Abstract
Therapy with mesenchymal stem cells (MSCs) is considered an attractive strategy for the repair or regeneration of damaged tissues. However, low survival of MSCs limits their applications clinically. Oxidized low-density lipoprotein (ox-LDL) is significantly increased in patients with hyperlipidemia and decreases the survival of MSCs. Bcl-2 is critically involved in important cell functions, including cell membrane integrity and cell survival. The present study was designed to test the hypothesis that ox-LDL attenuates the survival of MSCs through suppression of Bcl-2 expression. Bone marrow MSCs from C57BL/6 mice were cultured with ox-LDL at different concentrations (0–140 μg/mL) for 24 h with native LDL as control. Ox-LDL treatment substantially decreased the survival of MSCs dose-dependently and enhanced the release of intracellular lactate dehydrogenase (LDH) in association with a significant decrease in Bcl-2 protein level without change in BAX protein expression in MSCs. Bcl-2 overexpression effectively protected MSCs against ox-LDL-induced damages with preserved cell numbers without significant increase in LDH release. Treatment with N-acetylcysteine (NAC) (1 mM) effectively preserved Bcl-2 protein expression in MSCs and significantly attenuated ox-LDL-induced decrease of cell number and increase in the release of intracellular LDH. These data indicated that ox-LDL treatment resulted in a significant damage of cell membrane and dramatically decreased the survival of MSCs dose-dependently through inhibition of Bcl-2 expression. NAC treatment significantly protected MSCs against the damage of cell membrane by ox-LDL and promoted the survival of MSCs in association with preserved Bcl-2 expression.
Impact Statement
Therapy with mesenchymal stem cells (MSCs) is considered an attractive strategy for the repair or regeneration of damaged tissues. However, one of the critical limitations for therapies with MSCs is the poor survival in vivo after cell delivery. Oxidized low-density lipoprotein (ox-LDL) is present in circulation and some organ systems that is significantly increased in patients with hyperlipidemia or diabetes mellitus. The present study revealed (1) ox-LDL could contribute to the poor survival of MSCs because of decreased expression of Bcl-2, and (2) treatment with N-acetylcysteine could effectively preserve Bcl-2 protein expression in MSCs and rescue MSCs against ox-LDL-induced damage. Targeting ox-LDL or Bcl-2 and/or NAC treatment could preserve MSCs and enhance the therapeutic efficacy of MSC’s treatment with improved outcome.
Introduction
Cell-based therapy with stem cells, including mesenchymal stem cells (MSCs) and endothelial progenitor cells, remains a viable strategy for the repair or regeneration of damaged tissues.1–3 MSCs are readily available from many sources, including bone marrow, adipose tissue, brain, liver, kidney, pancreas, muscle, and lung.4,5 MSCs are considered an ideal source for cell therapy for multiple reasons, including their easy availability without ethical concerns and little (if any) immunogenicity for adverse immune reactions because of their inhibitory and/or modulatory effects on immune system. 1 However, one of the critical challenges to therapies with stem cells, including MSCs, is their poor in vivo survival after delivery to the target tissues. Studies have demonstrated that <1% of mesenchymal stromal cells remained in the organ systems, including lung, heart, kidney, liver, spleen, and gut, for longer than a week following systemic delivery.2,6 It has been shown that <10% of stem cells were present or viable in the target areas 4 weeks after in vivo delivery directly into the ischemic areas (via intracoronary or intramyocardial injection).2,6
Although the mechanism(s) for the dismal in vivo survival of transplanted stem cells is complex and has not been fully defined, oxidative stress has been widely believed as an important factor that significantly contributes to the poor survival of stem cells after in vivo delivery. 7 Oxidized low-density lipoprotein cholesterol (ox-LDL) is a natural and potent oxidant in circulation and within vascular wall and is significantly increased in hyperlipidemic state. 8 It has been shown that prolonged high-fat diet selectively decreased the numbers of lin−/CD117+ cells (a subpopulation of MSCs) in the bone marrow of aging mice because of generation of excessive reactive oxygen species (ROS). 9 Intravenous administration of ox-LDL significantly decreases the numbers of bone marrow stem cells and endothelial progenitor cells in mouse models. 10 Previous studies have demonstrated that ox-LDL substantially attenuated the function and survival of murine bone marrow multipotent adult progenitor cells (MAPCs) in vitro via multiple mechanisms such as excessive ROS production, decreased Akt signaling, and cell membrane damage.11–13 Thus, attenuation of excessive ROS production with N-acetylcysteine (NAC) only partially protected MAPCs against low-dose ox-LDL in vitro. 14
Cell membrane is critically involved in important cell functions and survival. The family of Bcl-2 proteins significantly contributes to the control of various vital cell functions such as cell membrane integrity and cell survival. 14 Bcl-2 is present in many cells, including cardiomyocytes, T cells, and stem cells, together with human embryonic stem cells, neural stem cells, as well as pluripotent adult stem cells.15,16 It has been revealed that ROS and oxidative stress play an important role in the regulation of Bcl-2 expression. 17 The present study was designed to test the hypothesis that ox-LDL could impair the survival of MSCs through suppression of Bcl-2 expression. There were three specific objectives for the present study: (1) to investigate the effect of ox-LDL on the survival of MSCs and the protein expression of Bcl-2 in MSCs in vitro, (2) to determine if overexpression of Bcl-2 could protect MSCs against ox-LDL, and (3) to determine if NAC could preserve the expression of Bcl-2 and protect MSCs against ox-LDL.
Materials and Methods
Preparations of LDL and ox-LDL
Both native LDL and ox-LDL were purchased from Yiyuan Biotechnologies (YB-001 and YB-002-1). Native LDL was obtained and prepared from the plasma of healthy human subjects with centrifugation (1.019–1.063 g/cc) as described.
18
To prepare a highly oxidized LDL, freshly prepared native LDL was oxidized using Cu2SO4 (5 μM) at 37°C. After 3 h, the oxidation of LDL was terminated by adding ethylenediamine tetraacetic acid (EDTA)-Na2 (final concentration of 0.25 mM). The degree of LDL oxidation was determined colorimetrically by measuring the production of thiobarbituric acid reactive substances (TBARS) using malondialdehyde as a standard as described.19–21
To ensure the quality of the preparations and reproducibility, the TBARS value for LDL preparation before the initiation of LDL oxidation was 0.5
Cell culture
Bone marrow MSCs from C57BL/6 mice were purchased from Cyagen OriCell (MUBMX-01001). MSCs were stable phenotypically, and stained positive for CD29, CD44, and Sca-1 and negative for CD117 and CD31. These cells have been shown to have a doubling time of <72 h and exhibit the capability of multidirectional differentiation, including bone, fat, and cartilage tissues as described.22–24 Cells were cultured in the complete MSC medium (Cyagen OriCell, MUXMX-90011) and were kept at a density of 2.5 × 104 cells/cm2 at 37°C with humidified gas mixture of 5% carbon dioxide and room air.
To determine the effect of ox-LDL on the survival of MSCs, the cells were cultured at a density of 4 × 104 cells/cm2 (8 × 104 cells/well in a 24-well plate) in the presence of highly oxidized LDL at different concentrations (0–140 μg/mL, final concentration) for 24 h. Native LDL was used as the control. The cell survival was then evaluated by Cell Counting Kit-8 (Bimake, B34304) 24 h after incubation with ox-LDL as described. 25
Cell membrane damage measurement
Intracellular lactate dehydrogenase (LDH) could not penetrate an intact cell membrane; thus, release of intracellular LDH from cells is a reliable marker for cell membrane damages. To determine the effect of ox-LDL on cell membrane integrity, MSCs were maintained at a density of 4 × 104 cells/cm2 (8 × 104 cells/well in a 24-well plate) in the presence of ox-LDL with the final concentrations from 0 to 140 μg/mL for 24 h. Same amount of native LDL was used as the control. The LDH Cytotoxicity Detection Kit (cat. #MK401; Takara) was used to measure the concentrations of LDH in the media of MSCs 24 h after culture with ox-LDL as per the manufacturer’s recommendation.
Measurement of intracellular ROS
Mouse bone marrow MSCs were placed in 35-mm glass culture dishes at a density of 4 × 104 cells/cm2. After 24 h of culture, culture medium was replaced with fresh medium without fetal bovine serum (FBS). The ROS detection reagent 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) (10 μM, Beyotime, S0033S) was added into the media for intracellular ROS measurement as per the manufacturer’s protocol. After 20 min of incubation at 37°C, the cells were treated with ox-LDL (with the concentrations of 0, 60, and 100 μg/mL) for 1 h with phosphate-buffered saline (PBS) as control with or without NAC (1 mM). After thoroughly washing with PBS, the cells were examined using a fluorescent microscope, and the DCFH-DA intensity was analyzed by ImageJ. Each experiment was conducted at least three times.
Western blot analysis for Bcl-2 and BAX expression in MSCs
Proteins were prepared from mouse MSCs using centrifugation at 14,000 g for 10 min after lysis with a cell lysis buffer (Cell Signaling #9803). Protein concentration was quantitatively measured with the bicinchoninic acid assay as described. 26 A total of 30 μg protein preparation for each sample was loaded into 10% acrylamide sodium dodecyl sulfate (SDS) gel for electrophoresis. The preparations were transferred to 0.45 mm polyvinylidene difluoride (PVDF) membranes (Immobilon-P) and then incubated with 5% nonfat milk. After blocking with milk for 1 h, the preparations were exposed to the primary antibody for total Bcl-2 (1:1000, Proteintech), BAX (1:2000, Proteintech), and beta actin antibody (1:2000, Proteintech), followed by incubation with horseradish peroxidase (HRP)-conjugated secondary antibodies. After exposure to the chemiluminescent substrate (Advansta K-12045-D10), the protein bands were detected with X-ray film exposure (Amersham Imager 600) and analyzed using ImageJ software. Each experiment was conducted at least three times.
Cell transfection with Bcl-2 plasmid
Mouse MSCs were transfected with BamHI/KpnI digested GV657-Bcl-2 plasmid prepared by Shanghai Jikai Gene Chemical Technology Co., LTD. MSCs were seeded in 6-well culture plates at a density of 1–4 × 105 cells/mL and cultured with standard conditions. When the cells reached 70–90% confluence, the cells were cultured in freshly changed media without FBS and incubated with a mixture containing diluted LipofectamineTM 3000 (Invitrogen, L3000-008, Carlsbad, USA) and GV657-Bcl-2 plasmid (with the ratio of 1:1). Western blotting analysis was used to determine the expression of constitutively active Bcl-2 in the MSCs 24 h posttransfection, confirming the successful transfection with Bcl-2 plasmid.
To confirm the involvement of Bcl-2 in cell membrane damage by ox-LDL, MSCs with Bcl-2 overexpression were used to culture with ox-LDL. Cell number and LDH release from the cells were evaluated 24 h after culture with ox-LDL with native LDL as control.
Effect of N-acetylcysteine (NAC) on bcl-2 expression and cell growth of MSCs
NAC has been known to protect MSCs against ox-LDL-induced damage through multiple mechanisms, including inhibition of ROS formation, increase in Akt signaling, and protection of membrane integrity.13,27 To determine if NAC could preserve the expression of Bcl-2 in MSCs, cells were incubated with NAC (1 mM, Sigma A9165) for 5 min before exposure to ox-LDL. Cell number and LDH release from the cells as well as Bcl-2 expression in MSCs were evaluated 24 h after culture with ox-LDL with native LDL as control as described above. Each experiment was conducted at least three times.
Statistical analysis
The data in the present study were presented as mean ± standard error of mean for experiments. Statistical analysis was conducted using one-way analysis of variance (multiple comparisons) (GraphPad Prism 10 for macOS 10.1.0 [264]; GraphPad Software, LLC) followed by t-test for two columns of data. A two-tailed p value of ≤0.05 was considered statistically significant.
Results
Ox-LDL significantly reduced the number of MSCs and increased LDH release from MSCs
To define the effect of ox-LDL on the survival of MSCs and LDH release from MSCs, mouse MSCs were treated with ox-LDL for 24 h at different concentrations (with the final concentration from 0 to 140 μg/mL) with native LDL as control. No significant change in the survival of MSCs was observed for the cells treated with native LDL with stable cell number after 24 h of culture. However, ox-LDL treatment significantly reduced the survival of MSCs dose-dependently as reflected with a dramatic decrease in the number of MSCs in culture system 24 h after exposure as shown in Figure 1A. Cell number started to decrease when the concentration of ox-LDL was increased to 80 μg/mL in the media with the maximal reduction in the number of MSCs at the ox-LDL concentration of 120 μg/mL. No further reduction in the number of MSCs was observed when the concentration of ox-LDL was above 120 μg/mL.
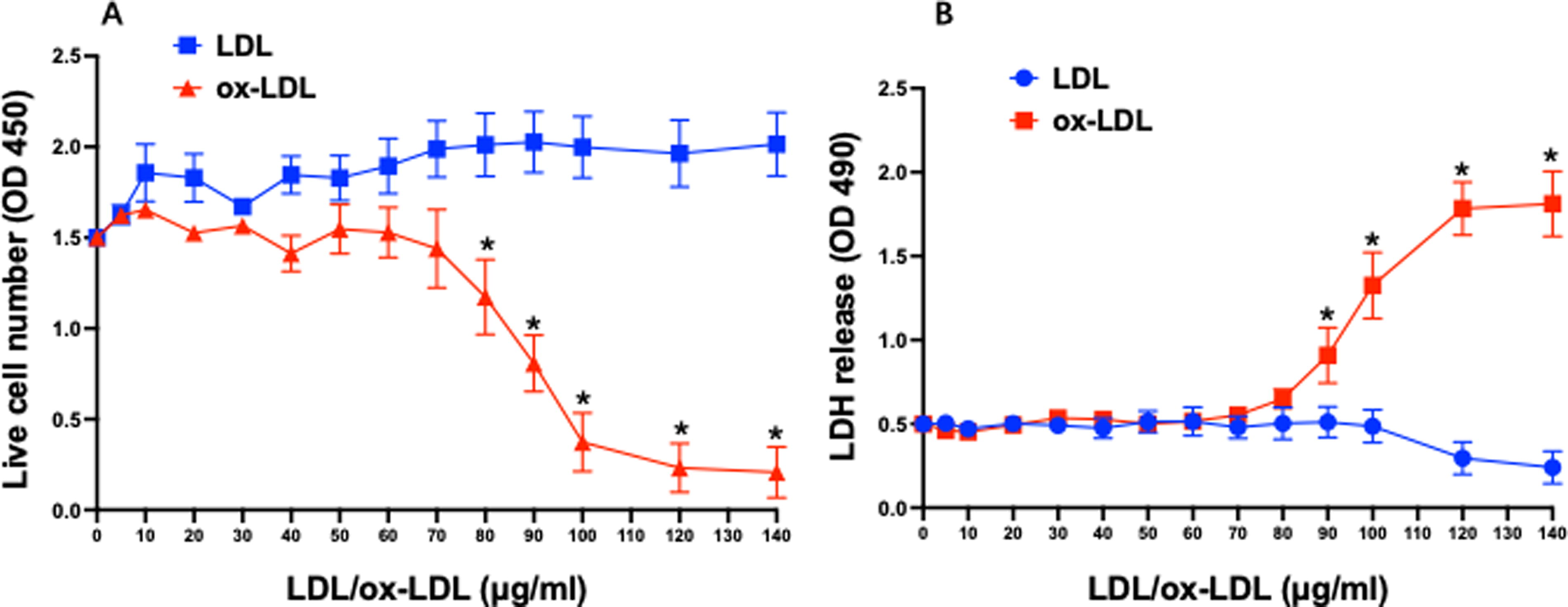
Effect of oxidized LDL on the survival of and LDH release from MSCs. Native LDL had no significant effect on the number of live MSCs with stable cell number after 24 h of culture. In contrast, cell number started to decrease when ox-LDL concentration was increased to 80 μg/mL with the maximal reduction in the number of MSCs at the ox-LDL concentration of 120 μg/mL
In line with the data on cell numbers, native LDL exhibited no significant effect on extracellular LDH levels after 24 h of treatment as shown in Figure 1B, suggesting an intact cell membrane. In contrast, ox-LDL treatment substantially increased the level of extracellular LDH in the culture media of MSCs dose-dependently. Similar to the data on cell number, extracellular LDH level began to rise when the concentration of ox-LDL was increased to 90 μg/mL and reached the maximum at the ox-LDL concentration of 120 μg/mL. No further increase in extracellular LDH level was observed with further increases in ox-LDL concentrations (Fig. 1B).
Bcl-2 expression played a critical role in cell damage of MSCs by ox-LDL
To evaluate the role of Bcl-2 expression in cell damage of MSCs by ox-LDL, Bcl-2 protein level was evaluated in MSCs after 24 h of ox-LDL exposure. Western blotting analysis revealed that ox-LDL treatment significantly suppressed the expression of Bcl-2 protein in MSCs as shown in Figure 2A. Bcl-2 protein level started to decrease significantly in the cells when the concentration of ox-LDL was increased to 100 μg/mL, whereas no significant change in BAX protein level was present in MSCs treated with ox-LDL (Fig. 2B). No significant effect on the expression of Bcl-2 protein was observed in MSCs treated with native LDL.
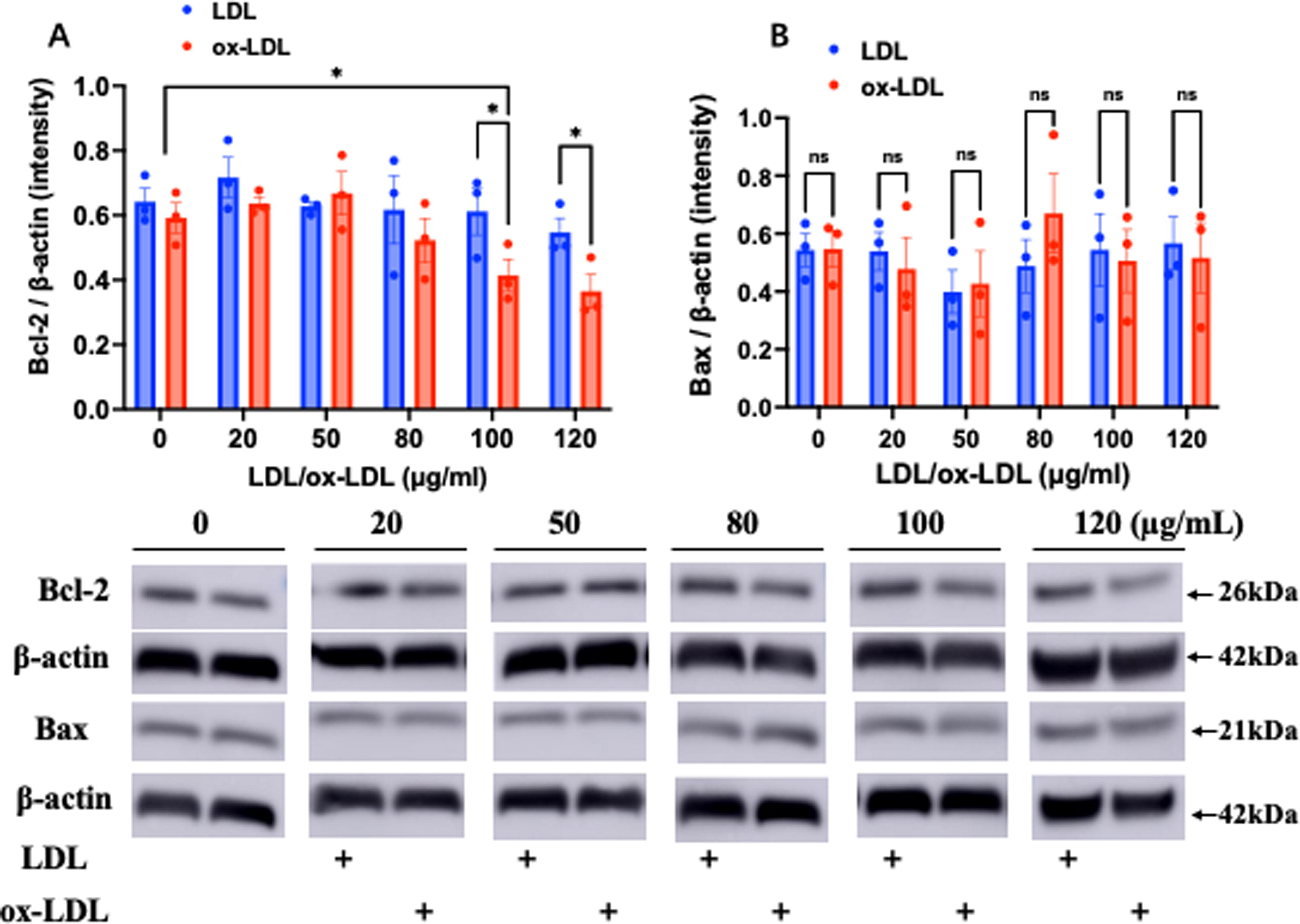
ox-LDL significantly attenuated Bcl-2 protein expression in MSCs. Western blotting analysis showed that Bcl-2 protein level was significantly decreased in a dose-dependent manner in MSCs treated with ox-LDL for 24 h, whereas native LDL had no significant effect on the protein expression of Bcl-2 in MSCs.
To determine if decreased expression of Bcl-2 protein was critically involved in mediating the effect of ox-LDL on the survival of MSCs and their membrane damage, experiments were repeated using MSCs that overexpressed constitutively active Bcl-2 (Fig. 3A, B). Indeed, overexpression of Bcl-2 effectively rescued the cells against ox-LDL with preserved cell numbers (Fig. 3C) without significant increase in extracellular LDH level (Fig. 3D) when MSCs were exposed to ox-LDL at the concentration of 100 or 120 μg/mL for 24 h.
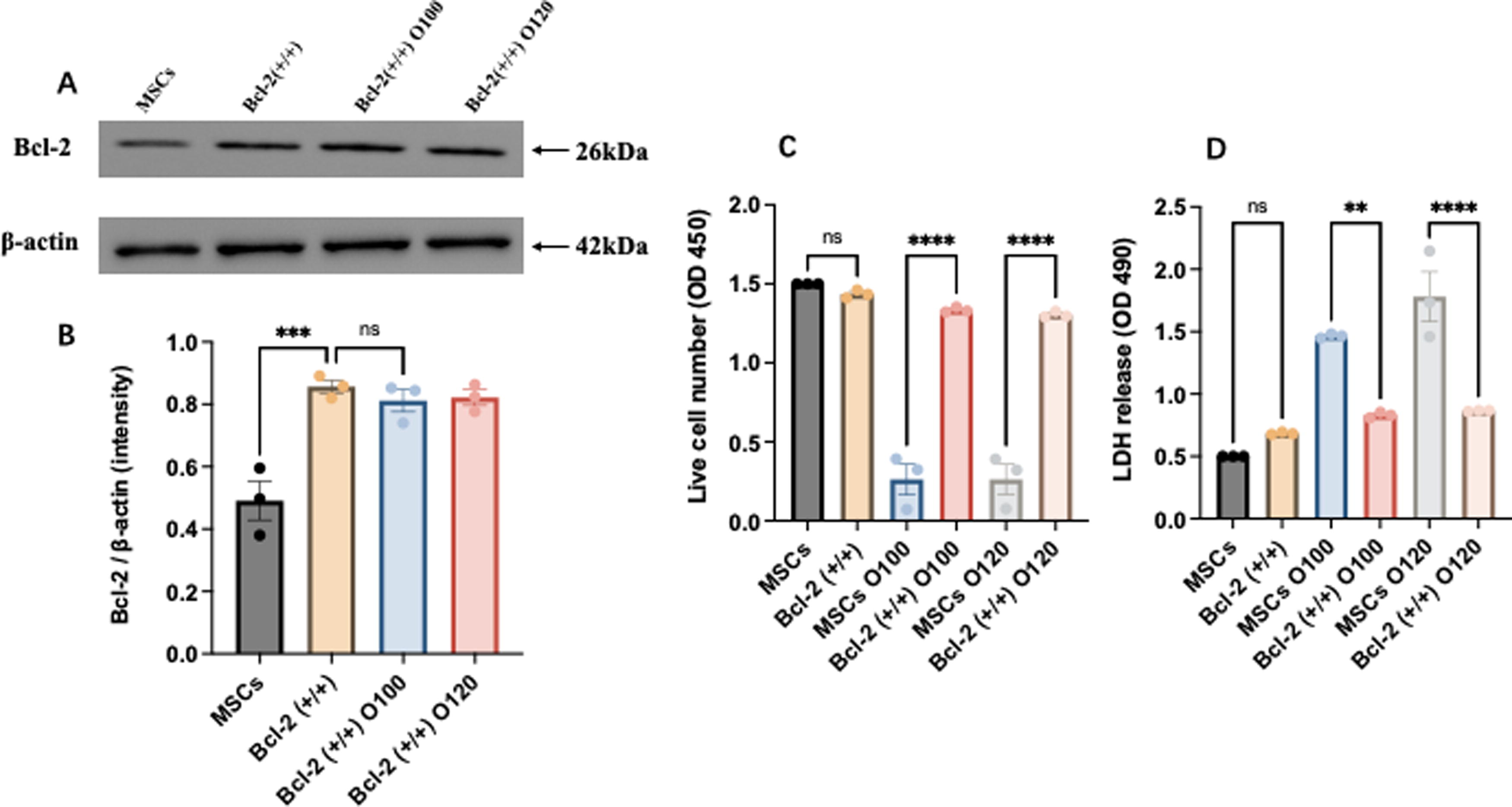
Bcl-2 overexpression effectively protected MSCs against ox-LDL-induced damage.
NAC treatment protected MSCs against ox-LDL in association with preserved Bcl-2 expression
It has been demonstrated that ox-LDL markedly decreased the survival of MSCs partially through membrane damages 12 and that NAC enhanced the survival of MSCs. 28 To determine if NAC could protect MSCs and preserve the expression of Bcl-2, the cells were pretreated with NAC (1 mM) for 5 min before incubation with ox-LDL. As shown in Figure 4, NAC treatment effectively preserved the protein expression of Bcl-2 in MSCs (Fig. 4A, B) and significantly attenuated the reduction of cell number by ox-LDL (Fig. 4C) and the increase in extracellular LDH level (Fig. 4D). Of note, a significant dose-dependent increase in intracellular ROS level was observed in MSCs exposed to ox-LDL. NAC treatment effectively attenuated excessive ROS production in MSCs incubated with ox-LDL (Fig. 4E).
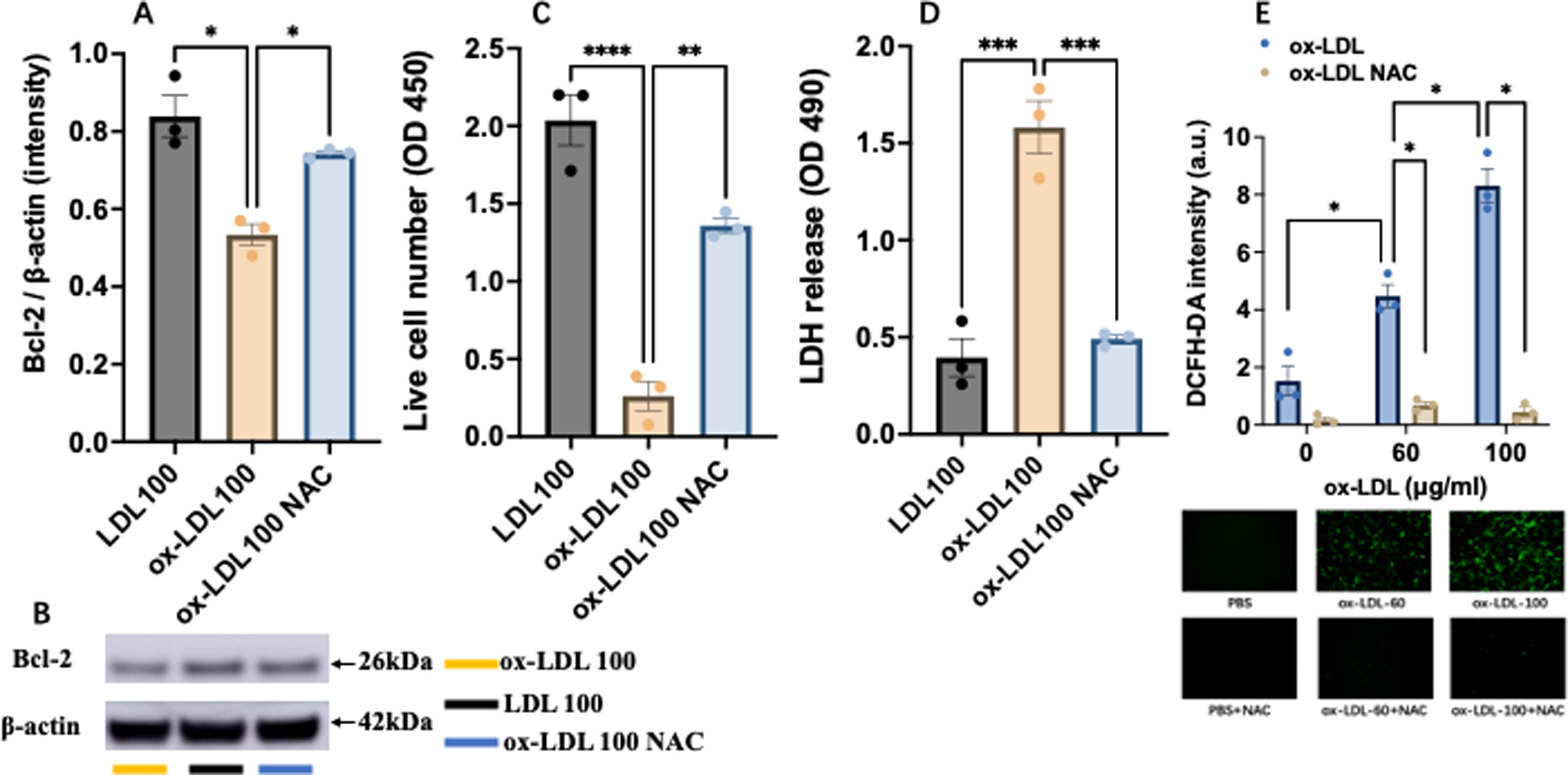
N-acetylcysteine (NAC) effectively preserved the expression of Bcl-2 protein and protected MSCs from ox-LDL-induced injury.
Discussion
The data from the present study demonstrated the following: (1) ox-LDL markedly attenuated the survival of MSCs dose-dependently, (2) ox-LDL significantly increased the release of intracellular LDH from MSCs dose-dependently, (3) ox-LDL dose-dependently suppressed the expression of Bcl-2 protein in MSCs, (4) Bcl-2 overexpression in MSCs effectively rescued MSCs against ox-LDL, and (5) NAC treatment effectively preserved the expression of Bcl-2 protein in MSCs exposed to ox-LDL and significantly prevented the decreased survival of MSCs by ox-LDL and intracellular LDH release from MSCs with ox-LDL treatment. These data suggest that ox-LDL decreases the survival of MSCs via cell membrane damage because of decreased expression of Bcl-2. Data also indicate that NAC may protect MSCs against ox-LDL-induced damage because of preservation of Bcl-2 expression in MSCs.
A wide range of ox-LDL concentrations were used in the present study to evaluate the effect of ox-LDL on the in vitro survival of MSCs as the levels of ox-LDL in blood and vascular wall vary widely in human subjects. The blood ox-LDL concentration has been reported to be 0.7 mg/dL (7 μg/mL) in healthy subjects. The blood level of ox-LDL was increased to 1.72 mg/dL (17.2 μg/mL) and 2.36 mg/dL (23.6 μg/mL) in patients with stable coronary artery disease (with an average LDL level of 125.8 mg/dL) and in the individuals with acute coronary syndrome (with an average LDL level of 143.0 mg/dL), respectively.29–31 The blood ox-LDL levels could be certainly further increased for the subjects with extremely elevated LDL levels such as in patients with familiar hyperlipidemia. It is demonstrated that ox-LDL can enter the vascular wall via ox-LDL receptor 1 32 and that LDL oxidation occurs locally within atherosclerotic lesions, 33 thus significantly contributing to high levels of ox-LDL in the vascular walls with atherosclerosis. In fact, a study using paired samples from patients with carotid atherosclerosis and undergoing carotid endarterectomy demonstrated that the ox-LDL level in atherosclerotic lesions was nearly 70 times higher than plasma ox-LDL level. 34 Studies have shown that resident vascular stem cells, including MSCs, in vasculature play an important role in vascular repair, remodeling, and angiogenesis. 35 These resident vascular stem cells could be potentially impaired by high levels of local ox-LDL in vascular wall in the subjects with hyperlipidemia and/or atherosclerosis. Thus, the concentrations of ox-LDL used in the present study were indeed clinically relevant.
In vivo cell survival is critical for the therapeutic outcomes of stem cells, including MSCs. Although the mechanisms for the poor survival of stem cells after in vivo delivery have not been fully defined, oxidative stress and excessive ROS are important contributing factors for cell death in vivo. 28 Ox-LDL generates a significant amount of ROS both in vivo and in vitro,36–38 and intravenous administration of ox-LDL has been shown to decrease the numbers of bone marrow stem cells and endothelial progenitor cells in animal models. 10 In vitro studies have also revealed that ox-LDL substantially decreased the survival of MSCs via multiple mechanisms, including (but not limited to) excessive ROS formation, inhibition of Akt-signaling, and cell membrane damage.11–13 Data from the present study also demonstrated that ox-LDL significantly decreased the survival of MSCs dose-dependently along with a significant increase of intracellular LDH release from the cells. These data are consistent with the previous observation that interrupted cell membrane integrity was critically involved in the reduction of the survival of MSCs by ox-LDL. However, a significant difference in the susceptibility to ox-LDL-induced damages was present for different types of subpopulations of bone marrow stem cells. It was reported that a concentration of 5–10 μg/mL ox-LDL substantially decreased the survival of rat MAPCs in vitro,13,14 whereas a concentration of 100–120 μg/mL ox-LDL was needed to achieve a similar level of effect on cell survival of mouse MSCs in vitro as shown in the present study. Future studies are necessary to define the mechanism(s) for the significant difference in the susceptibility to ox-LDL-induced damages for different types of stem cells.
One of the interesting findings from the present study was that ox-LDL substantially suppressed the expression of Bcl-2 protein in MSCs. Bcl-2 protein is a member of Bcl-2 family and is expressed in a variety of cells (including normal and cancer cells) and is involved in many important biological functions, including (but not limited to) cell autophagy, apoptosis, calcium homeostasis, mitochondrial dynamics, and energy metabolism.39,40 Bcl-2 also significantly contributes to maintaining the integrity of mitochondrial and cell membranes, thus, promoting cell survival and preventing cell death.39,41 It has been shown that exposure of neonate cardiomyocytes to hydrogen peroxide (H2O2) resulted in a rapid degradation of Bcl-2 in association with cardiac myocyte cell death. 42 ROS has been reported to sensitize T cells to apoptosis through inhibition of Bcl-2 expression. 17 It has also been demonstrated that treatment with antioxidant NAC effectively prevented ROS-induced death of 32D cells through regulation of Bcl-2 expression. 15 Overexpression of Bcl-2 significantly promoted the survival of human embryonic stem cells in response to stress in serum-free environment and enhanced the survival and function of MSCs and neural stem cells as well as pluripotent adult stem cells.16,43–45
The data from the present study revealed that Bcl-2 was also critically involved in mediating the effect of ox-LDL on the survival of MSCs and their membrane integrity. This finding may have important clinical implication because of the following reasons: (1) ischemic cardiovascular diseases, such as ischemic cardiomyopathy and ischemic stroke, are the leading cause of mortality and morbidity without effective therapy; 2) hyperlipidemia is a major risk factor for ischemic cardiovascular diseases; 3) ox-LDL level is significantly increased in hyperlipidemic states; and (4) ox-LDL is known to attenuate the survival and function of MSCs.12,13 Thus, Bcl-2 could be an important target to enhance the clinical outcomes of stem cell therapy in patients with hyperlipidemia. In addition, resident MSCs in vascular walls are believed to be critically involved in maintaining the integrity of vascular structures and function as well as disease development, including atherosclerosis.46–49 It has been shown that modified LDL and free cholesterol could have significant impact on the function of resident MSCs. 48
There have been significant advances in technology to improve the in vivo survival and subsequent efficacy of MSCs in atherosclerotic cardiovascular disease with some success, including (but not limited to) modification of the delivery of MSCs, 5 application of labeled cell spheroids, 50 and utilization of specific exosomes 51 from MSCs. NAC is considered an effective antioxidant and anti-inflammatory agent and has been shown to enhance the survival of MSCs in ischemic tissues in mice with type-2 diabetes mellitus in association with preserved expressions of important antioxidant enzymes and decreased infiltration of macrophages in the ischemic tissue, as well as improved circulatory and functional recovery in the ischemic muscle.28,52 NAC also protected MSCs against oxidative stress induced by ox-LDL and H2O2 in vitro via multiple mechanisms such as preservation of Akt-mediated signaling.13,27,53 The present study demonstrated that NAC effectively preserved the protein expression of Bcl-2 in MSCs and significantly enhanced the survival of MSCs and protected their cell membrane integrity against ox-LDL-induced damage. Thus, NAC could be used to enhance the survival of MSCs to achieve the optimal outcomes of MSC therapy in patients with hyperlipidemia and/or diabetes.
Study Limitations and Future Plan
There were a few limitations in the present study, including (1) it was an in vitro study. In vivo study using MSCs overexpressing Bcl-2 is needed to confirm if overexpression of Bcl-2 could enhance the survival of MSCs in hyperlipidemic mice and improve the therapeutic efficacy of MSCs. (2) The component composition of ox-LDL is very complex and has not been fully defined. It contains a mixture of a variety of fatty acid derivatives (both in the ester and free forms) and cholesterol with and without oxidative modifications, as well as proteins with oxidized amino acids and crosslinks.18,54 Thus, it is very challenging to clearly define and characterize the exact component(s) that may significantly contribute to the actions of ox-LDL on the survival of MSCs. Future studies are needed to analyze the chemical changes to LDL upon oxidation and identify the critical components(s) that renders the effect of ox-LDL on different cells and other diverse biological systems. (3) It is unclear how ox-LDL impairs the cell membrane integrity and compromise cell viability that requires further investigation, and (4) it is unclear how NAC upregulates the expression of Bcl-2 protein in MSCs and other cells, and further investigations are needed.
In conclusion, the present study demonstrated that ox-LDL significantly impaired the cell membrane integrity and dramatically decreased the survival of MSCs dose-dependently through inhibition of Bcl-2 expression. NAC treatment effectively protected MSCs against ox-LDL-induced cell membrane damage and enhanced the survival of MSCs in association with preserved Bcl-2 expression.
Footnotes
Authors’ Contributions
Design: J.-L.D. and X.L.; validation: J.-L.D. and X.L.; experiment: X.L., Y.L., and H.Y.; data analysis: X.L., Y.L., H.Y., and L.-L.M.; writing—original draft preparation: X.L.; writing—reviewing and editing, J.-L.D., Z.L., and G.D.; figure preparation: X.L.; supervision: J.D. and X.L.; and funding acquisition: X.L. All authors have read and agreed to publish the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The present study was supported by the National Natural Science Foundation of China to X.L. (Grant No. 81700246).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
