receiving alginate microspheres at the later time point.
The FBR occurred even though ultrapure alginate was used, which is reported to reduce the immunogenicity. 47 Our results agree with several studies showing that ultrapure alginate elicits an immunostimulatory reaction. 48 Commercially available ultrapure alginates can contain contaminants including immune stimulatory peptidoglycans and lipoteichoic acid, and new contaminants can be added during the purification process. 49 Moreover, the same response from the surrounding tissue was present with alginate microspheres regardless of whether the spheres contained human cells or not. Thus, it appears that the negative response noted with the alginate group was related to the alginate itself and not to the xenotransplant model.
Alginate has been widely used as a biocompatible cell delivery material in many applications due to its ease of gelation, tunable mechanical properties, biodegradability, permeability, and lack of cytotoxicity. Various efforts have also been used to chemically modify alginate to reduce its immunogenicity and make it more biocompatible and immune tolerable.50,51 Such modifications are complex, and examples include chemical PEGylation which involves attaching a PEG moiety to the alginate or creating modified alginate analogs.50,51 The studies presented here demonstrated that PVA-based polymers provide a much simpler alternative than trying to de-immunize alginate, which is not always effective.
The significance of the reduced FBR in PVA microbeads compared with alginate underlines the potential benefits of PVA microbeads for clinical implantation applications. The findings are interesting because earlier assessments of PVA sheets rather than microspheres demonstrated an FBR to the PVA. 52 However, the successful use of biomaterials is dependent on a complex series of molecular and cellular events that take place at the interface of the implanted material and the host immune and inflammatory responses, and not all PVA hydrogels are equivalent. Admittedly, data on the immune response to pure PVA are far less voluminous compared with alginate, as PVA is often combined with other hydrogels, minerals, or factors.53,54 Our studies show that PVA microbeads offer a superior biocompatible material for the potential application of hepatocyte transplants to treat liver disease. We propose that PVA-based core-shell applications may also be useful in the delivery of other cell-based therapies such as islets, among others.
In summary, the two encapsulated groups performed better than the unencapsulated cells. In the in vivo studies, as the length of the study reached 30 days, the PVA group outperformed alginate and unencapsulated groups in the functional test of albumin secretion likely due to the substantial FBR noted in the alginate groups, even in immunocompromised animals.
Footnotes
Authors’ Contributions
S.H.: Conceptualization and design, methodology, data collection and analysis, interpretation, article writing and review, project administration, and final approval of article. E.L.: Methodology, data collection and analysis, interpretation, article review, and final approval of article. A.W.: Methodology, data analysis and interpretation, article writing and review, and final approval of article. V.L.: Conceptualization and design, methodology, data interpretation, article review, final approval of article, and funding. L.S.-B.: Conceptualization and design, data analysis and interpretation, financial support, administrative support, article writing and review, and final approval of article. F.K.: Conceptualization and design, data collection and analysis, interpretation, article writing and review, and final approval of article.
Author Disclosure Statement
S.H., E.L., A.W., L.S.-B., and F.K. are employees of Likarda, Inc. V.L. is an employee of Takeda Pharmaceuticals. L.S.-B. is a founder and has equity in Likarda.
Funding Information
This study was supported and funded by Likarda, Inc. and Takeda Pharmaceuticals. The authors wish to thank Megan Hamilton for her assistance with animal transplantations and Karthik Ramachandran for his discussions concerning assay designs and results.
