Abstract
Background:
Tissue adhesives are increasingly being used as alternatives to traditional sutures and staples in surgical incision closure applications. Exofin Fusion, a novel cyanoacrylate-based adhesive with a mesh, has been developed to enhance surgical incision closure. This study investigates the microbial barrier effectiveness of Exofin Fusion.
Methods:
An in vitro assessment of 35 plates, including challenge organisms, negative controls, and positive controls, was conducted by an independent research organization, North American Science Associates, Inc (NAMSA), USA. Exofin Fusion was applied to the plates aseptically. Each plate was then inoculated with specific challenge organisms. Observations for visible growth or color changes in the media were made over a 21-day period.
Results:
The results demonstrated no growth or color changes on test plates for common pathogens such as Staphylococcus aureus, Staphylococcus epidermidis, Methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and Aspergillus brasiliensis throughout the 21-day period. Candida albicans showed no growth or color changes up to day 20; however, two out of five replicates exhibited growth on day 21.
Conclusions:
The study confirms the microbial barrier properties of Exofin Fusion, as it effectively prevents the penetration of challenge organisms for the entire test period. Despite the growth of C. albicans in two replicates on day 21, the adhesive demonstrates remarkable efficacy in protecting against microbial infiltration. These findings suggest the potential clinical utility of Exofin Fusion in surgical incision closure applications, highlighting its significance in healthcare settings.
Various surgical incision closure techniques are utilized in medical practice, ranging from traditional methods like sutures, surgical tapes, and staples to more modern alternatives such as ligating clips and adhesives. Adhesives, in particular, have gained substantial popularity owing to their minimally invasive application and user-friendly nature, all the while delivering favorable cosmetic outcomes. 1 Their utilization extends to reducing physical discomfort and alleviating patient anxiety, notably among pediatric cases where needle injections for anesthesia can be a source of fear.1,2 They find extensive use in both emergency and elective medical procedures.
In the United States, each year sees millions of individuals seeking emergency room treatment, a considerable proportion of which involve traumatic wounds and injuries necessitating wound care.3,4 Notably, these emergency wound care cases bear a notable risk of infection. Surgical incisions also have risk of surgical site infections (SSIs) which pose a substantial threat to patients undergoing various surgical procedures, carrying the potential for severe consequences. For instance, SSIs affect ∼1% of patients undergoing total hip replacement and 0.5% of those undergoing total knee replacement (TKR). Even more concerning is that nearly half of these SSIs involve deep tissues or the underlying joint. 5
Exofin Fusion is a novel cyanoacrylate-based topical skin adhesive along with a polyester mesh. It is commonly used in orthopedic procedures such as joint replacements and spinal procedures which involve long incisions. In the realm of orthopedic surgery, the predominant pathogen responsible for SSIs is Staphylococcus aureus, accountable for 39% of these infections, with 58% being meticillin-sensitive and 42% meticillin-resistant. Following TKR, coagulase-negative Staphylococci becomes the predominant infective pathogen, contributing to 30% of infections. Other common pathogens in these cases include Enterobacteriaceae (18.8%), Enterococcus spp. (8.0%), Pseudomonas aeruginosa (3.6%), Pseudomonas spp. (3.3%), Streptococcus spp. (2.8%), anaerobic bacilli (1.4%), anaerobic cocci (1.3%), Acinetobacter spp. (0.4%), and fungi (0.3%). 5
Preventing surgical incision infections hinges on the microbial barrier properties of closure materials. Sutures can act as pathways for skin flora migration, potentially causing infections. 6 The use of cyanoacrylate adhesives, such as octyl cyanoacrylate, has expanded in medical settings owing to their ease of use and quicker application and provides microbial barrier function.7,8 Research indicates that surgical incisions closed with cyanoacrylate exhibit substantially lower staphylococcal counts compared with those closed with sutures. 9 This has led to the widespread adoption of cyanoacrylate adhesives in surgical incision closure, showcasing products like octyl cyanoacrylate. 10 Cyanoacrylate-based topical adhesives with mesh surgical incision closure systems are commonly used in orthopedic procedures involving valuable implants. However, there is a lack of evidence in the English literature regarding whether these adhesive plus mesh systems provide any microbial barrier benefit and, if so, for how long.
This study aims to investigate the microbial barrier properties and duration of effectiveness of Exofin Fusion, an innovative cyanoacrylate-based adhesive plus mesh system, through an in vitro model. The ability of Exofin Fusion to protect against microorganism infiltration is crucial for surgical incision closure applications, particularly in procedures where a costly implant is placed beneath the surgical incision. This research builds upon the success of cyanoacrylate adhesives, such as octyl cyanoacrylate, in reducing infection risk, presenting a potential advancement in surgical incision management and patient care.
Materials and Methods
Test method
This study was carried out independently by a third-party research organization, North American Science Associates, Inc (NAMSA). NAMSA prepared the design of the study, collected data, analyzed, and interpreted the results. A total of 35 plates were required to test each challenge organism in five replicates, with an additional three plates each for negative controls. Five 100 × 15 mm D/E agar plates, per challenge organism were labeled. Five Cetrimide agar plates were labeled for P. aeruginosa. Altogether, 35 plates were labeled for seven challenge organisms. For negative control, three 100 × 15 mm D/E agar plates and three Cetrimide agar plates were labeled. For positive control, 21 (18) 15 × 100 mm D/E agar plates three per challenge organism and three Cetrimide agar plate were labeled. The liquid adhesive was applied onto 25 × 50 mm surface in the middle of the plate. The applicator was activated by twisting tube into the applicator. The bulb was squeezed, and the liquid adhesive was applied over the entire surface of the mesh and allowed to cure for 3 min.
Each plate was inoculated with 10 μL (1–5 × 106 Colony Forming Unit [CFU]/10 μL) of the respective challenge organism (Table 1).
Test Organisms and Their Inoculum Concentration
Test organisms:
Staphylococcous aureus
Staphylococcous epidermidis
Staphylococcus aureus (MRSA)
Pseudomonas aeruginosa
Esscherichia coli
Candiada albicans
Aspergillus brasiliensis
Target population: 1–5 × 108 CFU/mL
Inoculum population: 1–5 × 106 CFU
Inoculum volume: 10 µL
Incubation temperatures: bacteria incubated at 37 ± 2°C; yeast and mold incubated at 20–25°C.
After inoculation, the lid was placed on the plate, and the plate was sealed with Parafilm to avoid loss of moisture. All bacterial plates were incubated at 37 ± 2°C and yeast and mold plates at 20–25°C for 21 days.
Every third day, the plates were carefully removed from the incubator and an observation was made for any visible growth or any color change of the media from purple to yellow with the D/E agar plates and any color change from white to blue-green with the Cetrimide agar plates. The observations were documented in the worksheet. In the absence of growth or media color change, the plates were placed back in the incubator and documented in the worksheet. Plates that were observed to show growth or media color change were removed from the incubator and documented on the worksheet. The positive control plates were removed from the incubator once the growth or media color change was observed and documented on the worksheet. After 21 days of incubation, the test and control plates were removed from the incubator. The results were recorded as growth or no growth and media color change from purple to yellow for D/E agar plates and from pale white to greenish blue for the Cetrimide agar plates. If a color change or growth was observed in test or negative control plates, a gram stain was performed to determine if the growth organism had a gram stain reaction and morphology consistent with the challenge organism.
Acceptance criteria
Inoculum verification plates should yield a population of ∼1.0 × 106 CFU/aliquot.
Uninoculated media control plates or plates containing the uninoculated test article (negative control) should show no color change or growth of any test organisms.
Inoculated positive control plates should exhibit color change or growth of the challenge test organisms.
Results
Inoculum verification plates did yield a population of ∼1.0 × 106 CFU/aliquot.
There was no color change or growth of any test organisms on the uninoculated media control plates or plates containing the uninoculated test article (negative control).
Color change or growth of the challenge test organisms was observed on the inoculated positive control plates.
No growth or color change was observed on the Exofin®Fusion Skin Closure System plates tested with S. aureus, S. epidermidis, S. aureus (MRSA), P. aeruginosa, E. coli, and A. brasiliensis for 21 days (Table 2).
Observation Records After Every 3 Days for 21 Days
CC = color change; G = growth; NG = no growth; NCC = no color change.
Therefore, Exofin Fusion demonstrated antimicrobial barrier properties, preventing these challenge organisms from penetrating its surface into the agar. For C. albicans, no growth or color changes were observed on the test article up to day 20, but two (Figs. 1 and 2) out of five replicates demonstrated growth on day 21.
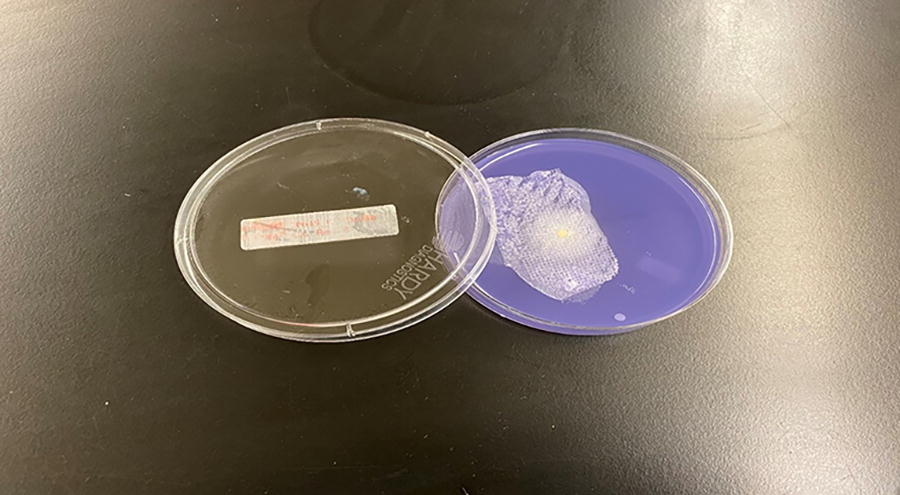
First Candida albicans replicate showing color change on 21st day.
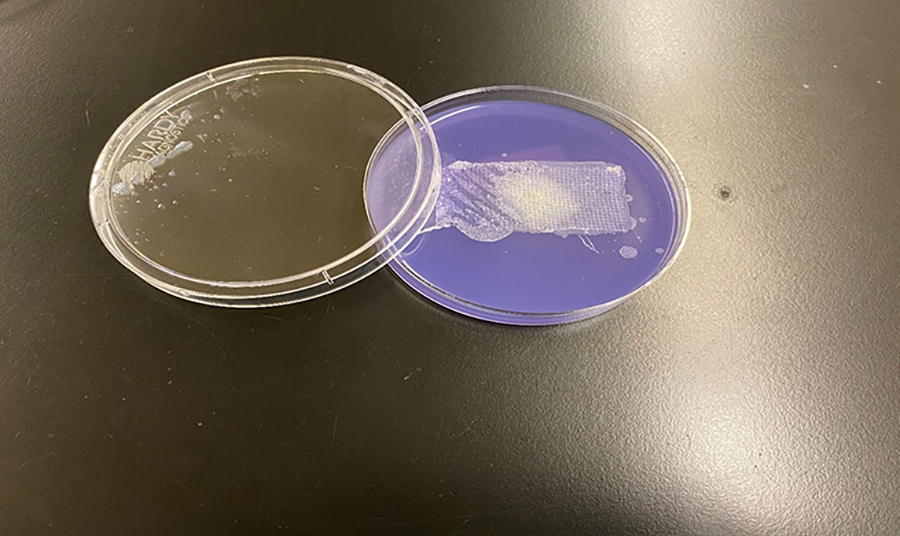
Second Candida albicans replicate showing color change on 21st day.
Statistical analysis
For the results on the 20th day, n = 35 (representing the number of test samples), x = 0 (indicating the number of observed failures), and g = 0.95 (denoting a predetermined level of confidence), we conducted a statistical analysis utilizing the binomial distribution. The goal was to calculate the probability of observing a failure (p). This analysis yielded a value of p = 0.009936. Complementarily, we also calculated the probability of achieving a success (q), which amounted to q = 0.99006.
For the results including 21st day with two positive tests for C. albicans n = 35 (representing the number of test samples) and x = 2 (indicating the number of observed failures), while maintaining a confidence level of g = 0.95, we can employ the binomial distribution to calculate the probability of observing a failure (p). The statistical analysis yielded a value of p = 0.022, which reflects the probability of observing a failure. Additionally, the complementary probability of achieving a success (q) can be calculated as q = 0.978.
The data indicated that Exofin Skin Closure System provided a barrier to microbial penetration with 95% confidence of 99% efficacy for 20 days and with 95% confidence of 97.8% efficacy for 21 days in this in vitro model.
Discussion
Surgical incision healing encompasses a series of intricately coordinated events. Epithelial cells, the only cells capable of regenerating during surgical incision healing, initiate the process immediately after surgical incision closure. Basal cells migrate across and into the incised or lacerated dermis, sealing the approximated surgical incision edges. 11 Within 48 h of injury, a barrier against external elements is established, and the fibroblast phase commences. However, during this period, the surgical site tissue has minimal tensile strength, relying solely on the surgical incision closure device to maintain its structural integrity. 12
Cyanoacrylate adhesives, including octyl cyanoacrylates, provide a less invasive and versatile alternative to traditional sutures for surgical incision closure. 9 They are favored for their high tensile strength and flexibility, making them suitable for various wounds, including facial lacerations and select torso and extremity lacerations. Octyl cyanoacrylate’s effectiveness extends to non-linear and long incisions, particularly for partial-thickness surgical s. It is advisable to complement full-thickness surgical incision closure with subcutaneous sutures. Octyl cyanoacrylate’s slow degradation rate and non-toxic nature make it a valuable option in many cases. 13
Octyl cyanoacrylate has undergone comprehensive investigation regarding its antimicrobial properties, both through in vitro and clinical studies. Research indicates lower infection rates in surgical incisions closed with topical octyl cyanoacrylate tissue adhesive.2,14,15 This favorable outcome is attributed to the substantial antimicrobial effect exhibited by octyl cyanoacrylate adhesive against gram-positive organisms and gram-negative organisms, as demonstrated in standard antimicrobial disc sensitivity tests.16–18 Octyl cyanoacrylate has also been reported to have high efficiency in preventing bacterial intrusion, except for P. aeruginosa. 19 Another proposed mechanism may be the absence of a foreign body implanted beneath the skin surface may contribute to its effectiveness, as octyl cyanoacrylate adhesive is applied topically rather than implanted. 20
Our study primarily aimed to evaluate the antimicrobial efficacy of the 2-octyl cyanoacrylate-based Exofin Fusion closure system against pathogens commonly linked to infections. Our study has confirmed that Exofin Fusion can successfully provide microbial barrier for 21 days. DERMABOND™ PRINEO Skin Closure System provided a flexible microbial barrier with 98.43% protection in vitro for 72 h against organisms commonly responsible for SSIs. 21 The results of our study unequivocally demonstrate that Exofin Fusion has potent antimicrobial properties against common pathogens responsible for infection of surgical incisions for substantially larger duration of 21 days. It was proposed that this antimicrobial effect is more pronounced against gram-positive organisms which may be because of strong electronegative charge present in the cyanoacrylate monomer, which interacts with the positively charged carbohydrate capsule often found in gram-positive organisms. 22 Recent research has revealed a broader antibacterial effect than previously reported, as both gram-negative and gram-positive bacteria are substantially eradicated. This effect is attributed to the low water content of 2-octyl cyanoacrylate, which leads to the diffusion of water from bacterial cells, rendering them barren and ultimately causing their death. 18
It is essential to acknowledge several limitations in our study. First, we did not compare the effectiveness of 2-octyl cyanoacrylate with standard antimicrobial compounds, which hinders our assessment of the relative size of inhibition zones. Future research should explore this aspect to gain a clearer understanding of 2-octyl cyanoacrylate’s efficacy. Additionally, the inclusion of only aerobic bacteria and fungi excluded anaerobic bacteria, which are frequently associated with a substantial number of post-operative infections. Future studies could incorporate anaerobes for a more comprehensive assessment of microbial barrier function. Moreover, the increased antimicrobial activity of liquid tissue glues against gram-positive organisms and E. coli is particularly noteworthy in surgical settings where contamination may occur during the procedure rather than post-operatively. 23
It is also important to recognize the differences between in vitro models and clinical settings. Our in vitro tests are more sensitive than animal infection models; even the introduction of a single microorganism (1 CFU) could lead to a positive test, whereas in vivo models typically require a higher number of microorganisms to induce infection because of the immune response and resistance present in animals. Existing research indicates that ∼80% of surgical incisions will become infected if colonized with 105 CFU/g of S. aureus and closed with the least reactive percutaneous monofilament sutures. 24 Additionally, the temperature ranges used for incubating bacterial and fungal cultures differ from the human body’s constant temperature of 37°C. Although our conditions align with laboratory protocols, this variance may influence microbial growth differently than in in vivo conditions. Lastly, although manufacturer’s recommended adhesive application protocol were followed, variations in adhesive volume during clinical use could impact the microbial barrier’s effectiveness, highlighting the need for further investigation in this area.
Conclusion
In conclusion, this study aimed to assess the microbial barrier properties of Exofin Fusion topical adhesive in vitro. The results obtained in these experiments provide compelling support for the hypothesis that Exofin Fusion tissue adhesive constitutes an effective barrier to bacterial penetration in the described in vitro model. These barrier properties extend to gram-positive and gram-negative species, as well as motile and non-motile species for up to 21 days.
Footnotes
Author’s Contributions
C.D.P.: Writing, editing, and reviewing. NAMSA: Conceptualization, methodology, and data collection and interpretation.
Funding Information
This study was funded by
Author Disclosure Statement
C.D.P. discloses that he works as a medical advisor for Chemence Medical Inc., the manufacturer of the Exofin Fusion Skin Closure Device. This affiliation may present a potential conflict of interest regarding the research findings and conclusions presented in this article. However, the author affirms that the research was conducted objectively, and the results reported are accurate and unbiased. Any potential influence from this affiliation on the research process or article content has been transparently disclosed.
