Abstract
Cells are inherently conferred with the ability to self-organize into the tissues and organs comprising the human body. Self-organization can be recapitulated in vitro and recent advances in the organoid field are just one example of how we can generate small functioning elements of organs. Tissue engineers can benefit from the power of self-organization and should consider how they can harness and enhance the process with their constructs. For example, aggregates of stem cells and tissue-specific cells benefit from the input of carefully selected biomolecules to guide their differentiation toward a mature phenotype. This can be further enhanced by the use of technologies to provide a physiological microenvironment for self-organization, enhance the size of the constructs, and enable the long-term culture of self-organized structures. Of importance, conducting self-organization should be limited to fine-tuning and should avoid over-engineering that could counteract the power of inherent cellular self-organization.
Impact Statement
Self-organization is a powerful innate feature of cells that can be fine-tuned but not over-engineered to create new tissues and organs.
The Power of Self-Organization
From a simple zygote, our stem cells choreograph their own assembly and differentiation, subsequently creating all the tissues and organs in the adult human body with high fidelity. This phenomenon is called self-organization, and cells are naturally conferred with this astonishing ability. For millennia, philosophers and biologists alike have recognized the power of self-organization. For example, Immanuel Kant first introduced the term in 1790 when he argued that there must exist an entity with parts that are capable of governing itself. Self-organization principles can be seen in many facets of our daily lives: from the crystallization of a salt solution to the flocking of birds.
Self-organization can be achieved by self-assembly, self-patterning, and self-driven morphogenesis, which are distinct processes driven by different factors.1,2 In self-assembly, cells change positions over time. In self-patterning, a homogeneous population of cells become heterogeneous in response to different spatial and temporal cues. In self-driven morphogenesis, cells change shape using intrinsic mechanics and cues. Although these principles can be found in many current tissue engineering strategies, we propose that to realize the full potential of the cells we use, scientists need to give greater credence to the cells' ability to self-organize themselves, thus ensuring that our engineering does not interfere with this powerful process.
Advances in Self-Organization
As the field of tissue engineering evolved, it has moved from an era of serendipitous discovery to one that aims to take inspiration from development and endogenous repair processes.3,4 For example, the precisely orchestrated spatial and temporal cues of development have been recapitulated to engineer diverse organs such as bone and liver.5,6 Likewise, by studying endogenous repair in vivo, 4 important molecules have been identified and used to enhance the regeneration of, for example, periodontal structures (i.e., alveolar bone, periodontal ligament, and root cementum) and bone in vitro.7,8 The field has also taken notice that three-dimensionality is important for directing cell fate, especially with regard to maturation, 9 and the importance of the extracellular matrix (ECM) 10 and cell–cell contact have both become well-established maxims. 11 Nonetheless, we propose that many tissue engineering strategies would be improved were scientists to fully appreciate the power of self-organization and design their constructs in a way that promoted rather than hindered the cell interaction needed to drive self-organization.
We would be remiss not to consider the role of self-organization in the formation of a patent vasculature. One of the major challenges within tissue engineering is the mass transport problem that occurs when a three-dimensional (3D) tissue is generated, where simple diffusion cannot meet the needs of the construct. Self-organization can create the mature and functional vasculature needed for a functional tissue 12 and it appears to be an optimal strategy when the vasculature is formed in tandem with the respective organ and not as separate entities because of the crosstalk involved.
For example, the renal glomerulus is highly vascularized and is connected to Bowman's capsule. It appears the latter, which is derived from the metanephric mesenchyme, is at least partly responsible for recruiting endothelial cells to the glomerulus, underlining the need to consider these two cell types of the nephron together.13,14 A similar situation exists in the liver, where the central vein and hepatic triads comprising the hepatic duct, hepatic portal vein, and artery are best generated together, not as separate entities, so the necessary crosstalk can occur. 15 There is a role for technology in promoting vascularization,12,16,17 although self-organization principles remain.
Self-organization, by definition, does not require the control of an external agent, but we have seen repeatedly that some inductive cues are almost always needed in vitro. Although muscle and cartilage have been regenerated by encouraging self-organization of muscle cells from a biopsy and mesenchymal stem cells (MSCs),18,19 respectively, more complex organs like the kidney, liver, or heart require the input of a so-called external agent.20,21 For example, the seminal discovery that Lgr5+ cells could self-organize after induction to form cystic structures like the intestinal crypt villus 22 was followed by examples in many other tissues.
Other types of organoids are self-organizing clusters of cells, such as human pluripotent stem cell (hPSC)-derived kidney organoids that develop vascular networks after transplantation and perform glomerular perfusion.14,23–25 With the right culture conditions, a single progenitor cell can reconstruct the self-formed ectodermal autonomous multizone or the renal ureteric bud through a process resembling branching morphogenesis.26,27 In addition, pancreatic islet organoid self-organization was further enhanced by the addition of other cell types like endothelial cells and MSCs, and matrigel.28–30 Beyond organs, one recent example of the power of self-organization was the formation of mouse blastocyte-like structures termed blastoids, which were formed from trophoblast and embryonic stem cells (ESCs) to morphologically and transcriptionally resemble embryonic day 3.5 blastocysts 31 (Box 1).
Box 1: Self-Organization Combined with Enabling Technologies Can Make a Blastocyst in a Dish
The organization of embryonic stem cells and trophoblast stem cells into a blastocyst-like structure termed a blastoid required two key technologies: a microfabricated microwell platform and the biochemical induction of key pathways that were informed by studying embryogenesis. High-throughput screening was used to determine that a Wnt activator and a cyclic adenosine monophosphate analog could induce the self-organization. 31 This blastoid was determined to resemble a blastocyst from embryonic day 3.5. The process of self-organization is defined as self-driven morphogenesis, because also internal cellular signaling is altered by the added small molecules. In addition, it requires symmetry breaking to form the inner cell mass, blastocyst cavity, and trophoblast. 60 This blastoid could be implanted in utero in mice forming patterned deciduae. However, it is currently not able to generate a complete embryo in a dish. The formation of a complete embryo is likely to require new technologies and a deeper understanding of the underlying biology.
Limitations of Self-Organization
Although self-organization is undoubtedly a powerful process, there is a limit to how well it can proceed. The lack of tissue-specific regenerative capacity, tissue complexity, and 3D connections with other tissues could limit self-organization. 32 For example, self-organization of the multilayered epithelium of the skin in vitro happens spontaneously by self-assembly. In addition, the self-organization of the pancreas or the retina in vitro is limited to the formation of immature beta cells or the optic cup.33,34 To enhance self-patterning and formation, self-organization should be enhanced. A complex tissue like a neuron that has many connections with different tissues and a limited regenerative repair capacity upon injury is even more difficult to self-organize. It can only be formed in parts and patterning factors such as Fgf, Wnt, and BMP are essential to induce self-organization by self-morphogenesis35–37 (Fig. 1).
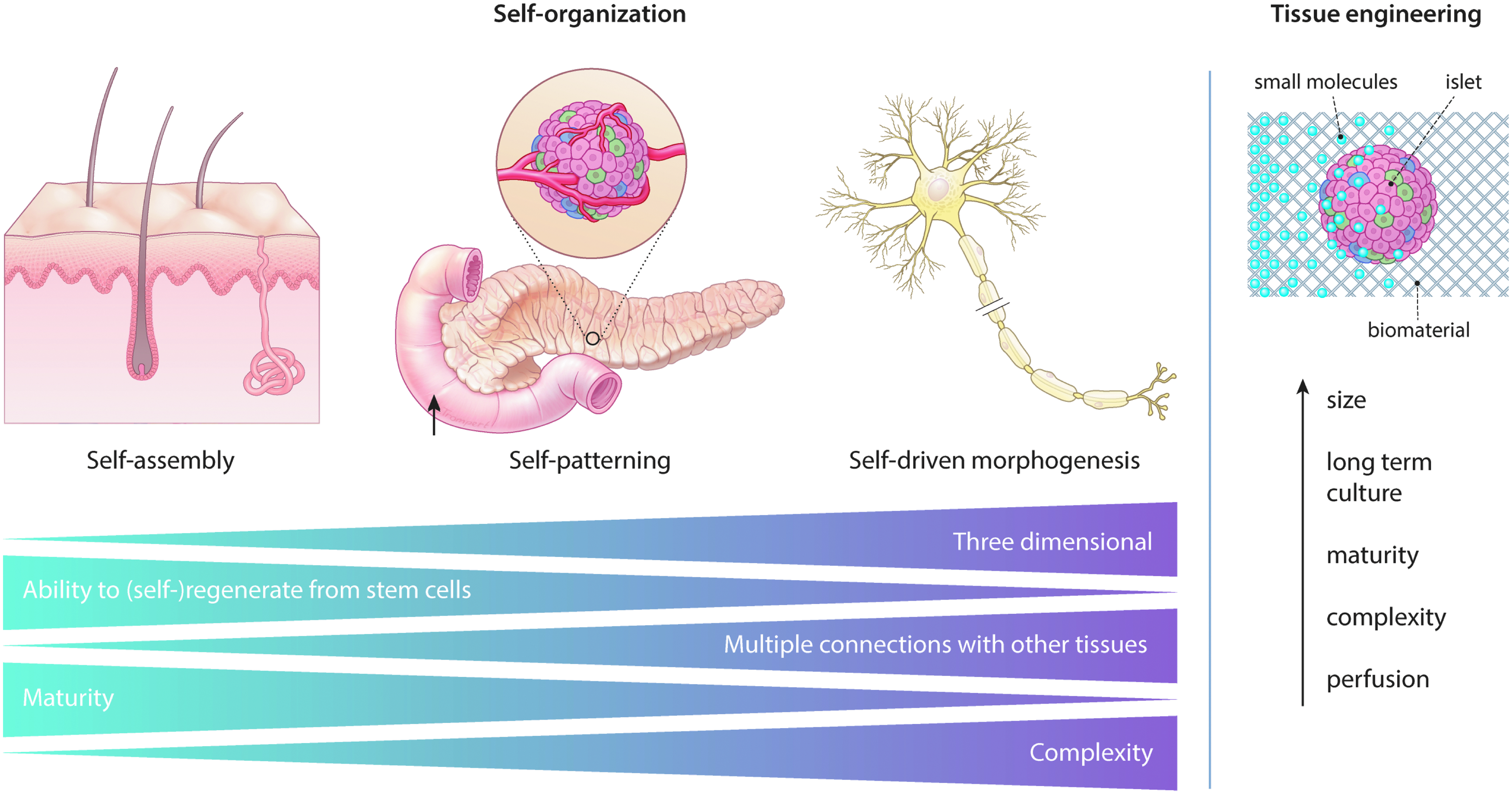
Self-assembly can induce the formation of the skin epithelium, self-patterning forms the pancreatic islets of Langerhans, and self-driven morphogenesis induces self-organization of a neuron. It becomes more difficult to induce self-organization as tissues become more complex, for example, with more cell types or less differentiation potential. Tissue engineers can fine-tune self-organization with their technologies like microfluidics and microfabrication that can provide small molecules and an engineered microenvironment to enhance the size, long-term culture, perfusion, maturity, and complexity of the self-organizing structures, without over-engineering by interfering with the process of self-organization itself.
Clearly from these observations, self-organization in vitro is accompanied by several important limitations. By tackling these limitations, self-organization could be enhanced. First, enough starting material is essential to induce self-organization. For example, human gastruloids, which are ESC-derived aggregates that stimulate gastrulation-like events, disappear on small micropatterns, because of too little tissue to induce self-organization. 38 Second, the maturity of in vitro self-organized tissue could be enhanced perfusing vasculature to release endogenous signals and provide external factors. 39 Third, mass transport problems should be prevented by enhancing perfusion and increasing the concentrations of oxygen and nutrients. Finally, the important crosstalk between cell types such as immune cells and the nervous system should be promoted by adding all the necessary cell types. The trick is to fine-tune the process of self-organization, while not interfering with the tissue's own ability to self-organize. Fortunately, tissue engineers have already developed many of the tools and technologies needed to lend a helping hand to self-organizing cells, without interference.
Boosting Cells' Intrinsic Capacity to Self-Organize: Small Molecules and Growth Factors
Well-considered tools in tissue engineering can be used to assist cell self-organization. The most prevalent example is surely the addition of small molecules, a strategy that has been widely used with success, for example, in the self-organization of skin or the optic cup organoids.33,40,41 Examples abound on the use of growth factors in directed differentiation of stem or progenitor cells to functioning and specified cell types, and by identifying these factors, self-organization can be improved, leading to the successful formation of mature tissue.
For example, pancreatic beta cell differentiation, which had long been impossible to recapitulate in vitro, can be made possible by adding harmine, a β-carboline alkaloid present in plants, which has been found to enhance beta cell proliferation and differentiation by targeting dual-specificity tyrosine-regulated kinase-1a (DYRK1A) and the nuclear factors of activated T cells (NFAT) family of transcription factors. 42 In addition, adding Wnt signaling antagonist Dickkopf-related protein 1 (DKK-1) could induce the formation of 3D optic vesicles from human-induced pluripotent stem cells that were normally not able to self-organize and differentiate toward retinal tissue. 43 Finally, to enhance connectivity of different neural parts, for example, the neuroepithelium-derived Purkinje cells, interneurons, granule cells, and cerebellar nuclei neurons from the rhombic lip, fibroblast growth factor 19 (FGF19), and stromal cell-derived factor 1 (SDF1) were supplemented. This led to the generation of the continuous connected cerebellar plate from human ESC-derived cerebellar progenitors, creating an early human cerebellum resembling the first trimester. 44
Caution, however, is needed with these approaches. The addition of growth factors or small molecules can also interfere with the important spontaneous formation of gradients that pattern developing tissues 45 and may disrupt the carefully orchestrated competition from different directional cues. 46
Engineered Microenvironment
An engineered microenvironment composed of biochemical and biophysical cues can be added to positively influence self-organization. For example, mechanical forces mimicking those found in development or in adult tissues can be provided by a biomaterial or bioreactor and this has been shown to contribute to the self-organization of a specific tissue.47–49 These microenvironments can provide boundaries for a specific tissue or mimic its ECM. For example, a decellularized matrix or mesh substrate can create a cell-adhesive microenvironment that induces self-organization and differentiation to generate chondrocytes from MSCs to enhance formation of cartilage.50,51 Gradients of morphogens can be established by microfluidics or microfabrication on the micro- or nanometer scale52,53 and combined with an engineered ECM expressing specific nanotopography or through 3D (bio) printing.54–56 However, one should be careful with designing biomaterials or matrices of poorly defined composition, heterogeneous nature, and batch-to-batch variability, which could inhibit the process of self-organization.
New Technologies to Enhance Self-Organization
To further enhance cell self-organization, new tools can be applied. These can be used to address the limitations related to self-organization. The size and long-term culture of organoids can be increased by perfusing a vasculature with microfluidics. This will prevent oxygen and nutrient deprivation because of a lack of functional vasculature. Recent successes were demonstrated with a perfusable chip containing a bioprinted 3D renal proximal tubule 57 and a microfluidic device that enhances primitive streak formation from hPSCs by inducing asymmetric cell patterning. 58 In addition, the maturity of the organoids or microtissues generated in vitro could be enhanced by creating structures on the micro- and nanoscale that allow for patterned functionalization of either physical, chemical, or biological cues on the same scale. By microthermoforming these structures, the right gradients of endogenous factors and external cues to further promote self-organization could be provided. For example, the formation of nephron-like structures such as podocytes, proximal tubules, loops of Henle and distal tubules in an organized, continuous arrangement from nephron progenitor cells derived from hPSCs could be enhanced by adding a small molecule like Noggin, BMP4 inhibitor, to the device. 25
By applying these technologies, the self-organization of tissue that is difficult to self-organize can be enhanced. This approach has recently been referred to as “narrative engineering,” 59 in which the engineering space is combined with biological (small molecules and ECM) and synthetic environmental control (e.g., perfusion, mechanical, and electrical stimuli). However, we caution tissue engineers to not over-engineer. We should be careful about interfering with precisely regulated processes and disrupting the inherent power of self-organization. We should limit our role to fine-tuning and be careful not to prevent cells from communicating with each other. We should avoid situations where single cells are seeded because tissues have complex cell–cell communication, and instead consider generating small aggregates or organoids as an intermediate. This will enhance their complexity and maturity by creating their preferred microenvironment. Just as the conductor of an orchestra aims to get the most out of their musicians, tissue engineers should orchestrate the cells in their constructs with an inspired, but light touch.
Footnotes
Acknowledgments
The authors thank Hang Nguyen (MERLN/M4I, Maastricht University, the Netherlands) for her help revising the article.
Disclosure Statement
No competing financial interests exist.
Funding Information
This research has received funding from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement No. 694801).
