Abstract
The use of biomaterials and biomaterial functionalization is a promising approach to support pancreatic islet viability posttransplantation in an effort to reduce insulin dependence for patients afflicted with diabetes mellitus type 1. Extracellular matrix (ECM) proteins are known to impact numerous reparative functions in the body. Assessing how endogenously expressed pancreatic ECM proteins are affected by posttransplant-like hypoxic conditions may provide significant insights toward the development of tissue-engineered therapeutic strategies to positively influence β-cell survival, proliferation, and functionality. Here, we investigated the expression of three relevant groups of pancreatic ECM proteins in human native tissue, including basement membrane (BM) proteins (collagen type 4 [COL4], laminins [LAM]), proteoglycans (decorin [DCN], nidogen-1 [NID1]), and fibril-forming proteins (fibronectin [FN], collagen type 1 [COL1]). In an in vitro hypoxia model, we identified that ECM proteins were differently affected by hypoxic conditions, contributing to an overall loss of β-cell functionality. The use of a COL1 hydrogel as carrier material demonstrated a protective effect on β-cells mitigating the effect of hypoxia on proteoglycans as well as fibril-forming protein expression, supporting β-cell functionality in hypoxia. We further showed that providing endothelial cells (ECs) into the COL1 hydrogel improves β-cell response as well as the expression of relevant BM proteins. Our data show that β-cells benefit from a microenvironment composed of structure-providing COL1 with the incorporation of ECs to withstand the harsh conditions of hypoxia. Such hydrogels support β-cell survival and can serve as an initial source of ECM proteins to allow cell engraftment while preserving cell functionality posttransplantation.
Impact statement
Expression analysis identifies hypoxia-induced pathological changes in extracellular matrix (ECM) homeostasis as potential targets to support β-cell transplants by encapsulation in biomaterials for the treatment of diabetes mellitus. A collagen-1 hydrogel is shown to attenuate the effect of hypoxia on β-cells and their ECM expression. The functionalization of the hydrogel with endothelial cells increases the β-cell response to glucose and rescues essential basement membrane proteins.
Introduction
The extracellular matrix (ECM) physically and biochemically supports tissues and organs. It consists of large fibrillar proteins, glycoproteins, and proteoglycans, as well as water, growth factors, enzymes, and other biomolecules. 1 The ECM is vital for cell survival and function as it provides biological, chemical, and mechanical cues to cells, which in turn, remodel the ECM to control tissue homeostasis. 1
Alterations and disruptions of the ECM homeostatic state adversely affect cells resulting in structural tissue damages, diseases, and potentially organ failure. 2 Mechanical disruption of the ECM that occurs during injury or is induced during transplant surgery can severely compromise cell survival and function, ultimately hindering cell engraftment. 1 This is a major issue in cell and tissue transplantation due to a number of underlying factors, including the hypoxic environment at the transplant site, the implant/transplant-triggered immune response, and pathological ECM remodeling posttransplantation.
The transplantation of Islets of Langerhans or isolated pancreatic β-cells is a promising therapy to treat type 1 diabetes mellitus. However, poor engraftment combined with hypoxic conditions of posttransplantation are responsible for the loss of more than 60% of the transplanted cells within 1 month, leading to the loss of insulin-independency and severe hypoglycemic events.3,4 Therefore, strategies to improve transplant survival and efficacy should include tissue-engineered methods to support the transplants' survival under hypoxic conditions and improve the ECM homeostasis of the transplant itself.
The isolation of the Islets of Langerhans from donor pancreases severs the vasculature connection between the islets and pancreas. During this process, collagenases are infused into the tissues, disrupting the interstitial matrix of fibrillar proteins, such as collagen type 1 (COL1).4–6 The process partially digests the double basement membrane (BM) surrounding the islets, which is composed of collagen type 4 (COL4), laminins (LAM), fibronectin (FN), and proteoglycans.6,7 BM proteins were reported to be differentially affected by the isolation procedure; perlecan and laminin α5 are completely lost, while COL4 and other LAM are still present after the process. 7 The resulting loss in ECM structure has been previously shown to negatively impact islet survival and function of posttransplantation. 8
Endothelial cells (ECs) are the main source of BM proteins in the native pancreas. 9 Most transplantation sites lack ECs. The reestablishment of EC presence via revascularization requires several days of posttransplantation. 10 During that time, β-cells experience severe hypoxic conditions leading to improper ECM remodeling. Hence, β-cells initially rely on their intrinsic ECM protein expression to survive in a hypoxic posttransplantation environment or on external support, for example, carrier materials such as collagen-based hydrogels.11,12
Although many studies have shown the importance of the ECM and how it affects β-cell function, there have been very few investigations on how hypoxia regulates the expression and homeostasis of endogenous ECM in pancreatic islets. 13 Therefore, we developed an in vitro hypoxia model to investigate the ECM expression of β-cell composed of pseudoislets and to monitor pathological ECM changes under hypoxic conditions (Supplementary Fig. S1). We showed that hypoxia differentially affects the expression of BM proteins, proteoglycans, and fibril-forming ECM proteins. The use of a clinically approved COL1 hydrogel as carrier material allowed the rescue of the proteoglycans and fibril-forming ECM proteins. Coculturing ECs with β-cell pseudoislets in the COL1 hydrogel improved the endogenous expression of BM proteins. The combination of biological cues together with ECs is a promising strategy to prevent or reduce hypoxia-related apoptosis and loss of β-cell functionality.
Materials and Methods
INS1E cell culture and pseudoislet assembly
The insulin producing rat insulinoma cell line INS1E was a kind gift from Prof. Maechler from the University of Geneva, Switzerland. INS1E cells were cultured in adapted RPMI 1640 (Gibco) containing 10 mM HEPES (Gibco), 50 μM 2-mercapto-ethanol (Sigma-Aldrich), 1 mM pyruvate (Gibco), 5% fetal bovine serum (Sigma-Aldrich), 100 iU/mL penicillin, and 100 μg/mL streptomycin, under standard humidified normoxic conditions (37°C, 5% CO2, 20% O2), and passaged at 80% confluency. To assemble pseudoislets, nonadherent 96-well U-bottom plates (Thermo Fisher Scientific) were used at a density ranging from 500 to 10,000 cells per well in a working volume of 100 μL/well and placed under normoxic conditions for 48 h before any experiment. Hypoxia experiments were performed under the following conditions: 37°C, 5% CO2 and 1% O2, which were chosen according to literature.14–16 In detail, pseudoislets were seeded in overnight preincubated hypoxia medium. Media was changed every other day. Every 24 h, images of the pseudoislets were obtained using a brightfield microscope (Zeiss), and the pseudoislet size was analyzed using ImageJ V 1.52p. The growth speed was assessed by applying a linear regression curve for each pseudoislet size from day 2 to 8, resulting in the average growth in micrometers per day.
Viability assay
Fluorescein diacetate (FDA)/propidium iodide (PI) (Sigma-Aldrich) staining was performed according to the manufacturer's protocol. In brief, pseudoislets were washed once with phosphate-buffered saline (PBS), incubated with 33 μL FDA and 300 μL PI per 1 mL RPMI 1640 for 15 min at normoxic conditions. After incubation, pseudoislets were washed three times with PBS before recording the fluorescence using a laser scanning microscope 710 (Zeiss). The fluorescence was recorded in the z-stack mode using maximum intensity projection. Images were evaluated by Zen Blue (Zeiss) and ImageJ V 1.52p software. The number of PI+ cells was normalized by the diameter of each pseudoislet.
Human umbilical vein endothelial cell culture and coculture pseudoislet assembly
Human umbilical vein endothelial cell (HUVEC) culture and coculture pseudoislet assembly was performed as recently reported. 17 The magnetizing of HUVEC and INS1E allows the proper and reproducible aggregation of pseudoislets by placing the HUVECs around the INS1E β-cells. In brief, HUVECs (PromoCell) were cultured in corresponding EC growth medium (PromoCell), passaged at a confluency between 80% and 90%, and used between passage 2 and 6. For coculture pseudoislets assembly, INS1E cells and HUVECs were magnetized overnight before seeding, using NanoShuttle™-PL (Greiner) at a concentration of 40 μL/mL according to the manufacturer's protocol. To obtain optimal glucose-stimulated insulin secretion (GSIS) functionality of the cocultured pseudoislets, INS1E cells were seeded first at a density of 500 cells per well in 50 μL INS1E media to ensure proper pseudoislet formation. HUVECs were added 24 h later at a density of 500 cells per well in 50 μL HUVEC media, resulting in a media ratio of 1:1, and placed atop the magnetic levitation plate for 30 min under standard culture conditions for another 24 h.
COL1 pseudoislet encapsulation
An average number of 48 pseudoislets or coculture pseudoislets were encapsulated in COL1 with a concentration of 6.0 mg/mL with a total volume of 250 μL following the manufacturer's instructions (Fraunhofer IGB, Stuttgart 18 ).
GSIS assay
Ninety-six hours after seeding, pseudoislets with 500 and 1000 cells per pseudoislet were grouped by 4 per well in 50 μL medium. Pseudoislets were washed twice using KREBS 1 × buffer containing 0.1% bovine serum albumin (Sigma-Aldrich) and 25 mM HEPES (Gibco). Then, pseudoislets were incubated for 30 min in KREBS 1 × buffer under normoxic conditions. Subsequently, KREBS 1 × buffer containing 3.3 mM glucose (Gibco) was added and the pseudoislets were incubated for 1.5 h. The pseudoislets were washed again twice and incubated with KREBS 1 × buffer containing 16.7 mM glucose for another 1.5 h. The supernatants were collected and stored at −20°C for further insulin detection using enzyme-linked immunosorbent assay (Mercodia). The GSIS index represents the fold increase in insulin secretion from 3.3 to 16.7 mM glucose. For the GSIS assay in COL1 hydrogel, all incubation times were doubled with regard to diffusion properties.
Histological analysis
Pseudoislets were washed with PBS (Gibco), fixed with 4% paraformaldehyde, embedded in HistoGel® (Thermo Fisher Scientific), and processed for paraffin embedding using a Shandon Citadel 1000 (Thermo Fisher Scientific) according to the manufacturer's protocol. Sections were cut with a thickness of 3 μm (Microtome RM2145; Leica), deparaffinized using xylene, and rehydrated by graded ethanol (100–50%) in VE-water. Adult human pancreatic tissues were commercially obtained from Novus biologicals (NBP2-30191; Novus Biological, Bio-Techne GmbH, Wiesbaden, Germany).
Immunofluorescence staining
TRIS-EDTA (pH9) and citrate (pH6) buffer were used for antigen retrieval. The antibodies were used according to the manufacturer's specifications and are shown in Supplementary Table S1. Sections were incubated for 10 min with 4′,6-diamidino-2-phenylindole (DAPI, 2 μg/mL in PBS; Sigma-Aldrich) prior mounting with Molecular Probes ProLong Gold Antifade solution (Invitrogen). Immunofluorescence (IF) staining images of pseudoislets and pancreatic tissues were obtained using an Axio Observer Z1 microscope (Zeiss) as well as laser scanning microscopes 710 (Zeiss) and 780 (Zeiss). The images were analyzed using Zeiss Zen Blue software and ImageJ V1.52p.
Terminal deoxynucleotidyl transferase-mediated nick-end labeling assay
Click-iT TUNEL Alexa Fluor Imaging Assay Kit (Thermo Fisher Scientific) was used according to the manufacturer's specifications and used as previously described. 17 In brief, the sections were deparaffinized and permeabilized with 0.25% Triton-X for 20 min. Terminal deoxynucleotidyl transferase reaction buffer was added to the slides for 60 min at 60°C. Subsequently, sections were incubated with Click-iT reaction buffer for 30 min at room temperature. The counterstaining was performed using DAPI.
Image analysis
PI+ cells were counted using an automated macro in ImageJ V 1.52p and normalized by the size of the pseudoislet. Blinded terminal deoxynucleotidyl transferase-mediated nick-end labeling (TUNEL)- and Ki67-stained images were quantified by two independent, unbiased observers. Cells were counted as TUNEL+ and Ki67+ when exhibiting a green (TUNEL or Ki67) and blue (DAPI) double staining. Ratios of TUNEL+ and Ki67+ cells were calculated by dividing the number of TUNEL+ and Ki67+ cells, respectively, by the number of total DAPI+ cells. Staining intensities were evaluated via the mean gray value (MGV) per pixel within the region of interest (ROI). Pseudoislet ROI was selected as DAPI+ area and for human pancreatic sections as insulin+ region. Fold changes were calculated by dividing the MGV of the hypoxia samples by the MGV of the normoxia samples. The standard deviation was calculated using the formula for the propagation of error.
Nuclear expression of FN was quantified by a custom-written macro using Microsoft Excel (Microsoft Corporation, 2018). The program validates, whether the single pixel values of the blue channel (DAPI) and the green channel (FN) exceed a set threshold value. If both thresholds were exceeded, this pixel was counted as nuclear FN expression. The pixel was counted as nonnuclear FN expression only when the green threshold was exceeded. The percentage of nuclear FN expression was calculated using the following formula: percentage of nuclear fibronectin expression = (number of pixels with nuclear fibronectin expression)/(number of pixels with nuclear fibronectin expression + number of pixels with nonnuclear fibronectin expression).
Overall fold changes for ECM protein expression between normoxic COL1 hydrogel and hypoxic COL1 hydrogel+HUVECs were calculated by multiplying the fold changes of (hypoxic COL1 hydrogel/normoxic COL1 hydrogel)*(hypoxic COL1 hydrogel + HUVECs/hypoxic COL1 hydrogel).
Raman imaging of pseudoislets
INS1E pseudoislets were prepared for Raman imaging as previously described. 19 In brief, hypoxic and control pseudoislets were placed in a microfluidic chip for noninvasive in situ Raman imaging. The microfluidic device stabilizes the pseudoislets during measurements, while supplementing nutrients via media perfusion. Spectral mapping was performed on a customized inverted WITec alpha300 R Raman system (WITec GmbH) equipped with a green laser (532 nm) and a CCD spectrograph with a grating of 600 g/mm. An incubation chamber (Okolab S.R.L.) was integrated in the setup to keep the pseudoislets constantly at 37°C. Images were acquired as triplicates at a laser power of 58 mW, an integration time per spectrum of 0.5 s and a pixel resolution of 1 × 1 μm.
Multivariate data analysis
Image analysis of the spectral map was performed with the Project Five 5.2 software (WITec GmbH). Raman data were pretreated whereby each scan was subjected to cosmic ray removal, polynomial baseline correction (to remove the glass background), and intensity normalization. True component analysis (TCA) was performed at the spectral range of 600–1800 cm−1. In brief, TCA is a nonnegative matrix factorization-based multivariate data analysis tool elaborating spectral components, which predominantly occur in the data set allowing to identify the localization of these components by false color intensity distribution heatmaps. Gray value intensities per pixel (MGV) were determined in ImageJ V 1.52p to semiquantify the distribution of the spectral components in normoxic and hypoxic conditions. Furthermore, TCA allowed to preselect ROI with similar spectral information representing nuclei, ECM, and mitochondria, which was extracted for further in-depth analysis of the molecular composition by principal component analysis (PCA) using Unscrambler X10.5 (Camo). PCA is a multivariate data analysis tool reducing the dimensionality of a set of spectral data on a vector-based approach. Each vector, the so-called principal component (PC), describes a variation in the spectra. Plotting PC values against each other visualizes a correlation or separation of two or more data sets.
Statistical analysis
Statistical analysis was performed using Origin 2019b (OriginLab) and GraphPad Prism version 6.00 for Windows (GraphPad Software). Results are shown throughout the entire article as mean ± standard deviation. All data sets are tested for normal distribution using Kolmogorov–Smirnov test; outliers were removed using Grubb's test with a confidence interval of 0.05. All n-numbers, applied tests, and corresponding significances for each result are listed in the figure legends.
Results
Assembly of pancreatic β-cells into glucose-responsive pseudoislets
To assess the ECM-related changes induced by hypoxic conditions on pancreatic islets, we established an in vitro model composed of INS1E β-cells that were assembled into pseudoislets to mimic the insulin-secreting endocrine function of the native Islets of Langerhans. The native pancreatic islets range is from 50 to 400 μm in diameter with an average size of 150 μm.20,21 Therefore, pseudoislets were formed by spontaneous aggregation with 500 to 10,000 cells per pseudoislets (Fig. 1). The proliferative nature of the INS1E β-cells lead to an overall increase in their size over time, monitored for a period of 8 days (Fig. 1A). The growth speed was impacted by the number of initially seeded cells: pseudoislets of 2000 (2k), 4000 (4k), and 10,000 (10k) cells per pseudoislet grew significantly faster than pseudoislets of 500 (0.5k) and 1000 (1k) cells per pseudoislet (Fig. 1B).
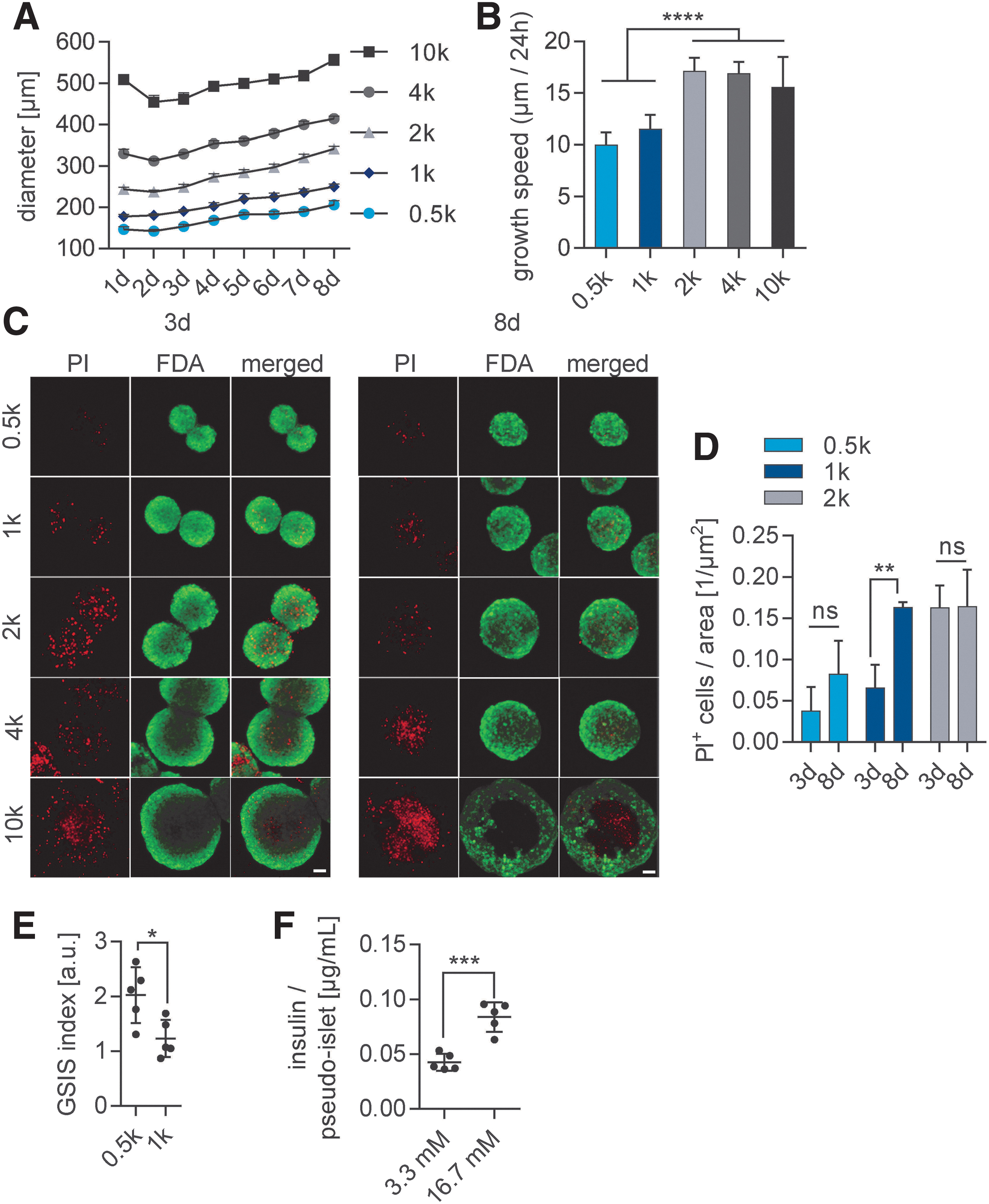
Pseudoislet size affects survival, growth, and functionality in vitro.
Diffusion of nutrients and oxygen are essential for the survival and physiological function of three-dimensional (3D) cell constructs.22,23 The lack of diffusion that occurs in large cell aggregates results in the formation of a hypoxic core. 24 Therefore, we assessed the influence of pseudoislet size on pseudoislet viability (Fig. 1C). We observed a hypoxic core depicted by PI-stained cells in large pseudoislets (4k and 10k pseudoislets) after 8 days in culture, which was not observed in smaller pseudoislets (0.5k, 1k, and 2k pseudoislets). The quantification of PI+ cells in 0.5k, 1k, and 2k pseudoislets showed a significantly higher dead cell ratio in 2k pseudoislets compared with 0.5k and 1k pseudoislets after 3 and 8 days (0.5k: 0.038 ± 0.026 [3 days] vs. 0.082 ± 0.04 [8 days], p < 0.0001; 1k: 0.0656 ± 0.024 [3 days] vs. 0.163 ± 0.006 [8 days], p < 0.01; 2k: 0.163 ± 0.022 [3 days] vs. 0.0164 ± 0.040, p < 0.0001). These data suggest that 0.5k and 1k pseudoislets allowed for proper nutrient and oxygen diffusion overtime, which is in line with previous studies showing that pseudoislets above 1000 cells per islet lead to an unstable aggregation (Fig. 1D).24,25
The major function of β-cells is to secrete insulin in response to glucose, which is also influenced by the pseudoislet size.26,27 Therefore, GSIS assays were performed to assess potential functional differences between 0.5k and 1k pseudoislets (Fig. 1E, F). The GSIS index revealed that 0.5k pseudoislets had a significantly higher glucose response compared with the 1k pseudoislets (Fig. 1E; 0.5k GSIS of 2.024 ± 0.51 compared with 1k GSIS 1.232 ± 0.340; p < 0.05). We showed that 0.5k pseudoislets were highly viable and glucose-responsive with an average basal insulin secretion of 0.0427 ± 0.0077 and 0.0839 ± 0.0134 μg/L per islet when stimulated with 16.7 mM glucose (Fig. 1F). The 0.5k pseudoislets were chosen for all further experiments, as they offer high reproducibility and stability over time to study the effects of hypoxia on endogenous β-cell ECM expression.
Hypoxia induces apoptosis and impairs pseudoislet functionality in vitro
Pseudoislets were subjected for a period of 48 h to hypoxic conditions (1% oxygen). Β-cell death, proliferation, and functionality were evaluated to validate the hypoxic effect in the in vitro model (Fig. 2). The expression of caspase-3, which is a downstream activator of the apoptotic pathway, significantly increased under hypoxic conditions (Fig. 2A). 28 To verify that the activation of caspase-3 signaled to its final target, we looked at the number of cells positive for TUNEL, which identifies DNA fragmentation in nuclei that occurs during late-stage apoptosis. 28 We identified a significant increase of TUNEL+ cells in the hypoxia in vitro model (Fig. 2B; 0.54% ± 0.76% normoxia vs. 10.45% ± 0.764% hypoxia; p < 0.0001), confirming the completion of apoptotic pathways. The proliferative capacity of INS1E β-cells was impaired by the hypoxic culture condition, demonstrated by a significant loss of Ki67 expression (Fig. 2C; 79.27% ± 7.89% normoxia vs. 43.3% ± 11.34% hypoxia; p < 0.001).
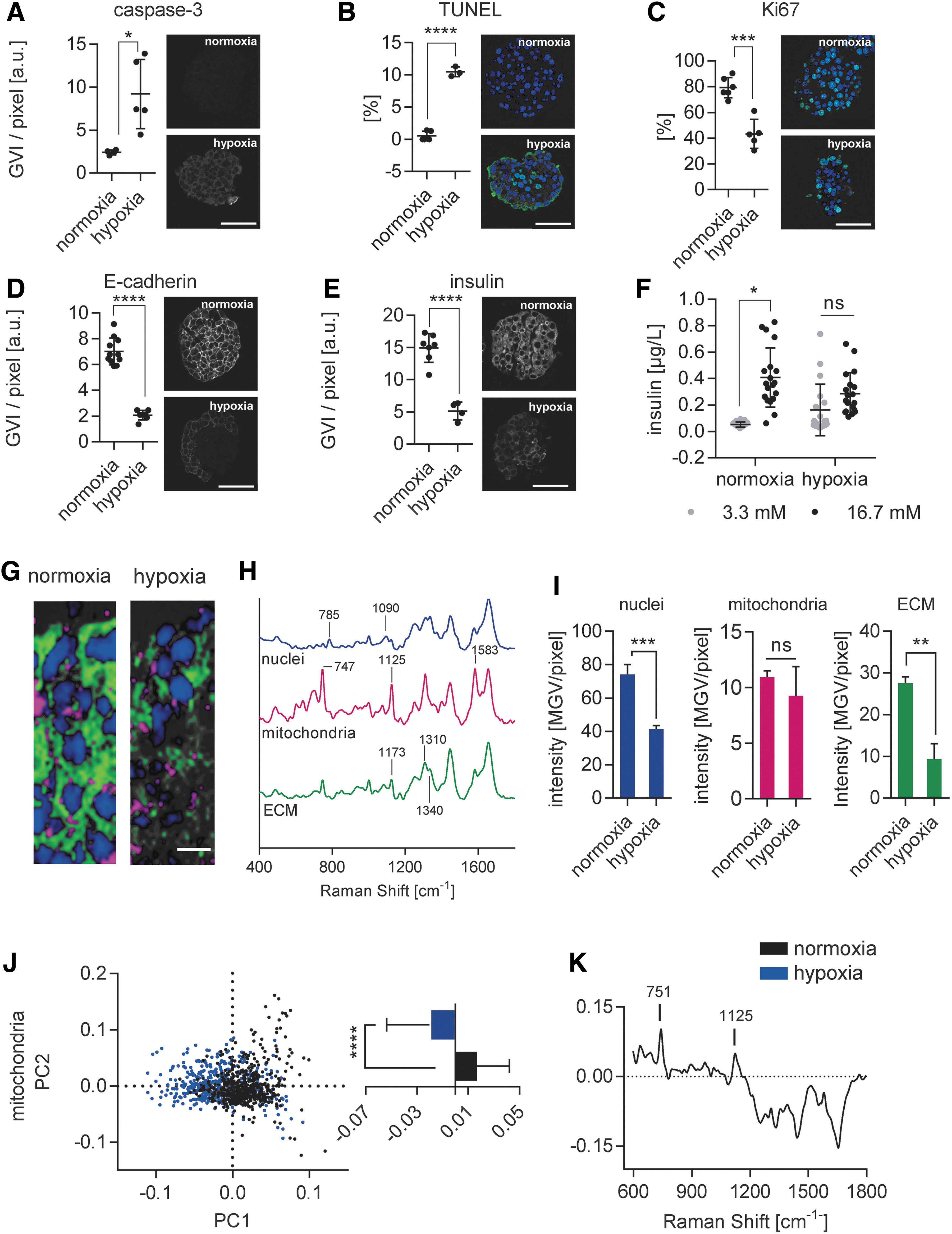
Hypoxia induces apoptosis and impairs pseudoislet functionality in vitro.
We assessed GSIS, insulin expression, and E-cadherin expression, which is an important cell–cell contact protein contributing to pseudoislet integrity and functionality under hypoxic conditions. 29 We showed that pseudoislets cultured under hypoxic conditions expressed significantly lower amounts of E-cadherin (Fig. 2D; 6321 ± 1086 gray value intensities (GVI)/pixel normoxia vs. 1940 ± 209 GVI/pixel hypoxia; p < 0.0001) and also expressed significantly less insulin (Fig. 2E; 14223 ± 2491 GVI/pixel normoxia vs. 4583 ± 1204 GVI/pixel hypoxia; p < 0.0001), contributing to a lack of response when stimulated by glucose (Fig. 2F).
To identify hypoxia-specific biochemical changes in situ within the living cells and cell-derived ECM in pseudoislets, Raman microspectroscopy and Raman imaging were used as noninvasive marker-independent techniques on living pseudoislets (Fig. 2G–K). 19 Raman imaging and TCA identified three main components in the pseudoislets that were previously assigned to nuclei, mitochondria, and ECM (Fig. 2G, H). Major peaks related to nuclei were identified as 785 cm−1 (DNA) and 826 and 1090 cm−1 (PO2 backbone). Mitochondria TCA was defined by intense peaks located at 747, 1125, and 1583 cm−1 describing heme vibrations in cytochrome C complexes.30,31 The ECM TCA was highly impacted by changes in collagens (1173, 1310, and 1340 cm−1).30,32,33 TCA quantification further revealed a significant decrease in the distribution of the nuclei component, indicating changes and/or loss of the nuclear biochemical spectral fingerprint, while there was no impact on the mitochondrial component (Fig. 2I).
For in-depth analysis of the TCA patterns within the mitochondrial component, Raman spectra were subjected to PCA. The multivariate data analysis of single spectra showed a separation according to the first principal component (PC1) (Fig. 2J). The loading of PC1 revealed that the major molecular differences responsible for the separation were 751 and 1125 cm−1, indicating that mitochondria of pseudoislets are more active under normoxic conditions (Fig. 2K). The negative range showed an overall higher protein signal in hypoxic pseudoislets, which may be the results of mitochondrial damage followed by the infiltration of cytoplasmic proteins.
Raman TCA quantification identified a significant alteration of the ECM proteins in hypoxic pseudoislets. This impact on crucial pancreatic ECM structures most likely contributes to the loss of β-cell function, as it has been previously shown that intraislet ECM interactions can modulate β-cell proliferation and survival. 34
Hypoxia differentially impairs the ECM protein expression in pseudoislets
To assess the effects of hypoxia on endogenous ECM expression in β-cells, we focused on three different groups of ECM proteins: BM proteins (LAM and COL4), the proteoglycans decorin (DCN) and nidogen-1 (NID1), and the fibril-forming proteins FN and COL1 (Fig. 3 and Supplementary Fig. S2). Comparative immunofluorescence staining verified the presence of the selected ECM proteins in both native human pancreatic tissue and pseudoislets cultured under normoxic conditions. However, semiquantification showed that hypoxia differentially regulated ECM expression. The expression of BM proteins LAM (Fig. 3A, B) and COL4 (Fig. 3C, D) was homogenous throughout pseudoislets cultured under normoxic conditions and significantly decreased in the hypoxia in vitro model (LAM: 12259 ± 985 GVI/pixel normoxia vs. 7628 ± 1241 GVI/pixel hypoxia, p < 0.001; COL4: 5980 ± 336 GVI/pixel normoxia vs. 3928 ± 900.97 hypoxia, p < 0.001).
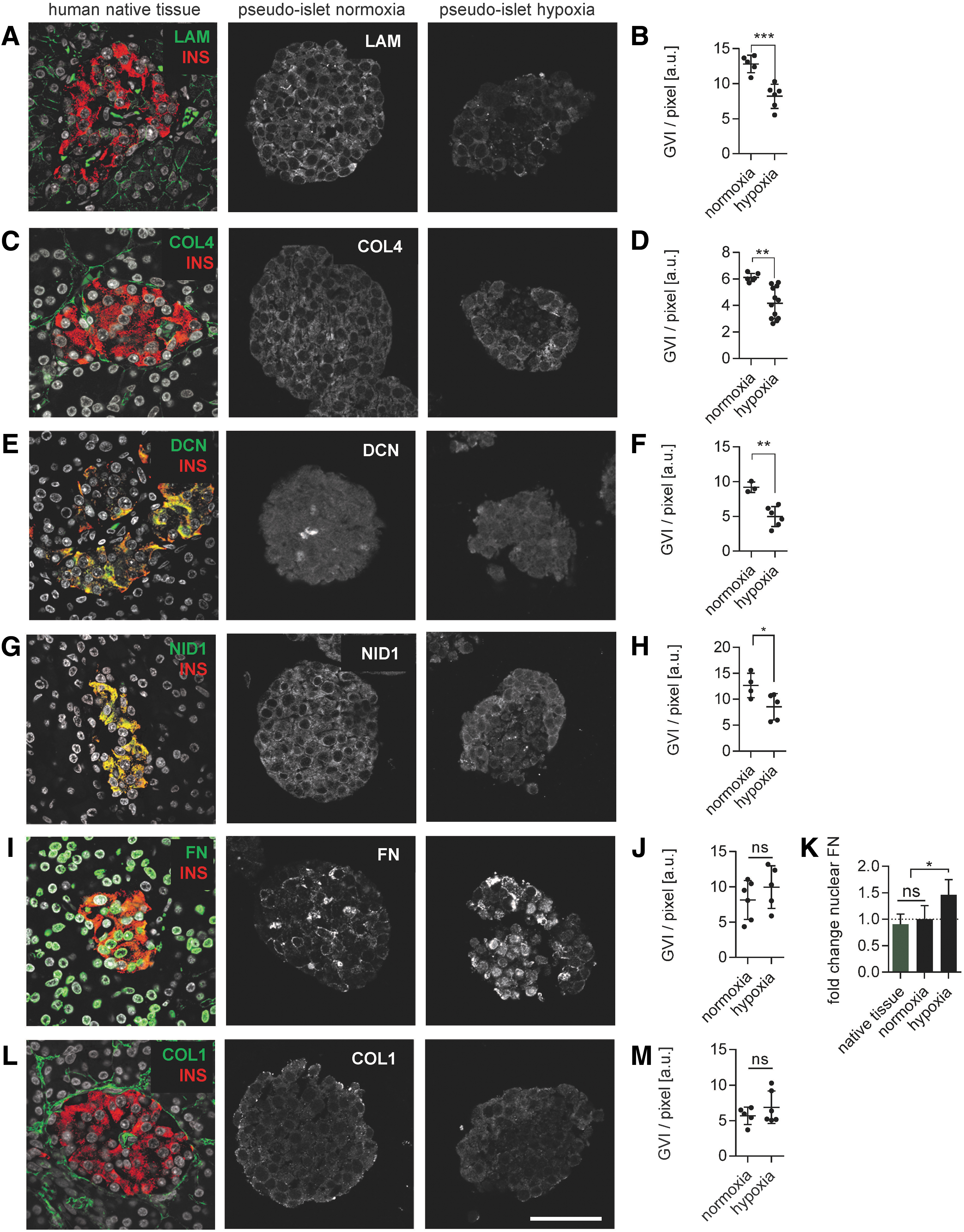
Hypoxia differentially impacts the expression patterns of BM proteins, glycoproteins, fibrillar, and fibril-associated ECM proteins. IF staining and quantification of different ECM proteins in human native tissue and in pseudoislets under normoxic and hypoxic culture conditions.
Similar to the BM proteins, the expression patterns of DCN (Fig. 3E, F; 8619 ± 436 GVI/pixel normoxia vs. 5464 ± 504 GVI/pixel hypoxia; p < 0.001) and NID1 (Fig. 3G, H; 12267 ± 1656 GVI/pixel normoxia vs. 7568 ± 1675 GVI/pixel hypoxia; p < 0.01) were homogeneous under normoxic conditions, but significantly decreased in the hypoxia in vitro model.
In contrast to BM proteins and proteoglycans, the expression levels of fibril-forming proteins (Fig. 3I–M) were not significantly impacted (FN: 7820 ± 2499 GVI/pixel normoxic conditions vs. 8454 ± 1840 GVI/pixel hypoxic conditions, p = 0.68; COL1: 5527 ± 1353 GVI/pixel normoxia vs. 5915 ± 1617 GVI/pixel hypoxia, p = 0.70). Although FN expression did not significantly decrease (p = 0.68), its cellular location shifted from cytoplasmic to nuclear after being subjected to hypoxia (Fig. 3I).
Changes in ECM composition have been described to promote transcriptional changes in the nucleus, potentially hinting toward cellular stress. 35 Therefore, we investigated whether cytoplasmic and nuclear expression of FN was present under hypoxic conditions (Fig. 3K). When normalized to the normoxia pseudoislet controls (1.0 ± 0.26), native human tissue did not show a significant fold change (0.91 ± 0.19, p = 0.75). In contrast, under hypoxic conditions, pseudoislets expressed significantly more nuclear FN (1.0 ± 0.26 normoxia vs. 1.46 ± 0.29 hypoxia; p < 0.01), reflecting increased cellular stress under hypoxic conditions.
Our data show that β-cells have the ability to express relevant pancreatic ECM proteins under normoxic conditions.34,36 However, hypoxia significantly impacted pancreatic BM proteins, glycoproteins, and FN, while COL1 expression remained stable.
COL1 hydrogel mimics native pancreatic tissue and attenuates hypoxic impact on pseudoislet functionality
To prevent ECM loss with concomitant loss of functionality under hypoxic conditions, we investigated the effect of encapsulating the pseudoislets in a COL1 hydrogel that is commercially available as an FDA-approved GMP-product and has been used to support β-cell function in normoxic conditions.37,38 Pseudoislets were encapsulated in a COL1 hydrogel, exhibiting similar COL1 expression patterns to native human adult pancreatic tissue (Fig. 4).
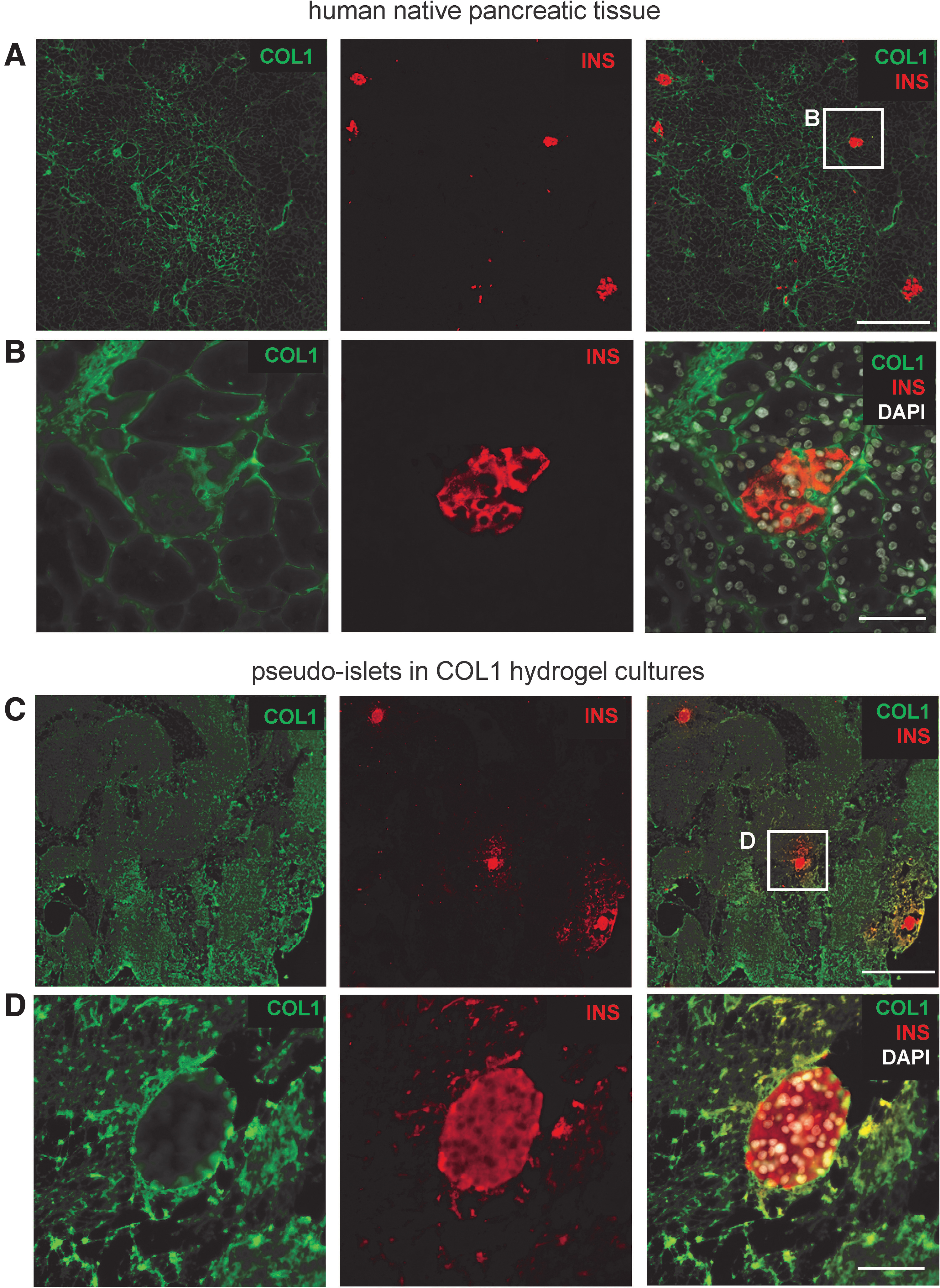
COL1 hydrogel mimics native pancreatic tissue.
The impact of COL1 hydrogel on pseudoislets under hypoxic conditions was investigated by comparing changes in apoptosis, proliferative capacity, basic β-cell functionality and ECM protein expression patterns with pseudoislets cultured in suspension under hypoxic conditions (Fig. 5A–F). The expression of the hypoxia-induced apoptosis marker caspase-3 (Fig. 5A; 4.41 ± 1.44 suspension vs. 1.77 ± 0.68 COL1 hydrogel; p < 0.05) as well as the number of TUNEL+ cells (Fig. 5B; 5.88 ± 1.67 suspension vs. 3.27 ± 0.76 COL1 hydrogel; p < 0.05) were significantly decreased in pseudoislets cultured in the 3D COL1 hydrogel when compared with pseudoislets grown in suspension cultures. Although the proliferative capacity between pseudoislets in suspension and COL1 hydrogel was comparable (Fig. 5C; 0.55 ± 0.14 suspension vs. 0.49 ± 0.15 COL1 hydrogel; p = 0.55), the expression of E-cadherin (0.31 ± 0.06 suspension vs. 0.75 ± 0.25 COL1 hydrogel; p < 0.01) and insulin (0.32 ± 0.09 suspension vs. 0.59 ± 0.12 COL1 hydrogel; p < 0.01) was significantly higher in pseudoislets in COL1 hydrogel (Fig. 5D, E). While hypoxia significantly decreased the overall insulin content of the pseudoislets in COL1 hydrogel, E-cadherin did not change significantly (Supplementary Fig. S3). Pseudoislets in COL1 hydrogel under normoxic and hypoxic conditions secreted significantly more insulin with 16.7 mM glucose stimulation than in the basal state (3.3 mM) (Fig. 5F; normoxia: 0.08 ± 0.03 basal state vs. 0.144 ± 0.04 16.7 mM glucose, p < 0.05; hypoxia: 0.09 ± 0.02 basal state vs. 0.16 ± 0.06 16.7 mM glucose, p < 0.05). These data demonstrate the beneficial effect of the COL1 hydrogel on pseudoislet functionality when comparing glucose diffusion and insulin release under hypoxic conditions.
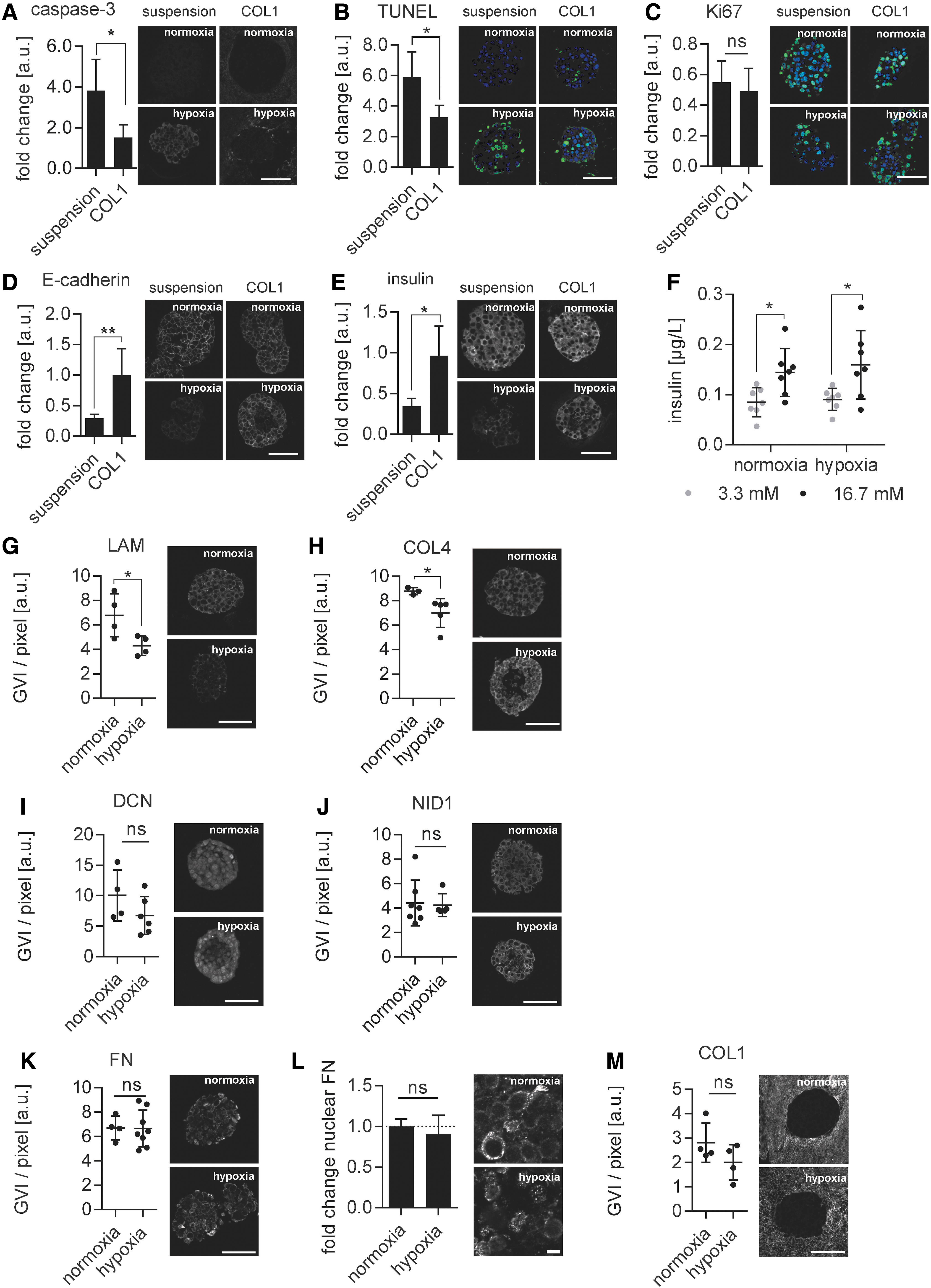
COL1 hydrogel improved pseudoislet functionality and minimized ECM changes due to hypoxia.
Exogenous biomechanical cues are known to impact cellular behavior. 1 Therefore, we investigated whether providing an external COL1 hydrogel changes the ECM protein secretion patterns in the pseudoislet cultures (Fig. 5G–M). Interestingly, only the secretion of LAM (6577 ± 1360 GVI/pixel normoxia vs. 4007 ± 594 hypoxia; p < 0.05) and COL4 (8553 ± 223 GVI/pixel normoxia vs. 7306 ± 708 GVI/pixel hypoxia; p < 0.05) significantly decreased, whereas the expression of DCN (9117 ± 2877 GVI/pixel normoxia vs. 6418 ± 1824 GVI/pixel hypoxia; p = 0.14) and NID1 (4451 ± 1488 GVI/pixel normoxia vs. 3996 ± 96 GVI/pixel hypoxia; p = 0.60), as well as fibril-associated (FN: 6655 ± 936 GVI/pixel normoxia vs. 7129 ± 1360 GVI/pixel hypoxia; p = 0.58) and fibrillar proteins (COL1: 9396 ± 2297 GVI/pixel normoxia vs. 6751 ± 2022 GVI/pixel hypoxia; p = 0.18) were not significantly impaired. In contrast to pseudoislets cultured in suspension, no change in nuclear FN expression was observed under hypoxia in pseudoislets encapsulated in the 3D COL1 hydrogel (Fig. 5L; 1.0 ± 0.09 normoxia vs. 0.90 ± 0.24 hypoxia; p = 0.46).
COL1 hydrogel functionalized with HUVECs supports endogenous pseudoislet BM protein expression
The encapsulation of pseudoislets in a COL1 hydrogel attenuated the hypoxic effect and preserved the expression of the glycoproteins DCN and NID1. However, the expression of BM proteins LAM and COL4 remained impaired. ECs are an important producers of BM protein in the pancreas. 9 Therefore, we established a co-culture of 1000 INS1E β-cells with 1000 HUVECs using magnetic levitation with HUVECs surrounding the β-cells as previously described. 17 After culturing control β-cell only pseudoislets and coculture pseudoislets in a COL1 hydrogel for 48h under hypoxic conditions, functionality, and ECM protein expression were assessed (Fig. 6). Coculture pseudoislets grown in the COL1 hydrogel were glucose responsive after 48h in hypoxia (Fig. 6A) and secreted significantly more insulin upon glucose stimulation compared with only β-cell-containing pseudoislets in COL1 hydrogels (Fig. 6B; 0.16 ± 0.06 COL1 hydrogel vs. 0.36 ± 0.24 COL1 hydrogel+HUVECs; p < 0.05), while the GSIS was not significantly changed (Supplementary Fig. S4A). Furthermore, evaluation of caspase-3, TUNEL, Ki67, E-cadherin, and insulin expression using IF staining did not show significant differences between the two groups (Supplementary Fig. S4B–F). The successful integration of HUVECs into the hydrogel was shown by significantly more CD31+ cells (Supplementary Fig. S4G; 0.07 ± 0.03 GVI/pixel COL1 hydrogel vs. 0.26 ± 0.11 GVI/pixel COL1 hydrogel+HUVECS; p < 0.01). The evaluation of the ECM protein expression in β-cell only pseudoislets and coculture pseudoislets in the COL1 hydrogel after 48h (Fig. 6C–I) revealed a significant increase in both LAM (1.24 ± 0.66 GVI/pixel COL1 hydrogel vs. 3.16 ± 0.96 GVI/pixel COL1 hydrogel+HUVECs; p < 0.01) and COL4 (1.62 ± 0.62 GVI/pixel COL1 hydrogel vs. 3.42 ± 1.24 GVI/pixel COL1 hydrogel+HUVECs; p < 0.01) as well as NID1 (1.23 ± 0.39 GVI/pixel COL1 hydrogel vs. 3.38 ± 0.85 GVI/pixel COL1 hydrogel+HUVECs; p < 0.001). Interestingly, both the overall expression of FN (2.55 ± 1.34 GVI/pixel COL1 hydrogel vs. 0.97 ± 0.33 GVI/pixel COL1 hydrogel+HUVECs; p < 0.05) and the normalized nuclear expression of FN (1.0 ± 0.15 COL1 hydrogel vs. 0.57 ± 0.20 COL1 hydrogel+HUVEC; p < 0.001) decreased significantly, while DCN (1.75 ± 0.33 GVI/pixel COL1 hydrogel vs. 1.44 ± 0.18 COL1 hydrogel+HUVECs; p = 0.12) and COL1 (5.10 ± 0.81 GVI/pixel COL1 hydrogel vs. 4.72 ± 1.37 GVI/pixel COL1 hydrogel+HUVECs; p = 0.64) were not affected by the HUVEC coculture. These data demonstrate the stimulative capacity of HUVECs on β-cells regarding functionality, BM protein expression, and decrease in hypoxia-induced cellular stress markers.
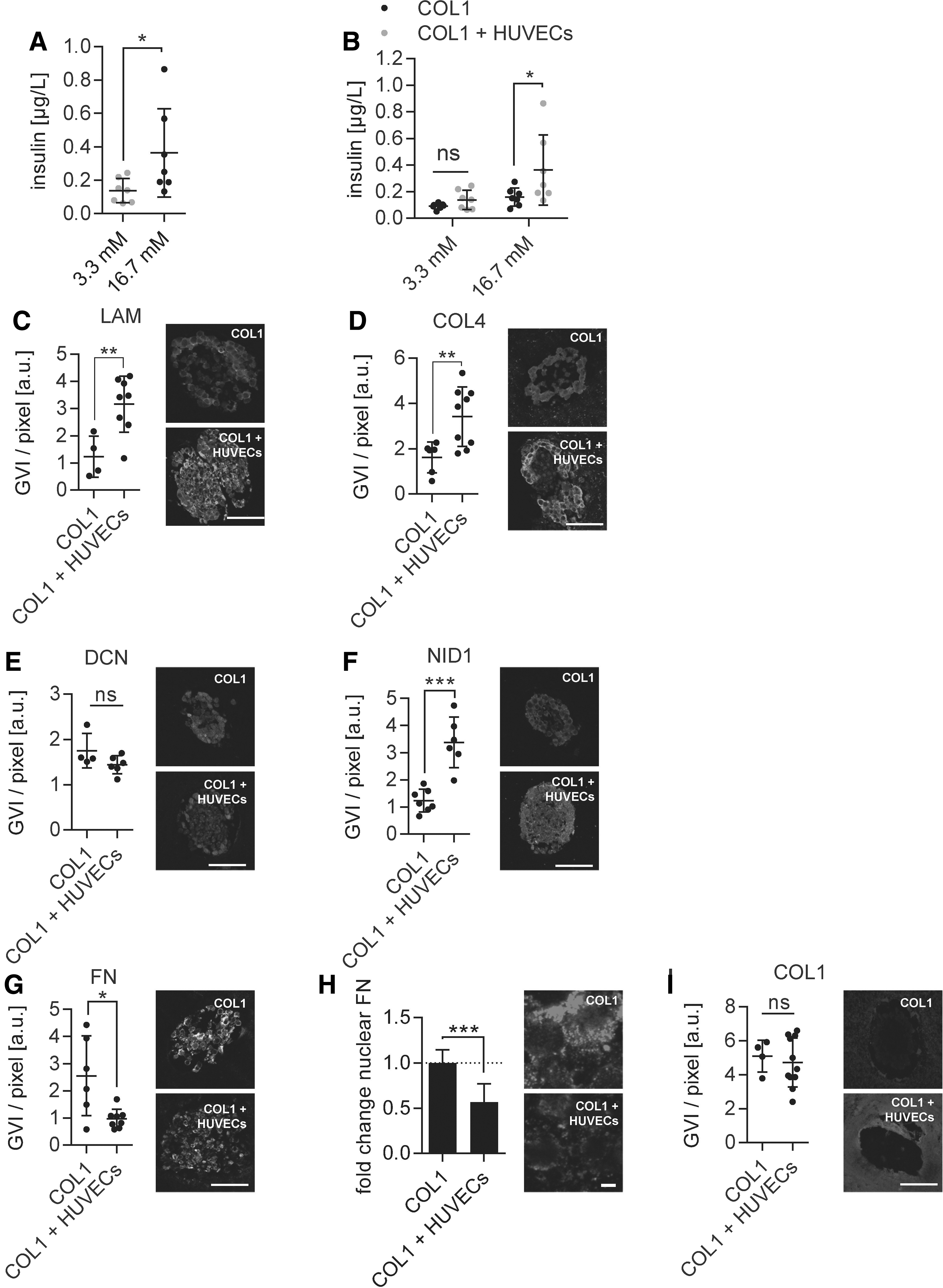
COL1 hydrogel with incorporated HUVECs stimulates insulin and BM protein secretion in β-cell-containing pseudoislets.
Discussion
In healthy native pancreatic tissue, pancreatic β-cell functionality is supported by a highly specialized composition of ECM proteins produced by various cell types, including fibroblasts and ECs.9,39 A dense vascularization of the pancreas further ensures highly oxygenated blood and nutrient delivery. 40 Using current protocols, during the Islets of Langerhans isolation procedure, the majority of the ECM and vascular access are destroyed, resulting in decreased islet survival and functionality.8,41 Posttransplantation, the Islets of Langerhans are subjected to an ischemic period leading to severe hypoxia, with a partial oxygen pressure ranging from 5 to 10 mmHg, corresponding to 1% oxygen. 42 The removal of the pancreatic ECM, which is accelerated by hypoxic conditions, accentuates the importance for β-cells to produce their own microenvironment. However, the role of β-cell ECM production in hypoxia had not been investigated yet.
In this study, we asked whether β-cells have the ability to secrete ECM proteins and how their production is impacted by a hypoxic environment that mimics the initial phase posttransplantation where no vascularization is present. We developed a functional rat-based model to successfully mimic hypoxic conditions to study the direct effect of hypoxia on β-cells. In line with previously reported studies, β-cell glucose responsiveness was lost under hypoxic conditions accompanied by increased apoptosis and necrosis markers, especially in the core region of the islets.16,43 Here, marker-free Raman imaging on living pseudoislets identified a significant hypoxia-induced alteration in ECM homeostasis. This impact on pancreatic ECM structures may contribute to the loss of β-cell function, as it had been previously shown that intraislet ECM interactions can modulate β-cell proliferation and survival. 34 The overall decrease in ECM signal within pseudoislets supports our hypothesis that hypoxia negatively impacts the ECM expression of β-cells.
The specific roles of the different types of ECM regarding Islets of Langerhans transplant survival and function are not fully understood. 44 Hence, we assessed the hypoxia-induced disruption of ECM homeostasis on six different ECM proteins. DCN is an important binding partner of collagens (mainly types I and III) that regulates collagen fibrillogenesis. The loss of DCN can lead to uncontrolled collagen fiber formation, which then results in pathological ECM development such as fibrosis and ultimately graft failure. 45 The hypoxia-induced decrease of DCN in the pseudoislets after 48 h expression can be a driver for early graft failure as a controlled fibrillogenesis of COL1 and COL3 may be impaired. Furthermore, DCN has been shown to positively affect angiogenesis and modulate the immune response,46,47 which are both highly important processes to achieve proper transplant engraftment.
The main purpose of COL1 and other parts of the exocrine interstitial matrix is the preservation of structural stability 48 as well as assuring β-cell survival and insulin expression by stimulating a variety of surface receptors. 49 Although COL1 overall expression remained stable, the quality or maturity of COL1 fibers might be impaired by the decreased DCN expression. Immature COL1 has not been described yet to act via the same integrins as fully formed COL1, potentially contributing to the loss of glucose responsiveness. 50
FN is another component of the interstitial matrix. 51 The impact of FN on β-cells is currently controversially discussed. Freshly isolated human Islets of Langerhans showed an improved glucose response on surfaces coated with FN. 37 Hadavi et al. recently published the positive effect of FN in combination with COL4 and LAM to stimulate β-cell functionality in vitro. 52 In contrast, Navarro-Alvarez et al. demonstrated that isolated human Islets of Langerhans, which were attached to FN-coated dishes, showed a clear tendency toward disintegration and loss of spherical structure. 53 Here, FN expression patterns exhibited a hypoxia-induced shift from cytoplasmic to nuclear expression, especially in the central region of the pseudoislets. Since FN is present in tissues to repair damaged cells, the increase in nuclear FN expression may be the first indicator for a hypoxia-induced cell damage. 54 Nuclear FN has been shown to be present in some tumor cells; however, the precise role of nuclear FN remains unclear. 55
In contrast to proteins of the interstitial matrix, the expression of ECM proteins associated with the BM were highly affected by hypoxic conditions. In the pancreas, Islets of Langerhans are located in the BM, a dense network of copolymerized LAM and COL4. These proteins are connected by NID1, which acts as key bridging protein between LAM and COL4 in native tissues. 56 Laminin α5 and COL4 have been reported to modulate β-cell differentiation and maturation from stem cells into mature β-cells and stimulate insulin expression in pseudoislet systems,57,58 highlighting the relevance of BM proteins for β-functionality in vitro. Since the expression of all proteins of the BM was reduced in our study under hypoxic conditions, this lack of BM proteins may be associated with functionality loss. Hence, the rescue of BM proteins may support β-cell function under hypoxic conditions posttransplantation.59,60
One approach to reduce hypoxia-induced damage and altered ECM expression is to provide exogenous ECM to the cellular transplant. The addition of a protective COL1 hydrogel surrounding the β-cell pseudoislets significantly reduced the negative impact of hypoxia. While apoptotic and necrotic cell numbers decreased, functionality markers increased, and glucose responsiveness was restored. The results of previous studies on the effect of COL1 as well as the reduced number of apoptotic markers and dead cells in this study point toward a combination of biological and mechanophysical support by the COL1 hydrogel.61–63
Assessment of the endogenously expressed ECM proteins showed that DCN and NID1 could be restored. DCN binds COL1 via the leucine-rich repeats five to six to regulate its assembly, structure, and biomechanical properties. 64 The rescue of DCN might therefore be facilitated by the supply of the COL1 hydrogel, which offers possible binding sites. However, an interaction between COL1 and NID1 has not been reported yet. Interestingly, the translocation of FN into the nucleus was prevented, supporting the observation that the COL1 hydrogel attenuates the cellular stress levels of the hypoxic environment.
Although the levels of NID1 could be restored, the LAM and COL4 did not show any signs of rescue. The persistent lack of BM proteins suggests that providing β-cells with only COL1 is not enough for a long-term restoration of glucose-responsiveness. Therefore, we hypothesized that stimulation of β-cells by ECs might enhance the expression of BM proteins, as ECs are the main producers of BM proteins in pancreatic tissues. 9 We encapsulated both β-cells and HUVECs into the pseudoislets and cultured them in COL1 hydrogels. We identified that upon encapsulation of β-cells together with HUVECs, we could both stimulate functionality of β-cells as well as rescue BM protein expression of LAM, COL4, and the linker protein NID1 (Table 1). In addition, overall FN expression was reduced, which might be precipitated by the highly significant decrease in nuclear FN, an indicator for reduced hypoxia-induced cellular stress. 29 Furthermore, FN regulates the fibrotic response along with COL1, 49 which is unfavorable for engraftment of the transplant. Consequently, a drop in FN content may have a beneficial long-term effect.
Overview of Extracellular Matrix Protein Expression Changes in Collagen Type 1 Hydrogel of β-Cell Pseudoislets and Coculture Pseudoislets After 48 h Under Hypoxic Culture Conditions (Normalization to Pseudoislets Cultured Under Normoxic Culture Conditions)
The up arrow indicates significant upregulation (p < 0.05) and the down arrow indicates significant downregulation (p < 0.05).
COL1, collagen type 1; COL4, collagen type 4; DCN, decorin; EC, endothelial cell; ECM, extracellular matrix; FN, fibronectin; HUVECs, human umbilical vein endothelial cells; LAM, laminins; NID1, nidogen-1; n.s., not significant.
In summary, our data demonstrate the supportive effect of the COL1 hydrogel protecting β-cell-composed pseudoislets under hypoxic conditions by reducing apoptotic effects and cell death, attenuating a loss of ECM protein secretion while rescuing the glucose responsiveness. We further showed how the combination of biological cues from ECs in a coculture system with COL1 hydrogel rescued BM protein expression and improved the glucose responsiveness. Importantly, COL1 is a bioactive base hydrogel that can be further modified and functionalized using growth factors, other ECM proteins or cell types to meet specific requirements, for example to improve transplant function,5,24,37,61–63,65 or to create a more robust in vitro model. Future studies could include the addition of α and δ cells into the pseudoislets. Also, the investigation of reoxygenation posthypoxia would be of interest as it may elucidate key biomolecules influencing the transplantation and healing process; however, it may be difficult to mimic the exact time response and oxygen percentages in vitro that are found during the healing processes in vivo. In addition, the addition of immune cells would be of high interest as the foreign body response to islet transplantation is a major issue requiring the use of immune suppressants posttransplantation. Many groups are working on the addition of immune cells into in vitro test systems in general, which has been difficult in the past due to the complexity of the immune system. Any addition to in vitro model complexity must be weighed against its increase in cost and the ability to determine valid readouts that can answer the scientific question being asked.
Conclusion
In this study, we developed an in vitro model mimicking the hypoxic posttransplantation environment of pancreatic islets for the purpose of investigating and mitigating the pathological effects of hypoxia on ECM homeostasis with a functionalized material. We identified a significantly reduced production of important ECM proteins such as COL4, LAM, DCN, and NID1 within β-cells accompanied with increased cellular death and loss in functionality. Furthermore, we evaluated a clinically approved COL1 hydrogel and demonstrated its protective effect on β-cells in hypoxia. We further functionalized the hydrogel with HUVECs, which prevented ECM loss and stimulated β-cell functionality. By establishing a COL1 hydrogel, including ECs, we created a carrier matrix that attenuates the hypoxia-induced disruption of ECM homeostasis in β-cells to support them during the first phase of ECM reestablishment of posttransplantation and therefore potentially increase the efficacy of the Edmonton protocol for diabetic patients.
Footnotes
Disclosure Statement
No competing financial interests exist.
Funding Information
This work was financially supported by the European Union (H2020-NMP10-2014-645991-2, DRIVE to Katja Schenke-Layland and Garry Duffy), the International Foundation for Ethical Research (to Aline Zbinden), Germany's Excellence Strategy (EXC 2180-390900677 to Katja Schenke-Layland), as well as the Ministry of Science, Research and the Arts of Baden-Württemberg (33-729.55-3/214 and SI-BW 01222-91 to Katja Schenke-Layland), and the Deutsche Forschungsgemeinschaft (INST 2388/33-1 and GRK 2543/1 to Katja Schenke-Layland).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
