Abstract
Four human acellular dermal matrices (hADMs) were characterized in a nonhuman primate abdominal wall repair model by evaluating host immune response, vascularization, and incorporation into host tissues. AlloDerm™ (electron beam–sterilized hADM [e-hADM]), AlloMax™ (gamma beam–sterilized hADM, freeze-dried [g-hADM-FD]), DermaMatrix™ (hADM, freeze-dried [hADM-FD]), and FlexHD™ (ethanol-treated hADM [EtOH-hADM]) were each implanted in an abdominal wall–bridging defect in nonhuman primates (n = 3 animals/time point, n = 36 animals). Immunohistochemical and histological assessments were conducted on biopsies from each hADM at 1-, 3-, and 6-months postimplantation to assess vascularization (hematoxylin and eosin [H&E], CD31, alpha smooth muscle actin [αSMA], collagen IV), inflammatory/immune response (H&E, CD3, CD20, CD68), and collagen turnover (H&E, matrix metalloproteinase-9 [MMP-9]). MMP-9 immunolabeling was similar among different hADMs at 1 month; however, hADM-FD and EtOH-hADM showed higher total mean MMP-9-immunopositive areas at approximately 16% compared with <1% for e-hADM and g-hADM at 6 months postimplantation. Cells that stained positively for CD68, CD3, and CD20 were generally higher for hADM-FD and EtOH-hADM compared with other hADMs. The mean CD31-immunopositive area, CD31 vessel density, CD31 vessel diameter, and collagen IV–immunopositive area increased over time. Among all the hADM types, e-hADM had the highest mean (±standard deviation [SD]) CD31-immunopositive area at 1.54% ± 1.01%, vessel density at 7.86 × 10−5 ± 3.96 × 10−5 vessels/µm2, and collagen IV–immunopositive area at 2.55% ± 0.73% 1-month postimplantation. The pattern of αSMA immunolabeling varied among the hADMs. Histology showed that overall inflammation was mild at 1 month. Overall fibroblast repopulation and collagen remodeling increased over time from 1 to 6 months postimplantation. Fibroblast infiltration was minimal to mild at 1 month, with e-hADM showing the highest mean (±SD) score at 2.00 ± 0.00 compared with other hADMs. Only hADM-FD was not completely replaced by neotissue formation at 6 months postimplantation. All hADMs promoted vascularization, cell infiltration, and incorporation into host tissue, which were associated with acute inflammation and immune responses, within a 6-month period. A trend toward relatively enhanced early vascularization in e-hADM compared with other hADMs was observed. Immunogenic responses among the hADMs in the present study showed a slight distinction toward more quiescent terminally sterilized hADMs (e-hADM, g-hADM-FD) versus aseptically processed hADMs (EtOH-hADM, hADM-FD).
Impact Statement
Human acellular dermal matrix (hADM)-derived surgical meshes are used for reconstructive procedures, and their vascularization is essential for host tissue integration and maintenance of newly regenerated tissues. Few in-depth comparative studies evaluating vascularization in hADMs have been conducted. This study directly compared four commonly used hADMs (AlloDerm™, AlloMax™, DermaMatrix™, and FlexHD™) in a functional nonhuman primate model of abdominal wall defect by evaluating host immune response, vascularization, and hADM incorporation into host tissues at different stages of angiogenesis. Knowledge of the vascularization properties and host tissue response timeline of different hADMs may impact material selection and surgical outcomes.
Introduction
Rapid vascularization is essential for successful integration and performance of decellularized extracellular matrix (ECM)-based technologies like acellular dermal matrices (ADMs). 1 Such products are commonly used in homologous applications, including burns, breast procedures, and hernia repair, because their biochemical and biophysical properties allow ready integration and subsequent repair or regeneration of strong and healthy tissues.1,2 As various cell types migrate into these matrices and begin the remodeling process, maintaining the health of newly regenerated tissues is contingent upon nutrient delivery. 3 Efficient ingrowth of blood vessels, or angiogenesis, is thus imperative for preventing necrosis.
Rapid vascularization is associated with consistent surgical outcomes, as well as lower seroma incidence and resistance to infections.4–6 Blood vessels act as a direct conduit for immune cell migration to the implant site, allowing for swift responses to infections should they arise. 7 Therefore, a thoroughly vascularized construct can benefit patients at high risk of infection due to immunocompromised status or other comorbidities. 7 The combination of nutrient delivery and immune presence may accelerate the overall tissue regeneration process, allowing for better outcomes. Chung et al. 5 found that prevascularization of AlloDerm™, an electron beam–sterilized human-derived acellular dermal matrix (hADM), before implantation in a rat abdominal wall defect model resulted in lower hernia recurrence than use of AlloDerm direct from packaging, thus highlighting the clinical importance of vascularization.
Different hADM products undergo unique and proprietary processing methods (Table 1), including exposure to a variety of processing solutions, terminal sterilization methods, or subjugation to freeze-drying, which all likely have varying effects on ADM structural and biochemical properties.9,10,12 During the manufacturing process, ADMs are disinfected to minimize endotoxins, bacteria, and viruses. 14 Aseptic processing methods involve the use of chemical agents (e.g., alcohols, detergents, antibiotics), whereas terminal sterilization methods can involve the use of electron beam and gamma irradiation.14–16
Comparison of Processing Methods for Select Human Acellular Dermal Matrices
e-hADM, electron beam–sterilized hADM; g-hADM-FD, gamma beam–sterilized hADM, freeze-dried; hADM, human acellular dermal matrix; hADM-FD, hADM freeze-dried; EtOH-hADM, ethanol-treated hADM.
Several studies have compared existing hADMs using benchtop research and different animal models.9–12,15,17,18 These comparative studies with varying objectives and methods tend to be inconsistent with regard to rates of cellular infiltration, levels of inflammation, and rapidity of vascularization. Furthermore, few studies have attempted to analyze differences in vascularization at different time points or to delineate the identity of infiltrating cell subtypes, thus limiting the potential for mechanistic insights.
The present study compared four commonly used hADMs (Table 1) in an African green monkey hernia defect model by evaluating host immune response, vascularization, and hADM incorporation into host tissues. These objectives were achieved using immunohistochemistry, representing early-, mid-, and late-stage angiogenesis, as well as histology, applied to biopsies from each hADM sample at 1-, 3-, and 6-months postimplantation.
Materials and Methods
Biomaterials
Samples of AlloDerm (Allergan Aesthetics, an AbbVie Company, Branchburg, NJ), AlloMax™ (Bard, Warick, RI), DermaMatrix™ (Synthes, West Chester, PA), and FlexHD™ (MTF Biologics, Edison, NJ) were prepared according to the manufacturers’ instructions for use. From here on, AlloDerm will be referred to as electron beam–sterilized hADM (e-hADM), AlloMax as gamma beam–sterilized hADM, freeze-dried (g-hADM-FD), DermaMatrix as hADM, freeze-dried (hADM-FD), and FlexHD as ethanol-treated hADM (EtOH-hADM).
Primate abdominal wall repair model
One of four types of hADMs (n = 3 animals/time point) were randomly implanted in a full-thickness abdominal wall–bridging defect in wild-caught African green monkeys (Chlorocebus sabeaus, C. athiops sabeus; Worldwide Primates, St. Kitts, Eastern Caribbean) (N = 36).19–23 Briefly, a bilateral longitudinal full-thickness defect in the abdomen (≈3 × 7 cm) was repaired with a single piece of hADM that was trimmed to the equivalent size of the defect. Animals were euthanized, and the hADM and surrounding host tissue were excised at 1-, 3-, and 6-months postimplantation for further assessments. All procedures were approved by the Allergan Animal Care and Use Committee, following guidelines by the American Association for Laboratory Animal Science.
Immunohistochemistry and histology
Explanted hADM samples were placed in 10% neutral buffered formalin, embedded in paraffin blocks, and sectioned onto glass slides. Slides were stained with hematoxylin and eosin (H&E). The immunohistochemistry markers are summarized in Table 2.
Immunohistochemistry Markers
αSMA, alpha smooth muscle actin; EC, endothelial cell; MMP-9; matrix metalloproteinase-9.
H&E-stained slides were scored (0–4) for levels of inflammation, fibroblast repopulation, revascularization, and hADM collagen remodeling by a third-party board-certified veterinary histopathologist, as shown in Table 3. Anti-CD3-, anti-CD20-, and anti-CD68-stained slides were scored (0–4) internally by a subject matter expert (Table 3).
Histopathology Scoring Criteria
Adapted from Bolden et al. 23
ADM, acellular dermal matrix.
Matrix metalloproteinase-9 (MMP-9)-, alpha smooth muscle actin (αSMA)-, anti-CD31-, and anticollagen IV–stained slides were scanned using the Aperio AT2 whole-slide scanner (Leica Biosystems, Wetzlar, Germany), and image analysis was conducted using Visiopharm software (Hørsholm, Denmark). Implant tissue was identified on each slide and traced as a region of interest (ROI). Staining artifacts, folds/tears in the tissue, and other nonimplant tissue areas were excluded from ROIs. ROIs were subjected to imaging filters specific to 3,3'-diaminobenzidine chromogen and deconvoluted to the red, green, blue value for each pixel. A threshold was used to define pixels associated with positive immunolabeling.
For MMP-9-stained slides, the immunopositive area within each ROI was measured, compared with the total ROI area and reported as a percentage (% immunopositive area = (immunopositive area within ROI [µm2]/total ROI area [µm2]) × 100).
For αSMA-, CD31-, and collagen IV–stained slides, a filling technique was used to create an area inclusive of the immunolabeled structure and its encompassed lumen. Each of these areas was counted as a single vessel and used to calculate the immunopositive (vascular) area, vessel density (vessel density = vessel count within implant/total implant area [µm2]), and vessel diameter (CD31 only, minimum luminal diameter of each vessel, average vessel diameter [µm] = sum of all vessel diameters [µm]/vessel count within implant). In cases where large areas of high intensity, nonspecific immunopositive labeling was present, a second ROI was used to exclude these areas from total immunopositive area and vessel count calculations.
Statistical analyses
Data were summarized using descriptive statistics. Quantile–quantile plots were used to demonstrate normality in the datasets. For each experimental variable, linear mixed-effects models were applied, with the hADM group, time, and hADM group by time interaction as fixed effects and the animal identifications as random effects. Vessel density and vessel diameter were analyzed using an analysis of variance mixed-effect model with Satterthwaite’s Method for multiple post hoc comparisons. Statistical analysis was conducted using R programming software.
Results
Immunohistochemistry
Total MMP-9 (cell-associated and secreted)–immunopositive area
MMP-9, a collagen type IV collagenase secreted by neutrophils and macrophages, has an important role in neovascularization through the degradation of ECM proteins in the blood vessels and release of vascular endothelial growth factor (VEGF). 30 MMP-9 expression showed strong, diffuse cytoplasmic labeling of individual cells (cell-associated), as well as weak to moderate labeling throughout the ECM (secreted). Secreted MMP-9-immunolabeling often surrounded individual or clusters of immunolabeled cells but was sometimes observed throughout the sample. MMP-9 immunolabeling was similar among different hADM types at 1 month. However, hADM-FD and EtOH-hADM had higher total mean (± standard deviation [SD]) MMP-9-immunopositive areas at 16.01% ± 26.54% and 16.64% ± 26.29% at 6 months postimplantation, respectively, compared with 0.84% ± 0.56% and 0.57% ± 0.38% for e-hADM and g-hADM-FD, respectively, although these results were not statistically significant (Fig. 1a). The total mean MMP-9-immunopositive area for e-hADM and g-hADM-FD decreased over time between 1- and 6-months postimplantation. In contrast, the total mean (±SD) MMP-9-immunopositive area for hADM-FD steadily increased to 38.38% ± 13.17% at 3 months, then declined to 16.01% ± 26.54% at 6 months. The MMP levels for EtOH-hADM decreased to 1.03% ± 0.56% at 3 months but increased to 16.64% ± 26.29% at 6 months postimplantation, likely due to sample variability. Representative images of MMP-9-positive immunolabeling are shown in Figure 1b.
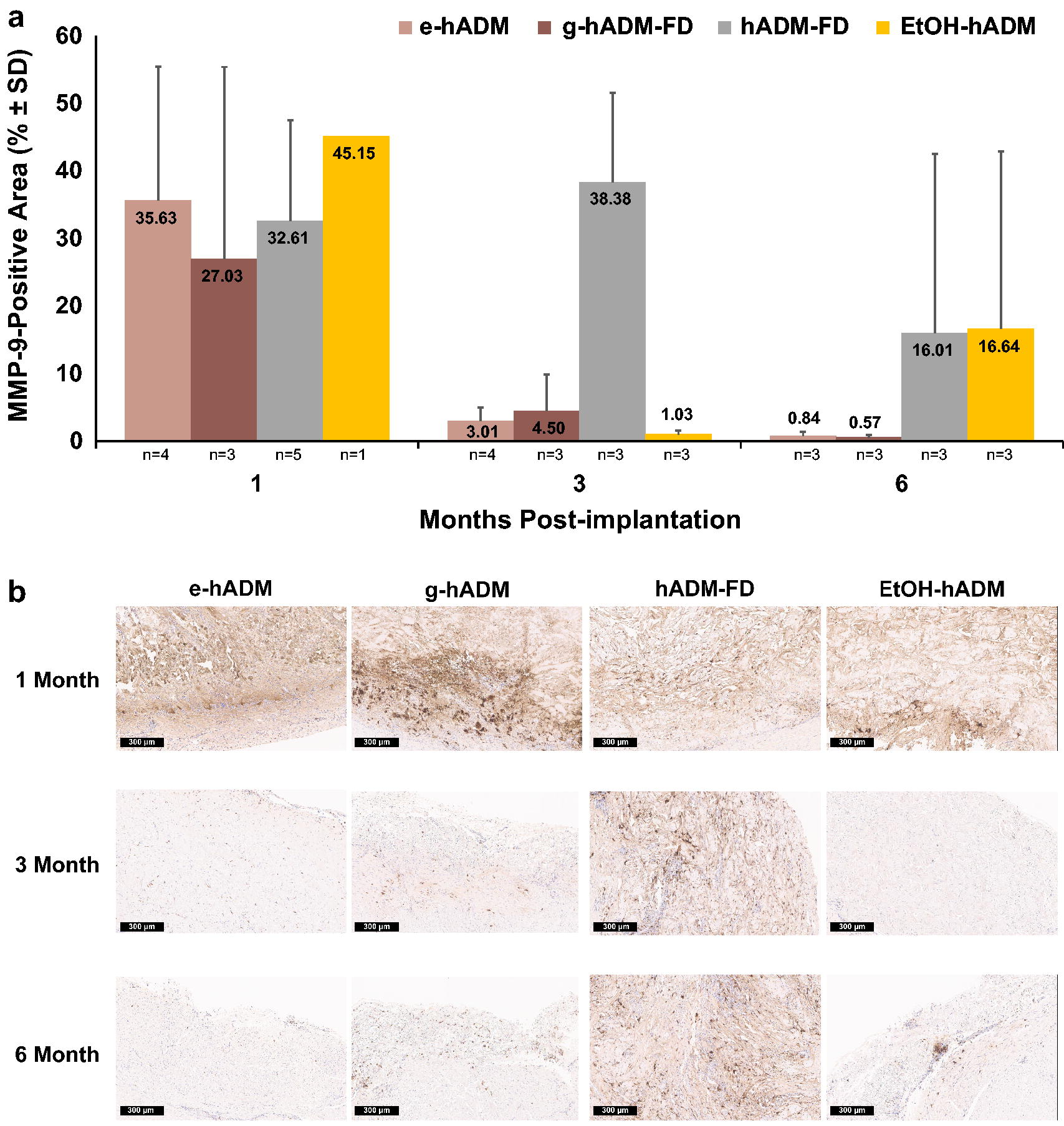
MMP-9 immunolabeling.
Immune cells (CD68, CD3, CD20)
Cells that stained positively for CD68, a cell marker for monocytes, macrophages, and multinucleated foreign body giant cells, were generally higher for hADM-FD and EtOH-hADM compared with e-hADM and g-hADM-FD for most time points (Fig. 2a). For e-hADM, g-hADM-FD, and hADM-FD, CD68-positive cells increased over time from mean scores of 0.9–1.1 at 1 month to 0.9–2.6 at 6 months postimplantation. For EtOH-hADM, mean scores decreased over time from 2.8 at 1 month to 1.7 at 6 months postimplantation.
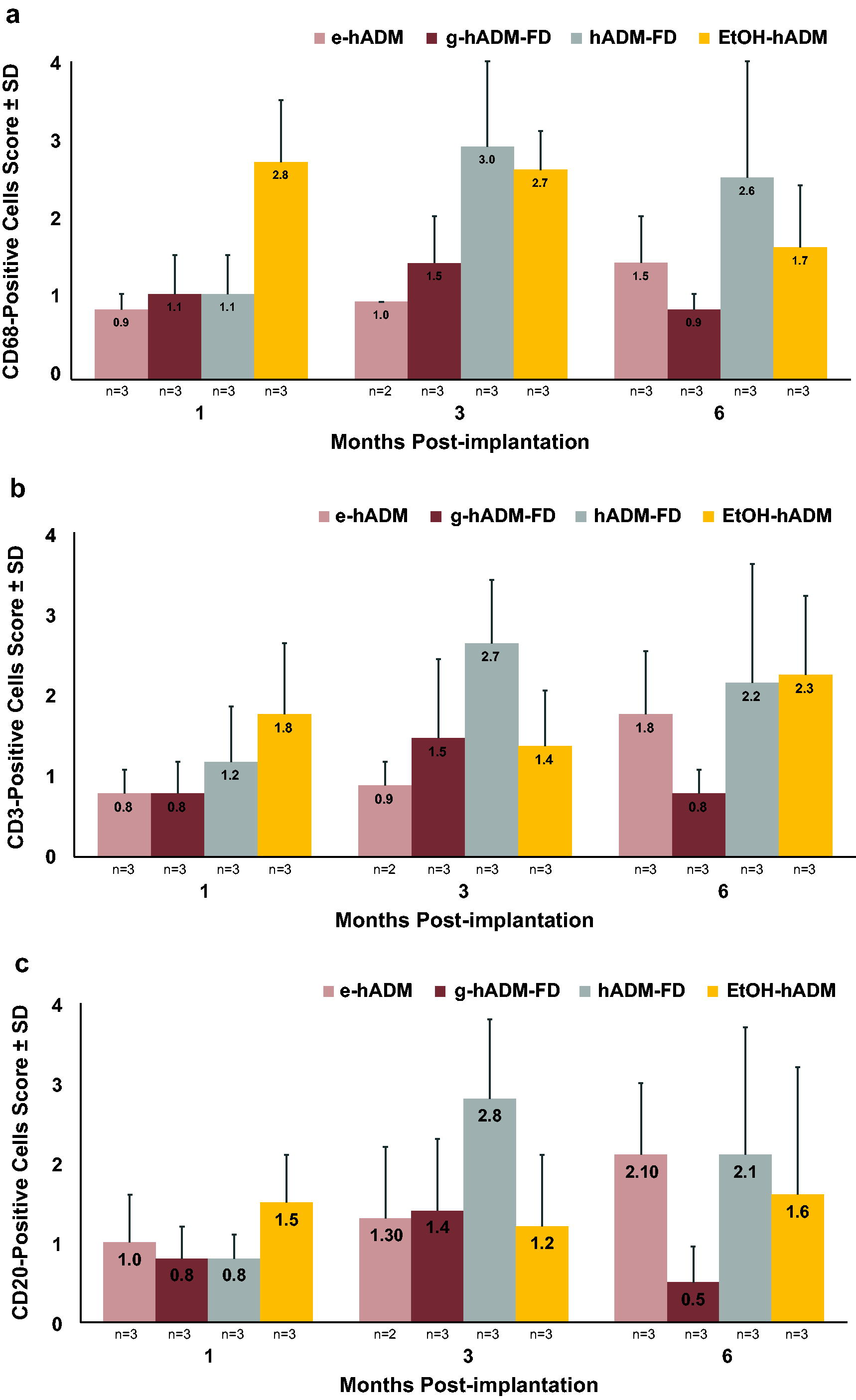
Immune cell response. Total mean (±SD) score for
Cells that stained positively for CD3, a T lymphocyte marker, were generally higher for hADM-FD and EtOH-hADM compared with e-hADM and g-hADM-FD for most time points (Fig. 2b). The pattern of CD3 positive cell staining varied among hADM types. Mean scores gradually increased over time from 1 to 6 months postimplantation for e-hADM; increased between 1 and 3 months, then decreased at 6 months postimplantation for g-hADM-FD and hADM-FD; and remained roughly constant between 1 and 3 months before peaking at 6 months postimplantation for EtOH-hADM. Cells staining positively for CD20, a B lymphocyte marker, showed a similar pattern to CD3 (Fig. 2c).
CD31-immunopositive area
CD31, a marker of endothelial cells (ECs), indicates early-stage angiogenesis.24,25 Among all the hADM types, e-hADM trended toward having the highest mean (±SD) CD31-immunopositive area at 1.54% ± 1.01% at 1 month, which remained steady at 1.50% at 3- and 6-months postimplantation (Fig. 3a). For all other hADM types, the CD31-immunopositive area increased over time from 0.56–0.96% at 1 month to 1.80–1.94% at 3 months, which remained steady at 6 months postimplantation.
CD31 and collagen IV immunolabeling.

(Continued).
CD31 vessel density
In general, vessel density was significantly different at 3 and 6 months compared with 1 month (p < 0.02; Fig. 3b). For all hADM types except e-hADM, the mean vessel density increased from 2.76 × 10−5–4.68 × 10−5 vessels/µm2 at 1 month to 7.46 × 10−5–7.71 × 10−5 vessels/µm2 at 3 months, then remained steady at 6 months postimplantation. Among all the hADM types, e-hADM showed the highest, though nonstatistically significant, mean (±SD) vessel density at 7.86 × 10−5±3.96 × 10−5 vessels/µm2 at 1 month, which decreased to 6.25 × 10−5 ± 1.54 × 10−5 vessels/µm2 and 6.13 × 10−5 ± 1.08 × 10−5 at 3- and 6-months postimplantation, respectively.
CD31 vessel diameter
Vessel diameter was significantly different at 3 and 6 months compared with 1 month (p = 0.005 and p = 0.019, respectively; Supplementary Fig. 1A). For all hADM samples, the mean vessel diameter gradually increased from 6.48–6.96 µm at 1 month to 7.16–8.01 µm at 3 months, and to 7.52–8.56 µm at 6 months postimplantation. At 3 months postimplantation, hADM-FD showed the lowest mean vessel diameter (±SD) at 7.16 ± 0.55 µm among all the hADM types (p < 0.05). Representative images of CD31 positive immunolabeling, vessel density, and diameter are shown in Figure 3c.
Collagen IV–immunopositive area
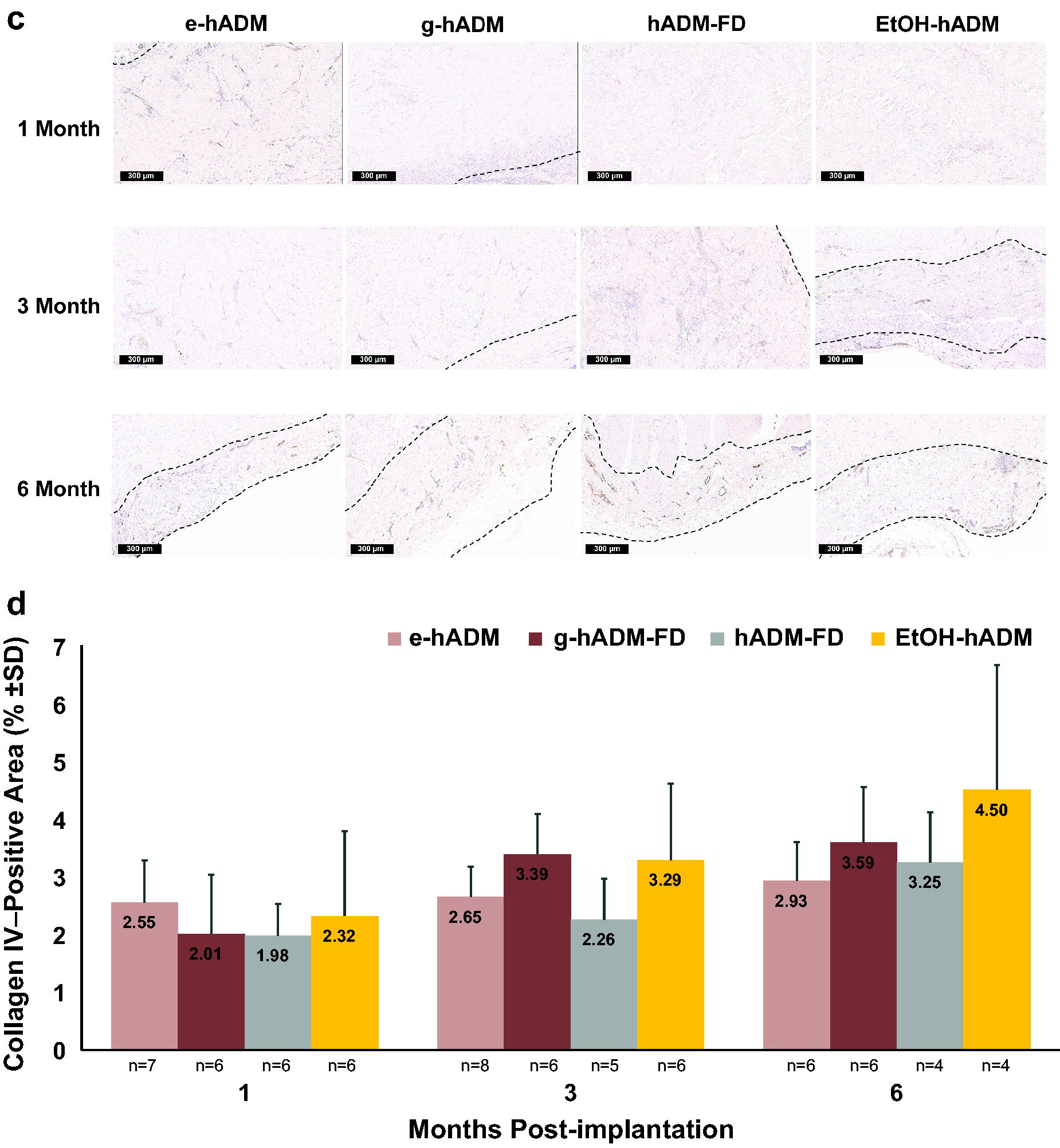
Collagen IV, which is secreted by ECs and fibroblasts, indicates midstage angiogenesis. 27 Among all the hADM types, e-hADM had the highest, though nonstatistically significant, mean (±SD) collagen IV–immunopositive area at 2.55% ± 0.73% at 1-month postimplantation (Fig. 3d). For all hADM types, the collagen IV–immunopositive area increased over time from 1.98–2.55% at 1 month, to 2.26–3.39% at 3 months, and to 2.93–4.50% at 6 months postimplantation.
αSMA-immunopositive area
αSMA, a marker of actively differentiating endothelial and smooth muscle cells, indicates late-stage angiogenesis.24,28,29 The trends of αSMA-immunopositive area over time varied among the hADM types. Although e-hADM had the lowest mean (±SD) αSMA-immunopositive area at 9.71% ± 7.49% at 1 month, αSMA-immunopositivity increased to 19.15% ± 3.85%, the highest among hADM types, at 6 months postimplantation (Supplementary Fig. 1B). The mean (±SD) αSMA-immunopositive area for g-hADM-FD increased from 17.78% ± 9.85% at 1 month to 25.83% ± 4.91% at 3 months, then decreased to 15.67% ± 7.16% at 6 months postimplantation. The mean (±SD) αSMA-immunopositive area for hADM-FD gradually increased over time from 11.70% ± 5.942% at 1 month to 13.63% ± 9.83% at 6 months postimplantation. In contrast, the mean (±SD) αSMA-immunopositive area for EtOH-hADM decreased over time from 17.55% ± 7.36% at 1 month to 11.22% ± 6.43% at 3 months, which remained steady at 6 months postimplantation.
Histology
Inflammation and inflammatory cells scores
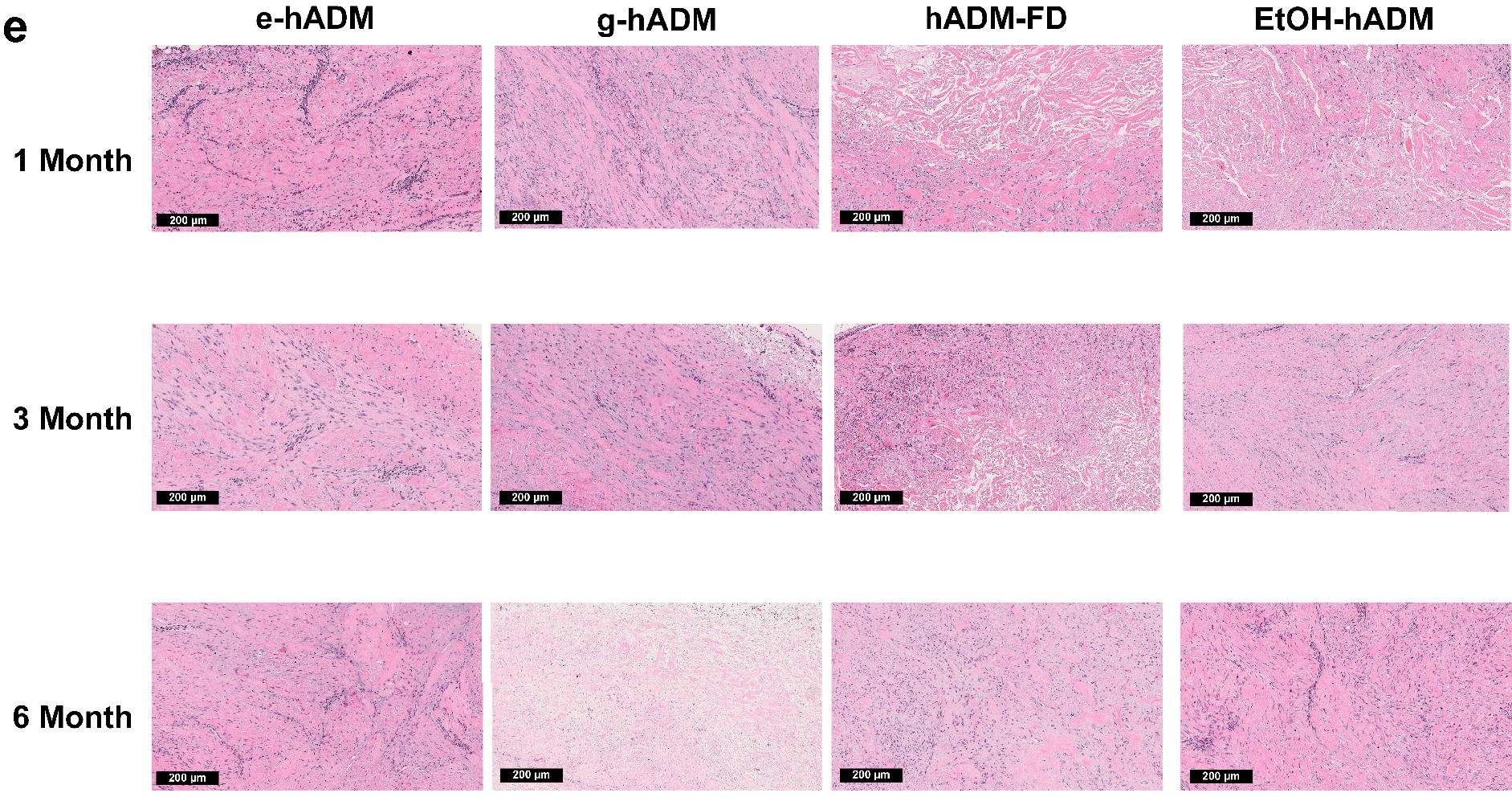
Mean (±SD) inflammation scores for e-hADM and g-hADM-FD decreased over time from 2.0 ± 0.8 and 2.0 ± 0.9, respectively, at 1 month, to 1.2 ± 0.4 and 1.0 ± 0.0, respectively, at 6 months postimplantation (Fig. 4a). In contrast, mean (±SD) inflammation scores for hADM-FD and EtOH-hADM increased from 2.0 ± 0.9 for both hADMs at 1 month to a peak of 3.0 ± 0.6 at 3 months and 2.3 ± 1.4, respectively, at 6 months postimplantation. Inflammation consisted of mostly lymphocytes, plasma cells, macrophages, and histiocytes, with fewer neutrophils, eosinophils, and multinucleated giant cells (Supplementary Fig. S2).
Histology scoring results. Total mean (±SD) score for

(Continued).

(Continued).
Fibroblast repopulation scores
At 1-month postimplantation, fibroblast infiltration was minimal to mild, with e-hADM showing the highest, though nonstatistically significant, mean (±SD) fibroblast repopulation score at 2.0 ± 0.0 compared with the other hADM samples (0.9 − 1.3; Fig. 4b). Fibroblast repopulation increased over time from 0.9–2.0 at 1 month, to 1.7–2.7 at 3 months, then remained steady at 6 months postimplantation. More specifically, scores increased for e-hADM and g-hADM-FD between 1 and 3 months, then decreased at 6 months postimplantation; scores for hADM-FD gradually increased from 1 to 6 months; and scores for EtOH-hADM increased between and 1 and 3 months, then remained steady at 6 months postimplantation.
Revascularization scores
At month 1 postimplantation, revascularization was minimal, with e-hADM and g-hADM-FD showing numerically higher mean (±SD) vascularization scores of 1.0 ± 0.6 and 1.3 ± 0.8, respectively, than hADM-FD and EtOH-hADM at 0.3 ± 0.4 and 0.7 ± 0.4, respectively (Fig. 4c). Revascularization scores increased over time from 0.3–1.3 at 1 month, to 0.9–2.0 at 3 months, and to 1.3–2.2 at 6 months postimplantation. More specifically, e-hADM, g-hADM-FD, and EtOH-hADM increased between 1 and 3 months, then remained steady at 6 months postimplantation; hADM-FD gradually increased from 1 to 6 months postimplantation.
Collagen remodeling scores
At 1-month postimplantation, collagen remodeling was minimal to notable, with g-hADM showing the highest, though nonstatistically significant, mean (±SD) collagen remodeling score at 2.00 ± 1.10 compared with other hADM types (Fig. 4d). For all hADM samples, collagen remodeling scores increased over time from 1.17–2.00 at 1 month, to 2.00–3.67 at 3 months, and peaked at 2.50–4.00 at 6 months postimplantation. Only hADM-FD was not completely remodeled at 6 months postimplantation, with a score of 2.50 compared with 4.00 for all other hADM samples. In all instances of hADM remodeling, the implants were replaced with collagen deposited at varying density levels, which demonstrates different levels of cellular organization. Representative H&E images are shown in Figure 4e.
Discussion
This study demonstrated similar levels of vascularization, as depicted by different markers of early-, mid-, and late-stage angiogenesis, across all four hADMs after 6 months of implantation in a functional nonhuman primate model. Notably, there was a trend for e-hADM constructs to promote faster vessel ingrowth in the first month, as demonstrated by CD31 and collagen IV immunostaining, which indicate early-24,25 and midstage 27 vascularization. Furthermore, although all the hADMs demonstrated collagen remodeling/turnover, hADM-FD was not completely remodeled by 6 months, and g-hADM-FD showed a trend toward having the fastest remodeling rate compared with other matrices at 1 and 3 months. None of the hADMs elicited an immune rejection response. Inflammation levels across all hADMs were moderate to low, although the two aseptically processed (AP)-hADMs (hADM-FD and EtOH-hADM) elicited somewhat more robust reactions than the terminally sterilized (TS)-hADMs (e-hADM and g-hADM-FD), as observed by the CD68 (monocytes, macrophages, and multinucleated foreign body giant cells marker) and CD3 (T lymphocyte marker) immunostaining results.
In this study, mean CD31 vessel diameter measurements peaked at 8.56 µm. Because previous studies have used a diameter <8 µm as a threshold for capillary vessels in macaques, 32 the majority of ingrown vessels likely represent capillaries. Immunolabeling for αSMA, which marks vascular smooth muscle cells that form concentric layers around later-stage, more mature vessels, 29 did not yield a consistent conclusion across time points or hADM types. However, αSMA is also known to stain myofibroblasts, mesenchymal stem cells, myoepithelial cells, actively differentiating ECs, and other cells;24,28 therefore, some nonvascular smooth muscle cell staining may have been observed.
CD31 indicates early angiogenesis because it stains ECs, the earliest players in new vessel formation.24,25 ECs typically sprout from existing blood vessels, migrate outward, and form a network of new solid cellular cores that eventually develop internal lumens with diameters that enlarge over time.25,33,34 Although overall revascularization via H&E staining at 1 month suggested elevated vessel presence in g-hADM-FD, followed by e-hADM, more targeted and quantitative metrics of early vascularization, the percentage of CD31-immunopositive area, and CD31 vessel density highlighted an enhanced vessel presence in e-hADM, ranging from 0.5 to 3 times that observed in other hADMs at 1 month. Other hADMs appear to catch up to, and in some cases surpass, e-hADM in CD31 and revascularization metrics by 6 months. These results suggest that early vascularization may be associated with the use of e-hADM.
The staining patterns of collagen IV, a basement membrane protein secreted to a limited extent by ECs and a marker of midstage angiogenesis, 27 support these observations. Collagen IV staining was higher in e-hADM than other hADM groups at 1 month, though this vascularity marker became more similar across hADM types by 6 months postimplantation. Collagen IV is also secreted to a greater extent by fibroblasts, 26 which migrate into acutely injured sites to assist in matrix remodeling and cell recruitment via secretion of MMPs, and cytokines, such as fibroblast growth factor (FGF), transforming growth factor-beta (TGF-β), and VEGF.35,36 Notably, fibroblast repopulation scores were also greater by up to 2× in e-hADM compared with other hADMs at 1 month. Early invasion of these cells has been shown to synergistically promote EC migration and proliferation, resulting in denser, faster-forming, and more stable vascular networks. 37 A subset of fibroblasts may ultimately become perivascular cells, a stabilizing component of the structure and diameter of early vessels.25,38 A trend toward stabilization of vessel diameters was observed in e-hADM between 3- and 6-months postimplantation. Together, the CD31, collagen IV, and fibroblast infiltration data support early vascularization in e-hADM at 1 month, which may affect tissue integration and infection resistance.
Variations in the ADM processing methods may account for some of the observed differences in early vascularization (Table 1).9,10,12 The use of chemicals (e.g., sodium hydroxide, acetone, detergents), irradiation, and freeze-drying may impact ADM-endogenous growth factors and other cytokines12,14 and contribute to the underlying differences in early vascularization. Sodium hydroxide, which has been shown to remove growth factors, is used to treat g-hADM.12,14 Carruthers et al. 17 specifically measured lower initial levels of basic fibroblast growth factor (bFGF), VEGF, and TGF-β per dry weight in g-hADM-FD compared with e-hADM. Freeze-drying, which has been shown to negatively affect growth factor functionality unless excipients are provided for stabilization, 39 is used for processing both hADM-FD and g-hADM-FD. 8
Processing methods also may affect ECM ultrastructure and biochemical components, ultimately impacting multiple aspects of cellular invasion and interaction. During processing, g-hADM-FD and EtOH-hADM are immersed in acetone and ethanol, respectively.9,11 Acetone and ethanol are commonly used as dehydration and fixative agents that can cause unnatural crosslinks, resulting in stiffer tissues that are potentially difficult to break down enzymatically.12,14 Sandor et al. 11 demonstrated that EtOH-hADM possesses greater mechanical strength out-of-package but faster benchtop degradation when subjected to collagenase compared with native dermal tissues. Surfactants/detergents, such as deoxycholate and Triton-X, may also result in selective loss of certain ECM components, such as glycosaminoglycans (GAGs).14,40 Triton-X and sodium deoxycholate are used for processing EtOH-hADM and e-hADM, respectively. 10 Although GAGs have been shown to be present in measurable quantities in e-hADM, the levels are lower than that in g-hADM-FD. 40 Although the present study used a prehydrated form of e-hADM, an earlier freeze-dried version of e-hADM previously demonstrated superior growth factor retention compared with g-hADM-FD, particularly of bFGF, VEGF, and TGF-β1. 17
Finally, both freeze-drying and sterilization processes can cause damage to ECM proteins and changes to ultrastructures, 14 such as collapse of open spaces through which cells might migrate. Pore collapse can be severe if these treatments are performed without appropriate supportive excipients (e.g., disaccharides), and particularly when ECM structures have already been damaged by other processing steps that cause protein elimination or denaturing.14,15,39 For example, g-hADM-FD is terminally sterilized via g-beam irradiation to sterility assurance level (SAL) 10−6, and e-hADM is sterilized via e-beam irradiation to SAL 10−3.9,11,41
Overall, g-hADM-FD represents the construct with the most expected process-derived damage considering its exposure to the combination of acetone, freeze-drying, and sterilization. In a previous study, g-hADM-FD was found to degrade more quickly in benchtop enzymatic testing compared with other hADM constructs that were exposed to only g-beam or only freeze-drying. 11 In this study, g-hADM-FD showed a trend toward having the highest collagen remodeling scores at 1 and 3 months, despite similar MMP-9 levels at early time points. EtOH-hADM exhibited the slowest remodeling of ADMs tested. Although this may be a function of its processing, and particularly crosslinks resulting from EtOH exposure, gross images also show a pattern of material bunching in about 50% of nonhuman primate samples. Bunching, which may be related to intrinsic material properties, can reduce the ADM surface area in contact with underlying tissues, thus reducing overall biological interactions, and potentially creating dead spaces for possible seroma formation.
Alterations in ECM ultrastructure due to various processing regimens may influence how hADMs are recognized by the immune system and specific cell types. Immunogenic responses among the hADMs in this study showed a slight distinction between TS-hADMs (e-hADM, g-hADM-FD) and AP-hADMs (EtOH-hADM, hADM-FD). While inflammatory scores were initially high across all constructs, likely representing a response to the initial surgical injury and ongoing reaction to a foreign implant, inflammatory scores subsided at later time points in the TS-hADMs. By comparison, inflammation scores remained at moderate or high levels in the AP-hADMs at 3- and 6-months postimplantation. The staining pattern of MMP-9, which is secreted by neutrophils and macrophages, 30 aligned with inflammation results.
Findings from other immune subtypes support prolonged inflammation in AP-hADMs compared with TS-hADMs. Although staining showed fluctuations over time, lymphocytes, which can react to ECM proteins that are sufficiently denatured, showed a trend to be slightly higher in the AP-ADM hADM-FD versus other hADMs at 6 months, which suggests prolonged inflammation. Cells staining positively for CD20, a B lymphocyte marker, trended similarly. CD68+ staining for monocytes, macrophages, and multinucleated foreign body giant cells was relatively lower in TS-hADMs than AP-hADMs across multiple time points. Multinucleated giant cells, an eventual byproduct of macrophages attempting to phagocytize persistent foreign materials, 42 were generally low across all hADMs, although hADM-FD showed relatively higher scores than other hADMs at 3 and 6 months postimplantation. From among the hADM types, hADM-FD had the slowest resorbing matrix, and thus may be the only hADM with sufficient remaining collagen to trigger phagocytosis.
Trends among other immune subtypes were less obvious. No clear pattern or differences were observed in histocytes or plasma cell responses among hADMs. Innate immune cells (neutrophils and eosinophils) were sparsely present across all time points, with consistently low histology scores averaging ≤0.5 on a 4-point scale. Among all the samples, e-hADM had 1- and 3-month histology scores of 1.00 and 0.86, respectively, for eosinophils and 1.14 and 1.00, respectively, for neutrophils. A small presence of these cells may influence angiogenic functions because these cells release substantial quantities of VEGF, bFGF, interleukin (IL)-10, and IL-17 and enhance EC proliferation.43,44 These results may therefore support early vascularization of hADM.
This study may be limited by several experimental factors. Subjects were wild-caught African green monkeys possessing inherent genetic heterogeneity, with statistical strength being further confounded by relatively small sample sizes (i.e., three animals/material/time point) and sample variability. Vessel density and diameter analyses were complicated by the possibility of multiple orientations within a planar cross-section, along with shortcomings of vessel identification algorithms and limits to antibody detection accuracy across species. Furthermore, the release of growth factors and interleukins was not specifically evaluated in this study, but future experiments can be performed. Nonetheless, this study is valuable for its direct comparisons of four commonly used hADMs in a clinically relevant animal model with more in-depth analysis of several mechanistic factors.
Conclusion
All hADMs demonstrated the ability to promote vascularization, cell infiltration, and incorporation into host tissues within a 6-month period. Slightly elevated inflammatory responses of AP-hADMs at later time points, mild differences in rates of hADM remodeling, and somewhat faster development of early-stage vessels in e-hADM were observed, all of which may be attributed to variable processing techniques. Although these differences may exist during early stages of hADM incorporation, they are important in differentiating the timeline during which ADMs may start providing appropriate mechanical and cellular immune support into surgical sites that are prone to necrosis and infection.
Data Sharing
Data reported in this article are available within the article and its supplementary materials. Additional data may be requested by contacting AbbVie Inc.
Footnotes
Acknowledgments
Statistical analysis was provided by Mufeng Hu and Yan Sun of AbbVie, Inc. Histopathology scoring was provided by CBSET, Inc (Lexington, MA), and immunohistochemistry staining and image analysis were provided by Inotiv, Inc. (West Lafayette, IN).
Authors’ Contributions
J.F.: Methodology, writing—original draft, and writing—review and editing. J.L.: Conceptualization, data curation, formal analysis, methodology, writing—original draft, and writing—review and editing. V.S.: Writing—original draft, writing—review and editing, and data analysis. M.G.S.: Conceptualization, methodology, project administration, supervision, writing—original draft, and writing—review and editing.
Disclosure Statement
J.F., J.L., V.S., and M.G.-S. are full-time employees of AbbVie.
Funding Information
Allergan Aesthetics, an AbbVie Company, funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided by Maria Lim, PhD of Peloton Advantage, LLC (an OPEN Health company) and funded by Allergan Aesthetics, an AbbVie Company.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
