Abstract
The loss of expression of chondrogenic markers during monolayer expansion remains a stumbling block for cell-based treatment of cartilage lesions. Here, we introduce sulfated alginate hydrogels as a cartilage biomimetic biomaterial that induces cell proliferation while maintaining the chondrogenic phenotype of encapsulated chondrocytes. Hydroxyl groups of alginate were converted to sulfates by incubation with sulfur trioxide–pyridine complex (SO3/pyridine), yielding a sulfated material cross-linkable with calcium chloride. Passage 3 bovine chondrocytes were encapsulated in alginate and alginate sulfate hydrogels for up to 35 days. Cell proliferation was five-fold higher in alginate sulfate compared with alginate (p=0.038). Blocking beta1 integrins in chondrocytes within alginate sulfate hydrogels significantly inhibited proliferation (p=0.002). Sulfated alginate increased the RhoA activity of chondrocytes compared with unmodified alginate, an increase that was blocked by β1 blocking antibodies (p=0.017). Expression and synthesis of type II collagen, type I collagen, and proteoglycan was not significantly affected by the encapsulation material evidenced by quantitative reverse transcription polymerase chain reaction (qRT-PCR) and immunohistochemistry. Alginate sulfate constructs showed an opaque appearance in culture, whereas the unmodified alginate samples remained translucent. In conclusion, alginate sulfate provides a three dimensional microenvironment that promotes both chondrocyte proliferation and maintenance of the chondrogenic phenotype and represents an important advance for chondrocyte-based cartilage repair therapies providing a material in which cell expansion can be done in situ.
Introduction
A
The cartilage extracellular matrix (ECM) is composed of 10–20% proteoglycans. 5 The core proteins of proteoglycans are heavily modified by glycosaminoglycans (GAGs) including chondroitin sulfate, keratan sulfate, and dermatan sulfate. These linear polysaccharides have sulfate and carboxylic groups, which contribute to the high fixed negative charge of the proteoglycans at physiologic pH. The GAG side chains also give the tissue its compressive and osmotic swelling properties by entrapping water. 3
Sulfation of molecules has purported effects on the biological activity of biopolymers. Chondroitin sulfate has been shown to have therapeutic benefits for hip and knee osteoarthritis.23,24 Heparan sulfate has a high affinity to a plethora of growth factors crucial for cartilage homeostasis.25,26 Freeman et al., studied the binding affinity of 10 different heparin-binding factors (including FGF, IGF, and VEGF) to alginate sulfate. 27 They found that except FGF most of the factors bind equally well or better to sulfated alginate compared with heparin. Re'em et al., showed that scaffolds containing a mixture of alginate and sulfated alginate caused attenuated transforming growth factor beta-1 (TGFβ-1) release and consequently improved chondrogenesis of entrapped mesenchymal stem cells compared with scaffolds lacking alginate sulfate. 28 The scaffolds used in these studies were made by freezing alginate sulfate into a macroporous scaffold. 27 In this study, we report for the first time the biological properties of a sulfated alginate hydrogel that is cross-linkable with calcium.
We hypothesized that modification of alginate with sulfate groups provides a more chondrogenic environment compared with pure alginate. To test this hypothesis alginate was modified with sulfate groups and chondrocytes were encapsulated in alginate and alginate sulfate at varying concentrations. Cell morphology, proliferation, gene expression, and synthesis of cartilage markers were analyzed.
Materials and Methods
Materials
Phosphate buffer saline (PBS), fetal bovine serum (FBS), cell culture media (DMEM-Glutamax), antibiotic-antimycotic, trypsin/EDTA, 4′,6-diamidino-2-phenylindole dilactate (DAPI), and Alexa 488 goat anti-mouse-conjugated IgG IgM (H+L) were from Invitrogen AG. Alginate Pronova UP LVG (low viscosity (20–200 mPaos) sodium alginate in which at a minimum 60% of the monomer units are α-
Preparation and characterization of sulfated alginate
Alginate was prepared at 0.5% (w/v) in water. The DOWEX Marathon C ion exchanger was charged with an equal mass of tetrabutyl ammonium bromide. Then, amounts of charged resin and alginate solution were mixed in a ratio of 1:10 (w/v), stirred overnight, filtered, and lyophilized. The yielded alginate tetrabutyl ammonium salt was suspended in water-free DMF at 1% (w/v) containing a five-fold excess SO3/pyridine per disaccharide unit. The mixture was stirred at room temperature for 1 h and the opaque solution was precipitated in acetone, brought to pH=12 (ethanolic NaOH) for 10 min, and then neutralized. The precipitate was filtered, dissolved in water, purified by dialysis, and lyophilized. The process of alginate sulfate synthesis is demonstrated in Figure 1. An important parameter to characterize the chemical composition of the alginate sulfate is the degree of sulfation (DSS). It gives the average number of sulfate groups per disaccharide repeating unit of alginate that is formed from β-
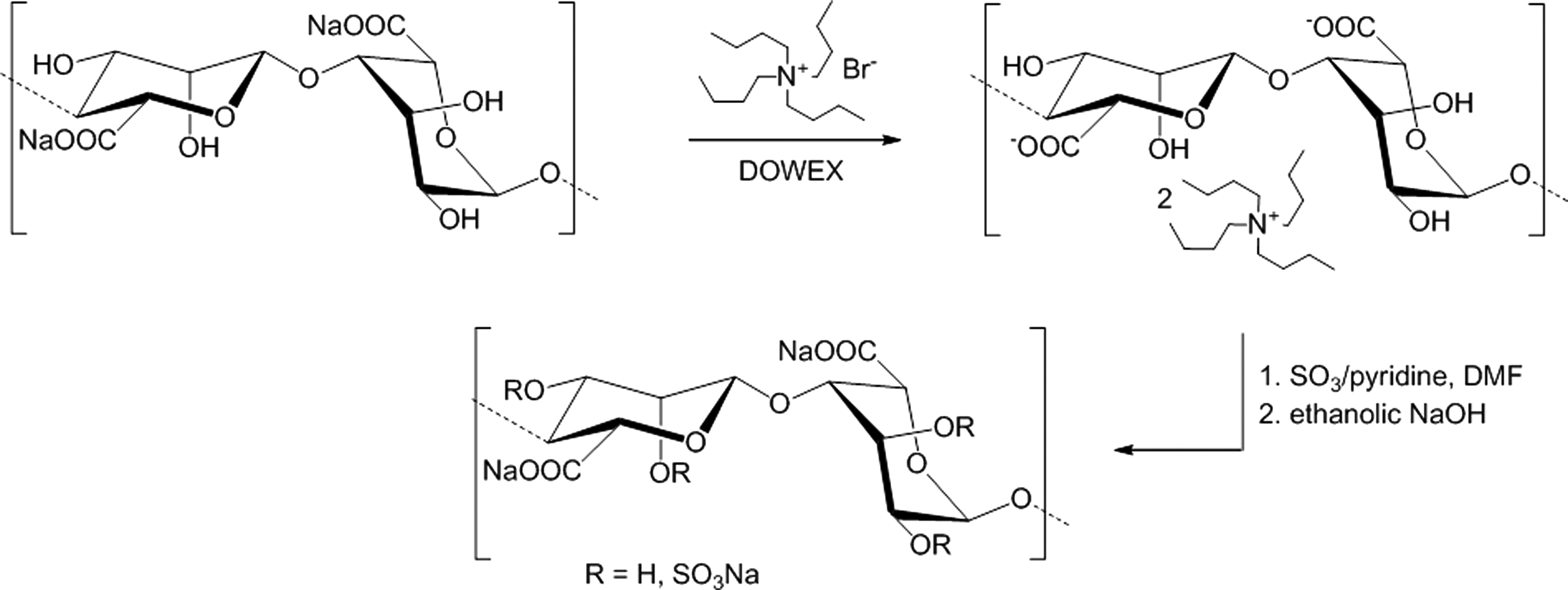
Reaction scheme for the synthesis of alginate sulfate. In a first reaction step sodium alginate is transformed into a tetrabutyl ammonium salt to improve the solubility in the reaction medium followed by the sulfation of free hydroxyl groups of alginate with SO3/pyridine
Cell isolation and hydrogel encapsulation
Chondrocytes were harvested from the knees of 6-month-old calves obtained from the local slaughterhouse as previously described.
31
Briefly, cartilage shavings were minced using a sterile blade and treated with 0.2% pronase in DMEM containing 1% antibiotic-antimycotic for 2 h at 37°C, 7% CO2 with gentle stirring. Following the pronase digestion, the tissue was washed 3×2 min with DMEM containing 1% antibiotic-antimycotic then incubated 6 h in 0.03% collagenase in DMEM supplemented with 1% antibiotic-antimycotic at 37°C, 7% CO2 with gentle stirring. Cells were separated from the digested matrix by filtering through a 100 μm cell strainer followed by a 40 μm cell strainer. Cells were counted and viability was determined using an automated cell counter (Countess™ Automated Cell Counter; Invitrogen AG). Cell viability in all isolations was above 90%. The isolated cells were seeded at 10,000/cm2 in DMEM supplemented with 1% antibiotic -antimycotic, 10% FBS, and 50 μg/mL
Cell morphology within alginate sulfate hydrogels
After a culture period of 7 or 21 days, gels were washed once with PBS, then fixed and permeabilized for 30 min in 4% formaldehyde and 0.2% Triton-X100 washed once with PBS, and stored in PBS containing 10 mM CaCl2. Encapsulated cells were stained with phalloidin-rhodamine to detect actin and DAPI for DNA. Images of the cells' cytoskeletal organization were acquired using a confocal microscope (Carl Zeiss AG/LSM 510, equipped with a 40×0.6 NA objective).
Assessment of cell proliferation within alginate sulfate hydrogels
To assess cell proliferation, a BrdU assay was performed using the Calbiochem® BrdU Cell Proliferation Assay (Calbiochem). The assay was performed according to the manufacturer's recommendations with slight modifications. Briefly, the gels were placed in media containing BrdU label to the recommended dilution in a 96-well plate for 24 h, post which the medium was carefully replaced with 270 μL of dissolving buffer (0.055 M sodium citrate, in 0.03 M EDTA, 0.15 M NaCl, pH 6.8) and the plate was rocked for 20 min at 1000 rpm at room temperature. A volume of 30 μL was transferred to a new 96-well plate and cells were centrifuged for 10 min at 1000 rpm, dried with an N2 stream, and fixed using the fixative solution supplied in the kit. Staining was then carried out by an Anti-BrdU antibody followed by HRP conjugation and substrate addition according to the manufacturer's protocol. Absorbance was measured at dual wavelengths (450–540) and the difference was blanked with the signal obtained from cell free gels using a plate reader (Synergy H1 Hybrid Reader; Biotek).
Hydrogel stiffness and cell spreading
A 2% (w/v) alginate solution was prepared in 0.15 M NaCl and 1.2% and 0.4% alginate concentrations were obtained by diluting the initial solution. A 2% (w/v) alginate sulfate solution was prepared in parallel. A volume of 30 μL of the above solutions was cast in the Q-gel casters (QGel SA) and gelled according to the working protocol. The compressive moduli (equation 1) were measured at the linear stress/strain region using the Texture Analyzer TA.XT plus by Stable Microsystems (Force resolution: 0.1 g, speed range: 0.01–40 mm/s, Range setting: 1–295 mm).
E: Compressive modulus
F: Measured force
A: Disc area
ΔL: Change in thickness
L: Gel thickness at equilibrium
To determine the effect of stiffness on cell spreading, cells were seeded in alginate of 0.4%, 1.2%, and 2% (w/v) concentration and 2% alginate sulfate and samples were imaged using transmission microscopy after 7 days in culture.
Molecular mechanisms of cell spreading and proliferation
To investigate whether cell proliferation/spreading in alginate sulfate hydrogels was integrin mediated, beta1 integrins were blocked with an antibody anti-beta1 integrin (AIIB2) and cell proliferation and spreading were assessed. Passage 3 chondrocytes were incubated with AIIB2 (1:200), or with a mouse IgG (1:200) as control, in serum-free medium for 30 min at 37°C. Cells were then encapsulated in alginate sulfate hydrogels and maintained for 7 days in culture in the presence/absence of AIIB2 or mouse IgG. At day 7, the proliferation assay was performed as described above.
Gene expression
At the end of the culture period, gels were washed once in PBS and incubated with 1 mL of dissolving buffer (0.055 M sodium citrate, in 0.03 M EDTA, 0.15 M NaCl, pH 6.8) for 10 min at 1000 rpm, 37°C. Cells were centrifuged for 2 min at 10,000 rpm, washed with PBS, and centrifuged for 2 min at 14,500 rpm. Pellets were lysed with 350 μL RLT Plus Buffer and then 540 μL RNA-free water and 10 μL Protease K solution (Qiagen) were added. Samples were incubated at 55°C for 10 min at 1000 rpm and RNA was isolated using the RNeasy Mini Kit (Qiagen) following the manufacturer's recommendations and quantified with a spectrophotometer (Nanodrop ND-1000). The measured 260/280 ratio was consistently 2.0±0.1 in all samples. Total RNA was reverse transcribed starting with 1 μg RNA and gene expression of COL2A1 (Col2), COL1A2 (Col1), SOX9, RUNX2, and CCND1 (Cyclin D1) were determined using quantitative real-time PCR (StepOnePlus; Applied Biosystems). Ribosomal protein L13 (RPL13) was used as a reference gene. An amount of 0.5% (w/v) BSA was added to the quantitative reverse transcription (qRT)-PCR mixture to prevent interference of the sulfate groups with the amplification reaction. 32 The Livak method was used for analysis of qRT-PCR data. 33 Lysates that were not directly processed were stored at −80°C. Genes investigated in this study are listed in Table 1.
qRT-PCR, quantitative reverse transcription-polymerase chain reaction.
Immunohistological staining
At the end of the culture time, discs were washed once with PBS, fixed with 4% paraformaldehyde, 0.2% Triton-X100 for 30 min then washed once with PBS, and stored in PBS containing 10 mM CaCl2 to prevent gel dissolution. Discs were embedded in Optimum Cutting Temperature compound and frozen on a dry ice block for 5 min and 6 μm-thick slices were cut using a microtome (CryoStar NX70; ThermoScientific). The slices were fixed in ethanol then washed with 10 mM CaCl2 in PBS. Antigen retrieval was performed using pronase at 1 mg/mL for 15 min at 37°C for Col2 and Col1 and using chondroitinase at 0.02 U/mL for 40 min at 37°C for proteoglycans. Samples were then blocked with 5% BSA in PBS for 1h at room temperature. Samples were incubated with primary antibodies specific against Col2 (II-II6B3, Developmental Studies Hybridoma Bank), Col1 (M38, Developmental Studies Hybridoma Bank) and proteoglycan hyaluronic acid-binding region (12/21/1-C-6, Developmental Studies Hybridoma Bank) at 1:10 dilutions in 1% BSA in PBS overnight at 4°C. Samples were washed 3× in PBS and incubated with 1:400 Alexa-488 goat anti-mouse secondary antibody for 1h at room temperature. Controls were prepared following the same procedure but omitting the primary antibody. Samples were imaged using a confocal microscope (Carl Zeiss AG/LSM 510, equipped with a 40× 0.6 NA objective). Images of the complete hydrogels were taken using a stereomicroscope (Leica WILD M650, equipped with a 6×objective).
Statistical analysis
Quantitative data were obtained from at least three independent donors and expressed as the mean±standard error. Statistical evaluation was carried out by analysis of variance (ANOVA) and post-hoc Tukey's tests where p values of less than 0.05 were considered significant. Statistical analysis was performed using OriginPro version 8.1.
Results
Morphology of chondrocytes encapsulated in sulfated alginate
Transmission and confocal microscopy were used to assess changes in the cytoskeletal organization (Fig. 2). The morphology of cells encapsulated in the different ratios of unmodified to sulfated alginate hydrogels was similar except for cells cultured in pure alginate sulfate (2%), which assumed a more spread morphology after 7 days in culture (Fig. 2E). Cells encapsulated in 1.5% alginate sulfate+0.5% alginate (Fig. 2D, I) exhibited minor spreading and protruding filopodia. Cells in 2% alginate (Fig. 2A, F), 0.5% alginate sulfate+1.5% alginate (Fig. 2B, G), and 1% alginate sulfate+1% alginate (Fig. 2C, H) had a typical round morphology as commonly observed for alginate encapsulated chondrocytes. Cell number was visually higher in the pure alginate sulfate hydrogel (Fig. 2E, J) and more cell division could be observed in the 1.5% alginate sulfate+0.5% alginate (Fig. 2D, I) hydrogels compared with hydrogels with lower alginate sulfate content.
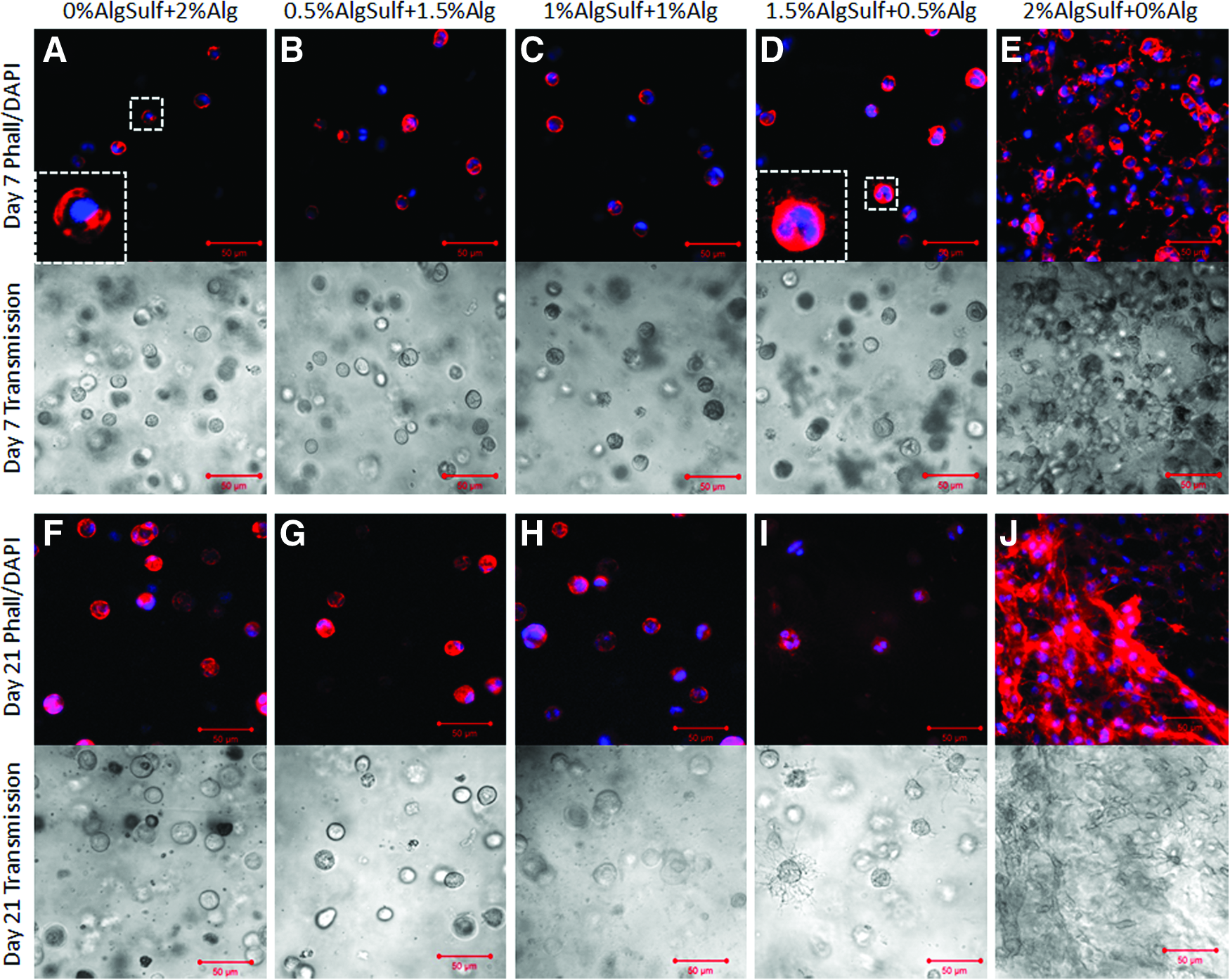
Phalloidin/DAPI staining and transmission imaging of P3 bovine chondrocytes encapsulated in alginate (Alg), alginate sulfate (AlgSulf) and mixtures of Alg and AlgSulf for 7 (
Assessment of cell proliferation within the hydrogels
Proliferation of chondrocytes encapsulated in 2% alginate sulfate, 2% alginate, and mixtures of the two components with 2% (w/v) total concentration was assessed using the Calbiochem BrdU proliferation kit. Proliferation of encapsulated cells measured by this assay corroborated the visual images of Figure 2. The 2% alginate sulfate hydrogels had five-fold higher proliferation when compared with 2% alginate (p=0.038) and the other mixtures (0.5% alginate sulfate+1.5% alginate (p=0.055), 1% alginate sulfate+1% Alg (p=0.059) and 1.5% alginate sulfate+0.5% alginate (p=0.11)) (Fig. 3).
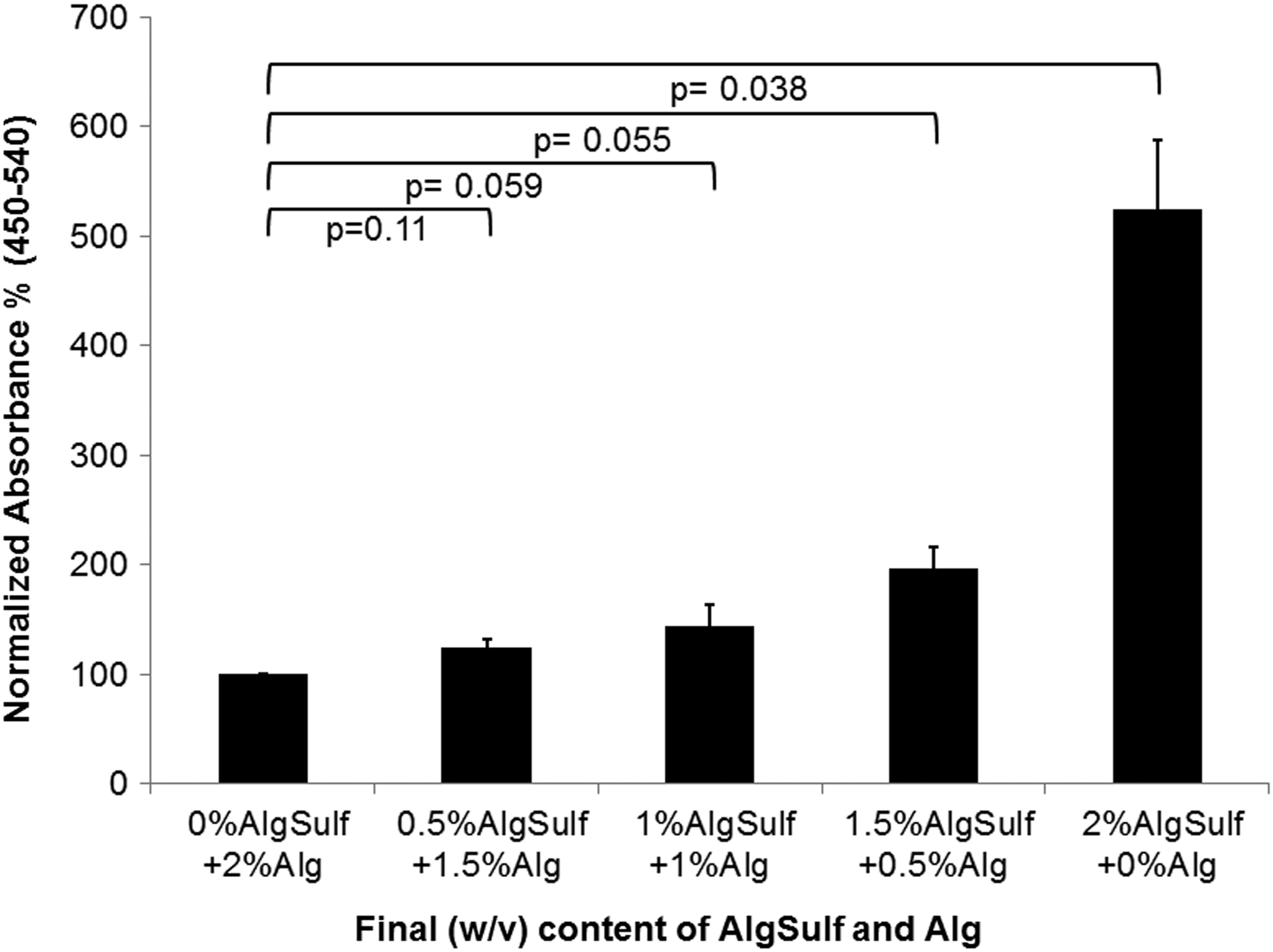
Cell proliferation in gels of various alginate sulfate (AlgSulf) contents measured using a BrdU assay after 7 days of cell encapsulation. Absorbance was normalized by relative quantitation of the absorbance value (450–540 nm blanked) of 2% alginate (Alg) gels (n=4).
Decoupling stiffness from cell spreading
To determine whether hydrogel stiffness was responsible for cell spreading, cells were encapsulated in alginate sulfate and alginate at a similar compressive modulus and cell spreading was qualitatively assessed (Fig. 4A). Alginate was prepared with increasing concentrations from 0.4% to 2% (w/v) where initial results revealed similar compressive moduli for 2% alginate sulfate and 0.4% alginate. The compressive modulus of 2% alginate (E=44.4±3.21 kPa) was significantly higher (p=1.2E-5) than 2% alginate sulfate (E=2.4±0.57 kPa), however 0.4% alginate had a similar compressive modulus (E=4.01±0.66 kPa) compared to 2% alginate sulfate (p=0.84). With regard to the cell morphology, no cell spreading was observed in pure alginate hydrogels (both 0.4% and 2%) while cells showed significant spreading in alginate sulfate hydrogels (Fig. 4B). This finding strongly suggests that the lower stiffness of the alginate sulfate hydrogels was not the cause of the extensive cell spreading in this material.
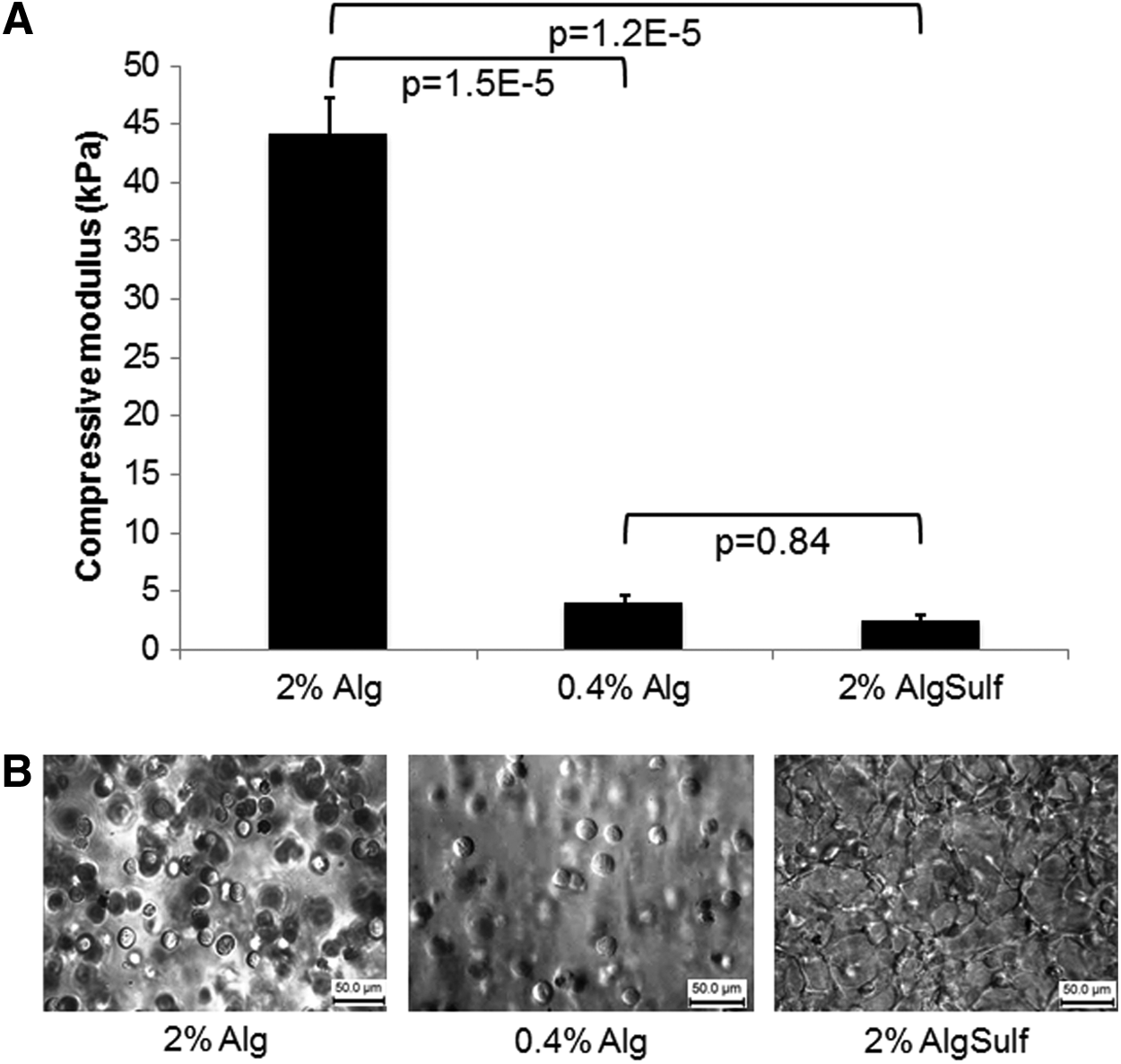
Mechanical properties of alginate and alginate sulfate (AlgSulf) hydrogels and effect on cell spreading.
RhoA and integrin signaling of chondrocytes in alginate sulfate
To explore whether cell morphology in alginate sulfate hydrogels was integrin mediated, we tested the effect of blocking beta1 integrins on cell spreading. Encapsulation within alginate sulfate hydrogels after incubation with the beta1 blocking antibodies showed a remarkable inhibition of cell spreading (Fig. 5A). We further investigated the effect of beta1 integrin blocking on cell proliferation. As shown in Figure 5B, anti-beta1 integrin antibody caused a statistically significant decrease in cell proliferation compared with untreated control (p=0.002), and to mouse IgG-treated control (p=0.014). This evidence indicates that integrin signaling plays a role in the physical interaction of the cells with the hydrogel and the increase in cell proliferation. Chondrocyte proliferation has been associated with RhoA-mediated signaling pathways. 34 Therefore, we also investigated whether beta1 integrin could act through a RhoA-mediated mechanism. For this purpose, cells within 2%, 0.4% alginate and 2% alginate sulfate maintained in culture in the presence/absence of beta1 integrin blocking antibody or nonspecific mouse IgG were assayed for the degree of RhoA activation. Interestingly, we found that RhoA activity was significantly higher in 2% alginate sulfate compared with pure alginate (both 2% (p=0.003) and 0.4% (p=0.026)). Moreover, beta1 integrin blocking produced a significant inhibition of RhoA activity compared with the nontreated control (p=0.017), while IgG did not have any significant effect (p=0.374) (Fig. 5C). To investigate whether the proliferation within alginate sulfate was controlled by genes regulating the cell cycle, expression of Cyclin D1 within alginate or alginate sulfate hydrogels was investigated (Fig. 5D). Cyclin D1 gene expression was upregulated two-fold in alginate sulfate compared with alginate (p=0.074). Those findings, taken together, indicate that beta1 integrin is responsible for cell protrusion and proliferation within alginate sulfate and could act through the RhoA GTPase pathway by upregulating Cyclin D1.
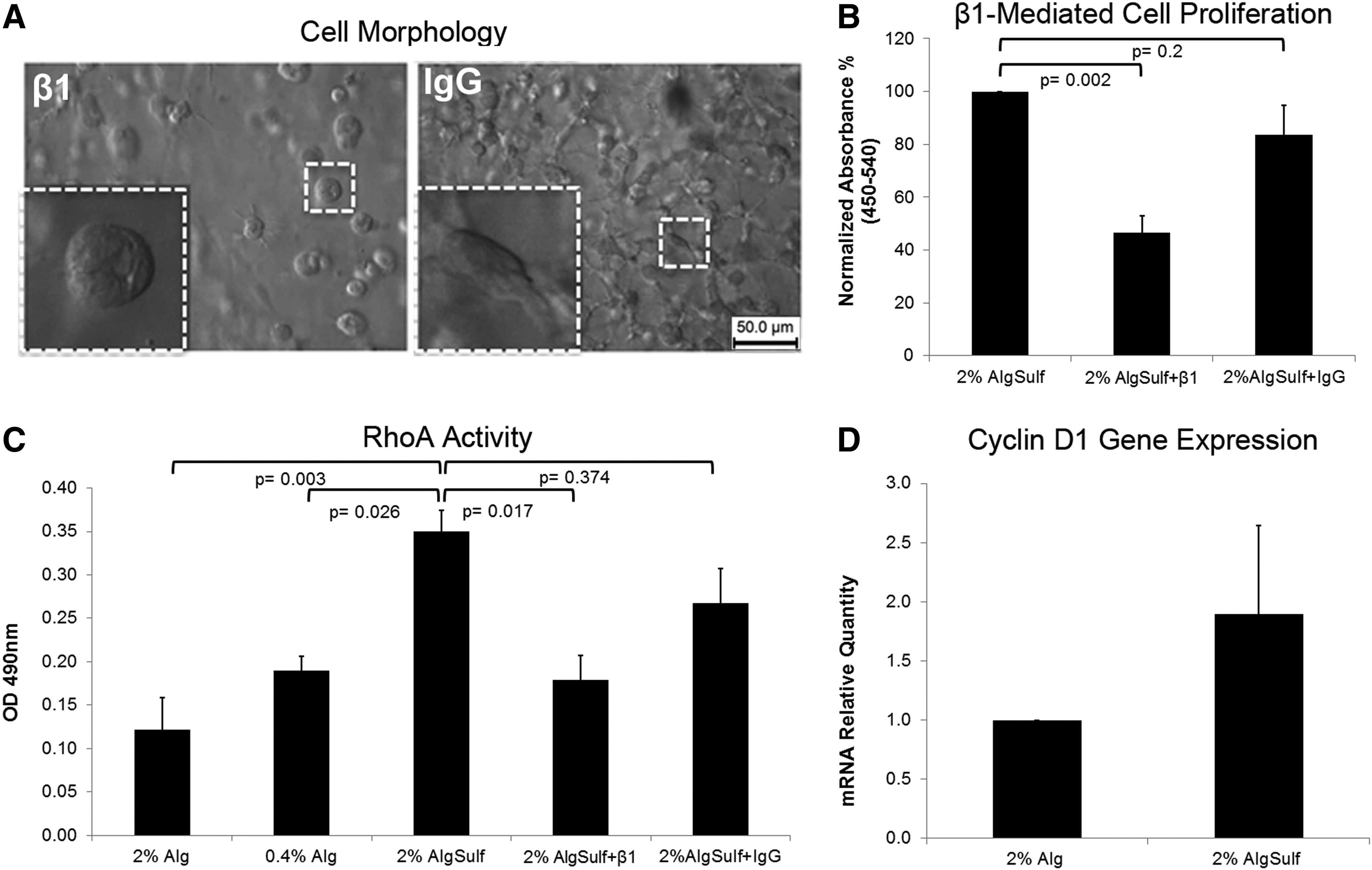
Integrin-mediated cell spreading and proliferation within alginate sulfate (AlgSulf) hydrogels
Expression of cartilage markers for dedifferentiated chondrocytes within alginate sulfate hydrogels
The expression of relevant cartilage markers showed no significant difference in expression between alginate sulfate and alginate indicating that the chondrocytes can proliferate without undergoing de-differentiation, which is typically associated with expansion in monolayer culture (Fig. 6). A two-way ANOVA showed that Col2/Col1 ratio was significantly higher in both alginate and alginate sulfate compared with cells expanded in monolayer (p=0.0004 and p=0.0001 respectively), whereas the difference between alginate and alginate sulfate was not significant (p=0.71). SOX9/RUNX2 ratio was also significantly higher in alginate and alginate sulfate compared with monolayer expanded cells (p=0.0047 and 0.01 respectively) and such a difference was not observed between alginate and alginate sulfate (p=0.89). Finally, the effect of culture time was only significant for Col2/Col1 ratio (p=0.016) and not significant for SOX9/RUNX2 (p=0.98).
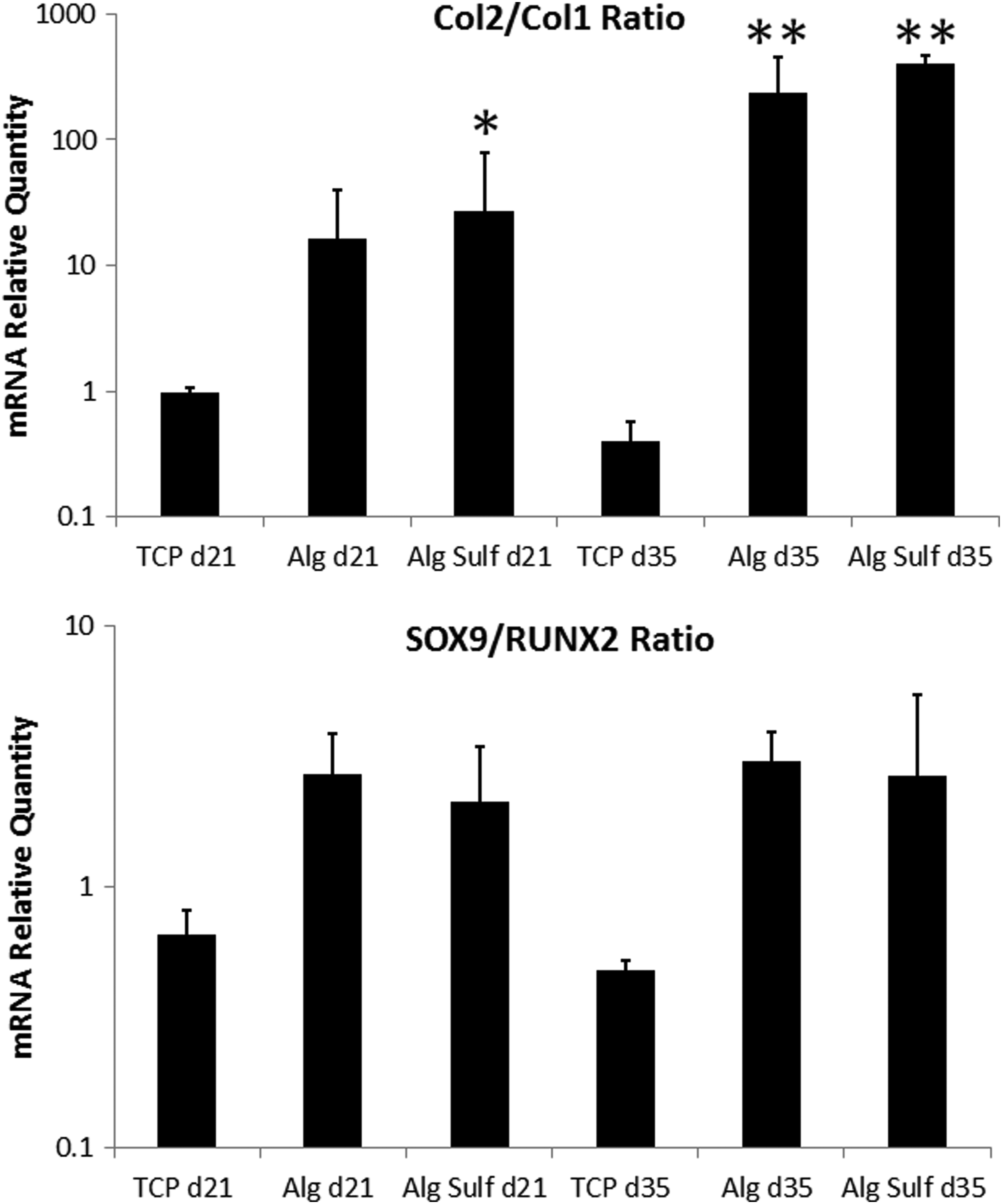
Relative gene expression of Col2/Col1 ratio and SOX9/RUNX2 ratio for bovine chondrocytes encapsulated in alginate (Alg), alginate sulfate (AlgSulf), or expanded on tissue culture plastic (TCP) for 21 or 35 days. Statistical significance is denoted with * for p<0.05 and ** for p<0.01 compared with TCP at the same time point, (n=3). RPL13 was used as a reference gene.
Immunohistological staining and gross appearance of alginate sulfate hydrogels
Encapsulation of bovine chondrocytes within the alginate sulfate hydrogels induced an increase in cell proliferation, and the cells were positive for Col2 and proteoglycan (Fig. 7). The alginate sulfate gels had a gross opaque appearance after 35 days in culture, whereas the unmodified alginate samples remained transluscent (Fig. 7).
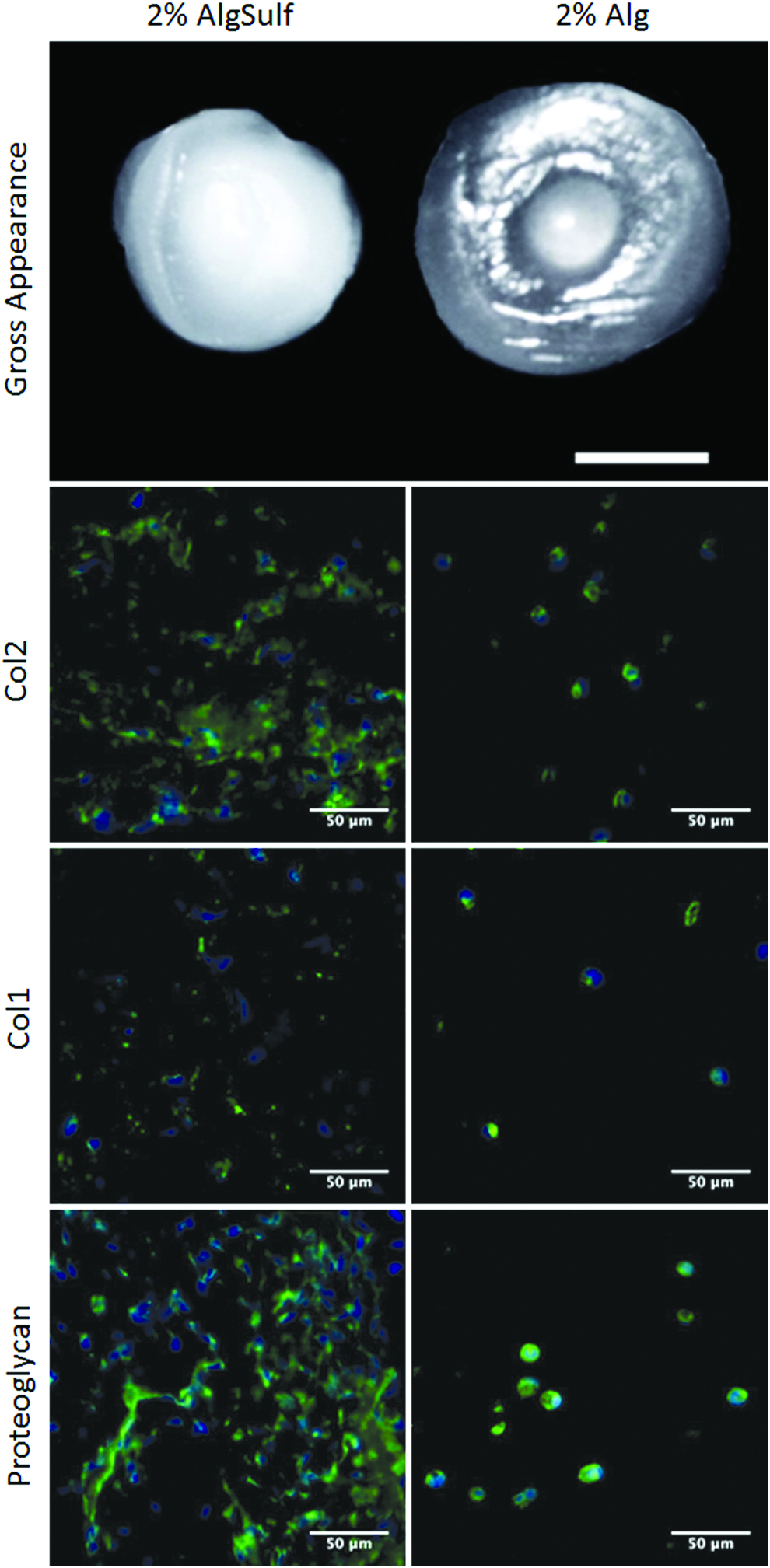
Gross appearance of alginate sulfate (AlgSulf, left) and alginate (Alg, right) 30 μL gels after 35 days in culture (scale bar 2 mm). Immunostaining of Col2, Col1, and proteoglycan in AlgSulf (left) and Alg (right) samples after 35 days in culture 40× magnification (scale bar 50 μm). Color images available online at www.liebertpub.com/tea
Discussion
In this work we studied bovine chondrocytes encapsulated within a novel hydrogel material made of sulfated alginate. The preparation of alginate sulfate hydrogels was only possible with DSs=0.8 whereas a higher degree of sulfation (DSs=2.6) did not form a gel using CaCl2 as a source for Ca2+ ions. This might be due to the strong electrostatic forces caused by the highly sulfated molecules that may inhibit ionic cross-links. Additionally, steric hindrance driven by the large sulfate moieties on the alginate chain may prevent the cross-linking of the sulfated alginate molecules. Surprisingly, cells encapsulated in alginate sulfate exhibited remarkable cell spreading after 7 days of culture accompanied by a strong increase in proliferation when compared with nonmodified alginate (five-fold, p=0.038). This was only significant for pure alginate sulfate when compared with alginate. Including as little as 0.5% alginate in the total mixture inhibited cell migration and forced cells to proliferate in nodes resulting in cell clusters (Fig. 2D). The unexpected increase in proliferation raised the question whether the lower stiffness of alginate sulfate was responsible for cell spreading and proliferation. However, cells encapsulated in nonmodified 0.4% (w/v) alginate, although having a similar compressive modulus to alginate sulfate (p=0.84), maintained a round morphology. We then studied the role of integrins in cell spreading and proliferation within the alginate sulfate hydrogels. Beta1 integrins have been shown to have a central role in mediating adhesion to the ECM proteins, motility, and proliferation of chondrocytes. 35 To investigate whether the mechanism of cell spreading/proliferation in alginate sulfate was beta1 integrin-dependent, cells encapsulated within hydrogels were maintained in culture for 7 days in the presence/absence of a beta1 integrin blocker or a nonspecific mouse IgG antibody. Blocking beta1 integrins reduced both cell spreading and cell proliferation. The large differences in proliferation rates between alginate and alginate sulfate encouraged us to further investigate whether the material was suitable for cartilage tissue engineering applications. To test this possibility, dedifferentiated bovine chondrocytes were encapsulated for up to 5 weeks within alginate and alginate sulfate and the expression of relevant cartilage markers was analyzed using qRT-PCR. Both alginate and alginate sulfate resulted in re-expression of Col2/Col1 and SOX9/RUNX2 ratios in dedifferentiated chondrocytes. Alginate sulfation had no significant effect on the expression of Col2/Col1 and SOX9/RUNX2 ratios. Similarly, at the protein level cells in both systems were not different. Alginate sulfate constructs attained an opaque appearance after 5 weeks in culture. This can be attributed to the higher cell proliferation in alginate sulfate resulting in an overall larger amount of synthesized matrix and not to higher protein synthesis per cell.
Sulfated GAGs are present in the ECM of most tissues; among these are heparin, heparan sulfate, chondroitin sulfate, keratin sulfate, and dermatan sulfate.3,36 The degree and regioselectivity of the sulfate units is orchestrated by a number of enzymes such as endosulfatases and sulfotransferases.37,38 The degree of sulfation of these molecules ranges from 0 to 3 in most cases. Chondroitin sulfate, which is the main sulfated component in cartilage, is typically sulfated on the 4th or 6th carbon of the N-acetylgalactosamine sugar, therefore resulting in an approximate degree of sulfation, which is close to 1. 36 Although this degree of sulfation is similar to the sulfated alginate material used in the present study, mature cartilage exhibits very limited proliferation. The reasons for low cell proliferation in mature normal cartilage are multifactorial; but involve the homeostasis of the tissue, the microenvironment, the age of the cells, and the composition and stiffness of the ECM components. When this balance is disturbed, such as in osteoarthritis or cartilage injury, chondrocytes may respond by increasing their proliferative rates in an attempt to repair the tissue. 3 The higher stiffness of normal cartilage compared to the sulfated alginate hydrogel used here (approximately two orders of magnitude) highly restricts cell migration and proliferation.
Sulfated GAGs have been shown to have important functions such as anticoagulant,39,40 growth factor and cytokine affinity,25,26 chondroprotective,23,24 neuroprotective, 41 and antioxidant properties. 42 Lately, Ronghua et al. 30 showed that alginate sulfate has anticoagulant properties comparable to heparin. However, the use of alginate sulfate for tissue engineering applications is limited to a few studies.28,43 In particular to our knowledge, a pure alginate sulfate hydrogel was not previously reported probably due to the sulfation method used. 27 Different sulfating agents including sulfuric acid-carbodiimide, 27 chlorosulfonic acid-formamide, 29 and various SO3/complexes44,45 have been used in the past to prepare sulfated alginate. Recently, we could show that SO3/pyridine is a mild sulfating agent for GAGs such as hyaluronan allowing proper control of the degree of sulfation, especially if a homogeneous sulfate group distribution and rather low DSS values are desired.46,47 We therefore decided to use SO3/pyridine for the sulfation of alginate. In our work, addition of 3 mM CaCl2 was necessary to maintain stability of the gel for the first 21 days of culture. CaCl2 levels have been shown to be important in maintaining the cartilage phenotype.48,49 CaCl2 was therefore removed from the cultures for the last 14 days and gels maintained stability, which might be attributed to the high cell proliferation and matrix synthesis.
Proliferation and spreading of bovine chondrocytes on collagen-based films were shown earlier to be reduced by blocking beta1 integrins. 31 Moreover, remodeling of 3D collagen fibers and formation of aggregates by mesenchymal stem cells was shown to be dependent on beta1 integrins. 50 We observed here that blocking beta1 integrins prevented cell spreading and reduced proliferation, suggesting that proliferation was integrin based and possibly through cell synthesized collagen. Further, RhoA GTPase activity was reduced with beta1 blocking indicating that proliferation was RhoA mediated (Fig. 5C). This is in agreement with previous findings, which reported that RhoA increased proliferation and delayed hypertrophic differentiation of chondrocytes. In particular, Wang et al. correlated the activity of RhoA with the increased expression of Cyclin D1, which is involved in regulating the cell cycle G1/S transition. 34 In this study, Cyclin D1 gene expression was upregulated two-fold in alginate sulfate compared with alginate, thus indicating that alginate sulfate possibly enhances proliferation by ultimately regulating expression of cell cycle genes (Fig. 5D). Beta1 dependent cell spreading and proliferation may occur in alginate sulfate in a direct manner (direct interaction of sulfated moieties with beta 1 integrins) or indirect manner (enhanced protein binding to sulfated substrata). It is though more probable that sulfation enhances cell proliferation and spreading via beta1 integrins in an indirect manner by allowing cell-synthesized matrix molecules, growth factors and media supplements to bind the hydrogel more effectively. To our knowledge there were no reports relating beta1 binding to sulfation patterns, however, such possible preferences cannot be underestimated. Assays evaluating the attachment of beta1-expressing cells versus beta1 mutant cells will help reveal possible beta1-sulfation patterns interactions.
Alginate sulfate has been shown to have a high affinity to most heparin-binding growth factors. 27 Moreover, a scaffold containing alginate sulfate was shown to improve chondrogenesis of encapsulated mesenchymal stem cells by gradual delivery of TGFβ-1. 28 Bovine chondrocytes were never reported in combination with alginate sulfate and we hypothesized that the high affinity to growth factors would result in a more chondrogenic environment for encapsulated chondrocytes. However, the focus of this study turned into the unexpected strong proliferation and spreading of encapsulated chondrocytes and whether chondrocytes would maintain their cartilage phenotype after long-term culturing. Chondrocytes have been traditionally reported to redifferentiate to a cartilage phenotype after dedifferentiation when cultured in 3D hydrogels such as agarose 19 or alginate.20–22 However, cell proliferation in these systems is limited and thus the regeneration capacity is low. 51 Collagen-based hydrogels were shown to induce cell proliferation at the expense of loss of the cartilage phenotype.51,52 Cells encapsulated in alginate sulfate hydrogels exhibited a high degree of proliferation while maintaining similar levels of cartilage markers. Although the current hydrogels are still mechanically inferior to native cartilage, we believe that subcutaneous implantation or longer culture periods would improve its mechanical properties. Moreover, the use of lower passage cells and addition of chondrogenic supplements can further improve the overall quality of the engineered tissue.
Alginate sulfate is a potential hydrogel for autologous chondrocyte implantation that allows cells to synthesize their own matrix, proliferate, and maintain their cartilage phenotype. In addition, it has a proven high affinity to important growth factors and can thus be loaded with relevant cartilage growth factors for improved performance. We also observed in preliminary studies that alginate sulfate supports the viability and proliferation of mesenchymal stem cells and induces neurite extension of encapsulated mice primary cortical neurons up to several millimeters in length (data not shown). In conclusion, alginate sulfate may prove to be an ideal biomaterial for engineering cartilage and other tissues.
Footnotes
Acknowledgments
The work was funded by the European Union Seventh Framework Programme (FP7/2007–2013) under grant agreement n° NMP4-SL-2009-229292 (Find&Bind). The type II collagen antibody II-II6B3 developed by T.F. Linsenmayer, type I collagen antibody M-38 developed by J.A McDonald, proteoglycan hyaluronic acid-binding region antibody 12/21/1-C-6 developed by B. Caterson, beta1 antibody AIIB2 and alpha5 antibody BIIG2 developed by C.H. Damsky were obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biology, Iowa City, IA 52242. We also thank Prof. Janos Vörös (Laboratory of Biosensors and Bioelectronics, ETH Zurich, Switzerland) for his valuable scientific input/use of lab space and Nadiia Kondratiuk for help with immunostaining.
Disclosure Statement
The authors declare that no competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
