Abstract
Human periodontal ligament stem cells (hPDLSCs) have been proposed as an alternative to conventional cosmetic fillers because they display an innate ability to synthesize collagen. The aims of this study were to determine the effects of water-soluble chitin (WSC) on the proliferation and migration of hPDLSCs, and to quantify collagen synthesis in vitro and in vivo compared with human adipose-derived stem cell (hADSC)s. hPDLSCs were isolated from healthy extracted teeth, and the cell proliferation and cell migration capacities of untreated hPDLSCs (control group) and WSC-treated hPDLSCs (test group) were compared. Insoluble/soluble collagen synthesis were also assessed, and collagen related markers were evaluated including lysyl oxidase (LOX), lysyl oxidase like (LOXL)1, LOXL2, and hydroxyproline. In vivo collagen formation was examined by transplanting hyaluronic acid as a cell carrier into the subcutaneous pockets of immunocompromised mice in the control and test groups; histology and immunohistochemistry analyses were performed 4 (n=4) and 8 (n=4) weeks later. There was a dose-dependent enhancement of hPDLSCs proliferation in the test group, and a concomitant reduction in cell migration. The amount of insoluble collagen formed was greater in the test group than in the control group (p<0.05), whereas soluble collagen formation was significantly reduced in the test group (p<0.05). The histology and immunohistochemistry results revealed that the amount of collagen formed in vivo was greater in WSC-treated hPDLSCs than in the control cells at 4 and 8 weeks (p<0.05), and histometric analysis at 8 weeks revealed that enhancement of collagen formation by hPDLSCs was greater than by hADSCs. These results indicate that WSC modulates the properties of hPDLSCs, rendering them more suitable for cosmetic soft-tissue augmentation.
Introduction
Human periodontal ligament (PDL) stem cells (hPDLSCs) are a type of MSC that can be obtained during the extraction of impacted third molars or premolars for orthodontic purposes, and they have been demonstrated to retain the basic characteristics and regenerative potential of MSCs.6,8–11 hPDLSCs have been reported to be highly proliferative cells that can differentiate into osteogenic, cementogenic, adipogenic, chondrogenic, neurogenic, and fibroblastic cells in specific environments.8,10,12–14 In particular, hPDLSCs appear to exhibit superior regenerative potential for collagen fibers and to persist longer than other MSCs or fibroblasts when transplanted ex vivo, suggesting that they could be used in plastic surgeries for cosmetic purposes, where enhanced collagen formation is considered to be essential. 6
Interestingly, the tissue engineering potential of hPDLSCs appears to be affected by the type of scaffold used. For instance, hPDLSCs generate cementum and PDL-like tissues when implanted with hydroxyapatite/tricalcium phosphate particles,6,10,13 whereas substantial collagen fibers are regenerated when they are transplanted with a collagen-based carrier. 6 Chitin, as an another type of cell carrier, has been applied to enhance collagen synthesis using the potential of hPDLSCs, since it exhibits excellent wound-healing property by promoting the formation of stable collagen fibers.15–18 A newly developed water-soluble chitin (WSC) was recently shown to be more effective in wound healing than conventional chitin and chitosan. 15 Moreover, tissue that healed in the presence of WSC solution exhibited the greatest tensile strength, and the arrangement of collagen fibers therein was similar to that of normal skin. 15 These observations prompted the authors to postulate that a combination of hPDLSCs and WSC can maximize collagen formation and be used in various applications in the cosmetic field.
The aim of this study was to determine the effect of combined treatment with hPDLSCs and WSC for collagen synthesis using chemical, biological, and histological analyses in vitro and in vivo.
Materials and Methods
Production of WSC
WSC was manufactured using the method described by Kurita et al. 19 Low-molecular-weight chitosan was used as the initial material, which was obtained from the shell of the Korean crab Chionoecetes opilio and manufactured in our laboratory. After 30 g of chitosan was dissolved in 800 mL of an aqueous solution containing 5% acetic acid, the chitosan solution was diluted by adding 800 mL of ethanol. A prepared mixture of 1000 mL of pyridine and 110 g of acetic anhydride was added to the chitosan solution. After stirring for 10 h at room temperature, the reaction solution was gradually poured into 3000 mL of acetone, followed by filtration of the collected precipitate. The filtered precipitate was dried and then dissolved in an aqueous solution containing 2% acetic acid, and the insoluble materials were removed by filtration. The pH of the remaining solution was adjusted to 12, and the solution was poured into 5000 mL of the mixed solution of acetone and water (acetone:water=7:1), followed by drying. In pilot studies, we have confirmed that the achievable maximum dosage of WSC was 0.3 mg/mL. At the time of usage, 0.3 g of dried WSC was dissolved in 100 mL of neutral water, the pH of the solution was adjusted to 7 using 0.5% citric acid, insoluble materials were removed by filtration, and the solution was subjected to dialysis using a membrane with a cutoff value of 10,000.
hPDLSC isolation and culture
The cell culturing protocol reported by Seo et al. 10 was performed with slight modifications. The experimental protocol was approved by the Ethics Committee of the College of Dentistry, Yonsei University (IRB No. 2-2008-0014), and written and informed consent was obtained from each subject before enrollment in the study.
hPDLSCs were isolated from healthy teeth extracted from three young adults (aged 11–19 years) for the purpose of orthodontic treatment. After washing with α-minimum essential medium (MEM) (GIBCO®) combined with 100 U/mL penicillin and 100 μg/mL streptomycin (GIBCO), PDL tissues were collected by scraping the root surface from the middle-third to the apex, and then minced. Small pieces of tissues were digested with a solution of enzymes four or five times at intervals of 20 min at 37°C; the enzyme solution contained α-MEM, 2 mg/mL collagenase type I (Waco Pure Chemical Industries), and 1 mg/mL dispase (GIBCO). Single-cell suspensions were obtained using a 70-μm strainer (BD Falcon™, BD Biosciences). Cells (5×105) were seeded into a T75 cell culture flask with culturing medium and incubated at 37°C in 5% CO2; the culture medium contained α-MEM, 15% fatal calf serum (GIBCO), 10 μmol/mL ascorbic acid 2-phosphate (GIBCO), 200 μmol/mL
Cell proliferation assay (bromodeoxyuridine incorporation and cell-cycle analysis)
Bromodeoxyuridine-incorporation assay
The proliferation of hPDLSCs was measured using a bromodeoxyuridine (BrdU) cell proliferation kit (Roche Diagnostics) according to the methods of Zheng et al. 20 Cells (5×104) were plated in 96-well plates, treated 6 h later with WSC at various concentrations (0.0003, 0.003, 0.03, 0.3, and 0 mg/mL), and cultured for 24 h. After the incubation period, a BrdU-labeling solution was added to each well to a final concentration of 10 μM, and cells were reincubated for 2 h at room temperature. After discarding the remnant labeling medium, the cells were fixed with FixDenat solution for 30 min, and incubated again with anti-BrdU-PDO working solution for 90 min at room temperature. Cells were rinsed with phosphate buffered saline (PBS) three times and the substrate solution was added. After sufficient color had developed, the absorbance of the incorporated BrdU was measured by a microplate spectophotometer (ELISA; Bio-Rad Laboratories) at 370 nm.
Cell-cycle analysis
Cell-cycle analysis was also performed to confirm the cell proliferation results found in the BrdU assay, and the optimal concentration (0.3 mg/mL) of WSC was selected for the group of WSC-treated hPDLSCs. After 1 day of treatment with WSC, hPDLSCs were harvested and passed through the 70-μm cell strainer (BD Falcon). After centrifugation at 90 g for 5 min, the cell precipitates were washed twice with PBS and resuspended in 1 mL of PBS with repeated vibration and mixing to ensure the production of a single-cell suspension. Cold dehydrated alcohol (2 mL) was then quickly mixed with the cell suspension to fix the cells at −4°C for 24 h. Finally, the cells were washed twice with PBS, incubated with propidium iodide (50 μg/mL; Sigma-Aldrich Biotechnology) and RNase A solution (5 μg/mL; Worthington Biochemicals) at 4°C for 30 min, and then applied to a flow cytometer (FACS Calibur™, BD Biosciences). One million cells were counted per sample, and the fractions of cells in the G1, S, and G2 phases of the cell cycle were analyzed with Cell QuestTM software (version 3.3; BD Immunocytometry Systems, BD Biosciences).
Cell migration assay
A cell migration assay was performed according to the protocol of the cell scratch assay with slight modifications. 21 hPDLSCs (1×105) were seeded on a 6-well plate (Nunclon™ Delta Surface, NUNC) and 2-well chamber slide (Nunclon) and cultured until 80% of confluence. The cell layer in the middle of the plate or slide was cautiously scratched using a cell scraper (SPL Lifesciences) to produce a cell-free strip, and then washed with PBS to eliminate floating cells. After treatment of WSC (0.3 mg/mL) for 3 days, cells were fixed with 2% paraformaldehyde (PFA; for Sigma-Aldrich Biotechnology). For microscopic observation, 6-well plates were stained with 0.4% trypan blue solution (GIBCO), and the representative ten areas of each well were taken under a inverted microscope (Olympus CK40; Olympus Optical), and the average distances that hPDLSCs moved were calculated. To confirm the migration of hPDLSCs, F-actin of cells in 2-well chamber slides were stained according to the previous report with slight modifications. 3 After fixation, the F-actin filaments were stained with phalloidin (Alexa flour 488; Invitrogen™, CA) at 1:500, and they were mounted with mounting medium containing with 4′-6-Diamidino-2-phenylindole (Vectashield®; Vector Laboratory, Inc.). The samples were visualized under a fluorescence microscope (Olympus BX41; Olympus Optical).
In vitro collagen synthesis assay (Sircol™ collagen and collagenous ECM formation assay)
Sircol collagen assay
Total soluble collagen in the cell supernatant was measured using the Sircol collagen assay kit (Biocolor Ltd.). To compare the fibroblastic potentials of hPDLSC with other type of stem cell, human adipose-derived stem cells (hADSCs) were also cultured. After 7 days, cell supernatants of hPDLSCs and hADSCs were collected by centrifugation at 260 g for 5 min. A 200-μL aliquot of the supernatant was added to 1 mL of the dye reagent provided with the kit, and the solution was incubated for 30 min at room temperature. After centrifugation at 9300 g for 10 min, the separated suspensions were discarded and the remnant pellets were dissolved in 1 mL of the alkali reagent provided with the kit. After ensuring thorough dissolution, the same aliquots were plated on 96-well plates and the relative absorbance was measured at 540 nm.
ECM-formation assay
ECM formation of hPDLSCs and hADSCs was detected by staining with picro-sirius red (Direct Red 80®, Sigma-Aldrich Biotechnology). hPDLSCs (8×104) were cultured with or without 0.3 mg/mL WSC in culture medium for 7 days. After thorough washing with PBS, cell lysis was performed by incubation with 2 mL of matrix solution for 5 min at 37°C; the matrix solution contained 0.5% Triton® X-100 (Sigma-Aldrich Biotechnology) and 20 mM NH4OH in PBS. The remaining matrix was washed with PBS once only. Picro-sirius red was added to each well, and the sample was incubated for 1 h at room temperature. After discarding the staining solution, wells were cautiously rinsed with PBS once, and then photographs of ten areas that were representative of each well were taken under a microscope, and the average areas that were stained red in the WSC-treated and control groups were calculated.
Quantitative real-time polymerase chain reaction
The finding of increased ECM synthesis prompted the authors to observe the changes in collagen synthesis at the mRNA and protein levels. Since collagen types I and III are the main types of human PDL tissue constituting ∼80% of all PDL fibers, 22 the quantitative real-time polymerase chain reaction (PCR) analysis were performed for these types of collagens. In addition, the gene expressions of lysyl oxidase (LOX), lysyl oxidase-like (LOXL)1, and LOXL2 were detected. hPDLSCs (1×105) were seeded on 6-well plates, and were treated with or without WSC (0.3 mg/mL) when they reached 80% of confluence. After 7 days, the expressions of collagen types I and III were confirmed by real-time PCR according to the manufacturer's instructions for each products. Cells were harvested using Trizol® (Invitrogen), and total cell RNA was prepared. Isolated total RNA was used as a template for the synthesis of cDNA with Oligo dT primer (Maxime RT Premix®, iNtRon Biotechnology). The subsequent PCR amplification reaction utilized the SYBR® real-time PCR kit (Premix Ex Taq™, Takara Bio), ABI 7300 real-time PCR system and software (Applied Biosystems), and specific primers (Table 1) under the following conditions: an initial step at 50°C for 2 min and 95°C for 10 min, followed by 3 steps of denaturation at 95°C for 15 s, annealing at 60°C for 30 s, and extension at 72°C for 30 s for 40 cycles, and then a final 10-min extension at 72°C. The relative levels of mRNA expression were quantified by comparison with the internal standard (β-actin). The amount of gene expression at real-time PCR was computed from the target threshold cycle (Ct) values and β-actin Ct values by means of the standard curve method: ΔCt=Ct of collagen type I or III−Ct of β-actin; ΔΔCt=ΔCt of target gene −ΔCt of control; normalized amount of gene expression=2−ΔΔCt. Each PCR was performed in triplicate with the same total RNA.
Western blot analysis
hPDLSCs (5×105) were seeded on 100-mm dishes, and were treated with WSC (0.3 mg/mL) when they reached 80% of confluence; cells in the control group were not treated. After 7 days, supportive western blot analysis was performed according to the manufacturer's instructions and a reported protocol
23
with slight modifications. Confluent monolayer cells on 100-mm dishes were lysed in protein extraction reagent (M-PER® Mammalian Protein Extraction Reagent, PIERCE) with a protease inhibitor cocktail tablet (Complete™ EDTA-free, Roche Diagnostics), and were collected with a cell scraper. After lysis, cells were incubated on ice for 60 min, followed by centrifugation at 9300 g at 4°C for 15 min, after which the supernatant was collected and stored at −20°C until use. After protein quantification of each sample with the BCA® protein assay kit (Thermo™ Scientific), cell lysates were solubilized with NuPAGE® LDS sample buffer (Invitrogen) at 70°C for 10 min, separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (10%), and transferred to a polyvinylidene difluoride membrane (Immobilon-P®, Millipore, Bedford, MA) using the Mini-PROTEIN® Tetra cell system (Bio-Rad Laboratories). The blots were blocked with 5% (w/v) dried skim milk (Difco™, Sparks) and TBST [20 mM Tris-HCl, 137 mM NaCl, and 0.1% (v/v) Tween 20] at 4°C overnight, followed by incubation for 2 h at room temperature with rabbit polyclonal antibodies: collagen type I (ab292, Abcam) at 1:5000, collagen type III (ab7778, Abcam) at 1:1000, hydroxyproline (ab37067, Abcam) at 1:1000, LOX (GTX100784, GeneTex) at 1:500, LOXL1 (GTX114532, GeneTex) at 1:1000, LOXL2 (GTX105085, GeneTex) at 1:1000, and β-actin (Cell Signaling Technology). The blots were incubated with anti
In vivo collagen synthesis assay (hPDLSC transplantation into immunocompromised mice and histological observations)
In vivo transplantation was performed to evaluate the collagen synthesis capacity. Since the use of an appropriate carrier is important to the potential of hPDLSCs,
6
we selected a hyaluronic acid (HA) component since this is one of the most common components used in biomedical surgeries,
24
and HA-based scaffolds have already been used for the culture and transportation of rat MSCs
25
and for the soft-tissue augmentation of hADSCs in reconstructive surgery.
5
For a single transplantation, 6×106 cells were mixed with 0.3 mL of HA filler (ReDexis®, Prollenium Medical Technologies), and then injected into subcutaneous pockets on the dorsal surface of 5-week-old immunocompromised mice. Each transplant was treated under the following four experimental conditions: (i) HA carrier only (control, Group 1); (ii) HA carrier pretreated with 0.3 mg/mL of WSC (Group 2); (iii) hPDLSCs combined with untreated HA carrier (Group 3); and (iv) hPDLSCs combined with an WSC-pretreated HA carrier (Group 4). Each group included four nude mice with four ectopic transplantations in each. The mice were killed after 4 weeks, and the implants were harvested. To evaluate the extended healing in comparison to hADSCs, additional transplantation experiments were performed for following groups using the aforementioned procedures (n=4): (i) HA carrier only (control, Group A); (ii) hADSCs combined with untreated HA carrier (Group B); (iii) hPDLSCs combined with untreated HA carrier (Group C); (iv) HA carrier pretreated with 0.3 mg/mL of WSC (Group D); (v) hADSCs combined with an WSC-pretreated HA carrier (Group E); and (vi) hPDLSCs combined with an WSC-pretreated HA carrier (Group F). The mice were killed after 8 weeks, and the implants were harvested. All animal experiments were carried out in accordance with the Guidelines and Regulations for the Use and Care of Animals of the Animal Care Committee of Medical College, Yonsei University. After being weighed on a balance (Adventurer, Ohaus Corp.; Fig. 3A) and fixed in 4% PFA for 3 days, they were washed with PBS before embedding in Tissue-Tek® O.C.T.™ Compound (Sakura Finetek Japan) for frozen section. For histological analysis, the sections (serially sectioned at 4 μm) were stained with hematoxylin
Immunohistochemistry analysis
In brief, after routine deparaffinization and rehydration, the antigen was retrieved using a microwave-based technique for 30 min in citrate buffer (pH 9.0). The endogenous peroxidase was blocked with 3% hydrogen peroxide for 5 min. The sections were incubated for 30 min at room temperature with primary antibodies: human-specific mitochondrial ribosomal protein (ab74285, Abcam) diluted 1:50, collagen type I (ab7778, Abcam) diluted 1:200, and collagen type III (ab292, Abcam) diluted 1:1000. After washing three times, immunodetection was performed using a commercially available kit (EnVision™ Detection System, Dako REAL™) according to the manufacturer's instructions. Slides were then counterstained with hematoxylin.
Statistical analysis
Each in vitro experiment was performed at least three times. Mean and standard deviations were calculated, and the statistical significance of differences between groups was examined by one-way analysis of variance and the t-test. Test results were considered statistically significant for probability values of p<0.05.
Results
WSC characteristics
The number-average molecular weight of WSC was 43,600, and the weight-average molecular weight was 94,800. Gel permeation chromatography revealed values of a and log K in the Mark-Houwink equation for WSC of 0.711 and −3.167, respectively. The degree of deacetylation was 51.6%. Various characteristics of the modified WSC are listed in Table 2.
Mn, number-average molecular weight; Mw, weight-average molecular weight; Pd, polydispersity; DA, degree of deacetylation; A, Mark-Houwink exponent (conformation); log K, Mark-Houwink exponent (volume); RG, radius of gyration; HR, hydrodynamic radius; IV, intrinsic viscosity.
Effects of WSC on the proliferation and migration of hPDLSCs
After 24 h of incubation, WSC at 0.03 and 0.3 mg/mL had significantly promoted the incorporation of BrdU into the hPDLSCs compared with the other groups (Fig. 1A; p<0.05). This effect appeared to be dose dependent with cell proliferation being maximally induced by 0.3 mg/mL WSC. Further, the cell-cycle distributions, as analyzed by flow cytometry, revealed that the proliferation index (S+G2+M phases) was significantly higher in the WSC-treated hPDLSC group, demonstrating that treatment with WSC enhances the transition process from the G1 to the S phase and further accelerates the DNA replication of hPDLSCs (Fig. 1B; p<0.05). On the other hand, the in vitro cell migration assay showed that the cell migration capacity was significantly decreased in WSC-treated hPDLSCs than in untreated hPDLSCs (Fig. 1C; p<0.05). In line with these results, the actin polymerization assay revealed that stained phalloidin (actin-specific-binding fluorescence) was significantly decreased in WSC-treated hPDLSCs.
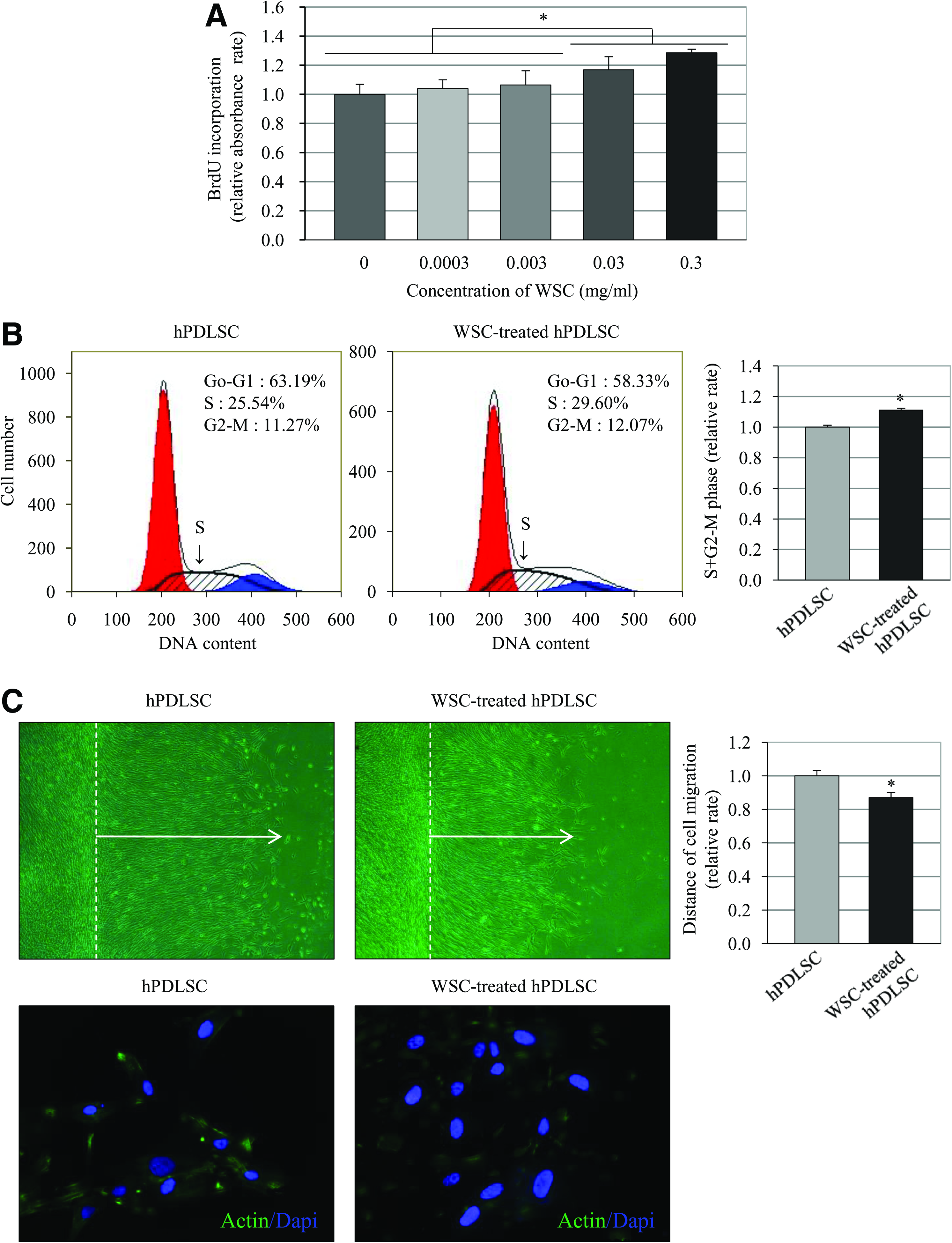
Influence of water-soluble chitin (WSC) on the proliferation and migration of human periodontal ligament stem cells (hPDLSCs).
In vitro analysis of collagen synthesis by hPDLSC and hADSC (ECM staining, Sircol assay, quantitative real-time PCR, and western blotting)
The authors observed that the Sircol assay for soluble collagen demonstrated that the amount of soluble collagen was significantly lower in the WSC-treated hPDLSC group than in the control group (Fig. 2A; p<0.05).
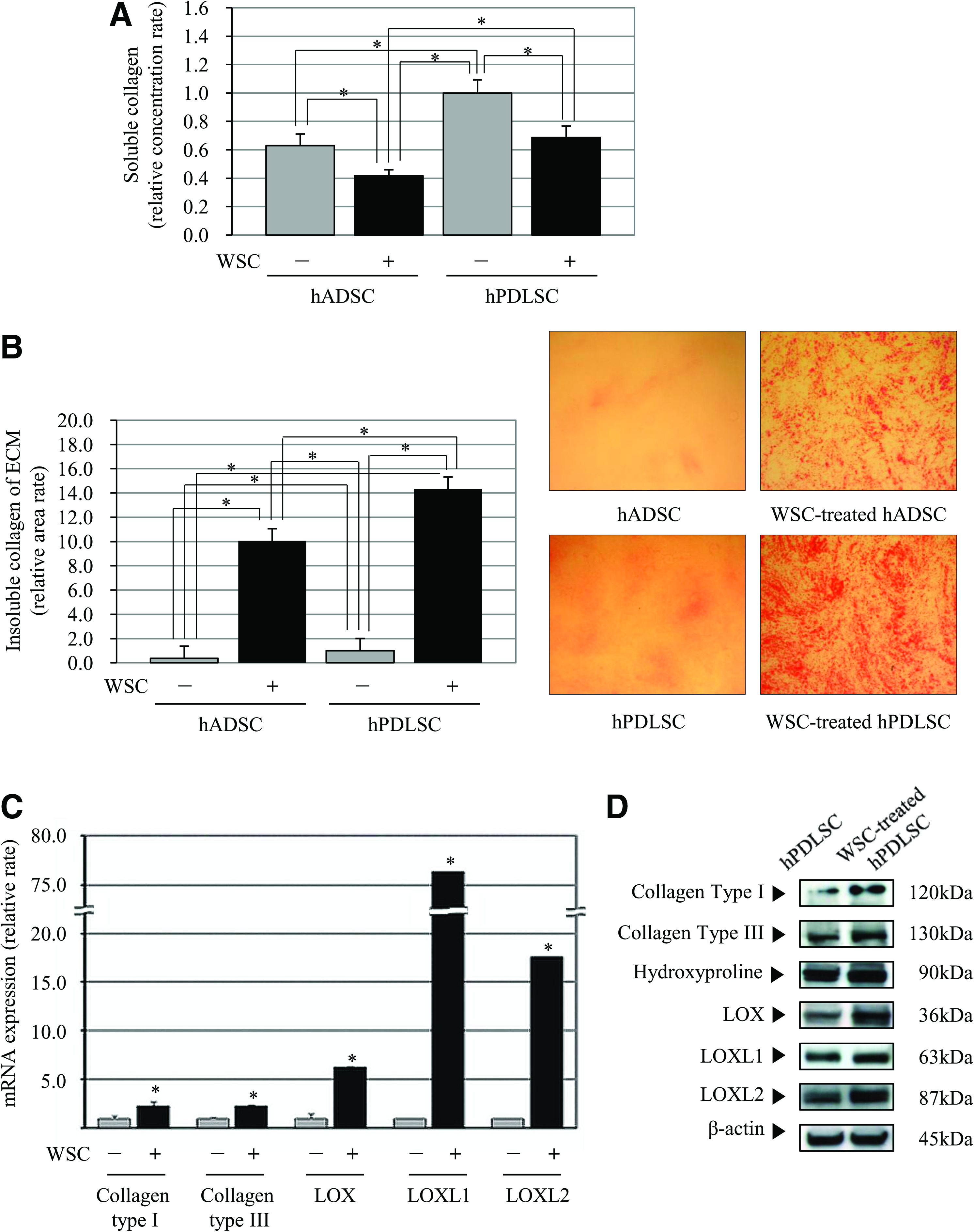
Enhanced collagen synthesis of hPDLSCs in vitro by WSC.
In contrast, the precipitations of ECM stained with picrosirius red after cell lysis, appeared far greater in the WSC-treated hPDLSC group than in the untreated group, suggesting that significantly more and a denser collagen matrix had been generated (Fig. 2B; p<0.05). Also, hADSCs showed less formation of red-stained ECM in comparison with the hPDLSC group. Although the treatment of WSC has significantly increased the ECM formation in hADSC and hPDLSC, the enhancement was significantly greater in WSC-treated hPDLSC.
The real-time PCR and western blotting were performed to determine the changes in collagen expression at the mRNA and protein levels including collagen type I, collagen type III, LOX, LOXL1, LOXL2, and hydroxyproline. PCR results showed that the expressions of mRNA for collagen types I and III were higher in WSC-treated hPDLSCs than in the control group (Fig. 2C). The relative mRNA expressions (β-actin versus collagen type I or III) demonstrated that the intensity of the band corresponding to WSC-treated hPDLSCs was significantly enhanced compared with the control group, indicating that the amount of collagen type I or III mRNA was significantly greater in the WSC-treated hPDLSCs than in the untreated hPDLSCs (Fig. 2C; p<0.05). These results were corroborated by those from the western blot assay (Fig. 2D).
In vivo study of collagen synthesis
We explored the regenerative potential of the combination of hPDLSCs or hADSC with WSC on collagen synthesis in a preclinical healing model. Histological analysis was performed 4 and 8 weeks after transplantation. The state of healing was uneventful with no implant exposure or complications observed at the surgical sites. The implants retrieved from the animals of 4 weeks healing appeared heavier and more rigid in WSC(+)/hPDLSC(+) group (group 4) than in the other groups.
The hematoxylin-eosin and picrosirius red staining revealed that the amount and density of collagen fibers and ECM were significantly greater in cell loaded groups 3 and 4 than in cell free groups 1 and 2 after 4 weeks. Quantification of the findings revealed that collagen-fiber synthesis was far greater in the presence of WSC, and was maximal in group 4 (Fig. 3B, C; p<0.05). This indicates that hPDLSCs can synthesize collagen regardless of the presence of WSC and that WSC promotes further synthesis of collagen fibers.
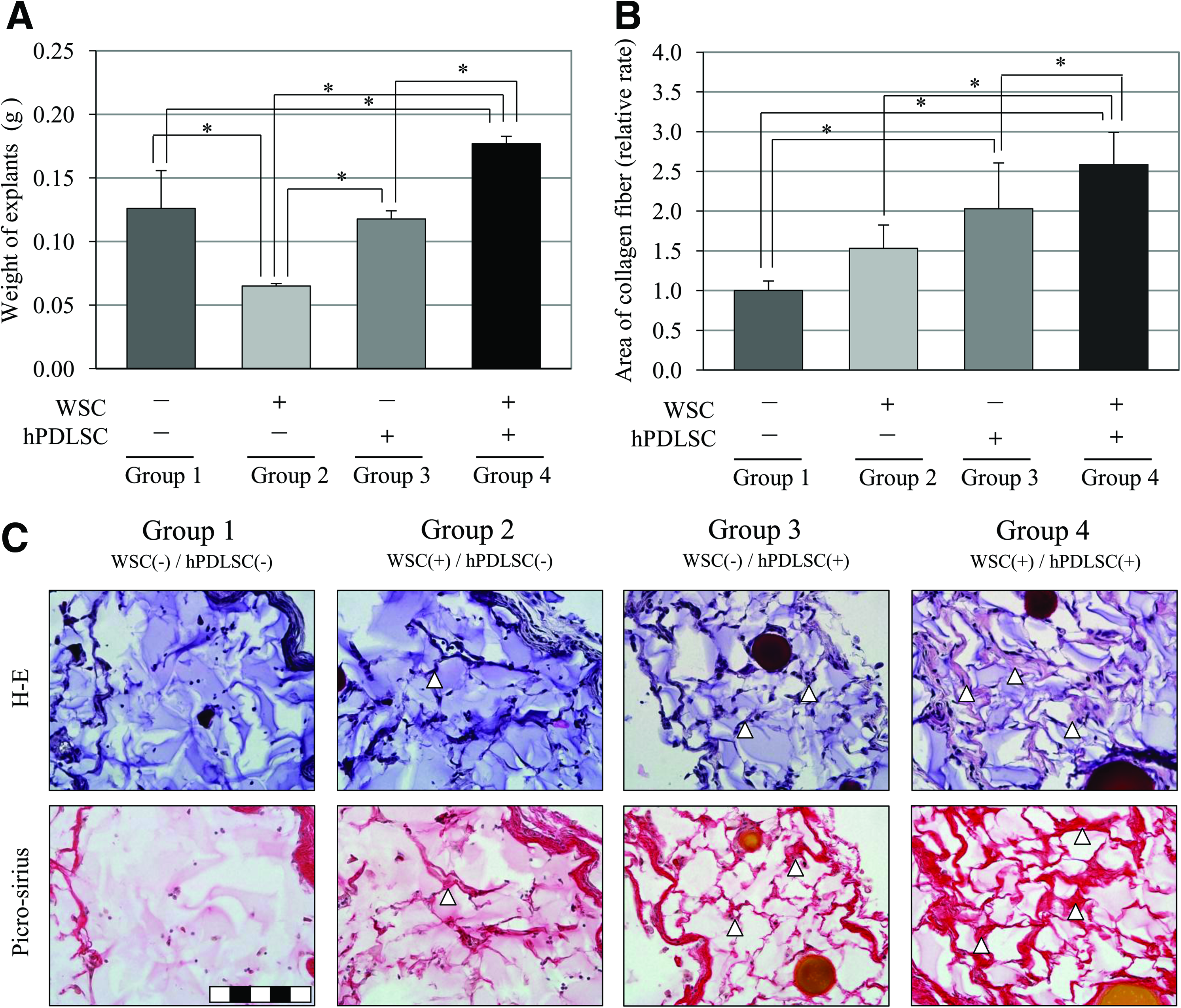
Regeneration of collagen fibers at 4 weeks by hPDLSCs in vivo.
Immunohistochemistry was performed to identify the constructed collagen fibers. The cells around the collagen fibers were positive for anti-human-specific mitochondrial antibodies after 4 weeks of healing (Fig. 4A), and thus it suggests that the regenerated collagen in groups 3 and 4 was induced by the transplanted hPDLSCs not by host cells. The augmented collagen tissues were then exposed to types I and III anticollagen antibodies in vivo, and WSC(+)/hPDLSC(+) group (group 4) revealed more positively stained cells than the other groups (Fig. 4B–E; p<0.05). Therefore, it appears that the accumulation of collagen fibers can be enhanced by the application of hPDLSCs and the combination of hPDLSCs and WSC, especially for collagen types I and III. These results are consistent with those of our in vitro study.
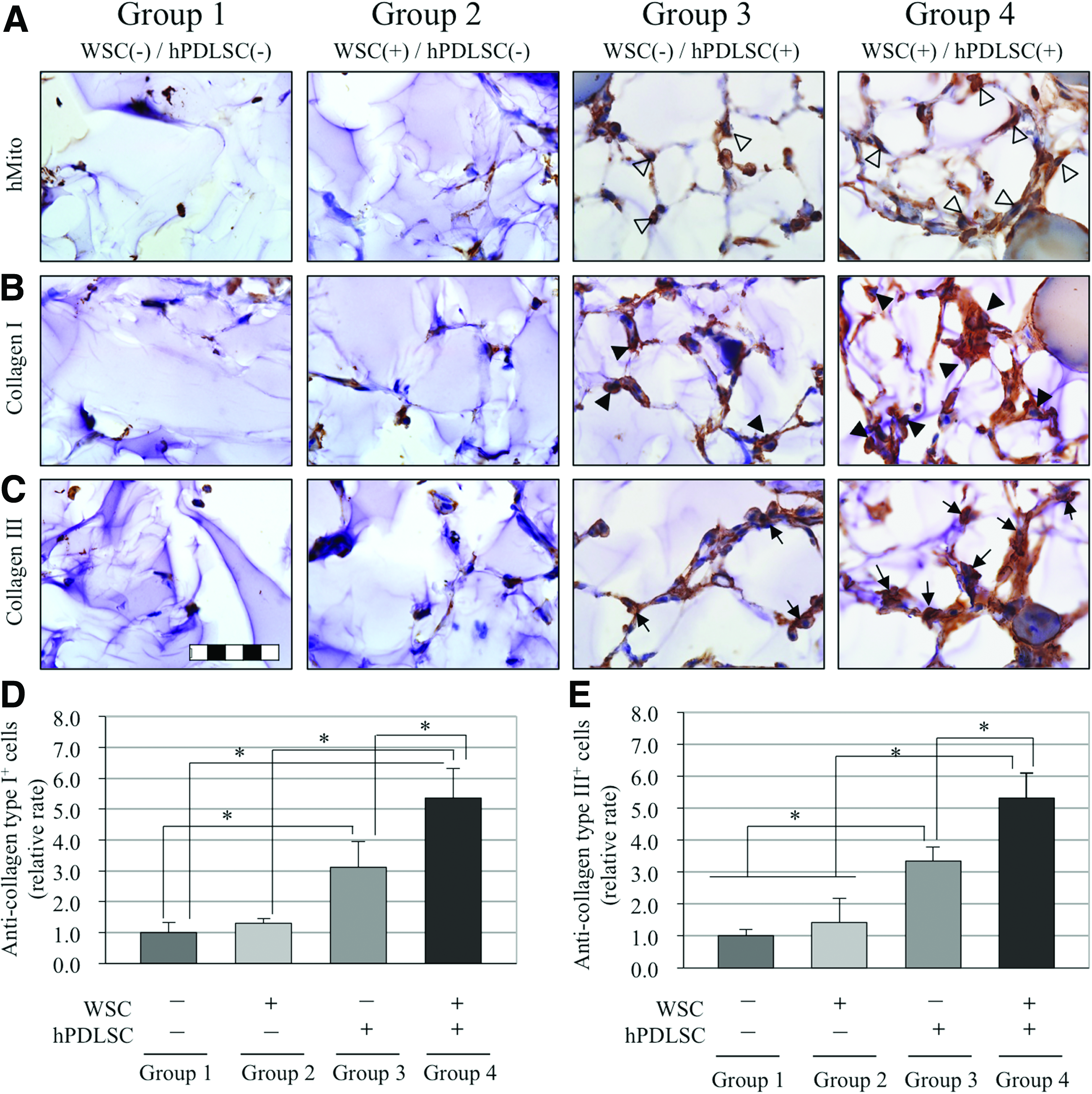
Immunohistochemistry of collagen types I and III. The regenerated collagenous tissues were originated from hPDLSCs and it was confirmed by immunohistochemical staining with an anti-human-specific mitochondrial antibody
To evaluate the extended healing, 8 weeks of healing period was provided (Fig. 5), and similar observations were obtained compared with 4 weeks healing results. Collagen formation by hPDLSCs was significantly greater than that by hADSCs, and the hPDLSCs-induced collagen formation was significantly enhanced by the application of WSC (p<0.05).
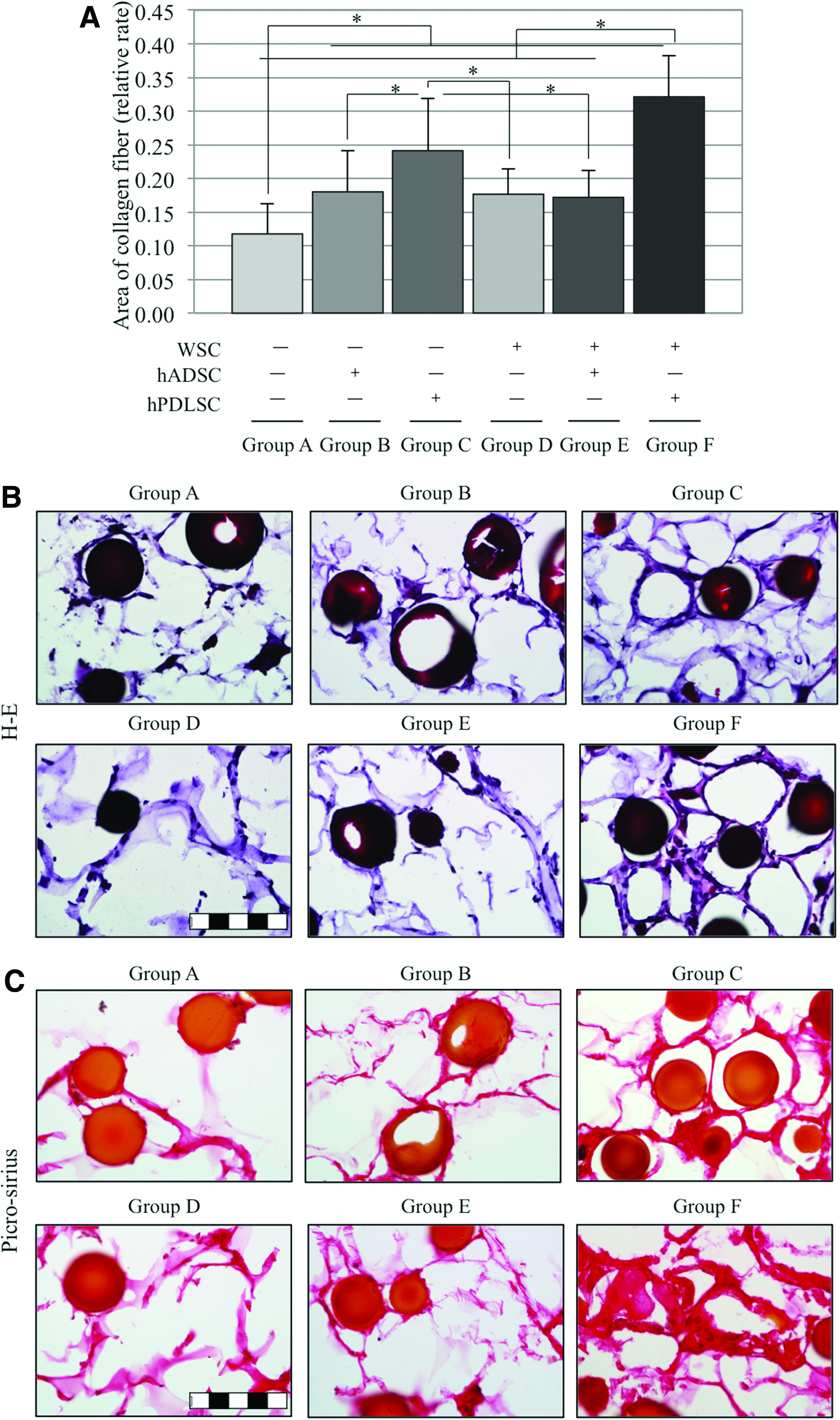
Regeneration of collagen fibers at 8 weeks by hPDLSCs and hADSCs in vivo.
Discussion
The results of this study showed that WSC somehow inhibited the migration of hPDLSCs, and concomitantly promoted their proliferation. Previously, the in vitro results were demonstrated, in which the proliferation of human PDL fibroblast cells increased in chitin contained growth medium, 26 and that the proliferation of periosteum-derived MSCs was significantly greater on chitin-coated glass coverslips. 27 Other studies have also shown that cell proliferation can be significantly improved by the application of chitin and its derivatives.28,29 However, few studies, if any, have investigated how cell migration is influenced by chitin or chitin derivatives, and the current understanding is that the influence of chitin or its derivatives in that migratory behavior is dependent on the type of cell. For instance, chitin or its derivatives have been shown to enhance the migration of vascular endothelial cells,29,30 whereas the migratory activity was significantly reduced in 3T6 fibroblasts treated with chitin, chitosan, and their oligomers and monomers. 30 In the field of esthetic and reconstructive tissue augmentation therapies, minimal cell motility is required for optimal outcomes, since an excessive cell mobility can result unexpected tissue formation outside the desired area. In the present study, we have demonstrated that expression of actin-specific-binding fluorescence in WSC-treated group was significantly decreased, implying that the actin polymerization was deeply downregulated by the application of WSC in hPDLSC. Although the reduced cell migration in our study is beneficial for clinical applications, the involved mechanism has yet to be established. Further studies are required to elucidate the exact mechanism underlying the relationship between WSC and the migratory activity of hPDLSCs.
In the WSC-treated hPDLSC group, we confirmed the presence of a significantly comparable collagen matrix, and then analyzed it using several methods in vitro. Our results demonstrated that insoluble collagens were upregulated and soluble ones were downregulated in the presence of WSC. This discrepancy can be explained by considering the process of collagen synthesis and maturation. Most major collagens, such as types I and III, form fibrils 31 that are essential in the formation and maintenance of tissue. During fibril formation, the solubility of collagen is reduced,31,32 since the fundamental element of fibril collagen is a soluble precursor, procollagen that converts into collagen molecules with the removal of a solvent molecule, C-propeptide, and then gathers and spontaneously assembles into insoluble fibrils.30,31 Thus, our results showing that more insoluble than soluble collagen was produced represent improvements in the general maturation of the collagen molecules. Interestingly, although soluble and insoluble collagen share similar cross-linking networks, the insoluble collagen exhibited a greater compression strength and lower degradation rate than the soluble form. 33 Therefore, the increase in insoluble and mature collagen synthesis by WSC-treated hPDLSCs in our study would be advantageous for stable collagen formation, and the combined application of WSC and hPDLSCs would present a plausible and successful alternative to plastic or cosmetic treatments that require mature and rigid collagen tissue formation.
We were curious as to whether the aforementioned phenomenon of collagen maturation would be reflected by consistent changes in DNA, protein levels, and ectopic transplantation in an animal model. We found that the gene and protein expressions of collagen type I and III were significantly upregulated in the WSC-treated group, and these changes were corroborated by in vivo experiments. The results of other studies into how collagen synthesis is influenced by chitin or its derivatives are consistent with those of our study. The expression of collagen type I mRNA was higher for human PDL fibroblasts treated with chitosan than for unstimulated cells. 26 The dose-dependent increase in the gene expressions of collagen types I and III was more dramatic for human anterior cruciate ligament cells cultured on chitosan-containing implantable material than in the nonchitosan group. 34 Further, Chitipack P, which is a chitin-coated polyester nonwoven fabric, promoted active collagen tissue regeneration, and exhibited greater production of collagen type I, III, and IV in the cat compared with the uncoated version. 35 Therefore, our results have verified the prominent formation of mature collagen both in vitro and in vivo that appears to be correlated with upregulation of the synthesis of collagen types I and III, which are the major components of PDL tissue.
To the best of our knowledge and based on the results from the present study, the application of hPDLSCs results in the synthesis of a significant amount of collagen, and this enhancement of collagen formation matured in a stationary and stable fashion when WSC was added to hPDLSCs as a combined application. Given the superior capability for collagen regeneration and easy accessibility of hPDLSCs, the combination of hPDLSCs and WSC not only provides easier and safer modalities for reconstructive cosmetic treatment without morbidity, but also maximizes the volume of collagen obtained after the treatment. Taken together with the 8 weeks healing results from the present study, hPDLSCs appear to have enhanced activity of collagen formation than hADSCs. This novel application of hPDLSCs and WSC could be an alternative treatment option to traditional esthetic fillers for collagen tissue regeneration. However, the present study performed only preliminary experiments for determining whether and how the collagen synthesis of hPDLSCs is influenced by WSC. For a more plausible and reliable application in the clinical setting, further long-term and comparative studies are needed to confirm the superiority of the combination of hPDLSCs and WSC over other conventional cosmetic fillers.
Conclusion
The findings of this study demonstrated that WSC significantly enhanced the proliferation and inhibited the migration of hPDLSCs, and the combined application of WSC and hPDLSCs increased collagen synthesis in vitro and in vivo, particularly collagen types I and III with simultaneous suppression of cell migration, suggesting the potential application of this combined material treatment for reconstructive soft-tissue augmentation in the cosmetic field.
Footnotes
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (KRF-2008-313-E00587), and by a grant of the Korea Health Technology R&D Project, Ministry of Health, Welfare & Family Affairs, Republic of Korea (A100443). The hADSCs were kindly provided by Pf. B-S. Kim. The author would like to thank Ms. J-M Lee for providing excellent data management.
Disclosure Statement
No competing financial interests exist.
