Abstract
Aims:
The present work aimed to examine impact of tanshinone IIA on intestinal barrier in sepsis and to explore the underpinning mechanisms.
Materials and Methods:
Sepsis induction in Sprague-Dawley (SD) rats was conducted via cecal ligation and puncture (CLP), with subsequent intraperitoneal injection of tanshinone IIA. Intestinal permeability was examined 12 h post-operation using the fluorescein isothiocyanate dextran method. Blood and distal ileum tissue samples were collected for Enzyme-Linked Immunosorbent Assay (ELISA) analysis of oxidative stress and inflammatory markers. Histopathologic examination was performed using hematoxylin and eosin staining and the Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) assay. Immunofluorescence and immunoblot were performed for protein detection. In vitro, Caco-2 cells were administered lipopolysaccharide (LPS) followed by tanshinone IIA treatment, and pregnane X receptor (PXR) and cytochrome P450-3A4 (CYP3A4) protein levels were assessed.
Results:
In sepsis model rats, tanshinone IIA dose-dependently reversed the increased intestinal permeability, bacterial shift rate, ileum Chiu’s score, apoptosis level of ileal mucosa, the elevated serum and ileal Malondialdehyde (MDA), Interleukin-1 beta (IL-1β), and Tumor Necrosis Factor-alpha (TNF-α) amounts, and the enhanced ileal expression levels of Proto-oncogene c-Fos (c-Fos) and tryptase proteins. In addition, tanshinone IIA restored the decreased serum and ileal Superoxide Dismutase (SOD) levels and reversed the reduced ileal expression levels of claudin-1, Junctional Adhesion Molecule (JAM), occludin, and ZO-1. In vitro, tanshinone IIA restored PXR and CYP3A4 levels following LPS stimulation.
Conclusion:
Tanshinone IIA exerts a protective effect in murine CLP-induced sepsis. The underlying mechanism may involve activation of the PXR-CYP3A4 pathway in murine intestinal epithelial cells.
Sepsis is a syndrome involving infection and organ dysfunction and represents the primary factor causing death in intensive care unit patients. 1 The intestinal barrier is important in sepsis occurrence; it encompasses adhesion layers, intestinal mucosal epithelium, and intercellular tight junctions that can prevent intestinal bacteria from translocating and endotoxins from entering the blood and other organs. 2 Occludin and claudins are the most prominent proteins that compose tight junctions in the gut, 3 and their dysregulated expressions contribute to dysfunction of tight junctions, disordered barrier, and increased permeability of intestinal mucosal epithelium. 4 To date, effective measures in preventing the septic damages to intestinal mucosal structure and maintaining intestinal barrier function are lacking.
Oxidative stress represents an important mechanism of tissue injury in sepsis. 5 Sepsis leads to ischemia, hypoxia, and activation of xanthine oxidase, which generates excessive oxygen-free radicals and damages the intestinal mucosa. 6 Oxidative stress also promotes pro-inflammatory effects, for example, endothelial damage, neutrophil recruitment, and cytokine release. 7 Tryptase, an inflammatory mediator secreted by mast cells, exacerbates inflammation, induces microvascular leakage, and causes intestinal mucosal cell apoptosis.8–10 Tanshinone IIA preserves the intestinal mucosal barrier in sepsis. Zhu et al. found that tanshinone IIA can ameliorate microcirculatory disruption and tissue damage by suppressing Reactive Oxygen Species (ROS) production in the rat model of cecal ligation and puncture (CLP)-induced sepsis. 11 Another animal experiment showed that tanshinone IIA inhibits inflammatory response and reduces intestinal injury by lowering the levels of nuclear factor-κB and Tumor Necrosis Factor-alpha (TNF-α) in intestinal tissue samples from rats with experimental sepsis. 12 Reportedly, tanshinone IIA also inhibits apoptosis in intestinal epithelial cells in rats with experimental sepsis, thereby ameliorating intestinal injury. 13 However, few reports have examined the role of intestinal barrier in sepsis, as well as the mechanism of tanshinone IIA, in this disorder.
Current evidence suggests that tanshinone IIA is a pregnane X receptor (PXR) agonist14,15 that can be utilized for the therapy of inflammatory bowel disease, regulating intestinal barrier function and reducing the permeability of intestinal mucosa by activating PXR. 16 Furthermore, repression of a cytochrome P450 enzyme, cytochrome P450-3A4 (CYP3A4), downstream of PXR, was shown to affect the nuclear localization of PXR in sepsis. 17 In the present study, the sepsis rat model induced by CLP was established. The role of different dosages of tanshinone IIA on intestinal permeability in rats with sepsis was investigated by detecting the fluorescein isothiocyanate dextran (FITC-D, 3–5 kDa) leaking out to plasma, as well as the bacterial translocation; the protective effect of tanshinone IIA on intestinal injury was investigated by examining the histopathologic changes, apoptosis level, and the expressions of tight junction proteins. The levels of inflammatory factors (TNF-α and Interleukin-1 beta [IL-1β]) and peroxide markers (Superoxide Dismutase [SOD] and Malondialdehyde [MDA]) were also examined to uncover the underlying mechanism. Most importantly, the effects of tanshinone IIA on PXR and CYP450-3A4 in intestinal epithelial cells were analyzed. Previous studies have demonstrated that activation of the PXR and CYP450-3A4 pathways enhances the expression of tight junction proteins in intestinal epithelial cells and reduces intestinal mucosal permeability, thereby further elucidating the mechanism by which tanshinone IIA protects intestinal mucosal barrier function in sepsis.
Materials and Methods
Animal and grouping
A total of 60 male Sprague-Dawley (SD) rats (7 to 8 weeks old, 200 ± 20 g) were obtained from Beijing Vital River Laboratory Animal Technology (Production License No. SCXK [Jing] 2012-0001; Animal Quality Certificate No. 11400700104944). Animal experiments were approved by our university (ZSLL-2017-176) on December 30, 2017, and followed current Guidelines for the Care and Use of Laboratory Animals Advocated by the National Institutes of Health. All the animals underwent housing at 20°C–25°C with 40%–70% relative humidity under a 12-h photoperiod with ad libitum food and water, for 6 days before sepsis modeling. Subsequently, the animals underwent randomization into 5 groups of 12, including the sham, model, low-tanshinone IIA (sulfotanshinone sodium injection, The First Biochemical Pharmaceutical, China, lot# 1412303), moderate-tanshinone IIA, and high-tanshinone IIA groups (Supplementary Figure S4).
Caco-2 cells and treatment
Human colon carcinoma Caco-2 cells were obtained from Procell (China) and underwent culture in Dulbecco’s Modified Eagle’s Medium (Gibco, USA) to the logarithmic phase. Then, the cells underwent seeding in a 6-well plate at 5 × 105/well and incubation for 24 hours at 37°C in a CO2 incubator. Subsequently, cells underwent pretreatment with 3.4 mmol/L tanshinone IIA and 10 μmol/L ketoconazole (a PXR antagonist 18 ; TargetMol, USA, catalog No. T0679) and an 8 h incubation at 37°C in a CO2 incubator. Subsequently, 100 μg/mL lipopolysaccharide (LPS) (Sigma, USA, catalog No. L2880) was added to these cells, and incubation continued for 36 hours.
Establishment of the sepsis model
Sepsis model induction was conducted by CLP. 19 In brief, the animals were fasted but allowed ad libitum water for 12 hours before operation. Anesthesia was conducted by intraperitoneal (IP) administration of chloral hydrate (300 mg/kg body weight). Next, all rats were shaved on the abdomen, followed by iodophor disinfection. Sham rats then underwent abdominal cavity opening and immediate closure, without ligation or perforation. The animals in the remaining four groups were subjected to 2–3 cm ventral midline incision made to expose intestinal mesentery and cecum. No.3 silk thread was used to ligate a large part of the cecum in a circular form, followed by puncture in two locations using an 18-gauge needle in approximately 1 cm of the distal cecum; the distance between the pinholes was 1 cm. Next, the cecum was gently compressed for extrusion of feces. Finally, the incisions were sutured layer-by-layer, and all rats were subcutaneously injected with Normal Saline (NS) (3 mL/100 g body weight).
Examination of intestinal permeability
Anesthesia utilized chloral hydrate (IP, 300 mg/kg) 11 hours after CLP operation. The ileal end located 20 cm above the ileocecal section was ligated with a 4/0 silk thread. Next, 1 mL of 25 mg/mL FITC-D (Sigma, Lot # FD4, Batch# BCBP6015V) was injected into the distal segment of the ileum. After 1 hour, 1 mL anticoagulated blood was obtained from the IP vein and submitted to a 10-min centrifugation. Plasma was used for determining FITC-D levels on a fluorescence spectrophotometer based on standard curves. Plasma FITC-D concentration was used to indicate intestinal permeability. 20
Analysis of bacterial translocation in viscera
Totally, 0.3 g of liver, spleen, and mesenteric lymph node samples were extracted in a sterile environment, respectively, and subjected to examinations. Each liver, spleen, and mesenteric lymph node specimen was divided into six samples. The samples were incubated with a specific medium and cultured for the identification of bacterial species.
Preparation of ileum tissues
A part of each ileum tissue sample was homogenized at 13,500 rpm using a homogenizer (Fluko, Germany; PRO200) for preparing a 10% ileum tissue homogenate. Each homogenate underwent a 15-min centrifugation at 3,000 rpm, followed by supernatant collection.
The remaining ileum tissues underwent fixation with 10% formalin, with subsequent dehydration with alcohol and xylene. After paraffinization and embedding in wax blocks, 4 µm-thick sections were obtained from the tissue samples placed on slides using a rotary slicer (RM2235, Leica, Germany).
ELISA
Specific Enzyme-Linked Immunosorbent Assay (ELISA) kits (Nanjing Jiancheng Bioengineering Institute, China) were used for determining the levels of MDA (batch# 20150613) and SOD (Batch# 20150610). The ELISA kits used for detecting IL-1β (Batch# 2301B41235) and TNF-α (Batch# 238250423) were purchased from Multisciences (Lianke) Biotech (China). Serum and ileal MDA, SOD, IL-1β, and TNF-α levels were assayed as per the manufacturer’s recommendations.
Assessment of pathologic changes in ileal mucosa
A part of the ileum tissue sections was submitted to hematoxylin and eosin (H&E) staining, with subsequent analysis under an optic microscope (BX43, Olympus, Japan). Evaluation used the Chiu’s score in a blinded manner. 21
Detection of ileal mucosa cell apoptosis
Another part of the ileum tissue sections was used for assessing ileal mucosa cell apoptosis by a Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) Assay Kit (Roche, USA, catalog# 11684817910) as directed by the manufacturer. Three high-power fields at 200× were analyzed per slide to determine the apoptosis index (AI), that is, amount of apoptotic cells per 100 cells.
Immunofluorescence staining
The remaining ileum tissue sections underwent dewaxing using xylene and rehydration with alcohol. Antigen repair utilized 0.01M citrate buffer (pH 6.0) and a microwave. Subsequently, slides underwent overnight incubation with primary antibodies against claudin-1 (1:20; Santa Cruz, catalog No. sc-166338, Batch# C1111), Junctional Adhesion Molecule (JAM) (1:100; Abcam, UK, catalog No. ab180821, Batch# GR227174-1), occludin (1:30; Santa Cruz, catalog No. sc-271842, batch# A0314), ZO-1 (1:50; Boster, China, catalog No. PB0231, Batch# GR208941-1), c-Fos (1:50; Santa Cruz, catalog No. sc-253, Batch# C0215), and tryptase (1:100; Abcam, catalog No. ab2378, Batch# GR93210-3) at 4°C, respectively. Next, slides underwent a 60-min incubation with fluorescent donkey anti-rabbit secondary antibody (Life, USA, catalog No. A21206, Batch# 1110071, excitation/emission: 488/520 nm) and fluorescent goat anti-mouse secondary antibody (Life, catalog No. A21424, Batch# 1588453, excitation/emission: 555/562 nm) at 37°C, at a dilution of 1:800, respectively. The slides underwent three Phosphate-Buffered Saline (PBS) washes and counterstaining with 4',6-Diamidino-2-Phenylindole (DAPI) for 10 minutes at ambient, followed by sealing with glycine in PBS. The fluorescence of slides was visualized by rotating disc-type confocal microscopy (Olympus).
Western blotting
Total protein from ileal tissue homogenates and Caco-2 cells was obtained with RIPA lysis buffer with protease/phosphatase inhibitors. Protein quantity was obtained by the BCA method. Equal amounts (20 μg) of total protein from ileal or Caco-2 cell samples were resolved by Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE), with subsequent transfer onto a polyvinylidene difluoride membrane. After blocking with 5% skimmed milk in Tris-Buffered Saline with Tween (TBST) for 2 hours, incubation was carried out with primary antibodies targeting claudin-1, JAM, occludin, ZO-1, c-Fos, tryptase, PXR (Affinity, USA, catalog No. DF6478), CYP3A4 (Affinity catalog No. DF7001), and Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH) (Abcam, catalog No. ab37168), respectively. Then, secondary antibodies were added for 2 hours, and immunoreactive bands were detected after treatment with the enhanced chemiluminescence reagent. Quantity One and BandScan software (Bio-Rad, USA) were used for quantitation.
Statistical analysis
Data are mean ± standard deviation. SPSS 22.0 (SPSS, USA) was used for data analysis. One-way Analysis of Variance (ANOVA) with post hoc Bonferroni test was performed for comparisons. p < 0.05 indicated statistical significance.
Results
Tanshinone IIA improves ileal permeability
Plasma FITC-D concentration was substantially elevated in the model group versus sham rats (Fig. 1A). However, tanshinone IIA starkly decreased plasma FITC-D concentration dose-dependently versus the model group (p < 0.05). An increase in the positive bacterial culture rate was observed in the model group in comparison with sham rats (p < 0.05), whereas tanshinone IIA dose-dependently reduced the positive bacterial culture rate versus the model group (p < 0.05) (Fig. 1B). These findings indicated that tanshinone IIA markedly improved ileal permeability.
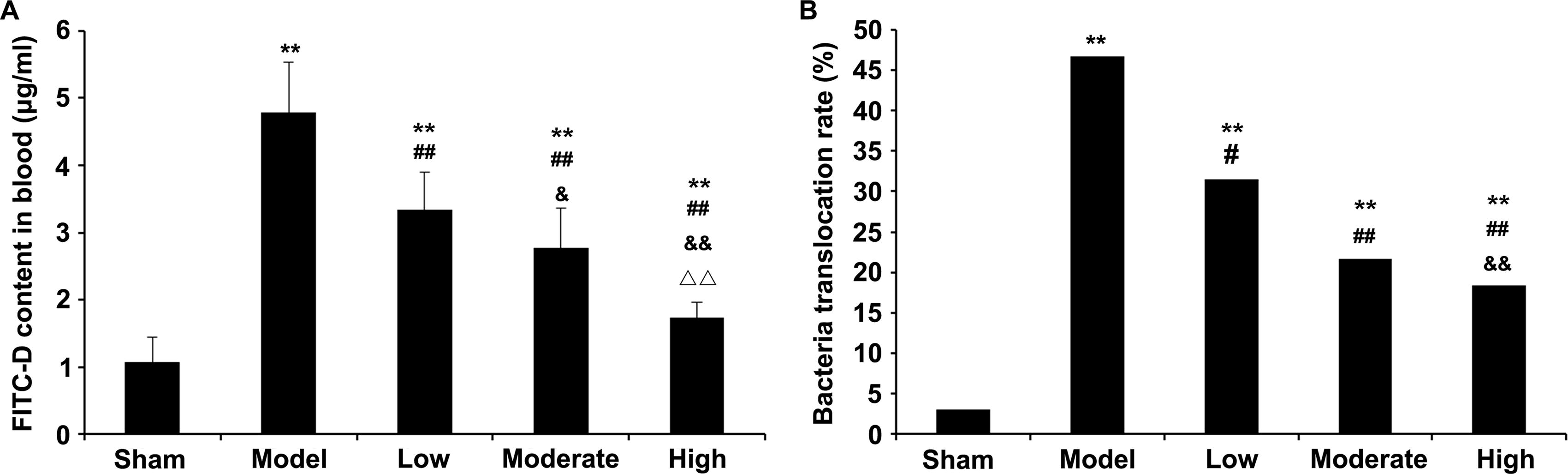
Effect of tanshinone IIA on ileal permeability. Rats were randomized into five groups, including the sham, model, and low-, moderate-, and high-dose tanshinone IIA (sulfotanshinone sodium injection) groups. Ten minutes and six hours after sepsis modeling (including sham operation in the sham group and CLP in the remaining four groups), tanshinone IIA was administered by intraperitoneal (IP) injection in the appropriate groups. Twelve hours post-operation, intestinal permeability was examined by the FITC-D method
Tanshinone IIA alleviates the pathologic changes induced by sepsis in the ileum
As shown in Figure 2, the normal intestinal mucosa and nonobvious inflammatory response were detected in sham animals. In the model group, mucosal epithelial cells were degenerated and detached. Low, medium, and high doses of tanshinone IIA produced a dose-dependent alleviation of the inflammatory response. Versus the sham group, Chiu’s scores in all other groups were starkly increased (p < 0.01). Chiu’s scores were markedly lower in both the moderate and high groups versus the model group (p < 0.01).
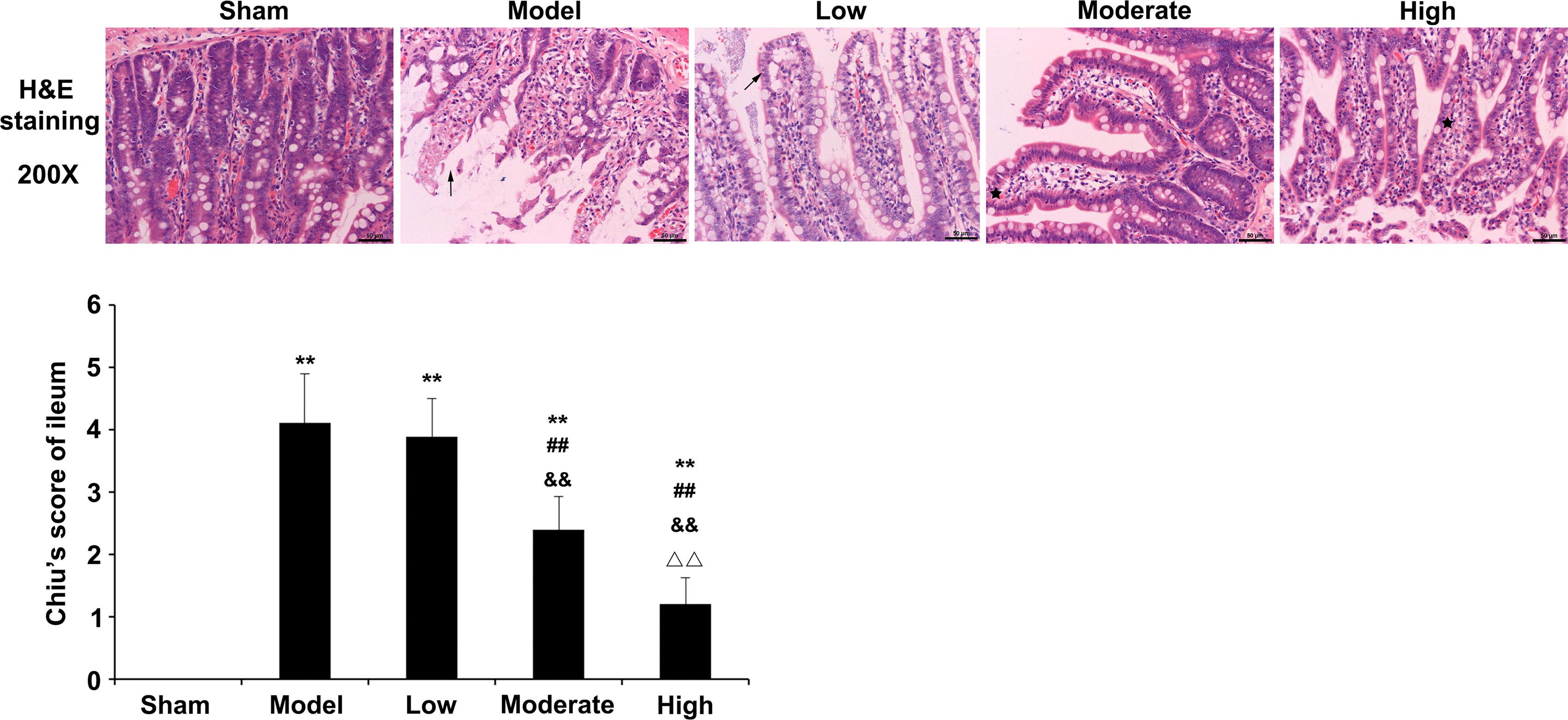
Effects of tanshinone IIA on the pathologic changes of ileum tissues and ileal cell apoptosis. After the rats were sacrificed, distal ileum tissue samples were collected and sectioned. The sections were stained with hematoxylin and eosin (H&E), examined under an optical microscope, and evaluated using Chiu’s score. Scale: 50 µm. n = 12 for the sham group; n = 9 for the model and low-dose tanshinone IIA groups; n = 10 for the moderate- and high-dose tanshinone IIA groups. Data are mean ± standard deviation. **p < 0.01 versus sham group; ##p < 0.01 versus model group; &&p < 0.01 versus low-dose tanshinone IIA group; ΔΔp < 0.01 versus moderate-dose tanshinone IIA group.
Tanshinone IIA prevents apoptosis in ileal cells
As shown in Figure 3, the vascular structures surrounding the ileum in sham rats were normal. In the model group, villi in the ileal mucosa were damaged, and apoptotic cells were markedly increased. Furthermore, in the low-, medium-, and high-dose tanshinone IIA groups, the ileal mucosal villi showed reduced damage, and the degree of ileal mucosal epithelial cell apoptosis was also alleviated. Apoptotic rates in ileal mucosal epithelial cells were greater in all groups versus sham animals (p < 0.01). However, versus the model group, a marked reduction in AI was observed for ileal mucosal epithelial cells in all tanshinone IIA groups (p < 0.01).
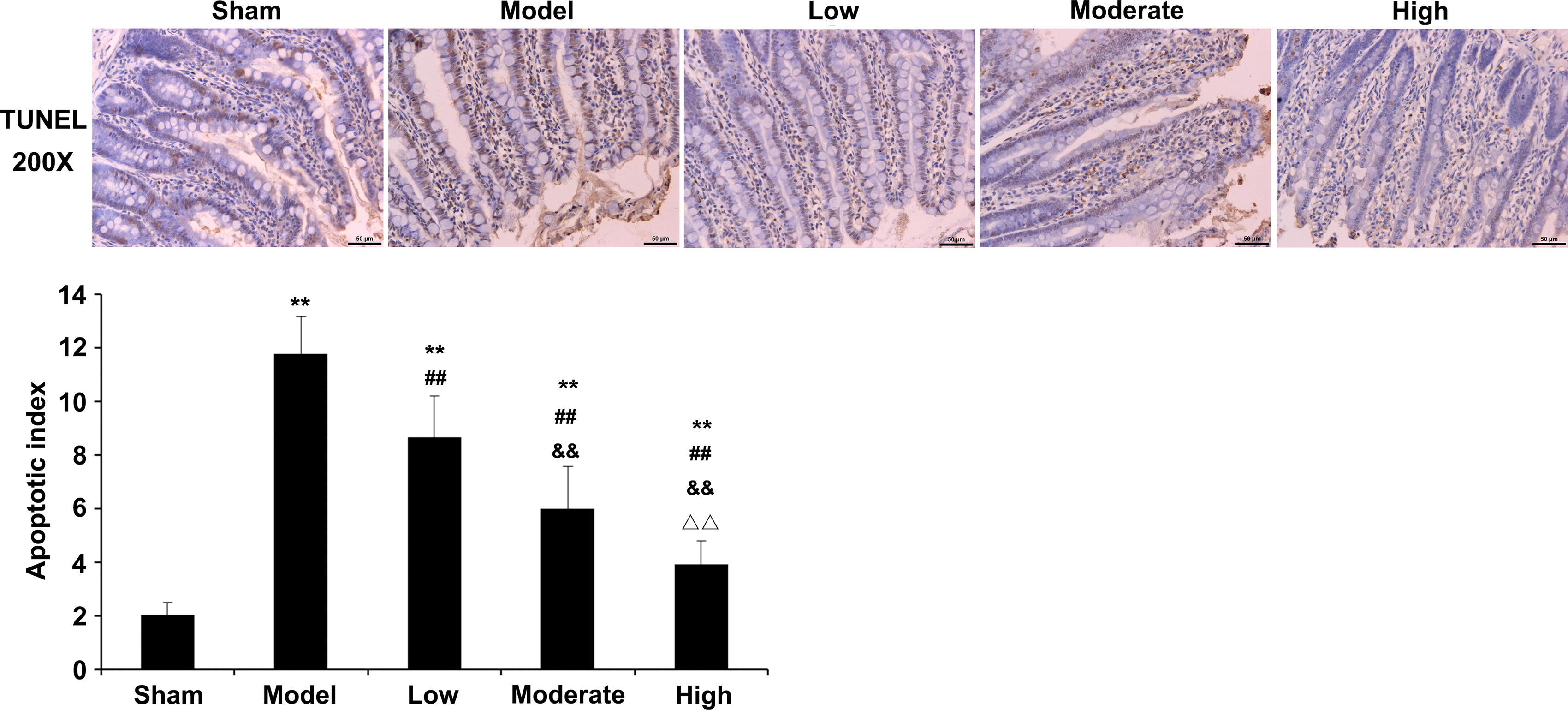
Effects of tanshinone IIA on the pathologic changes of ileum tissues and ileal cell apoptosis. Ileum tissue sections were also used for the analysis of apoptosis of ileal mucosal cells by the TUNEL assay. Scale: 50 μm. n = 12 for the sham group; n = 9 for the model and low-dose tanshinone IIA groups; n = 10 for the moderate- and high-dose tanshinone IIA groups. Data are mean ± standard deviation. **p < 0.01 versus sham group; ##p < 0.01 versus model group; &&p < 0.01 versus low-dose tanshinone IIA group; ΔΔp < 0.01 versus moderate-dose tanshinone IIA group.
Tanshinone IIA decreases MDA, IL-1β, and TNF-α levels and increases SOD contents in serum and ileal samples from sepsis rats
Serum and ileal MDA, IL-1β, and TNF-α levels were starkly heightened in the model group versus sham animals (p < 0.01, Fig. 4). After intervention with tanshinone IIA, serum and ileal MDA amounts were markedly lowered in the moderate- and high-dose groups (p < 0.01), whereas SOD amounts were markedly increased in all tanshinone IIA groups (p < 0.05). In addition, IL-1β and TNF-α amounts in serum and ileum were decreased substantially in all tanshinone IIA groups (p < 0.01) in comparison with the model group. Moreover, substantial decreases in serum IL-1β and serum and ileal TNF-α amounts were observed in the high group versus the low group (p < 0.05, p < 0.05, and p < 0.01, respectively).
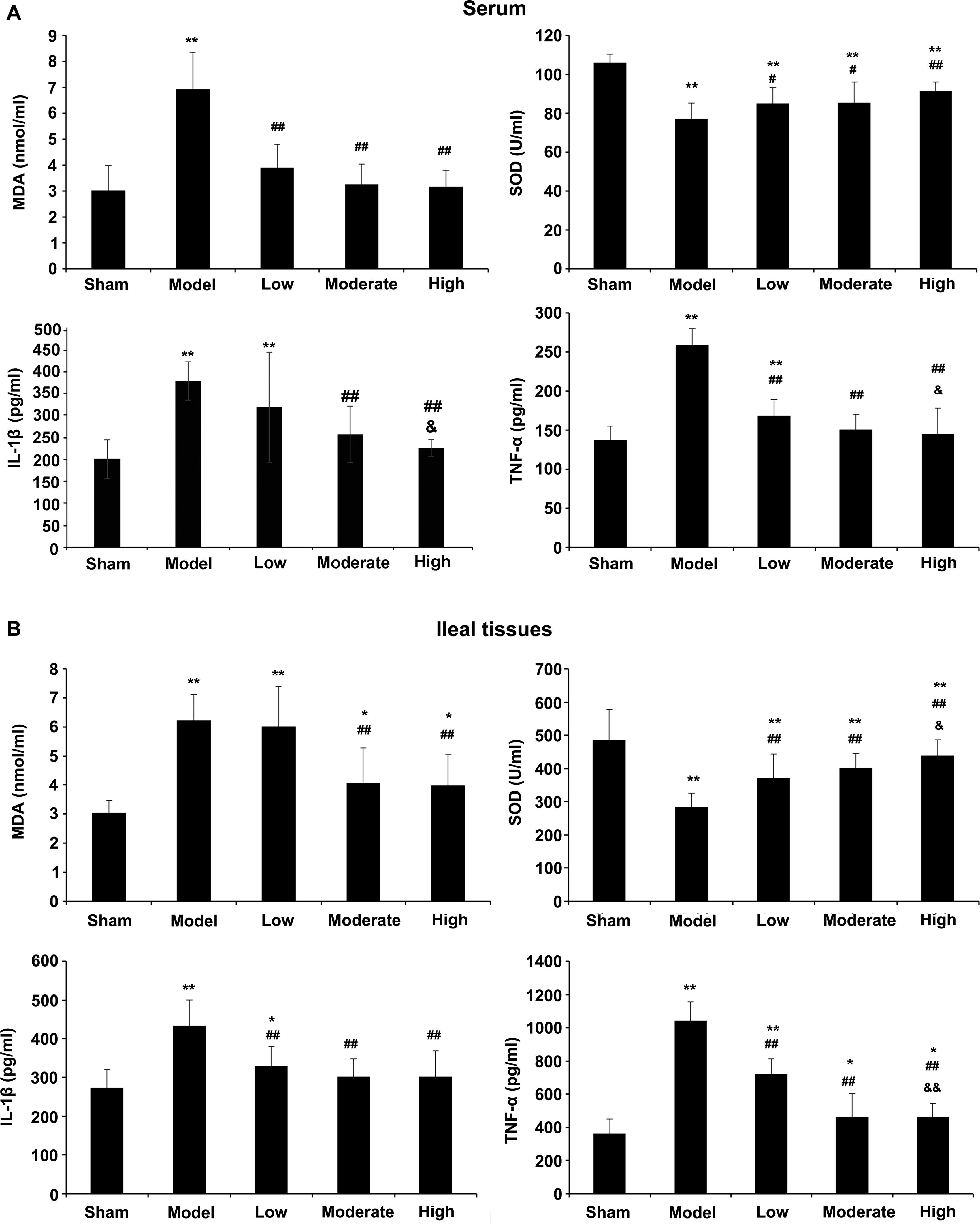
Effects of tanshinone IIA on oxidative stress indexes and inflammatory markers. Twelve hours post-operation, MDA, SOD, IL-1β, and TNF-α levels in serum samples
Tanshinone IIA increases the expression levels of the tight junction proteins claudin-1, JAM, occludin, and ZO-1, but decreases the expression levels of the inflammatory proteins c-Fos and tryptase in the ileum of rats with sepsis
As shown in Supplementary Figures S1 and S2, the highest intensities of ileal claudin-1, JAM, occludin, and ZO-1 were observed in the high group; although these intensities were still greater in the moderate and low groups versus the model group, they were lower than detected in sham animals. In addition, immunofluorescence also revealed that the highest intensities of c-Fos and tryptase were in the model group; meanwhile, reduced intensities were noted in all tanshinone IIA groups, similar to values obtained for sham rats (Supplementary Fig. S3). The above findings indicated that in rats with CLP-induced sepsis, tanshinone IIA could reverse decreases in ileal claudin-1, JAM, occludin, and ZO-1 amounts, as well as increases in ileal c-Fos and tryptase levels.
Western blotting confirmed that tanshinone IIA affected claudin-1, JAM, occludin, ZO-1, c-Fos, and trypsin amounts. As shown in Figure 5A, quantitative analysis revealed that claudin-1, JAM, occludin, and ZO-1 protein amounts were starkly reduced in the model group versus sham rats (p < 0.01). Tanshinone IIA increased the expression of these tight junction proteins (p < 0.01) and substantially reduced the elevated levels of the c-Fos and trypsin proteins caused by sepsis (p < 0.01).
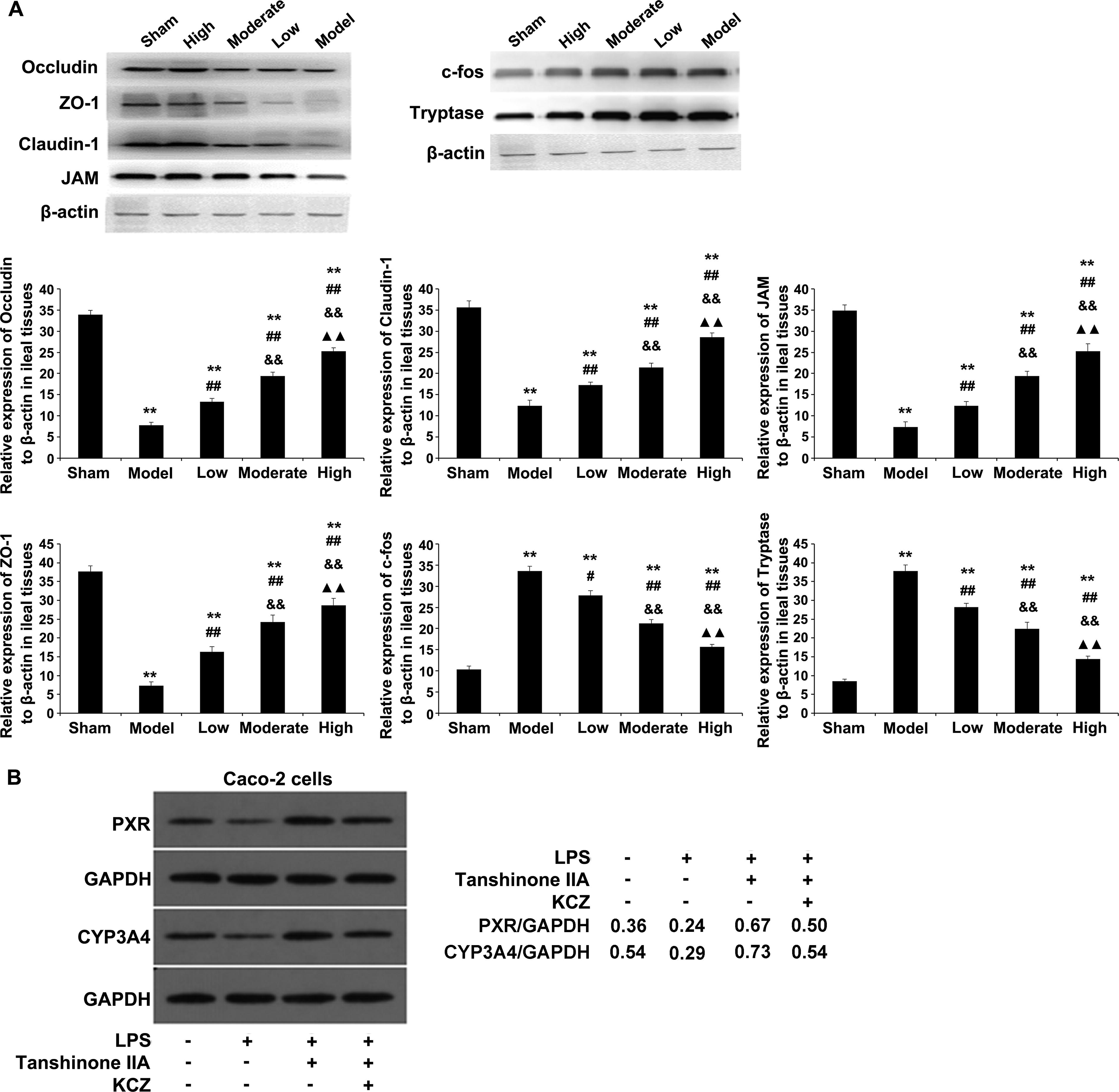
Effects of tanshinone IIA on protein expression levels confirmed by immunoblot.
Tanshinone IIA upregulates the PXR and CYP3A4 proteins in Caco-2 cells
Tanshinone IIA’s effects on PXR and CYP3A4 levels were examined by immunoblot in vitro (Supplementary Figure S5). The data indicated that tanshinone IIA markedly reversed the downregulation of the PXR and CYP3A4 proteins induced by LPS in Caco-2 cells (Fig. 5B).
Discussion
The current work investigated the effect and underpinning mechanism of tanshinone IIA in CLP-induced sepsis in rats and cultured cells. The findings revealed that tanshinone IIA inhibited the inflammatory response dose dependently. Tanshinone IIA ameliorated intestinal mucosal epithelial cell apoptosis and pathologic changes, increased the expression tight junction proteins, reduced the expression of inflammatory proteins, improved the permeability of intestinal mucosa epithelial cells, and reduced bacterial entry. The mechanism of tanshinone IIA may involve PXR/CYP3A4 signaling.
The tight junction, a crucial part of the intestinal mucosal barrier, when deficient or injured, leads to increased permeability of the intestinal mucosal epithelium, translocation of intestinal bacteria and endotoxin, and release of inflammatory factors. Tight junctions comprise transmembrane proteins, including claudins, JAM, occludin, and ZOs. 22 A lack of claudin-1 expression could damage the intestinal barrier. 23 The JAM protein plays a major role in accelerating neutrophilic granulocyte infiltration and aggregation in inflammatory sites and injured tissues. Occludin primarily regulates the structure and function of tight junctions. Abnormal distribution of occludin was observed in the pathologic state (induced inflammatory factors, ischemia, hypoxia, and oxidative stress), which further results in abnormal tight junction structure and function and increases intestinal mucosal epithelial cell permeability, thereby promoting the entry of bacteria, endotoxins, and macromolecular substances into the blood. 24 ZO is a peripheral membrane protein. A stable connective system comprises ZOs (ZO-1, 2, and 3), cytoplasmic occludin, and actin skeleton. 25 In this study, downregulated claudin-1, JAM, occludin, and ZO-1 proteins in the intestinal mucosa were detected in rats with sepsis. The damaged epithelial cells and abnormal expression levels of tight junction proteins were also detected in the model group by immunofluorescence staining. However, tanshinone IIA substantially improved the altered claudin-1, JAM, occludin, and ZO-1 levels dose dependently. The above data indicated that tanshinone IIA markedly improves the defects of tight junction proteins, thereby protecting the structural and functional properties of intestinal mucosal epithelial cells.
The c-Fos gene is considered an immediate early gene and a marker of injury, inflammation, and cell apoptosis. Its increased expression has been associated with gastric mucosal barrier dysfunction, the production of oxygen-free radicals, and ischemia–reperfusion injury. 26 Tryptase not only promotes neutrophil accumulation and the release of inflammatory factors 27 but also stimulates mast cells to induce the release of inflammatory mediators 28 and leads to the injury of intestinal mucosa IEC-6 cells by specific activation of the PAR-2 receptor. 29 In this study, rats with sepsis exhibited elevated c-Fos and trypsin levels in ileal mucosal epithelial cells. Tanshinone IIA downregulated c-Fos and trypsin dose dependently, thereby decreasing inflammation and apoptosis in intestinal mucosal epithelial cells and further protecting intestinal mucosal barrier function from the effects of sepsis.
PXR belongs to the nuclear receptor superfamily and is mostly detected in the liver, intestine, stomach, and kidney tissues. This protein is regarded as a key transcription factor in the regulation of CYP3A4, which substantially affects a majority of metabolic functions, including inhibition of inflammatory cytokine secretion and anti-lipid peroxidation. 30 Therefore, PXR and its downstream CYP3A4 play vital roles in biological regulation and detoxification in the body’s defense mechanisms. A previous study demonstrated that suppression of CYP3A4, downstream of PXR, impacts the nuclear localization of PXR in sepsis. 17 Activation of PXR can decrease intestinal mucosal permeability and enhance the protein expression of tight junctions expressed in intestinal epithelial cells, further improving intestinal mucosal barrier function. 16 Reportedly, tanshinone IIA suppresses the mitochondrial damage induced by Hydrogen Peroxide (H2O2) via PXR activation, further reducing cell apoptosis and exerting a protective effect on cells against injury induced by inflammatory mediators.14,15 The current study also found tanshinone IIA upregulated PXR and CYP3A4, thereby indicating that inhibition of inflammatory response in intestinal epithelial cells, antioxidation, maintenance of intestinal mucosa, and protection of intestinal mucosal barrier function by tanshinone IIA are associated with PXR/CYP3A4. However, the molecular effects of tanshinone IIA on PXR/CYP3A4 and its clinical efficacy require further investigation. Tanshinone IIA is the most abundant and representative structure of the tanshinone of Salvia miltiorrhiza. Previous studies have found that tanshinone IIA possesses various functions, such as anti-oxidation, anti-coagulation, dilation of blood vessels, altering blood viscosity, acceleration of blood circulation, improvement of microcirculation, anti-inflammation, and regulation of immune function. Overdosing may result in coagulation dysfunction, potentially increasing the risk of bleeding. Clinical studies should emphasize the dose–toxicity relation.
Conclusion
Tanshinone IIA exerts a protective effect in murine CLP-induced sepsis. The underlying mechanism may involve activation of the PXR/CYP3A4 pathway in murine intestinal epithelial cells.
Footnotes
Data Availability
All data generated or analyzed during this study are included in this article and supplementary information files.
Authors’ Contributions
L.L. and R.J. conceived and supervised the study. X.X. and X.W. conducted animal experiments. J.Z. and W.L. conducted cell experiments. N.X. and Y.X. analyzed data. S.P. conducted literature review. All authors gave final approval to the version to be published.
Funding Information
This work is supported by National Natural Science Foundation of China (81774070), Zhejiang Chinese Medicine Prevention and Treatment of Serious Disease Key Planning Project (No. 2012ZGG001), and Natural Science Foundation Project of Zhejiang province (LY14H290006).
Author Disclosure Statement
The authors report that there are no competing interests to declare.
