Abstract
Severe coronary artery disease is often treated with a coronary artery bypass graft using an autologous blood vessel. When this is not available, a commercially available synthetic graft can be used as an alternative but is associated with high failure rates and complications. Therefore, the research focus has shifted toward the development of biodegradable, regenerative vascular grafts that can convert into neoarteries. We previously developed an electrospun tropoelastin (TE)–polyglycerol sebacate (PGS) vascular graft that rapidly regenerated into a neoartery, with a cellular composition and extracellular matrix approximating the native aorta. We noted, however, that the TE-PGS graft underwent dilation until sufficient neotissue had been regenerated. This study investigated the mechanisms behind the observed dilation following TE-PGS vascular graft implantation in mice. We saw more pronounced dilation at the graft middle compared with the graft proximal and graft distal regions at 8 weeks postimplantation. Histological analysis revealed less degradation at the graft middle, although the remaining graft material appeared pitted, suggesting compromised structural and mechanical integrity. We also observed delayed cellular infiltration and extracellular matrix (ECM) deposition at the graft middle, corresponding with the area’s reduced ability to resist dilation. In contrast, the graft proximal region exhibited greater degradation and significantly enhanced cellular infiltration and ECM regeneration. The nonuniform dilation was attributed to the combined effect of the regional differences in graft degradation and arterial regeneration. Consideration of these findings is crucial for graft optimization prior to its use in clinical applications.
Impact Statement
This study revealed that the nonuniform dilation of electrospun tropoelastin–polyglycerol sebacate grafts was due to regional differences in graft degradation and arterial regeneration. Our results provide an important basis for future vascular graft designs incorporating mechanical and biological strategies to achieve balanced graft integration that minimizes dilation, which is a necessary requirement prior to their transition to large animal models.
Introduction
Coronary artery disease, caused by atherosclerosis, can be treated with coronary artery bypass grafts (CABGs), which have been shown to enhance survival rates and lower occurrences of major cardiovascular events compared to percutaneous coronary intervention. 1 CABG surgery frequently uses autologous grafts harvested from the internal thoracic artery, saphenous vein, and radial artery. In severe situations when autologous grafts are not available due to preexisting pathological conditions, synthetic vascular grafts fabricated from materials such as polytetrafluoroethylene and polyethylene terephthalate can be used as alternatives. 2 However, these nondegradable, nonregenerable vascular grafts are associated with a high failure rate due to thrombosis, intimal hyperplasia, calcification, and infection. 3
Recent focus has shifted toward the potential use of biodegradable and regenerative vascular grafts, which would ideally integrate into the existing vessel, following implantation, to form a neoartery with long-term mechanical and biological functionalities similar to the native artery. 2 Among biodegradable polymer-containing implants, polyglycerol sebacate (PGS)-based vascular grafts have achieved more rapid and superior arterial regeneration compared with other materials such as polycaprolactone (PCL) and polyglycolic acid (PGA).4–8 Porous PGS vascular grafts have rapidly regenerated into neoarteries containing a functional endothelium, mature smooth muscle cells (SMCs), and an extracellular matrix (ECM) composed of collagen, elastin, and glycosaminoglycans. 4 However, unlike the native artery with distinctly organized elastin in the intima–media and collagen in the adventitia layers, the regenerated ECM components were evenly distributed across the whole neoartery. Our previous work reported on the implantation of a nonporous, electrospun vascular graft made from tropoelastin (TE), the monomeric protein precursor of elastin, and PGS (TE-PGS; 50:50 ratio), in mice for 8 months. This graft facilitated the advanced regeneration of a neoartery with an adventitia layer containing matured collagen and an intima–media layer composed of organized elastin-based structures, including an internal elastic lamina and multiple elastic lamellae, which showed substantial resemblance to the native artery structure. 9
Following implantation, we noted that the TE-PGS graft experienced continued dilation for 18 weeks, after which time the luminal diameter stabilized. Graft dilation can result in sluggish or turbulent blood flow that can predispose the myocardium to ischemia, 10 prompt aneurysm formation, or cause graft rupture. Dilation is a common issue in biodegradable vascular grafts made from materials with fast and moderate degradation rates such as nonwoven polylactic acid (PLA) fiber mesh with poly(l-lactide-co-ε-caprolactone) (PLCL) sealant, 11 nonwoven poly-l-lactic acid fiber mesh with PLCL sealant, 12 braided PGA with PGS coating, 13 polydioxanone (PDS), 14 and PGS6. The cause of dilation is speculated to be the imbalance between graft degradation and neotissue formation.6,11 However, a detailed mechanism has been underexplored. This study aims to understand the progression of dilation in TE-PGS vascular grafts. We report that regional differences in TE-PGS graft degradation and regeneration between the graft proximal and middle contribute to a nonuniform dilation. These results provide crucial insights for future TE-PGS graft optimization and modification to enable the engineering of a viable, clinically relevant, elongated vascular graft.
Materials and Methods
Synthesis of PGS
PGS was synthesized using a modified published method. 15 Sebacic acid (0.1 mol) (99%, Merck) and 9.3 g glycerol (0.1 mol) (>99%, AlfaAesar) were mixed and melted at 140°C in a round-bottom flask for 10 min. The temperature was then reduced to 120°C before a vacuum of 20 mbar was applied. The reaction was kept under vacuum at 120°C for 48 h to obtain a pale yellow, viscous liquid. A sample of the polymer was dissolved in dimethyl sulfoxide, and its 1H nuclear magnetic resonance spectrum was confirmed using a Bruker NEO 300 MHz NMR spectrometer (Bruker).
Fabrication of TE-PGS graft
The TE-PGS graft was fabricated using the method previously described. 9 To prepare a solution of 10 w/v% TE and 10 w/v% PGS, the materials were fully dissolved in 1,1,1,3,3,3-hexafluoroisopropanol (HFP) by placing the container on a rotating rack at 4°C overnight. The solution was warmed to room temperature for 30 min before electrospinning. The electrospinning apparatus consisted of a moving needle-holder system, a mandrel-based collector, and a control unit. To fabricate a TE-PGS vascular graft, a 20% (w/v) TE-PGS in HFP solution was loaded into a 1 mL syringe connected to an 18-gauge needle via tubing. The needle was mounted onto a horizontally moving holder with a raster distance of 10 cm and a raster speed of 8 cm/s. A syringe pump delivered the solution at a flow rate of 1 mL/h. The electrospun fibers were collected on a Teflon-coated mandrel with a diameter of 0.7 mm, which was rotating at 1000 rpm. The distance from the needle tip to the mandrel was 15 cm. High-voltage power supplies (Gamma High Voltage Research, Inc.) supplied +15 kV to the needle and −15 kV to the mandrel, creating a 30 kV field. After electrospinning, the TE-PGS graft was left on the mandrel collector and cured in an oven at 160°C for 16 h to stabilize the TE and cure the PGS. The graft was then transferred to a biosafety hood, soaked in 80% ethanol for 1 h, and manually removed from the mandrel collector. Finally, the grafts were air-dried overnight in a biosafety hood.
Animal implantation
All experimental procedures involving animals were approved by nationwide children’s hospital institutional animal care and use committee. Animal care complied with the National Institutes of Health (NIH) guide for the care and use of laboratory animals. TE-PGS grafts were implanted into C57BL/6J (wild type) (n = 10) sourced from the Jackson Laboratory. Female mice aged 8–12 weeks were used for all procedures. The TE-PGS grafts were implanted as infrarenal abdominal aorta (IAA) interposition grafts using microsurgical techniques, as previously described. 16 Briefly, mice were anesthetized using ketamine/xylazine, with ketoprofen as a paranesthesia analgesic. A midline laparotomy incision was made from below the xiphoid to the suprapubic region. Intestines were wrapped in sterile saline-moistened gauze and retracted. The inferior vena cava was transected away from the aorta, and microvascular clamps were used to obtain vascular control of the aorta. The aorta was cut, and scaffolds were anastomosed proximally and distally with 10–0 polypropylene running sutures. The intestines were returned, and the skin was closed using a 6–0 black polyamide monofilament suture. Animals were moved to a recovery cage with a warming pad until becoming fully mobile. After 8 weeks of implantation, the mice were sacrificed, and implanted grafts were removed and fixed in formalin.
Ultrasound measurement of graft diameter and compliance
The patency of the grafts was monitored at different time points, including preoperatively and at 1, 2, 4, 6, and 8 weeks postoperatively, using high-frequency ultrasound (Vevo2100, FUJIFILM VisualSonics). To minimize any discomfort to the mice, they were anesthetized with 1–3% isoflurane and placed supine on a heated stage. Before imaging, hair in the abdominal region was removed using chemical depilatory cream (Nair, Church & Dwight). An 18–38 MHz frequency linear transducer (MS400, FUJIFILM VisualSonics) was used to capture long-axis brightness (B-) mode, motion (M-) mode, and electrocardiogram-gated kilohertz visualization images of the IAA at locations proximal, mid, and distal to the graft. The diameter of the IAA was measured using B-mode images, while compliance was determined by measuring systolic and diastolic diameters using M-mode images. 17 All measurements were taken in triplicate and averaged.
Histological and immunofluorescent staining and analysis
Sample blocks were sectioned (5 µm) transversely from the graft proximal to the graft distal. Slides then underwent deparaffinization in two changes of xylene for 10 min each, followed by rehydration using a sequence of ethanol solutions (100%, 100%, 95%, 95%, and 70%) for 2 min each. The rehydrated samples were then stained using different methods. For hematoxylin and eosin (H&E) staining, the rehydrated samples were first incubated in Harris’s hematoxylin for 5 min to stain nucleic material purple. Excess hematoxylin was removed by washing in tap water until clear. The slides were then stained with alcoholic eosin for 30 s, and the excess solution was removed by washing in tap water for 2 min. The samples were then dehydrated by washing three times in 100% ethanol for 1 min each, air-dried, and cover-slipped with dibutylphthalate polystyrene xylene (DPX). For Picrosirius red (PSR) staining, the deparaffinized tissue slides were first stained with Harris’s hematoxylin for 10 min, washed in tap water for 10 min, and then stained in PSR solution for 60 min. The samples were then briefly washed in acidified distilled water, dehydrated in three changes of absolute ethanol for 2 min each, and mounted in DPX before being coverslipped. For Verhoeff–van Gieson (VVG) staining, the samples were overstained in hematoxylin for 25 min, rinsed in running tap water until clear, and then differentiated in 2% aqueous ferric chloride until elastic fibers appeared black against a clear background. This was followed by washing in 95% ethanol for 30 s to remove iodine coloration and then washing in tap water. The samples were then counterstained with van Gieson’s stain for 3 min, dehydrated in absolute ethanol, cleared, and mounted in DPX before being coverslipped. All histology samples were imaged using an Olympus VS120 slide scanner (Olympus).
All analyses of images obtained following PSR and VVG staining were conducted using Fiji (ImageJ 1.53t) software. To assess collagen content the total neoartery area was first calculated by converting images of PSR stained sections to 8 bit then applying a threshold of 0–230. Then the area that corresponded to collagen staining was extracted using the Color Deconvolution plugin with the FastRed FastBlue diaminobenzidine vector, which separated collagen staining into the FastRed channel. Following conversion to 8 bit, a threshold of 0–170 was applied to determine collagen area. Collagen content in the neoartery was then described as the percentage of collagen area/total neoartery area.
To assess elastin content within the intima media as a percentage of the entire neoartery, the total neoartery area was first calculated by converting images of VVG stained sections to 8 bit and then applying a threshold of 0–170. The polygon selection function was then used to manually outline the border between the intima-media and the remaining graft. The area corresponding to the residual graft and adventitia was then removed from the image using the clear outside function. The remaining image was then converted to 8 bit and thresholded from 0 to 125 to obtain the stained elastin area. Elastin content was then described as the percentage of intima–media elastin area/total neoartery area.
To determine the number and thickness of elastic lamellae in each sample, two lines were randomly drawn across the entire intima–media region of each image of VVG stained sections, and the number and thickness of elastic lamellae crossing the lines were counted and measured using the measure function in Fiji.
For immunofluorescence staining, samples were deparaffinized and subjected to heat-based antigen retrieval in a 95°C water bath for 5 min using citrate-based antigen unmasking solution (H-3300–250, Vector Laboratories, 1:100). Primary antibodies including alpha smooth muscle actin (α-SMA) (ab5694, Abcam, 1:200), inducible nitric oxide synthase (iNOS) (ab3523, Abcam, 1:200), CD206 (ab64693, Abcam, USA, 1:1000), CD31 (ab182981, Abcam, 1:2000), and matrix metalloproteinase 2 (MMP2) (ab86607, Abcam, 1:200) were diluted in 1% bovine serum albumin (BSA) in phosphate buffered saline (PBS) and used to stain the samples at 37°C for 2 h in a humidified chamber. Samples were then washed three times in PBS, 10 min each. Secondary antibodies, goat antirabbit IgG H&L (ab175471, Abcam, 1:200) and goat anti-mouse IgG H&L (ab175473, Abcam, 1:200), both conjugated with Alexa Fluor 568, were diluted 1:200 in 1% BSA in PBS and incubated with the tissue slides at room temperature for 1 h before washing three times with PBS, 10 min each. The samples were then stained for nuclei using Hoechst 33342 (Merck) followed by three washes in PBS, 10 min each. The slides were then mounted in a Fluoromount-G mounting medium. Imaging was performed using a Nikon C2 confocal microscope. Stained markers were visualized using a 561 nm laser. Hoechst 33342 stained nuclei were imaged using a 405 nm laser.
Statistical analysis
Ordinary one-way analysis of variance and unpaired t-tests were performed using GraphPad Prism version 9 software. Tukey’s multiple comparison tests were used to determine the significant difference between different conditions. Statistical significance was accepted at values of p < 0.05. Exact p-values are indicated in the figures.
Results
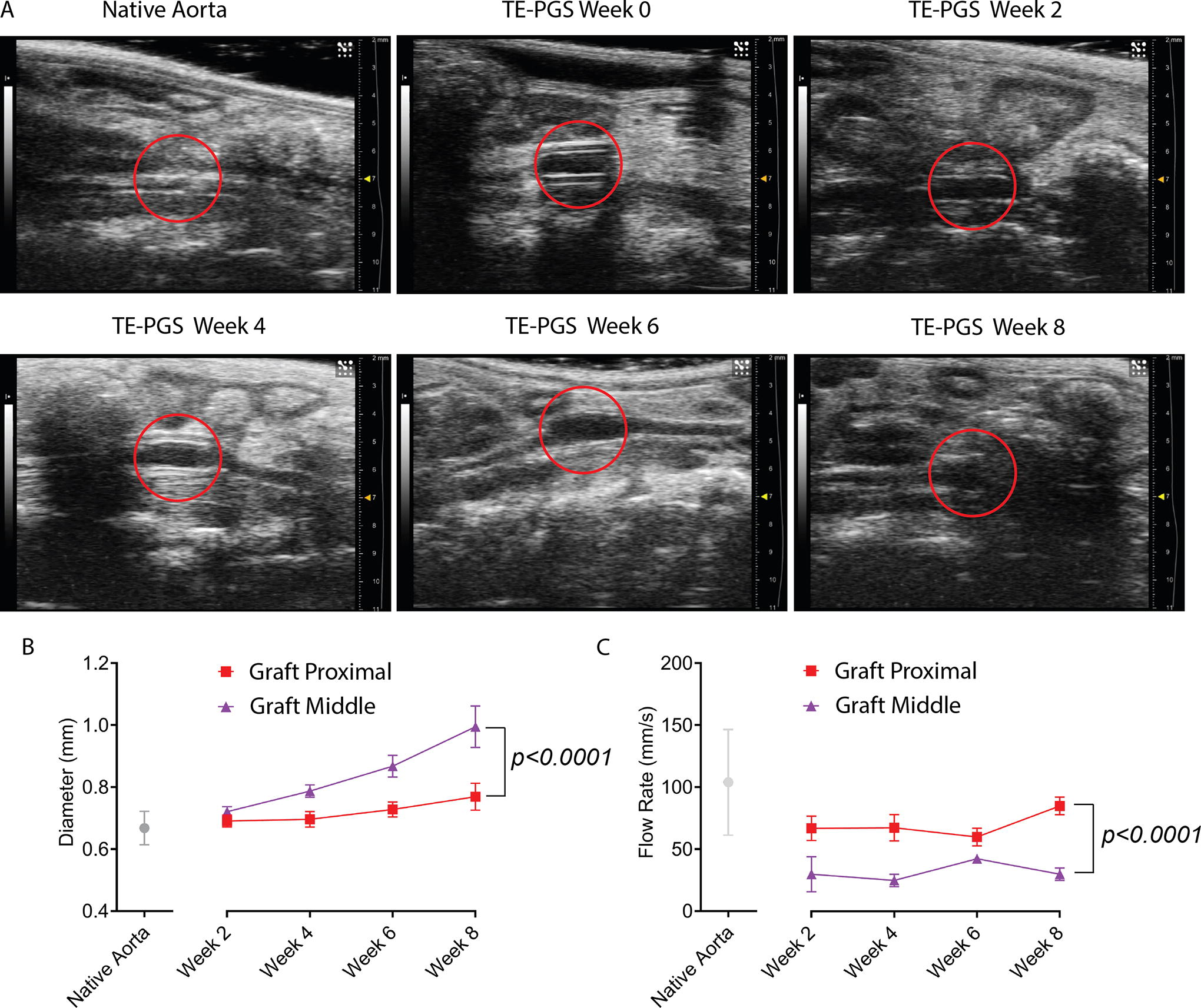
Nonuniform dilation of TE-PGS graft
Implanted TE-PGS grafts were monitored using high-frequency ultrasound for 8 weeks (Fig. 1A). The TE-PGS graft appeared rigid following implantation. During the 8 weeks of implantation, the graft displayed nonuniform dilation, which was most pronounced at the graft middle and decreased towards the graft proximal (anastomosis) sites. Ultrasound images showed that the average native aorta diameter was 0.67 ± 0.05 mm and remained unchanged over the implantation period. In contrast, the diameter at the graft proximal site showed a gradual increase from 0.69 ± 0.02 mm at 2 weeks to 0.77 ± 0.04 mm at 8 weeks. The graft middle had a faster dilation rate, showing a diameter increase from 0.72 ± 0.01 mm at 2 weeks to 0.99 ± 0.05 mm at 8 weeks (Fig. 1B). The flow rate at the graft middle was significantly lower than at the graft proximal site at all time points, but flow rates at both locations did not seem to change over the implantation period (Fig. 1C). By 4 weeks, the flow rate was 85 ± 5 mm/s at the graft proximal site and 30 ± 4 mm/s at the graft middle, both significantly lower than 104 ± 40 mm/s for the native aorta.
Regional differences in TE-PGS graft degradation and regeneration
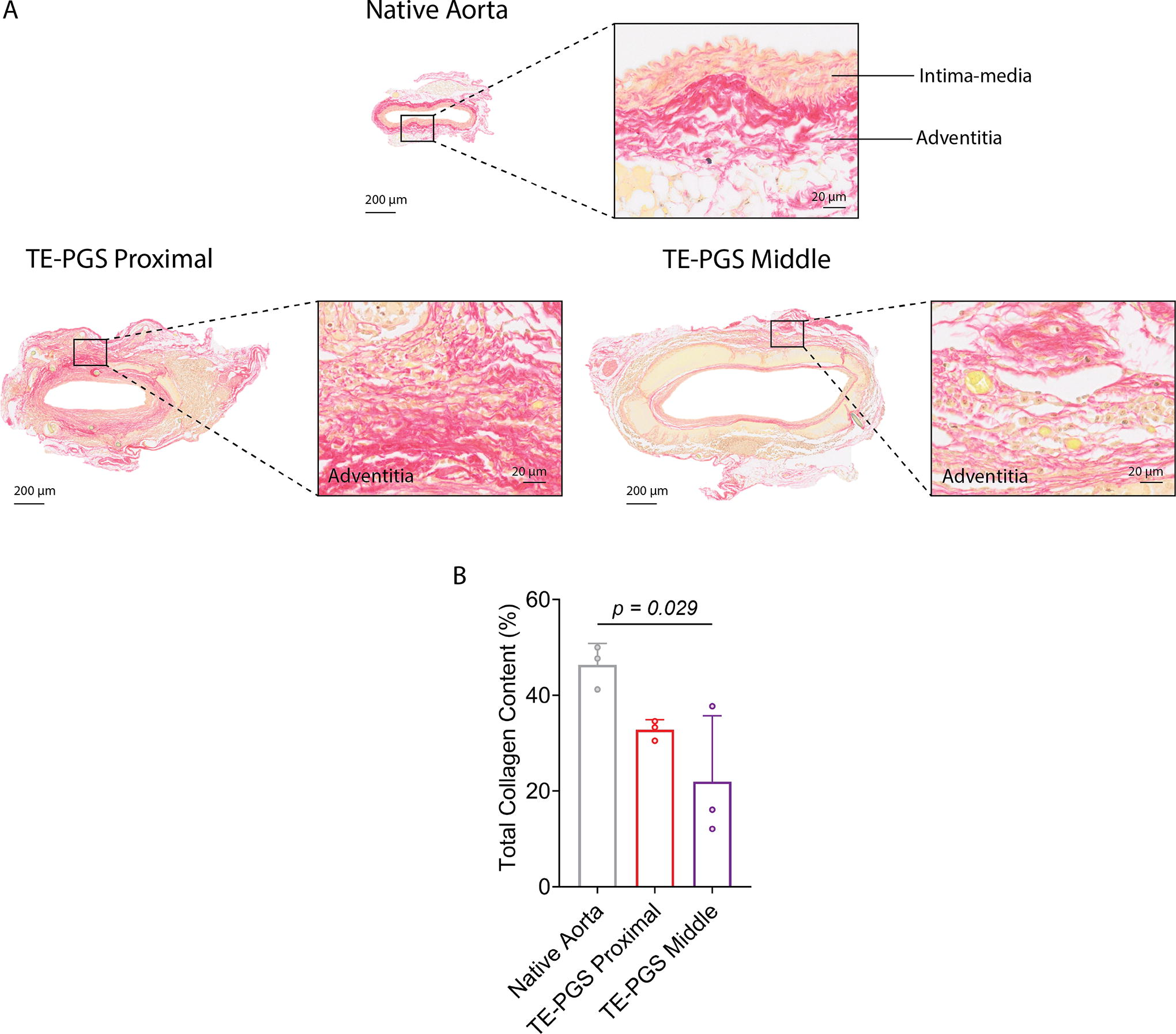
As we observed a nonuniform dilation of the TE-PGS graft, we hypothesized that graft degradation and ECM regeneration were nonuniform processes across the grafted region. We adopted a serial sectioning strategy to allow a histological comparison of the implanted graft site from the proximal through to the middle and onto the distal regions. H&E staining of the explanted native aorta showed it contained two distinct layers, an intima–media layer and an adventitia layer. In contrast, the TE-PGS graft sites at 8 weeks postimplantation consisted of three distinct layers, an intima–media layer, the remaining graft layer, and an adventitia layer (Fig. 2A). In addition, we observed sutures remaining at both the proximal and distal regions of the graft, which restrained graft dilation at both ends. The average area of the remaining graft layer increased from 9.3 × 104 ± 5.6 × 104 µm2 at the graft proximal to 22 × 104 ± 9 × 103 µm2 at the graft middle and decreased to 8.5 × 104 ± 3.9 × 104 µm2 at the graft distal (Fig. 2B), which corresponded to 17 ± 8%, 40 ± 5%, and 20 ± 7%, respectively, of the total neoartery area (Fig. 2C). In contrast, the area of the adventitial layer decreased from 57 ± 8% at the graft proximal to 35 ± 7% at the graft middle and increased to 61 ± 6% at the graft distal (Fig. 2D), while the intima–media layer area remained constant across the grafted area (Fig. 2E). Given the similarity in graft degradation and morphology at the graft proximal and distal regions, further studies concentrated on the proximal and middle regions of the graft.
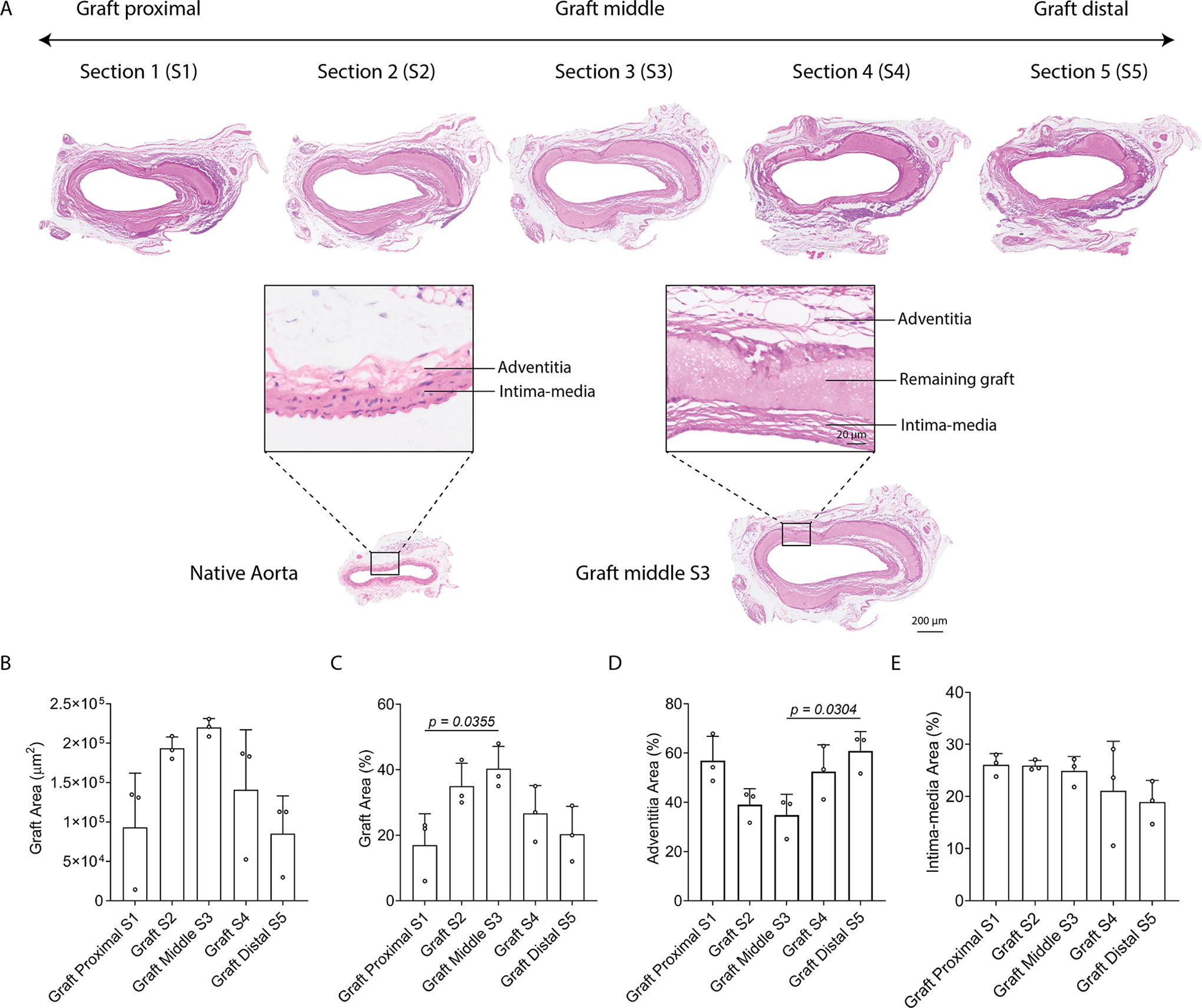
We observed substantial differences in the geometry and morphology of the graft middle and proximal sites (Fig. 3). At the graft middle, the remaining graft showed nonuniform degradation on the outer surface interfacing with the adventitia layer, with many pits present. In contrast, the inner surface interfacing with the intima-media layer displayed a smooth morphology. At the graft proximal sites, the majority of the grafted material had degraded, leaving behind cell-infiltrated graft fragments. Between these fragments, channels had formed. Surrounding the graft fragments, ECM regeneration was observed.
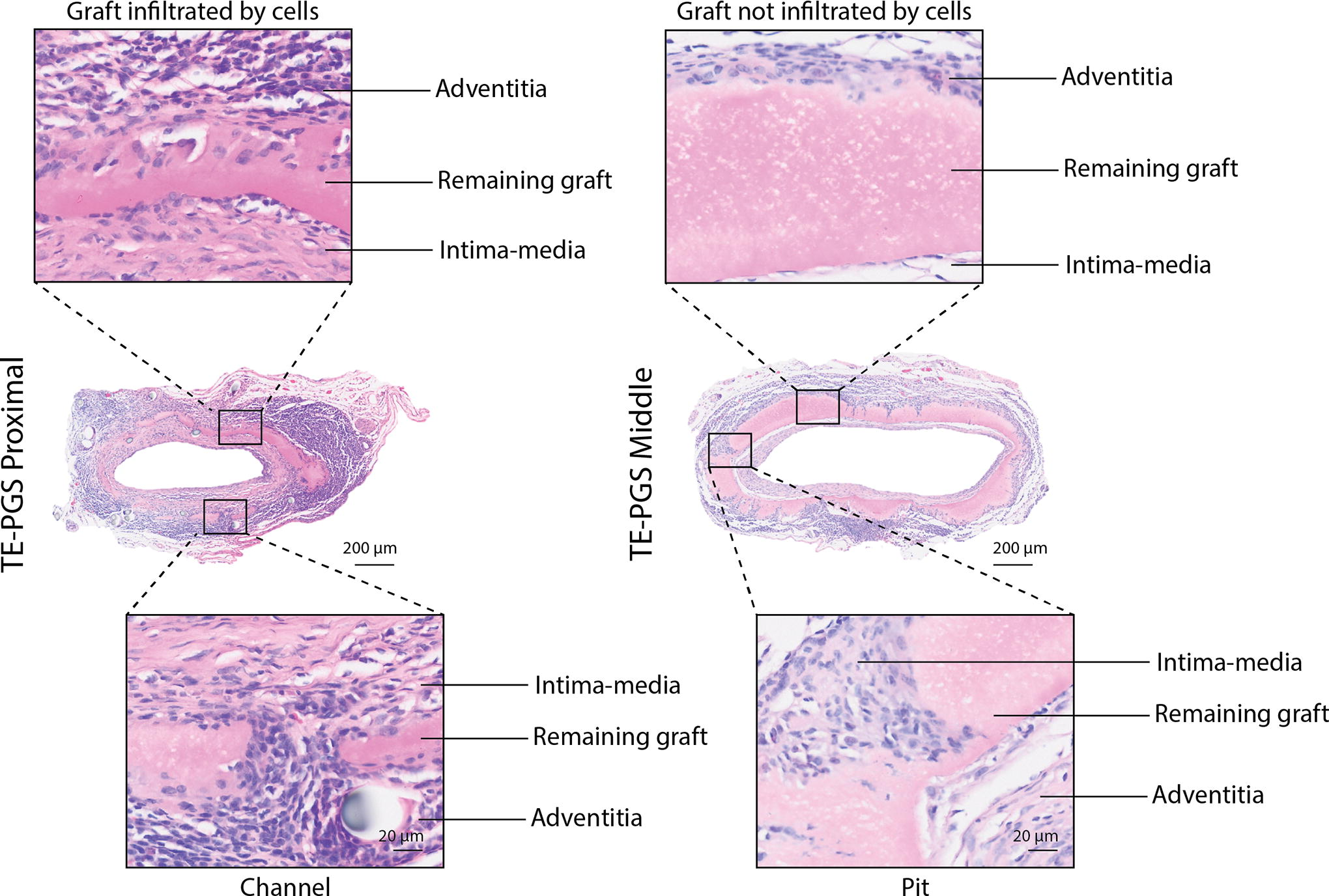
H&E staining of the explanted TE-PGS graft at the proximal and middle sites showing areas of graft infiltrated by cells and formed channel at the graft proximal and areas of graft not infiltrated by cells and formed pit at the graft middle.
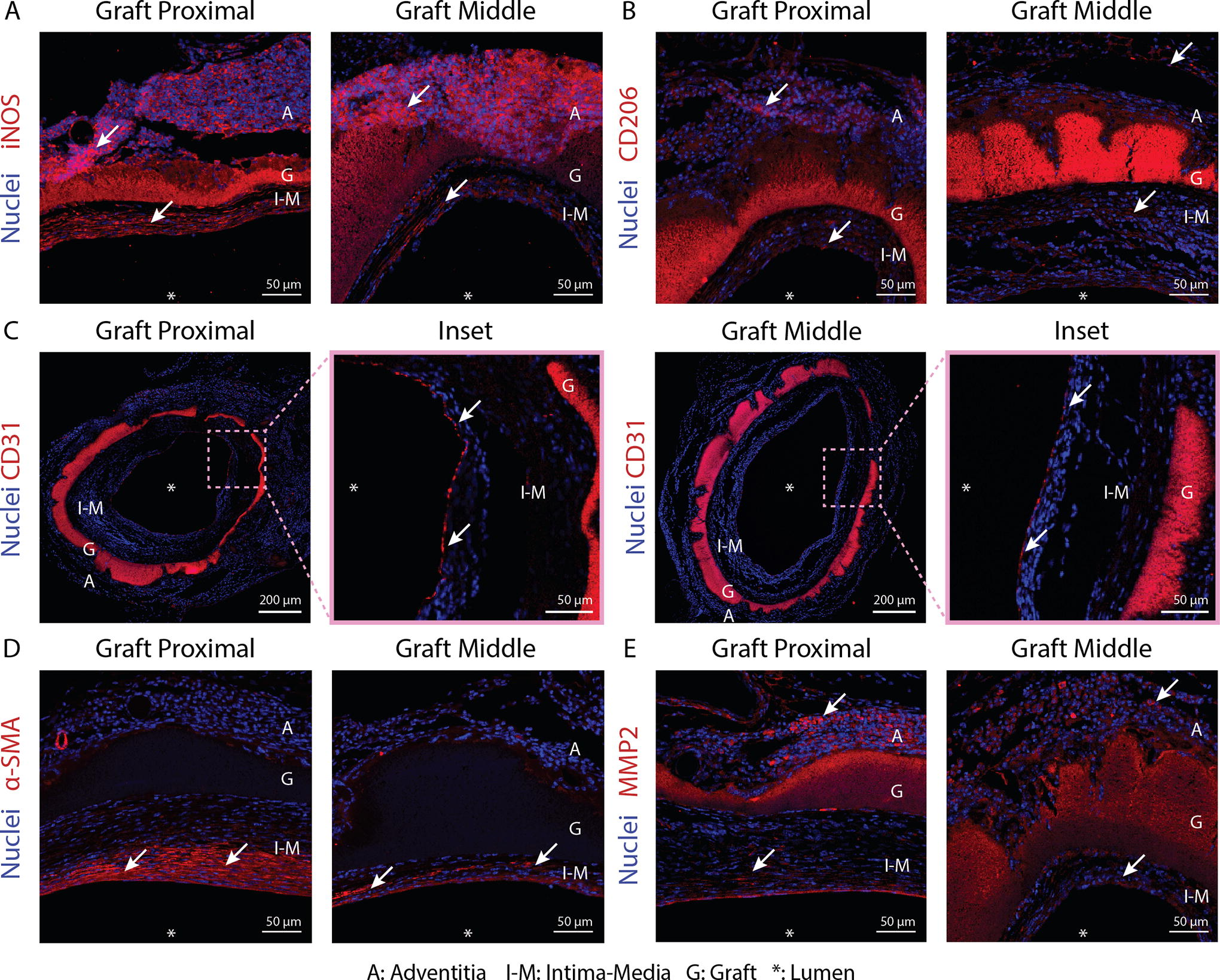
To understand the potential contribution of different cell types to graft degradation and regeneration, we used cell-specific markers to immunofluorescently stain for M1 macrophages (iNOS), M2 macrophages (CD206), endothelial cells (CD31), and SMCs (α-SMA). We observed that iNOS+ M1 macrophages were the predominant cell type in the adventitial and intima-media layers of both the graft proximal and middle regions, where they were found in close proximity to the graft including within the pits of the graft middle (Fig. 4A). CD206+ M2 macrophages were also found in these regions but at much lower numbers (Fig. 4B). For vascular cells, CD31+ endothelial cells covered the entire lumen at both the graft proximal and graft middle (Fig. 4C), while α-SMA+ SMCs were mostly observed in the intima–media at both graft proximal and middle (Fig. 4D).
Immunofluorescent staining for
As the graft contained TE, which is known to be degraded by members of the MMP family of enzymes, we stained for the presence of MMP2 (Fig. 4E). MMP2 positive staining was primarily seen in the adventitial layer and appeared to be more pronounced at the graft proximal than the middle. We observed a small amount of MMP2 staining in the intima–media at both the graft proximal and middle as well as a continuous layer of MMP2 expression in the endothelium at the graft proximal.
Collagen regeneration at graft middle and graft proximal sites
PSR staining indicated collagen had mostly been deposited within the graft adventitia (Fig. 5A). The quantification of PSR staining by percentage of total graft area showed collagen made up 46.3 ± 3.7% of the native aorta, 22.0 ± 11.2% of the graft middle, and 32.8 ± 1.7% of the graft proximal sites (Fig. 5B). These amounts correspond to 47.4% and 70.8% of total collagen being regenerated at the graft middle and the graft proximal sites, respectively, compared with the amount of collagen in the native aorta. The regenerated collagen in the graft appeared to be fibrillar and circumferentially aligned, whereas the collagen in the native aorta was dense and had a matured basketweave morphology.
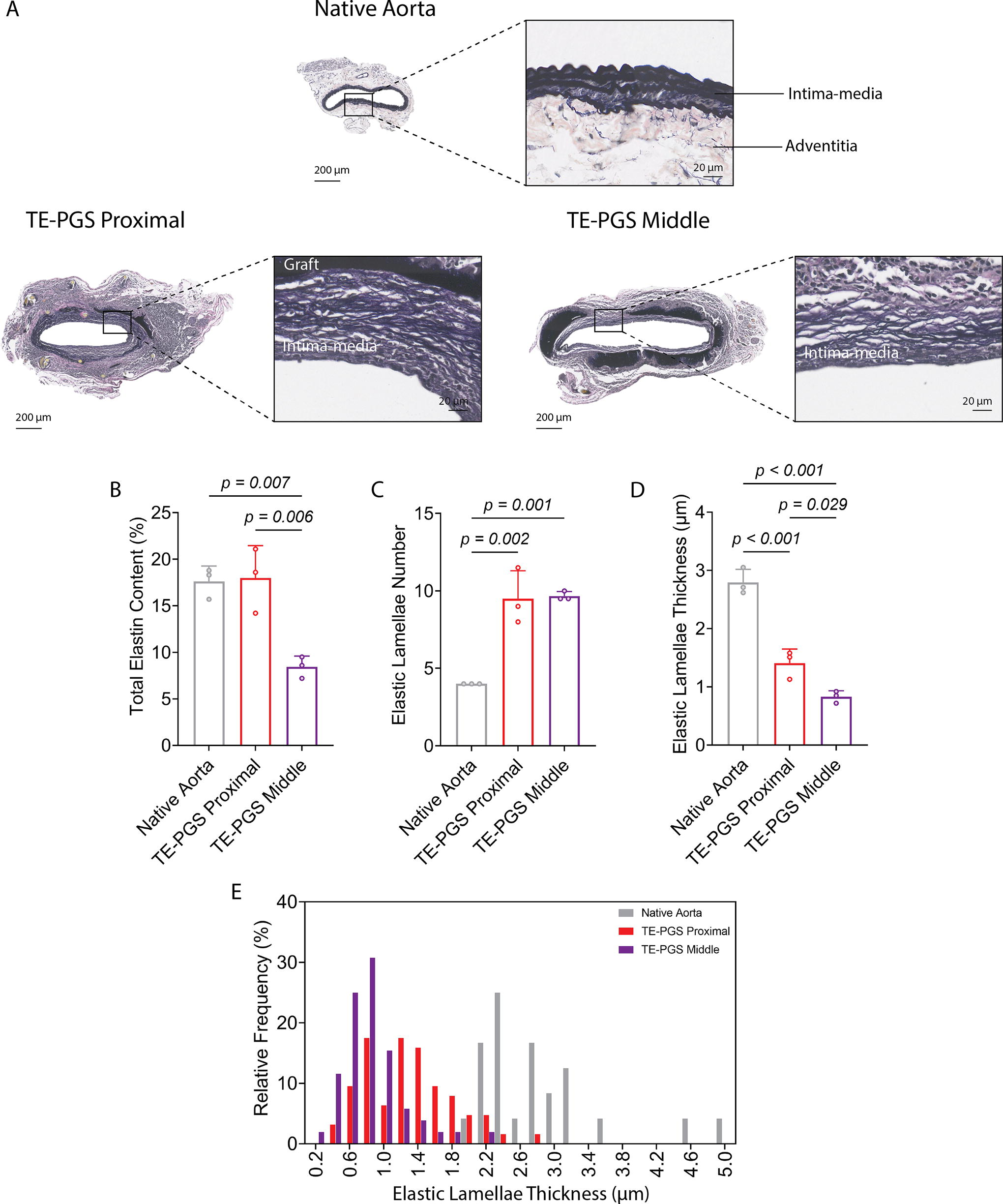
Organized elastin regeneration at graft middle and proximal sites
VVG staining for elastin indicated that substantial, continuous, and wavy elastic lamellae had developed within the intima–media regions of both the graft middle and graft proximal (Fig. 6A). The percentage of elastin within the cross-sectional area was 17.6 ± 1.4% in the native aorta and 18.0 ± 2.8% at the graft proximal site, both significantly higher than 8.4 ± 0.9% in the graft middle (Fig. 6B). About 10 elastic lamellae were found in the intima–media of the graft middle and proximal sites, which was significantly higher than four elastic lamellae in native aorta intima-media (Fig. 6C). The average thickness of the regenerated elastic lamellae at the graft proximal site was 1.4 ± 0.2 µm, which was about half the thickness of the native aorta elastic lamellae (2.8 ± 0.2 µm) (Fig. 4D). Interestingly, we found the average thickness of the regenerated elastic lamellae at the graft middle was 0.8 ± 0.1 µm, ∼60% thinner than at the graft proximal sites. A size distribution analysis showed a shift toward thicker elastic lamellae from graft middle to graft proximal sites (Fig. 6E).
Discussion
Vascular graft dilation is known to greatly influence hemodynamics, resulting in reduced blood flow rate, and generating turbulent flow that in turn contributes to further graft dilation. In this study, we observed a nonuniform dilation after aortic interposition implantation of TE-PGS vascular grafts in mice where the dilation was more pronounced at the graft middle compared to the graft proximal and distal. The difference in dilation between the graft proximal/distal and middle sites can be attributed to combinatory factors including graft degradation, cellular infiltration, and ECM regeneration. 18
The implanted TE-PGS grafts were fabricated with a consistent TE-PGS ratio and a uniform microstructure. 9 Therefore, similar surface erosion-based degradation mechanisms, including enzymatic degradation19,20 and phagocytosis, 9 might be expected at both the graft proximal and graft middle sites. In contrast, we observed faster degradation at the graft proximal. Within this region, we also observed increased staining for macrophages and more pronounced expression of the TE degrading metalloproteinase MMP2 compared with the graft middle region. Macrophages are known to secrete MMP2 21 and the enzymes lipase 22 and esterase, 23 which have been shown in a previous study to be the main enzymes responsible for PGS surface erosion. As macrophages are the dominant cells in the adventitial layer of the neoartery and more prominent in the graft proximal, it is likely that their enrichment in this region resulted in increased enzymatic and phagocytic activity, corresponding to faster graft degradation.
We observed pit formation on the outer surface of the graft middle and the presence of channels in the graft proximal region. The pits appeared to contain M1 macrophages. This is consistent with our earlier TE-PGS graft study, where iNOS+ M1 macrophages were found to be primarily responsible for graft degradation evidenced by their accumulation at the adventitia in close association with the degraded outer graft surface. 9 Here we further show that the degraded pit regions on the outer surface of the graft middle contain these phagocytes. This localization of phagocytic activity likely contributes to uneven graft degradation which in time leads to the formation of channels as seen at the graft proximal.
At the same time as graft degradation occurred, arterial regeneration was apparent through the accumulation of vascular cells and the deposition of ECM on both the inner and outer surfaces of the graft, forming intima-media and adventitia, respectively. Near the lumen, CD31+ endothelial cells had formed a complete endothelium at both graft proximal and graft middle. Interestingly, MMP2 expression was observed in a continuous layer within the endothelium at the graft proximal. Endothelial cells are known to express MMP2 during vascular remodeling. 24 In addition, aligned α-SMA+ SMCs were present in the intima–media at both graft proximal and graft middle. SMCs are known to secrete, organize, and remodel elastin to form elastic lamellae in response to aortic pulsatile pressure.25,26 VVG staining for elastin indicated that continuous and wavy elastic lamellae had developed within the intima–media region in this study, consistent with our previous results. 9 The formation of these organized elastic lamellae was likely driven by concentrically layered SMCs in the neointima interacting with TE and TE-based species that had been released from the graft. 9 The remodeling of the graft from amorphous to organized elastin was also likely facilitated by MMP activity, 27 consistent with the presence of MMP2 in the intima–media. PSR staining also showed that TE-PGS supported the regeneration of collagen in the adventitia, likely due to the presence of fibroblast-like cells and M2 macrophages. 9
Regional differences in graft degradation can be attributed to the delayed presence of phagocytic macrophages at the graft middle region. Macrophages that degrade the graft materials can migrate from either the adjacent arterial tissue via the anastomosis sites or from the surrounding connective or adipose tissues.28–30 The increased distance between the graft middle and the flanking arterial tissue means macrophage accumulation within this region takes longer to occur resulting in less pronounced phagocytic and enzymatic activity as evidenced by the presence of degraded pits in the graft middle compared to channels at the graft proximal. At the same time, we observed that the graft middle region also contained less collagen and thinner elastic lamellae compared with the graft proximal. This was likely due to migrating SMCs and adventitial cells also arriving later to the graft middle than to its proximal regions, 31 retarding elastin remodeling and organization and collagen accumulation. Together, the compromised structural and mechanical integrity of the graft middle due to pit formation coupled with the reduced deposition of a robust ECM led to more pronounced dilation at the graft middle compared with its proximal sites.
In this study, we implanted a short 2 mm long TE-PGS vascular graft, whereas the length of clinically used vascular grafts can reach up to 15 cm. 32 Increased length would likely further exacerbate the effects of regional imbalance in graft degradation and regeneration, posing concerns about long-term graft stability and performance. Therefore, targeted optimization strategies to promote the retention of graft structural and mechanical integrity and ECM regeneration should be implemented. Both mechanical and biological strategies to reduce dilation and improve TE-PGS graft performance could be utilized in future studies. Mechanical strategies should be aimed at increasing the physical resistance of the graft to dilation. Increasing the graft’s mechanical strength could be achieved by simply adding a sheath as reinforcement. Several studies have attempted to prevent PGS-based vascular graft dilation by adding a PCL sheath layer with a uniform thickness. These studies have shown promising results with reduced graft dilation in the short term4,5,33; however, a long-term study indicated graft dilation by 12 months. 34 This strategy could be improved by using a sheath with a thickness gradient where the sheath thickness gradually increases from the graft’s proximal to the middle region. In addition, biological-based approaches could aim to accelerate arterial regeneration at the middle of the graft by promoting the migration of vascular cells from adjacent tissues or the recruitment of circulatory arterial cell progenitors and immune cells with strategies to increase their differentiation efficiency toward mature arterial cells. This could be achieved through graft surface modifications that enhance cell attachment.35,36
Conclusion
This study elucidated the mechanism of TE-PGS vascular graft nonuniform dilation occurring after aortic interposition implantation in mice. The results suggest that regional differences in graft degradation and regeneration between graft proximal and middle sites led to the observed dilation. Our results provide an important basis for optimizing the graft design using mechanical and biological strategies to mitigate nonuniform dilation, thereby preparing TE-PGS graft for future evaluation in large animal models.
Footnotes
Acknowledgments
The authors acknowledge the technical and scientific assistance of Sydney Microscopy & Microanalysis, the University of Sydney node of Microscopy Australia.
Authors’ Contributions
Z.W., S.M.M., B.S.H., T.S., C.K.B., and A.S.W. conceptualized the project. Z.W., S.M.M., J.Z., K.M.B., T.Y., Y.M., and N.T.H.P. performed experiments and acquired data. Z.W., J.Z., and K.M.B. performed data analysis. Z.W., S.M.M., and A.S.W. drafted the article. Z.W., S.M.M., H.Z., K.M.B., T.Y., Y.M., N.T.H.P., B.S.H., T.S., C.K.B., and A.S.W. revised the article.
Disclosure Statement
A.S.W. is the founding scientist of Elastagen Pty. Ltd., now sold to Allergan, Inc., an AbbVie company.
Funding Information
This project is funded by the Australian Commonwealth Government Research Training Program Tuition Fee Offset and Stipend Scholarship (Z.W.) and National Health and Medical Research Council Investigator Grant 1195827 (A.S.W.).
