Abstract
Fabrication and application of engineered complex tissues composed of different types of cells is a crucial milestone in the next phase of tissue engineering. The delicate organization structure of each tissue component and its physiological connections enable all the functions in the human body. In this study, cell sheet-based engineering allowed us to fabricate a complex myofiber sheet tissue using motor neurons derived from human-induced pluripotent stem cells. In contrast with previous studies of other groups, a myofiber sheet with a biomimetic aligned structure was produced from human myoblasts using a striped-patterned thermoresponsive dish, which enabled manipulation of the sheet tissue by simply lowering the culture temperature. The myofiber sheet was transferred onto a gel that promotes functional maturation of human myofibers, resulting in production of contractile human muscle tissue. Just by seeding motor neurons onto the sheet tissue, all the neurons physically contacted to the aligned myofibers, and autonomously elongated in parallel to the myofiber orientation. In addition, the neurite outgrowth was enlarged by coculturing on the myofiber sheet. The presence of the neurons enhanced clustering of myofiber acetylcholine receptors (AChRs), typically found at the neuromuscular junctions (NMJs). Consequently, contraction behaviors of the myofiber sheet were regulated by neuronal signal transduction through NMJs. Muscle contraction was induced when the motor neurons were stimulated by glutamic acid, and effectively blocked by administration of
Impact Statement
A complex tissue containing skeletal myofibers and induced pluripotent stem cell-derived motor neurons was fabricated from human cells based on the cell sheet engineering technology. A micropatterned thermoresponsive culture dish and a fibrin-based gel substrate enabled production of aligned, and functionally matured myofiber sheet tissue. The motor neurons were autonomously oriented simply by seeding on the aligned myofiber sheet tissue. Induction and inhibition of the muscle contraction were effectively controlled by neuronal signal transduction. Considering the potential scalability and manipulability of the neuron–muscle sheet tissue, our methodology will contribute to future applications of regenerative medicine for locomotion apparatus.
Introduction
Tissue engineering enables the production of native-like tissues that can potentially be used as in vitro tissue models for biological studies and drug discovery.1,2 Although animal models have long been the main approach for drug discovery and prediction of pharmacokinetics, they have limitations in advanced biological studies to understand the more complex mechanisms in the human body.3,4 Therefore, the complex structures composed of different cell types found in native tissues need to artificially replicate the human mechanisms for the next generation of research in pathophysiology and drug discovery. On the contrary, the recent advances in induced pluripotent stem cell (iPSC) technology have opened up an entirely new era of possibilities for tissue modeling. The latest technologies are currently able to produce a variety of human cell-based tissue models, and even use cells that can only be obtained sparingly from the human body (e.g., neurons, cardiomyocytes).3–6 Moreover, since it is now possible to obtain some disease-specific iPSCs from patients, iPSC technology has even greater potential for modeling of disease-specific tissues.7–9
Muscle tissue engineering can now produce artificial muscle tissues to better understand myogenesis, including development, growth, and regeneration. Since skeletal muscles contribute to metabolic, neuromuscular, and dystrophic disorders, a functional muscle tissue model will be a powerful new tool to elucidate the mechanisms of muscle diseases and facilitate the discovery of new drugs for their treatments.10–13 However, many previous studies have mainly reported the production of rodent muscle tissues, because it is still difficult to engineer functionally mature human muscle tissue. Furthermore, unlike in native muscle, most engineered muscle tissues lack the physiological functions including neuromuscular junctions (NMJs). In native muscle, the NMJs connect physiologically between a motor neuron axon and a myofiber, and neuronal signal transduction through the NMJs induces muscle contraction. Although some research groups have attempted to engineer NMJs within their tissue constructs, most of the studies have succeeded in the production of NMJs using rodent primary neurons.8,14–19 On the contrary, the iPSC technology has great potential to engineer human NMJ models with motor neurons derived from human iPSCs.7–9 However, due to these substantial difficulties in functional maturation of human primary muscle cells and human iPSC-derived cells, successful human cell-based NMJ models have rarely been reported by a few research groups.7,9,20–23
Native skeletal muscle has a highly oriented structure of parallel bundles of myofibers that are comprised of multinucleated, striated, and contractile muscle cells. To mimic this aligned structure, several research groups, including our own, have reported innovative strategies using various kinds of microfabricated materials.24–29 However, due to the difficulty in stimulating physiological maturation on these materials, only a cylindrical-shaped hydrogel-based culture system has succeeded in the production of functional human muscle tissue with the biomimetic aligned structure.27–29 In their culture system, a gel environment not only forms an aligned structure but also promotes functional maturation of the myofibers, included within the gel. Although techniques such as these are very crucial for producing aligned human muscle tissue, there have been no other significant approaches reported. For that reason, most human NMJ models still lack a biomimetic structure. In this study, based on our original tissue engineering, “cell sheet-based engineering”, human contractile myofiber sheet tissue was engineered with an aligned structure. The thermoresponsive substrate allows confluent cells to be released from the surface by lowering the culture temperature and to be manipulated as a sheet-shaped assembly, a “cell sheet”.30,31 Using this cell sheet fabrication system, which differs from previous studies of other groups, our aligned myofiber sheet produced by a micropatterned thermoresponsive substrate was transferred onto a fibrin-based gel for functional maturation of human myofibers. This methodology exhibits potential application for studies of neuromuscular diseases including impaired NMJ functionality in future research.
Materials and Methods
Preparation of micropatterned thermoresponsive substrate
A photoinduced polymerization method was applied to fabricate polymer patterns on a thermoresponsive surface, as reported previously.26,30 To produce the micropatterns from a hydrophilic polymer on the surface, acrylamide (AAm) was polymerized with a photoinitiator by irradiation of visible light onto a culture surface. A commercially available thermoresponsive culture dish, UpCell® dish (CellSeed, Inc., Tokyo, Japan), was used in this study. 30 AAm aqueous solution (50 w/w%) (Wako, Tokyo, Japan) containing a water-soluble photoinitiator camphorquinone (7,7 dimethyl-2,3-dioxobicyclo[2.2.1] heptane-1-carboxylic acid) (1 w/w%) (Tokyo Kasei, Tokyo, Japan) was poured onto the substrate, and visible light was irradiated for 7 min onto the substrate through a photomask containing stripe-shaped patterning. Consequently, a hydrophilic polymer poly AAm was grafted spatioselectively as stripe-shaped micropatterns (the width of nonirradiation and irradiation regions: 50 μm) on a thermoresponsive polymer-grafted substrate.
Protein adsorption onto micropatterned culture surfaces
To confirm the formation of micropatterns on the substrate, fibronectin was immobilized on the surface, then the amount immobilized on the two different polymer patterns was estimated from fluorescence intensity. 32 Rhodamine-conjugated bovine fibronectin (10 μg/mL in phosphate-buffered saline [PBS]; Cytoskeleton, Denver, CO) was incubated with the culture substrate at 37°C for 2 h. After being washed thoroughly with PBS, the amount immobilized was relatively evaluated from the microscopic images captured using a fluorescence microscope (ECLIPSE TE2000-U; Nikon, Tokyo, Japan).
Differentiation of human iPSC into motor neuron
Human iPSC line 201B7 purchased from RIKEN (Tsukuba, Japan) was cultured in Primate ES Cell Medium containing 5 ng/mL basic fibroblast growth factor (bFGF; ReproCELL, Yokohama, Japan) on mitomycin C-treated mouse embryonic fibroblasts (ReproCELL) at 37°C. Differentiation of the iPSCs into motor neurons was induced through the procedure as previously reported by Shimojo et al.
33
Human iPSC colonies were harvested using CTK solution (ReproCELL) and suspended in Primate ES Cell Medium without bFGF. The suspension was cultured in a gelatin-coated dish for 2 h to remove the feeder cells from the medium. The remaining iPSC colonies were transferred into a normal culture dish and then cultured overnight. On day 1, the medium was changed to human embryoid body (hEB) medium (Dulbecco's modified Eagle's medium [DMEM]/F-12 [Sigma-Aldrich, St. Louis, MO] with 5% KSR [Thermo Fisher Scientific, Waltham, MA], 2 mM
Coculture of aligned myofibers and motor neurons using fibrin-based gel
Human skeletal muscle myoblasts isolated from the upper arm or leg muscle tissue were obtained from Lonza (CC-2580; Walkersville, MD). The myoblasts were seeded at a density of 5.0 × 104 cells/cm2 onto the micropatterned thermoresponsive substrate (the seeding area: 15 × 15 mm2) and then cultured in muscle cell growth medium (SkGM-2; Lonza) until reaching a 100% confluence. Next, a mixture of fibrinogen (from bovine plasma, 10 mg/mL, 2 mL; Sigma-Aldrich), thrombin (from bovine plasma, 20 U/mL, 500 μL; Sigma-Aldrich), Matrigel (500 μL; BD Biosciences, San Jose, CA), and CaCl2 solution (8 mM, 1 mL) was poured onto the cells (900 μL per dish). 26 Before the formation of the fibrin-based gel, a square-shaped silicon ring was inserted into the mixture to prevent shrinkage of the gel during the maturation of the myogenic cells. The mixture was incubated on the culture dish for 30 min at 37°C, and then a differentiation medium (DMEM containing 2% horse serum [HS; Thermo Fisher Scientific]) was added to the culture dish. The medium contained 20 μg/mL aprotinin (Sigma-Aldrich) as an antifibrinolytic agent. After 1–3 days of culture in the medium, the muscle cells were incubated at 20°C for 30 min to release them from the thermoresponsive surface. Using tweezers, the released construct was removed and reversed, laying it back onto the dish. Human iPSC-derived motor neurons were seeded onto the aligned muscle cells on top of the gel. The coculture tissue was cultured in motor neuron maturation medium with 20 μg/mL aprotinin. To produce a randomly oriented myofiber sheet as the control sample, human myoblasts were cultured on a nonpatterned thermoresponsive culture dish and transferred onto a fibrin-based gel through the same tissue fabrication procedure.
Electrical pulse stimulation for maturation of engineered myofibers
After 1 week of the culture in motor neuron maturation medium, recombinant rat agrin (R&D Systems) was added to the medium to promote expression and clustering of acetylcholine receptors (AChRs) on the myofibers. 15 The concentration of agrin was increased gradually (0.1 nM at day 1, 0.5 nM at day 2, and then 1.0 nM after day 3). In addition, the sheet tissues were electrically stimulated to enhance muscle maturation as reported previously. 26 The tissue construct was placed in a six-well plate, and then two carbon electrodes (C-Dish; IonOptix, Milton, MA) were immersed into the medium. The electrical pulse stimulation (EPS) (voltage: 10 V, frequency: 1 Hz, duration time: 10 ms) was continuously applied using an electrical pulse generator (IonOptix) for 1 h followed by incubation without EPS for 3 h. This procedure was carried out repeatedly for 2 weeks in motor neuron maturation medium containing agrin.
Immunofluorescence staining
Myofiber sheet tissues were fixed with 2% paraformaldehyde in PBS overnight at 4°C. After fixation, samples were washed with PBS and then incubated in blocking solution (5% bovine serum albumin with 0.2% Triton-X 100; Sigma-Aldrich) for 10 h. 26 The coculture tissues were washed with PBS, and then treated with primary antibodies (neurofilament [NF; 1:500], islet [1:500], sarcomeric α-actinin [1:500], laminin [1:500]; Abcam, Cambridge, MA) at 4°C overnight. After being washed with PBS, the tissues were treated with fluorescently labeled secondary antibodies (1:800; Thermo Fisher Scientific) or Alexa Fluor 488-conjugated α-bungarotoxin (Thermo Fisher Scientific) at RT for 2 h. For nuclei staining, tissues were incubated with Hoechst33258 (Dojindo Laboratories, Kumamoto, Japan) at RT for 2 h. Images were acquired using a confocal laser microscope (FV1200; Olympus, Tokyo, Japan). To measure the diameter of myofibers, immunofluorescence images of α-actinin–positive myofibers were taken under five representative fields, and all data were expressed as mean ± standard deviation (n = 3).
Analysis of tissue orientation
The orientation of myofibers and neurons in the sheet tissue was evaluated with a directional histogram constructed using the “directionality” function in the NIH ImageJ software. 26 The parallel alignment from the axis of the stripe patterns was denoted as 0°, and the perpendicular alignment was denoted as 90°. A flat histogram indicated a completely isotropic orientation, and a histogram with a clear peak indicated a respective orientation.
Analysis of areas of neurite outgrowth and AChR clusters
Neurite outgrowth was visualized by immunostaining with NF to quantify the expansion of neurons. AChR clusters on myofibers were also stained fluorescently using AlexaFluor488-conjugated α-bungarotoxin to analyze the areas of AChR clusters.8,15 Fluorescence images were randomly captured at five locations per sample. The areas from three independent samples were measured and averaged using NIH ImageJ software. The areas of NF were normalized to the total areas of the captured images (the area ratio: NF-positive area/image). The areas of AChR clusters and myofibers were measured using ImageJ software, and the AChRs areas were normalized to the areas of the myofibers, which were fluorescently visualized by staining with sarcomeric α-actinin (the area ratio: AChR area/myofibers area).
Displacement measurement of unidirectional muscle contraction
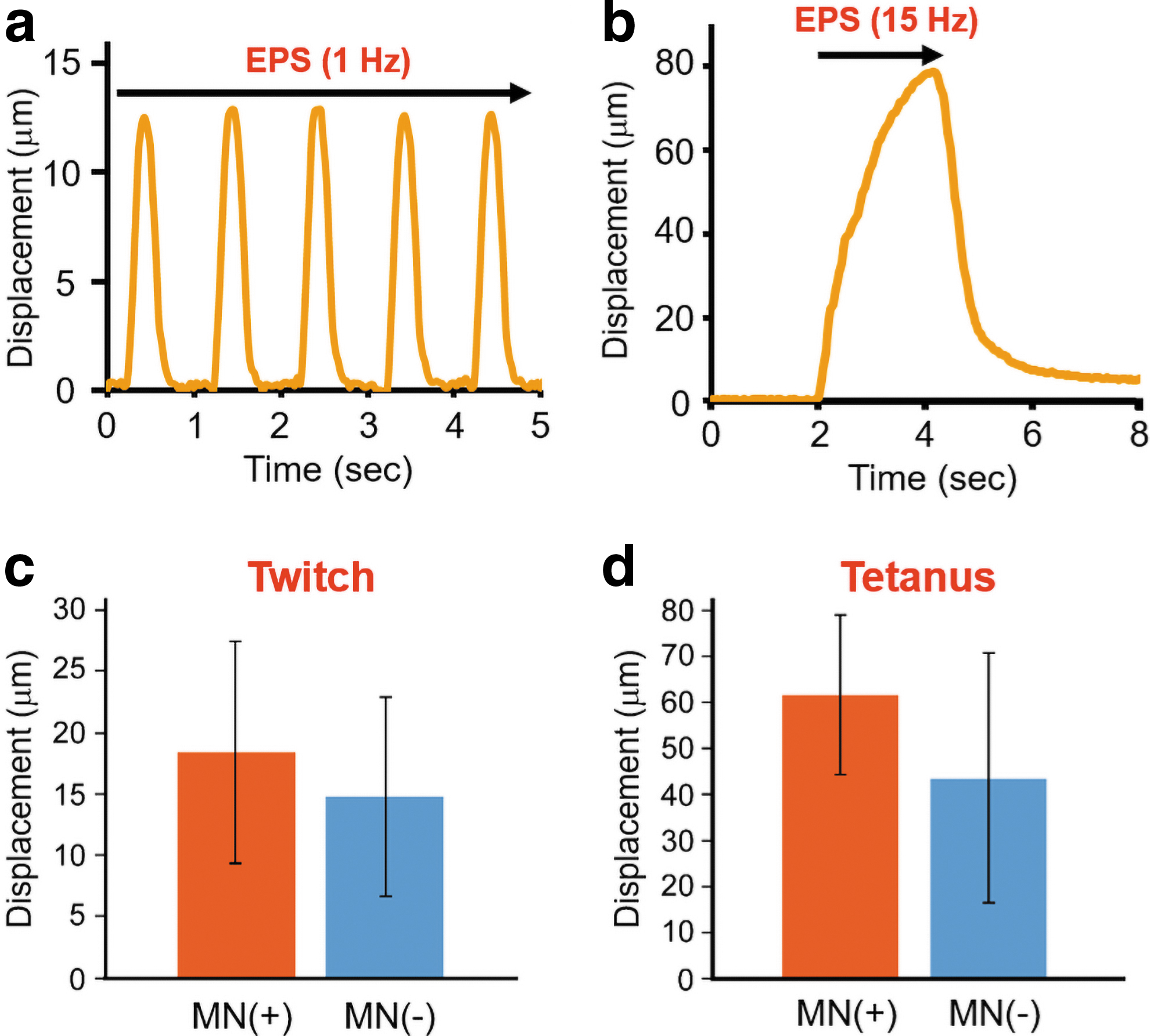
To observe the contractile ability, the coculture tissues were electrically stimulated (voltage: 10 V, frequency: 1 or 15 Hz, duration time: 10 ms). The EPS-induced muscle contraction was monitored using a CCD camera (30 frames/s; Basler Ace, Basler AG, Germany) through phase contrast microscopy. Cell shortening distances were calculated using the motion analysis software ViewPoint (Glenallan Technology, Inc., Clinton, NY). 26 Even if the coculture tissues were produced under the same culture conditions, the specific conditions of cell sources (donor, passage number, seeding density, etc.) affected the maturation level of muscle tissues. 34 Since myoblasts freshly harvested from donors produce more highly functional myofibers, 35 relative contraction displacements in this study were evaluated in comparison with the control samples prepared at the same time.
Pharmacological treatments for induction and blockage of muscle contraction
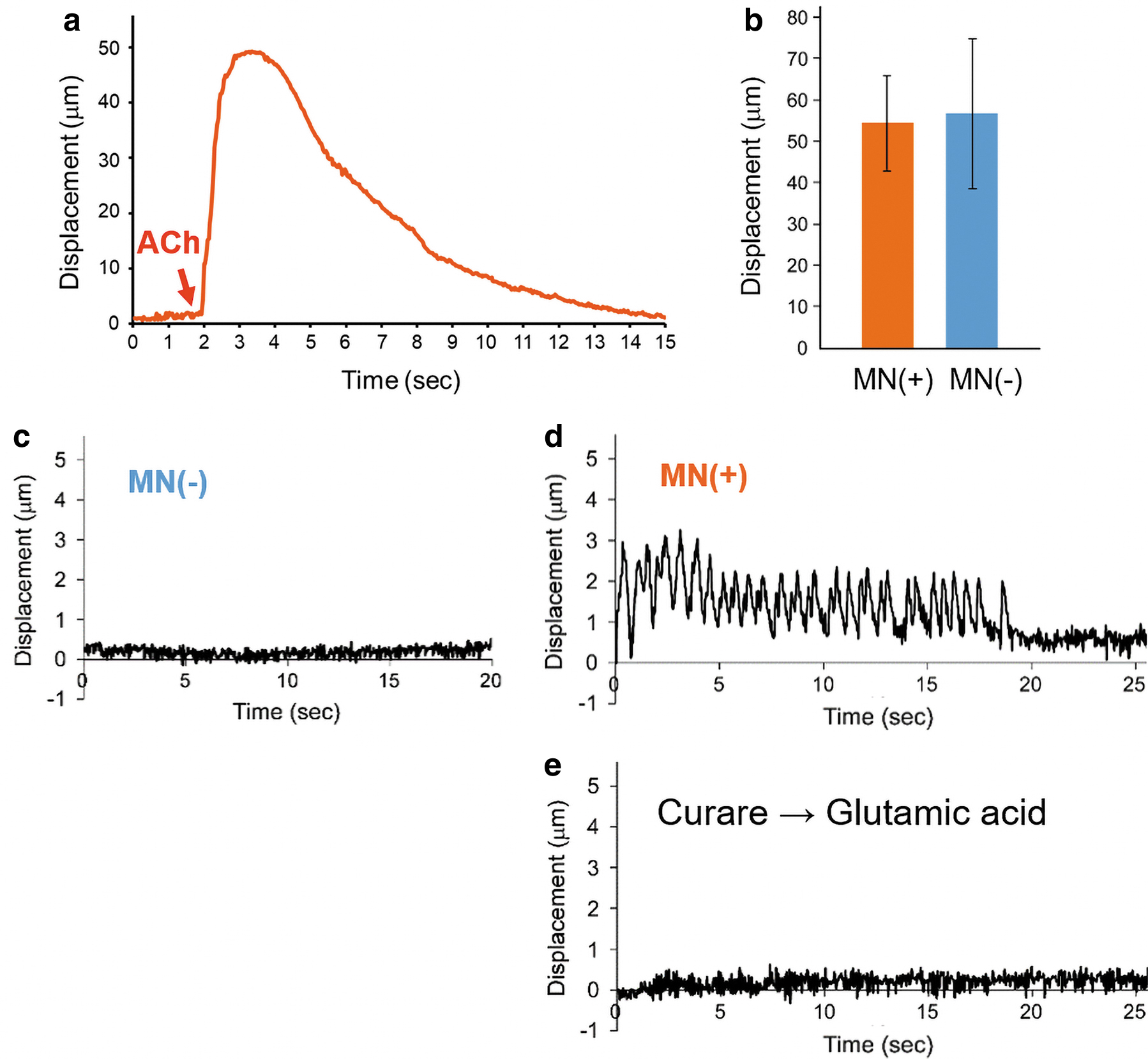
Evidence of chemically induced contractile ability of the myofibers was found by adding acetylcholine chloride solution (10 μM, 100 μL; Sigma-Aldrich) to the medium (4 mL). To stimulate the motor neurons in sheet tissues,
Statistical analysis
Data are expressed as the mean ± standard deviation. Statistical analysis was performed with Student's t-test when comparing two groups (Figs. 1, 4, 5, and 7). Statistical significance was considered as *p < 0.05. Statistical processing was performed using JMP Software.
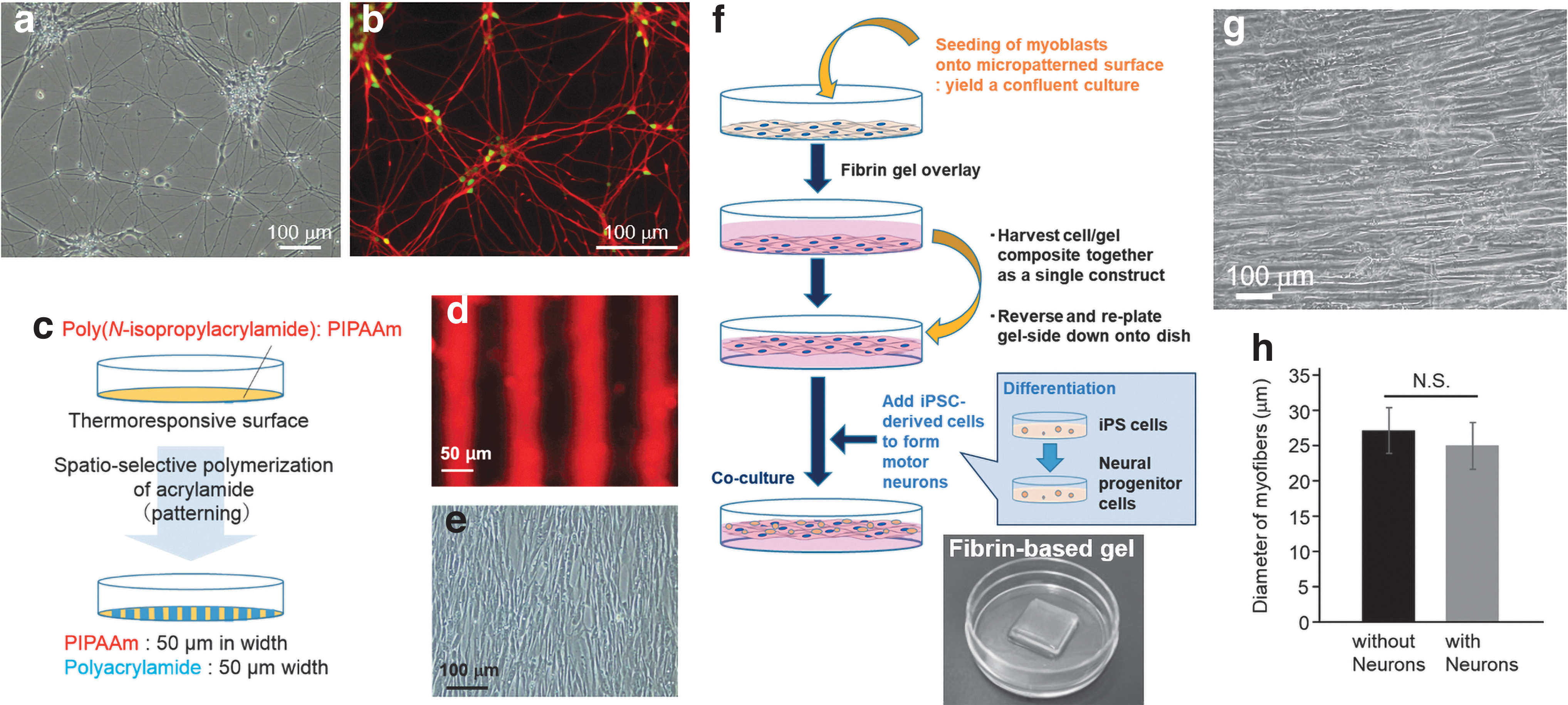
Results
Engineering of human-aligned myofiber sheet tissue with iPSC-derived neurons
Human motor neurons were differentiated from human iPSCs through the previously reported method. 33 When the iPSC-derived neurons were seeded onto iMatrix-coated dishes, NF- and islet-expressing motor neurons expanded on the culture dishes (Fig. 1a, b). Human myofiber sheets were produced based on a technique reported in our previous study.26,36 First, stripe-shaped micropatterns were fabricated on a thermoresponsive surface through a photoinduced polymerization of AAm (Fig. 1c). The two resultant patterns displayed different affinities for adhesive proteins such as fibronectin (Fig. 1d). Since human primary myoblasts can recognize the difference in cell–surface interaction, they aligned themselves along the striped patterns (Fig. 1e). Myoblasts reached confluence while maintaining their orientation, and then the aligned myoblasts were transferred from the micropatterned substrate onto a fibrin-based gel to promote myogenic maturation of the cells (Fig. 1f). After the gel formation, they were cultured in a differentiation medium (2% HS containing medium) to induce differentiation into myotubes. The micropatterned thermoresponsive culture substrate was used because it has a unique function that enables the release of adherent cells from the surface by lowering the temperature.32,37 In fact, the aligned muscle cells were released from the culture surface by lowering the culture temperature to 20°C. The gel with the transferred cell sheet was reversed, laying it back onto the dish, so that the aligned cells were now on top of the gel. Next, the human iPSC-derived motor neurons were seeded onto the aligned muscle cells, and cultured for 1 week in the motor neuron maturing medium (KBM medium). 33 As shown in Figure 1g, myoblasts differentiated into multinucleated myotubes while maintaining the aligned orientation on the gel surface. Although it was expected that the coculture with neurons would positively influence the myofibers, there was no significant difference in the diameter of the myotubes, with and without neurons (Fig. 1h).
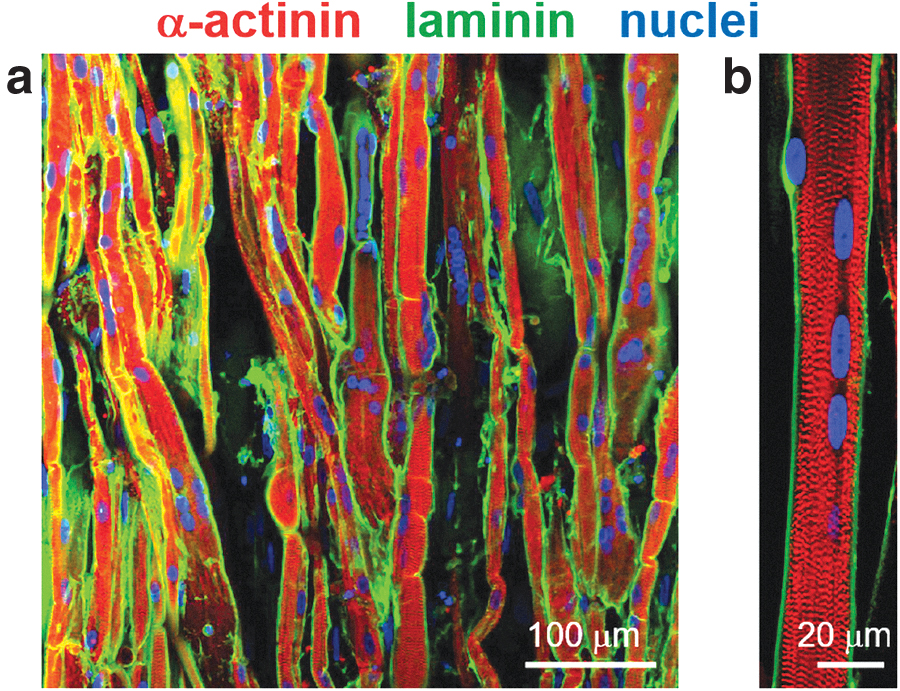
After 3 weeks of coculture, sarcomere structures formed in the aligned myofibers (Fig. 2a). This indicated that these aligned myofibers had matured sufficiently to actuate muscle contraction. In this study, for maturation of motor neurons, the KBM neural stem cell medium was used in the production of the neuron–muscle complex tissues. This medium was designated only for culture of iPSC-derived motor neuron, as reported previously. 33 However, since differentiation of myoblasts is often induced by culturing in serum-free medium, in this study this chemically defined medium was used to induce the differentiation of myoblasts into mature myofibers. Furthermore, fluorescence images showed that laminin was uniquely localized around the aligned myofibers. Since the same basement membrane-like laminin structure can be found in native skeletal muscle,26,27 the laminin localization suggested that our culture system provided an appropriate environment for maturation of the engineered human myofibers. In addition, cell nuclei were localized at the gap between the sarcomere and the membrane-like laminin layer (Fig. 2b). In native muscle, cell nuclei migrate from the center of a myofiber to the outside of the sarcomere structure during myogenic maturation. Therefore, the self-arrangement of myofibers produced in this study was expected to form native tissue-like membrane microstructures within the sheet tissue.
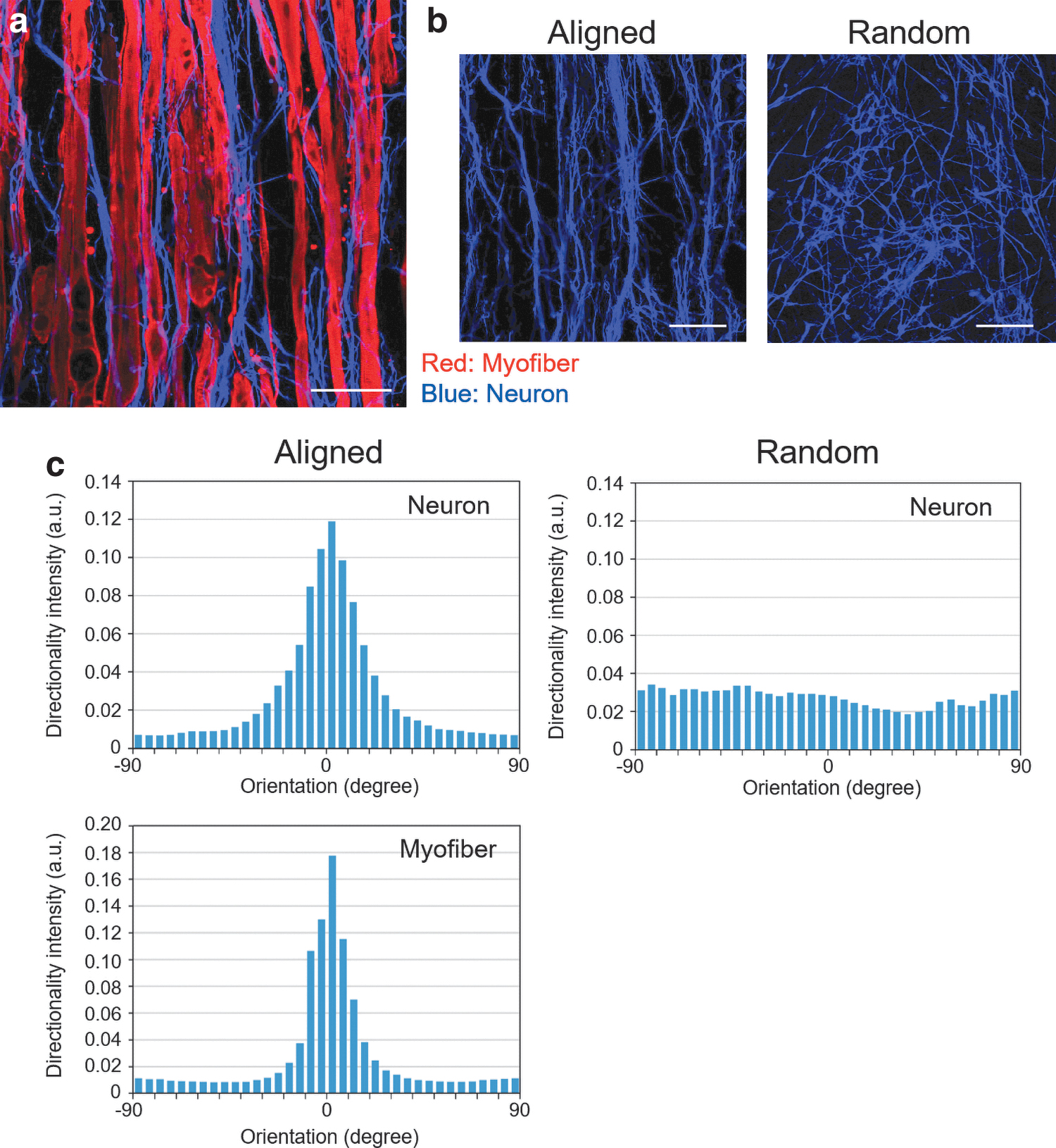
Alignment of neurite outgrowth directed by aligned myofibers
In the coculture tissue, neurons seeded directly onto myofiber sheet autonomously expanded in the same direction as the myofiber orientation (Fig. 3a, b). To confirm that the expansion was related to the alignment of myofibers, the orientation was evaluated using the “directionality” function in the ImageJ software. 26 In the coculture sheet composed of randomly oriented myofibers, fluorescence images and a histogram indicated that neurons formed randomly oriented networks. On the contrary, in the group of aligned myofiber sheet tissue, both histograms, indicating both neuron and myofiber orientations, showed clear peaks at the same position (Fig. 3c). This result obviously indicated that the expansion of neurons was directed by the myofibers and followed the aligned orientation of the myofibers.
Synergistic effects of iPSC-derived neurons and myofibers in coculture sheet tissue
In our coculture method, all neurons had direct contact with the myofibers and expanded on the myofiber sheet as their adhesion substrate. This is advantageous to effectively produce coculture effects induced by the physical contact between neurons and myofibers. Indeed, neurite outgrowth was much more intensive on the aligned myofibers (Fig. 4a–c) than on the representative substrate in the neuron culture (Fig. 1d, e). This indicated that the presence of myofibers enhanced neurite outgrowth through cell-to-cell interaction. Consequently, the neurons formed oriented networks in the same direction as the alignment of the myofibers. In addition, when neurons were seeded onto aligned myofibers before the gel formation, the neurons expanded on the myofibers adhering on a culture dish, similar to that on the cell/gel construct. Therefore, regardless of apical-basal cell polarization, the cell-to-cell interaction enhanced the neurite outgrowth on the aligned myofibers.
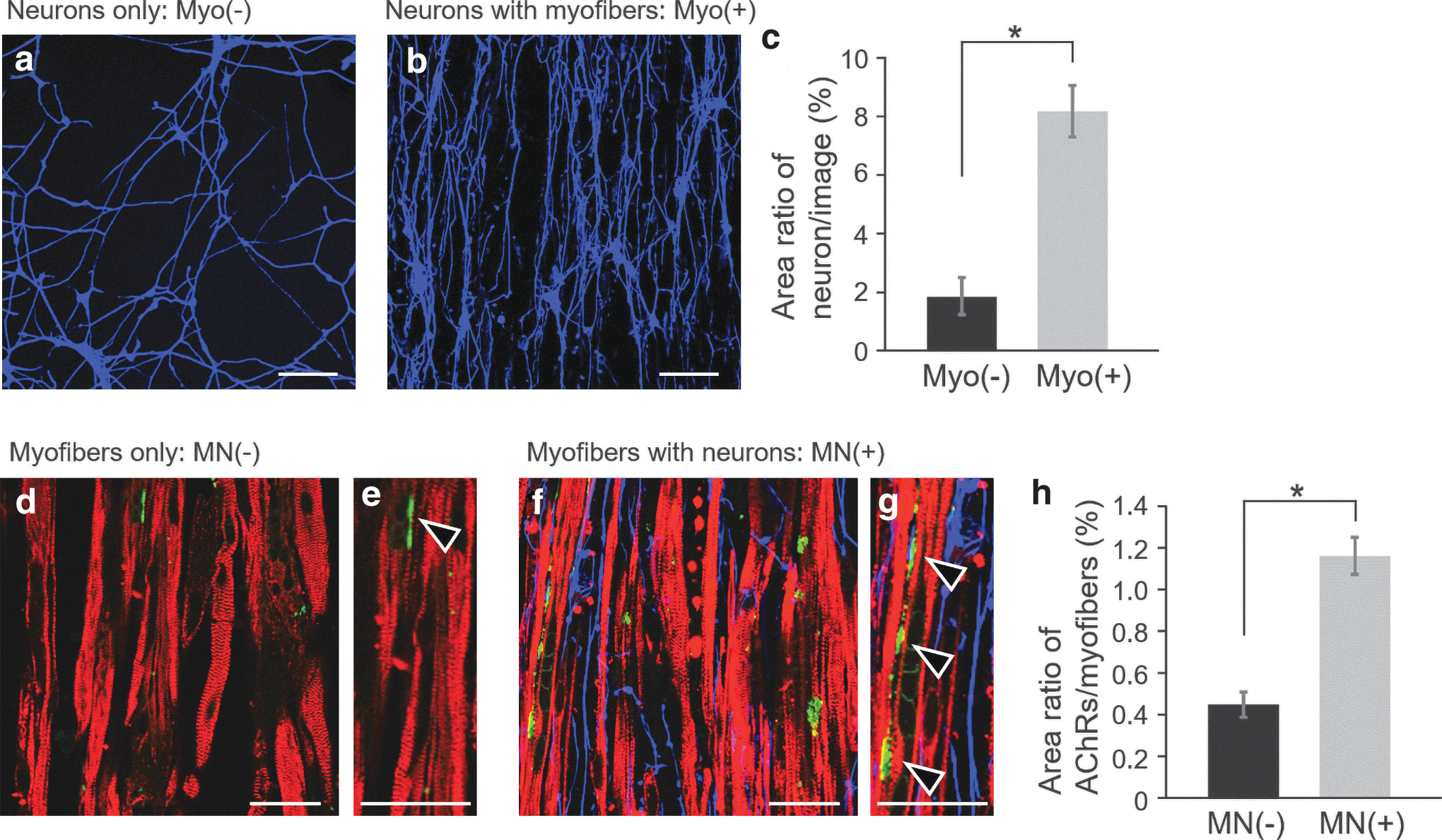
Enhancement of neurite outgrowth and AChR clustering by coculture of neurons with myofibers in in vitro culture.
In native muscle, on the contrary, physiological connections between motor neurons and myofibers are necessary to maintain the myofiber functions. From this viewpoint, it was expected that the presence of neurons would positively influence the maturation of the engineered myofibers. 23 In fact, although it triggered no enlargement of the myofibers (Fig. 1h), the expression and/or clustering of AChRs on myofibers were significantly enhanced by coculturing with neurons (Fig. 4d–h). AChRs are committed in neural transmission through NMJs, and must be clustered for the development of functional connectivity of NMJs. 38 As shown in Figure 4d–g, the presence of neurons caused an increase in the areas of AChR on myofibers. Since the clustering of AChRs is known to increase with the maturation of myofibers,8,15 it can be assumed that coculturing with neurons promoted the maturation of myofibers within the muscle tissues.
Contractile ability of human myofiber sheet tissue cocultured with neurons
As shown in Supplementary Videos S1 and S2, our myofiber sheet tissue contracted unidirectionally according to the frequency of EPS. When the myofibers were simulated at a frequency of 1 Hz, they showed a twitch contraction (Fig. 5a and Supplementary Video S1). As the frequency increased to 15 Hz, a tetanic contraction caused the myofiber sheet tissue to shorten more dynamically (Fig. 5b and Supplementary Video S2). These contractile behaviors relative to the EPS frequency indicated that our muscle tissue was electrophysiologically functional. The contractile behaviors of the coculture tissues were not significantly different from that of the muscle tissues without neurons (Fig. 5c, d). Therefore, the NMJ formation was probably not necessary for the functional maturation of the myofibers, while the coculture of neurons promoted AChR clustering on the myofibers (Fig. 4d–h).
Formation of NMJ within coculture tissues
To morphologically confirm the formation of NMJs, both NFs and AChRs were stained fluorescently.7,15,23,39 Fluorescence images showed neurite outgrowth terminated on the myofibers, sometimes colocalizing with AChR clusters (Fig. 6). These images suggested that NMJs were formed in this engineered neuron–muscle complex tissues. However, this result may only demonstrate the physical contact between neurons and AChRs on the myofibers. Therefore, physiological testing is also required to confirm the propagation of signals across the NMJs.
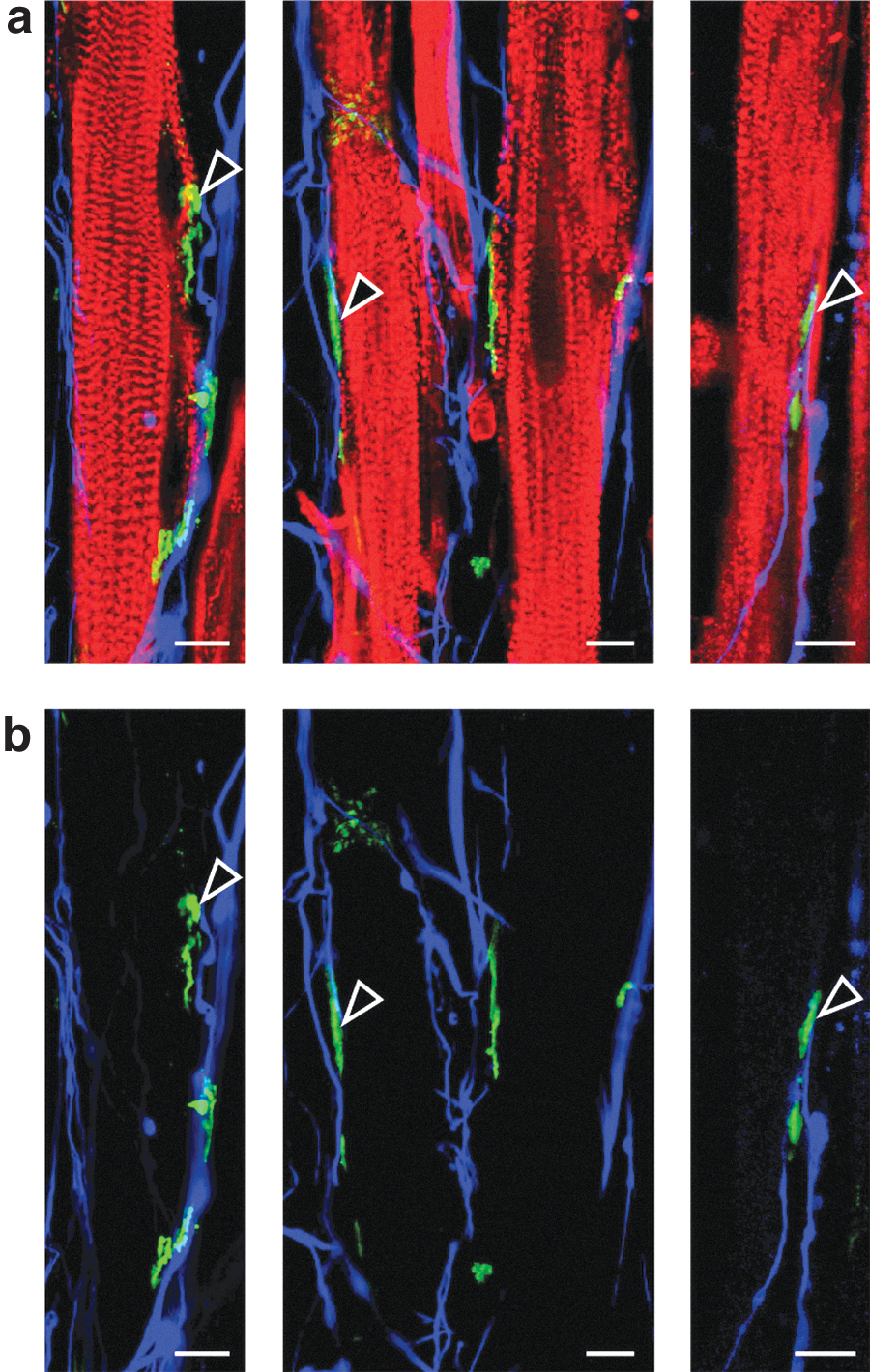
Immunostaining images of colocalization of AChR clusters and neuron terminations on the engineered myofibers.
In the NMJ system, neurotransmitters (e.g., glutamic acid) stimulate motor neurons, and then acetylcholine (ACh) is released from vesicles at the presynaptic terminal. The released ACh then binds to an AChR cluster at the postsynaptic myofiber membrane. Thus, to evaluate the functionality of AChRs on the myofibers, ACh solution was added to the culture medium of the engineered tissues.20,23,27 As shown in Supplementary Video S3 and Figure 7a, myofibers contracted in response to the ACh addition and then gradually relaxed after several seconds. This suggested that the coculture tissue was chemically stimulated by ACh through binding to AChRs on the myofibers. Since the ACh-induced contraction behaviors were not significantly different between muscle tissues with and without neurons (Fig. 7b), the coculture with neurons influenced AChR clustering, but induced no enhancement of the muscle contraction. Next, motor neurons in the coculture tissue were stimulated by the addition of glutamic acid.9,16,18,20 Whereas no contraction was induced in the myofiber sheet tissue without cocultured neurons (Fig. 7c), the chemical stimulation caused twitch contractions of the myofibers only in the coculture tissue (Fig. 7d). The chemical stimulation was repeatedly induced by multiple applications with glutamic acid (Supplementary Video S4). To further validate the presynaptic activation of motor neurons, a postsynaptic blocker,
Discussion
This study demonstrated that our methodology was successful to produce human cell-based tissue, consisting of a functional skeletal myofiber sheet and iPSC-derived motor neurons. Although human cell-based NMJ models are advantageous for understanding the complex physiological phenomena in the human body, most previous studies have only reported the production of rodent NMJ models. 40 Although some defined culture systems have achieved production of human neuron–muscle complex tissues for physiological and pathological studies of NMJs, only a few groups have adopted these systems.7,9,20–23 The human NMJ models hitherto have all reported a lack of biomimetic morphology. Human muscle cells must be cultured in an appropriate environment to be functionally matured; however, aligned muscle cells regulated by various microfabricated substrates have been unable to become functionally matured.14,15,17 Particularly, in the production of these NMJ models, human iPSC-derived neurons are not functionally connected to immature muscle tissues. Therefore, culture substrates appropriate for their maturation (e.g., Matrigel) are useful, even though they have no function for aligning myofibers.7,22,23 In this study, based on the cell sheet transfer technology, human innervated muscle tissue was produced with the biomimetic aligned structure using a micropatterned substrate. We succeeded in the maturation of the human muscle sheet tissue on the fibrin-based gel. Human motor neurons seeded onto the prealigned myofiber sheet tissue oriented spontaneously parallel to the aligned underlying myofiber layer. As a consequence, this human cell-based tissue had native-like morphological and physiological characteristics, including the aligned muscle structures, unidirectional contractile ability, and functional NMJ formation.
Our engineered myofibers showed a native tissue-like laminin structure. Since it is well known that this unique membrane structure is important for the maintenance of mature myofibers, the close similarity with the structure observed in this study suggested that our muscle tissue has the same biological aspects as native skeletal muscle. Moreover, in native skeletal muscle, the muscle-specific membrane structures including cell membrane, basement membranes, and AChR clustering are closely related to NMJ formation.41,42 Therefore, the membrane-like microstructure found in this study may be advantageous for AChR clustering and subsequent NMJ formation. Localization of cell nuclei at the outermost surface of the engineered myofibers is also a noteworthy result (Fig. 2b). This self-arrangement of cell nuclei can be seen in the mature myofibers of native muscle. Taken together, our tissue engineering approach can provide a culture environment that allows myofibers to self-arrange into a native tissue-like microstructure. Furthermore, the gel substrate provided myofibers with an elastic environment suitable for muscle contraction. Since the elasticity supports flexible displacement during the contraction of myofiber, in this study the neural stimulation by glutamic acid and its blockage by curare were clearly demonstrated, even if these contractions were relatively small compared with the EPS-induced contraction.
There is enormous potential for the use of iPSC in tissue modeling for studies of NMJ development and motor neuron diseases (MNDs), even though there are some issues still to be resolved. In recent studies, iPSC-derived motor neurons obtained from amyotrophic lateral sclerosis (ALS) patients were used to engineer a disease-specific NMJ model. 9 Spinal muscular atrophy (SMA)-specific NMJ systems were developed using iPSCs from SMA patients, and the resulting tissue model would provide new insights into the physiology and pathology of SMA.7,8 Coupling iPSC technology and human NMJ systems can potentially establish a platform for generating various kinds of disease-specific NMJ models. Achievement of this study could be applied to produce some disease models with the biomimetic morphology. Currently, only a cylindrical-shaped hydrogel-based system has succeeded to produce human NMJ models with small and unmodifiable aligned tissue structures.9,20 Our tissue engineering technique can scale up the sheet-shaped tissue simply by enlarging the micropatterned substrate. Due to its manipulation capability, multiple sheet-shaped tissues can be layered to produce a thicker three-dimensional tissue or rolled up to produce a bundle-like structured tissue. Combined with these advances in tissue engineering, the iPSC-based NMJ models could be upgraded for fabrication of patient-specific customized tissue modeling and drug development studies.
Conclusion
In this study, we established a novel strategy for producing an in vitro human NMJ model by coculture of aligned myofibers with iPSC-derived motor neurons. The combined use of the micropatterned substrates and the fibrin-based gel supported the formation of native-like aligned structures of contractile myofibers. In addition, the physically contacting neurons formed oriented networks in the same direction as the myofiber orientation. Consequentially, the neurons were functionally associated with the clustered AChRs on the myofibers, and muscle contraction was controlled by neuronal signal transduction through NMJs. Our methodology, reported in this study, allowed our muscle sheet tissue to have native-like morphological and physiological characteristics, including the aligned structures, contractile ability, a basement membrane-like laminin structure, and functional NMJ formation. Although the functionality of the NMJ should be improved to enhance its responsiveness to neuron transmission, our cell sheet-based tissue engineering method has considerable potential for the production of a human NMJ model for applications in studies of neuromuscular pathophysiology, including NMJ development and MNDs. Moreover, combined with iPSC technology, our tissue engineering technique will allow modeling of patient-specific diseases for personalized neuronal disease research applications.
Footnotes
Acknowledgments
We gratefully acknowledge Ms. Asako Iida, Ms. Kasumi Sugiyama, and Ms. Mika Tejima (Tokyo Women's Medical University) for their technical assistance. We also thank Mr. Allan Nisbet for his useful comments and editorial assistance.
Authors' Contributions
H.T., F.O., and N.T. conceived and designed the experiments. F.O. and H.T. performed the experiments. F.O. and H.T. analyzed the data. H.T., T.S., and N.T. wrote the article.
Disclosure Statement
T.S. is a stakeholder in CellSeed, Inc.; Tokyo Women's Medical University receives research funds from CellSeed, Inc.
Funding Information
This work was supported by Grant-in-Aid for Young Scientists (A) (JSPS KAKENHI Grant No. 16H05909) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
